Abstract
The early initiation of antiretroviral therapy (ART) in HIV-infected patients shortly after the initiation of treatment for Pneumocystis pneumonia (PCP) has not been fully validated in a clinical setting. We retrospectively extracted all patients diagnosed with HIV-related PCP (HIV-PCP), including those with severe cases, who were treated with first-line ART in our hospital. The HIV-PCP patients were divided into two groups: an early ART group (patients who commenced ART within 21 days after the start of PCP treatment) and a deferred ART group (patients who started ART after 22 days). We compared the incidence of AIDS progression or death, the virological suppression rate, and changes in the CD4+ cell count at 24 weeks after the initiation of ART between the two groups. In addition, we analyzed the incidences of immune reconstitution inflammatory syndrome and grade 3 or 4 laboratory and clinical adverse events within 24 weeks as safety outcomes. Ninety-one HIV-PCP patients (36 in the early ART group and 55 in the deferred group) were included in this study. We found no significant difference in the incidence of AIDS progression or death between the two groups. Virological outcomes tended to be better in the early ART group but were not significantly different. Increases in the CD4+ cell counts at 24 weeks were comparable in both groups, suggesting that the safety was not significantly different. Analysis of the propensity-score matched cohort was performed to adjust for selection bias, and no significant difference was found in any outcome. Our results suggest that early ART introduction can be considered for untreated HIV-positive patients with PCP on the basis of efficacy and safety.
Introduction
Pneumocystis jirovecii is a pathogenic microorganism that causes Pneumocystis pneumonia (PCP) in a host with cellular immunodeficiency. It naturally infects only humans 1,2 and exists mainly outside type I alveolar epithelial cells. 3 Details of its transmission mechanism remain unclear, although it is thought that airborne spread between humans is the primary route of transmission. 4 Most people are exposed during infancy 5 and are usually cured by their own innate immunity mediated by macrophages. 6 However, the pