Abstract
Menopause is a high-risk period for osteoporosis, which may be exacerbated by HIV and/or antiretroviral therapy (ART). Our goal was to study the impact of switching from tenofovir disoproxil fumarate (TDF) to tenofovir alafenamide (TAF) on bone mineral density (BMD) in peri- and early postmenopausal women living with HIV. This is a randomized international multicenter study of an early versus delayed (48-week) switch. BMD was measured by dual energy X-ray absorptiometry scan. Thirty-four women were enrolled: 19 in the immediate and 15 in the delayed switch arm from September 2017 to April 2019; 30 completed the 96-week protocol. The study closed for futility during the COVID-19 pandemic. The median (intraquartile range [IQR]) age was 51 years (47, 53), with a median (IQR) of 16.5 years (14, 23) since HIV diagnosis, median (IQR) 14 years (11, 20) of ART, and mean 8.6 years TDF. At enrollment, TDF was used in combination with a boosted protease inhibitor (n = 7), a non-nucleoside reverse transcriptase inhibitor (n = 13), an integrase inhibitor (n = 11), or more than one ART class (n = 3). The median (95% confidence interval [CI]) percentage change in BMD at the lumbar spine from 0 to 48 weeks in the immediate switch group was 1.97% (−1.15 to 5.49) compared with a median (95% CI) decrease of 2.32% (−5.11 to 0.19) in the delayed arm. The median (95% CI) percentage change in BMD from 0 to 96 weeks was 2.33% (0–4.51) in the immediate arm compared with 0.70% (−3.19 to 2.47) in the delayed arm. We demonstrated a trend to increased BMD at the lumbar spine after a switch from TDF to TAF in peri- and early postmenopausal women living with HIV.
Clinical
Introduction
The HIV population is aging with more than half older than 50 years of age in North America and Europe. Consequently, the focus in care is increasingly focused on preventing and managing comorbidities. 1 –4 Osteoporosis is more common in persons living with HIV than in the general population 5 and increases with age. 6,7 HIV may independently contribute to low bone mineral density (BMD) through a combination of viral, immune, inflammatory, and treatment-related effects on bone metabolism. 8 –13
Statistically significantly greater declines in BMD are seen in persons with HIV initiating antiretroviral therapy (ART) that includes tenofovir disoproxil fumarate (TDF) compared with abacavir, 14 and smaller BMD declines are observed in those initiating nucleoside reverse transcriptase inhibitor sparing regimens. 15 BMD losses may continue or plateau with prolonged TDF use. 16,17 Observational cohort studies report an increased fracture risk in those on TDF, especially with increasing age. 5,18,19
Tenofovir alafenamide (TAF), another prodrug of tenofovir, causes less BMD loss than TDF, and a switch from TDF to TAF is associated with a statistical increase in BMD. 8,20 –22 The clinical significance of this change is unknown as the studies are underpowered and too short duration to address fracture risk and have typically studied BMD changes in a younger male population. 6,23 BMD assessment with dual energy X-ray absorptiometry (DXA), although established for the diagnosis of osteoporosis and fracture risk assessment in postmenopausal women, 24 –26 is not validated in young men. Thus, it remains unclear whether use of TAF will decrease bone loss and fracture rates in older women with HIV.
Given sample size considerations and time to fracture endpoints, clinical validation studies will not be possible. Demonstration of changes in BMD that correlate with fracture risk in a peri- and early postmenopausal women population may provide better supportive evidence.
Menopause transition may be a vulnerable period when changes in BMD loss are enhanced. 27 Studies have shown increased immune activation during this period, which could enhance bone loss. 28 We hypothesized the clinical relevance of the impact of this switch might be better understood in a population in whom fracture risk has been validated with BMD.
Our goal was to use a randomized controlled clinical trial to determine the impact of switching from TDF to TAF in peri- and postmenopausal women living with HIV.
Materials and Methods
Study population
Participants were women living with HIV in the perimenopausal or early postmenopausal period receiving a TDF-containing regimen in one of five Canadian and two Italian participating HIV treatment centers.
Inclusion criteria
Cis gendered women, aged 45–55 years, with documented HIV-1 infection, in the perimenopausal period (typical symptoms) or up to 10 years of menopause (as documented by loss of menses for 12 months by history and chart review) and receiving an ART regimen containing TDF with stable viral suppression (plasma HIV-1 RNA <50 copies/mL) for >24 weeks were included.
Exclusion criteria
Women with high 10-year fracture risk at baseline (>20%) based on country-specific fracture risk assessment tool (FRAX) 29 or current treatment with active bone medications—bisphosphonates, denosumab, calcitonin, raloxifene, teriparatide, strontium, hormonal replacement therapy or hormonal contraceptives or current use of systemic steroids (inhaled steroids permitted), chemotherapeutic or aromatase inhibitors, or on dialysis or renal insufficiency (creatinine clearance <50 mL/min) were excluded.
Study design
The study is an open-label randomized multicenter strategic trial >96 weeks of older women on an ART regimen containing a regimen that includes TDF: Immediate switch of the TDF to TAF while maintaining the other ART agents. Delayed switch, with switch of the TDF to TAF at 48 weeks while maintaining the other Antiretroviral (ARV) agents.
Randomization
A computer-generated randomization list was created before study onset by a programmer at the CIHR Canadian HIV Trials Network (CTN). Site coordinators accessed the web randomization system through a personal code. Participants were randomized to switch at baseline (immediate) or 48 weeks (delayed) in 1:1 arm per group with permuted blocks of random size 4 and 6. Randomization was stratified by country.
Primary endpoint
The primary endpoint was percentage change from baseline in BMD (lumbar spine) at weeks 48 and 96.
Justification of primary endpoint
BMD assessment with DXA is well established for the diagnosis of osteoporosis and fracture risk assessment in postmenopausal women. A diagnosis of osteoporosis is made in postmenopausal women and women over age 50 years who have a BMD 2.5 or more standard deviations (SDs) below the normal BMD for young healthy white women (T-score ≤ −2.5), at the femoral neck, total hip, or lumbar spine. In the younger premenopausal population, the diagnosis of osteoporosis requires a BMD that is 2 or more SDs below the average BMD for age for women (Z-score ≤ −2) plus a fragility fracture.
Secondary endpoints
Percentage change from baseline in BMD at hip at weeks 48 and 96;
maintenance of viral suppression (HIV-1 RNA <50 copies/mL) at weeks 24, 48, and 96;
change from baseline in functional measures: grip strength to be measured by standardized methods and a calibrated dynamometer, and short physical performance battery;
changes in renal function: estimated glomerular filtration rate, creatinine, and urine albumin at weeks 24, 48, and 96; and
safety (clinical and laboratory adverse events) at weeks 24, 48, and 96.
Ethics approval
The study was approved by the University Health Network research ethics board, the community advisory committee of the CTN, and the research ethics boards at all participating centers. All participants gave written informed consent in English, French, or Italian. This study is registered with Clinical Trials.gov.
Study procedures
The study procedures were chart and medical history to document HIV test results, and demographic data including age, race, HIV risk factor, smoking history, exercise habits, prior fractures, calcium and vitamin D intake, illicit drug use, alcohol, steroid use, ARV history, past medical illness, medications, menstrual history, menopausal symptoms, weight, and body mass index (BMI). The country-specific FRAX calculator estimated the 10-year fracture risk. Routine investigations included viral load, CD4, hematology, biochemistry, lipids, and renal monitoring. Muscle grip strength was obtained using a dynamometer. 30 Short performance physical battery (SPPB) test assessed walking speed and balance. 31,32 DXA scan was performed at baseline.
Planned follow-up visits were at weeks 4, 12, 24, 36, 48, 52, 60, 72, 84, and 96 at which time points the participants had routine investigations as per the study site, and completed targeted physical examinations. Adverse events were captured at each study visit, as were changes in clinical conditions or medications. All participants completed DXA scans, quality-of-life questionnaires, and functional measures at weeks 48 and 96. DXA for each participant was performed using the same machine and at the same body position at each visit.
Sample size considerations
In total 128 women, 64 per group, were planned to be enrolled to detect a difference of 1% in the annual change in spine BMD at 48 weeks between those randomized to immediate switch to TAF and those randomized to delayed switch to TAF with a two-sample t-test, assuming a SD of 2%, 80% power, and a significance level of p < .05. The 1% difference was considered clinically important. The expected rate of loss in BMD in peri- and postmenopausal women is 1%–3% per year.
A standing Data Safety Monitoring Committee (DSMB) of the CTN was responsible for reviewing the study every 6 months for ongoing safety concerns, recruitment progress, and protocol violations.
Analysis
The original plan was to compare the impact of immediate versus delayed switch on percentage change in BMD spine using a generalized linear mixed model. Because our recruitment fell well short of our target, summary statistics for BMD (g/cm2) at the lumbar spine and total hip were tabulated for each study arm at baseline, week 48, and week 96 as well as change relative to baseline (absolute and percentage). And 95% confidence intervals (CIs) for the median change in BMD within each study arm was calculated.
Similarly, summary statistics were tabulated for each study arm at baseline for 10-year absolute fracture risk by FRAX fracture risk, SPPB, grip strength, and laboratory values. No formal comparison between groups was performed given the limited sample size. All analyses were conducted using SAS 9.4 (SAS Institute, Inc., Cary, NC).
Results
Thirty-four women were enrolled: 19 in the immediate and 15 in the delayed switch arm between September 2017 and April 2019; 30 completed the 96-week protocol. The study closed to follow-up in March 2021. Two participants in the delayed arm inadvertently switched to TAF at baseline and one participant in the delayed arm remained on TDF for the entire 96 weeks. Given the small number of randomized participants, an on treatment analysis was performed rather than intention to treat. The study closed prematurely for futility as recommended by the DSMB consequent to the COVID-19 pandemic and the inability of additional participants to attend study sites for imaging and other study procedures.
The baseline demographic characteristics including factors that could affect bone health of the 24 Canadian and 10 Italian women living with HIV are given in Table 1. These women enrolled in the peri- (47%) or early postmenopausal (53%) period. The median age was 51 years, 58.8% were black and 38.2% were white, 12% were current and 18% were former smokers, and 17.6% had a history of hepatitis C infection. The median BMI was 26 kg/m2. At baseline, 15% used calcium supplements and 59% vitamin D supplements. No participant had ever received therapy for osteoporosis and 8.8% had previously received hormonal replacement therapy.
Baseline Demographics and Factors That Could Impact Bone Health
IQR, intraquartile range.
Characteristics of the HIV disease and ART is described in Table 2. The median duration of HIV infection was 16.5 years, with 14 years of ART and 8.6 years of TDF use. The median CD4 cell count was 570 cells/mm3 (nadir 168) and viral load was suppressed in all cases. At enrollment, TDF was used in combination with a ritonavir-boosted protease inhibitor (n = 7), a non-nucleoside reverse transcriptase inhibitor (n = 13), an integrase inhibitor (n = 11), or more than one ART class (n = 3).
Baseline HIV Status and Antiretroviral History
ARV, antiretroviral; Int Inh, integrase inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; PI/R, protease inhibitor boosted by ritonavir; TDF, tenofovir difumarate.
The baseline values for functional status are given in Table 3. Twenty-one percent reported a prior fracture. Twenty-one percent reported having one or more falls in the previous 6 months. The baseline median score on the SPPB test was 11, with 23% in the intermediate- and 77% in the high-performance level. The median baseline grip strength was 27.3 kg (normal). At baseline, the median 10-year probability for a major osteoporotic fracture by FRAX was 4.4% (2.7, 5.0).
Physical Health at Baseline
The baseline BMD data and Z and T scores by DXA are outlined in Table 4 according to International Society of Clinical Densitometry guidelines. The baseline median (intraquartile range) BMD at the lumbar spine was 0.916 (0.804, 1.053) g/cm2 and at the total hip was 0.908 (0.841, 1.0) g/cm2. Based on the Z and T scores, and fracture history, 7 of 33 (21%) participants would meet criteria for osteoporosis and an additional 13 of 33 (39%) participants would meet criteria for low bone mass/osteopenia at baseline.
Baseline Bone Mineral Density by Dual Energy X-Ray Absorptiometry
Z-scores were used for all premenopausal women younger than age 50 years. T-scores were used for all postmenopausal women and those over age 50 years.
BMD, bone mineral density.
The changes in BMD over the course of the study are shown in Figure 1. The median (95% CI) percentage change in BMD from 0 to 48 weeks in the immediate switch group at the lumbar spine was 1.97% (−1.15 to 5.49) compared with a median (95% CI) decrease of 2.32% (−5.11 to 0.19) in the delayed arm. The median (95% CI) percentage change in BMD from 0 to 96 weeks was 2.33% (0–4.51) in the immediate arm compared with 0.70% (−3.19 to 2.47) in the delayed arm. Respective values at the femoral neck and total hip are presented.
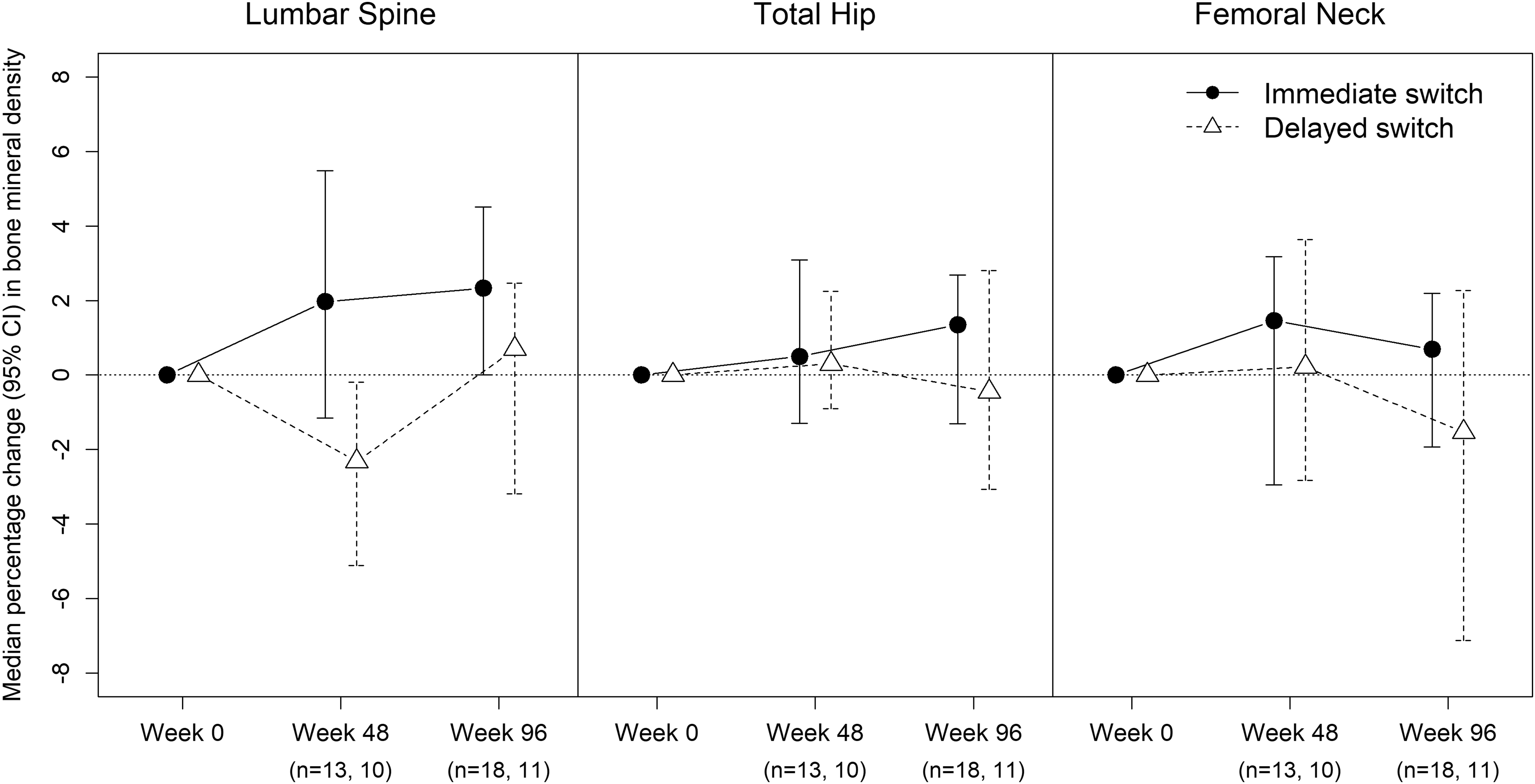
Median percentage change (95% confidence interval) in bone mineral density at baseline, week 48, and week 96 comparing those with immediate versus delayed switch from tenofovir disoproxil fumarate to tenofovir alafenamide fumarate.
Serum creatinine was normal in all participants at baseline. There were no significant changes in any of the parameters of renal function over the study (data not shown). No subject had loss of viral control and no serious adverse events were reported.
Discussion
We designed a randomized open-label controlled clinical trial to determine whether a switch from an ART regimen containing TDF to one containing TAF would improve bone health in peri- and early menopausal women living with HIV. Given the delays in regulatory approval of the study with our international sites, the timing of the licensure and commercial availability of TAF, and the restrictions imposed by the COVID-19 pandemic on the ability to recruit and follow participants, the study closed prematurely for futility and we were unable to meet our study recruitment and objective plans. We, however, describe our findings and the bone health in a cohort of aging HIV-positive women.
Osteoporosis is a major comorbidity of women in the general population 6,33 with an estimated lifetime risk of an osteoporotic fracture approximately one in three women over the age of 50 years. The likelihood increases with additional risk factors such as smoking, race, steroid use, lack of physical activity, and low BMI. 24,34 Women living with HIV may be at additional risk due to ART such as TDF especially when used in combination with a ritonavir- or cobisistat-boosted agent. The menopause transition period may be an especially vulnerable time as estrogen depletion may result in increased intestinal permeability, immune activation, and inflammation that may contribute to low bone density.
In our study, despite living with HIV for >16 years and having received 14 years of ART, including a mean of 8.6 years of TDF, the bone health of participants in the cohort was good with a median hip BMD of 0.9 g/cm2. The proportion meeting criteria for osteoporosis (21%) or low bone mass (39%) was similar to that observed in the general population of perimenopausal women. 34 Modifiable risk factors that could contribute to poorer bone health were present in a proportion of the women.
Our participants were relatively active with >75% exercising regularly, and 77% had high performance scores on SPPB testing, and grip strength (a measure of frailty) was normal. 30 Despite this, 20% reported falls in the previous 6 months, which could result in fracture especially in the setting of lower BMD. Seven percent reported previous fractures, all traumatic.
We detected a subset of peri- and postmenopausal women living with HIV with physical functional performance impairment in the intermediate range as assessed with SPPB. This typical geriatric tool is recommended for screening for frailty 35 and may be a marker of higher risk of falls and subsequently fractures.
There are conflicting data as to the risk of fracture in persons living with HIV. A recent systematic review and meta-analysis that included 13 studies reported a pooled incidence rate ratio of 1.58 (95% CI 1.25–2.0) for all fractures and 1.35 (95% CI 1.10–1.65) for fragility fractures among persons with HIV relative to controls. Smoking, white race, and older age were predictors of fragility fracture. 18 In contrast, an analysis from the Women's Interagency Study demonstrated that the unadjusted incidence of fracture did not differ between HIV-infected and high-risk HIV-uninfected women (1.8 vs. 1.4/100 person-years, respectively, p = .18).
Using multivariate models, white (vs. African American) race, prior hepatitis C virus infection, and higher serum creatinine, but not HIV serostatus, were statistically significant predictors of incident fractures. Among the cohort of women living with HIV in that study, older age, white race, current cigarette use, and history of AIDS-defining illness were associated with higher incidence of new fractures. 36
The role of TDF relative to TAF and the value of a switch to TAF on BMD is previously published. A meta-analysis of >8000 participants in clinical trials comparing TAF and TDF 20 included 83% male and 59% white participants with a mean age of 41 years. Across all follow-up time points, the risk of bone fractures was 1% lower for ritonavir- or cobisistat-boosted TAF compared with ritonavir- or cobisistat-boosted TDF (95% CI −2% to 0%, p = .04).
However, there was no difference in the risk of bone fractures when comparing unboosted TAF and unboosted TDF (risk difference = 0%, 95% CI 0% to +1%). In the subgroup of studies where TAF was compared with boosted TDF, percentage decreases in total hip BMD were 1.98% lower with TAF than with TDF (95% CI 1.63%–2.34%, p < .001), whereas with unboosted TDF regimens the mean difference was 1.48% (95% CI 1.14%–1.81%, p < .001).
In a switch study of 1443 HIV-infected participants, median age 41 years, 11% women, there was a statistically significant improvement in total hip and spine BMD, mean 1.47% and 1.56%, respectively, at 96 weeks. 21 In a multicenter European cohort (n = 167, 13% women) of age >60 years, the switch resulted in increases in BMD at the spine (2.24%) and total hip (1.33%) compared with decreases of 0.1% and 0.7%, respectively, (p < .0001) for those remaining on TDF >48 weeks.
In our cohort, we observed a trend toward improved BMD with a switch from TDF to TAF in perimenopausal women living with HIV. However, given the premature study closure, our small sample size and with the relatively normal BMD at baseline, we had inadequate power to show a statistical difference. We also note that the lumbar spine is the recommended monitoring site when treatment for osteopenia is initiated, and as it responds quickly to therapy it is not surprising that this is the area where a trend of improvements was seen.
Although our detected differences were small, the use of TAF was safe in peri- and early postmenopausal women living with HIV who are at a period of high risk for bone loss and osteoporosis. We acknowledge that our definition of peri- and early postmenopause was loosely defined limiting the generalizability of our findings and stresses the need for better documentation of menopausal status in routine clinical records.
Conclusions
With the aging of the HIV-infected population, we need to continue to optimize health and decrease the risk and consequences of comorbidities. Osteoporosis is a major comorbidity for aging women, and may be higher in women living with HIV. The additional impact of TDF on bone health in aging women compared with traditional risk factors is unclear, but collectively the literature shows improvements in BMD with switches to TAF. Our cohort had reasonably good bone health despite years of HIV and TDF therapy, but we did observe a trend toward increased BMD with a switch from TDF to TAF. The perimenopausal period is an important time to assess reversible parameters affecting bone health.
Footnotes
Authors' Contributions
Conceptualization of the study was done by S.W., G.G., and A.M.C.; methodology was carried out by J.S.; formal analysis was taken care by J.S. and T.L.; investigation was done by S.W., G.G., F.S., A.D.P., E.M., and S.T.; writing original draft was carried out by S.W.; writing review and editing were done by S.W., G.G., A.M.C., J.S., T.L., F.S., A.D.P., E.M., S.T., and R.C.; project administration was taken care by R.C.; and funding acquisition was done by S.W. and G.G. All named authors have read and approved the final article and agree to be accountable for the accuracy and integrity of the part of the study they have participated in.
Author Disclosure Statement
S.W. has served on advisory boards, spoken at CME events, and received research funds from ViiV Healthcare, GSK, Merck, Gilead, and Janssen pharmaceutical companies; A.M.C. has done consulting and received research funds from Amgen, Inc.; F.S. has received research funds from Gilead, ViiV Healthcare, and Merck; G.G. has served on advisory boards, spoken at CME events, and received research funds from ViiV Healthcare, Gilead, Merck, and Janssen; and A.D.P. has received research funds from Gilead, ViiV Healthcare, Merck, and Janssen, and has done consulting for Merck and ViiV Healthcare.
Funding Information
The study was funded by an investigator-initiated grant from Gilead Sciences, Protocol: BEING-W-IN-CA-311-3963.
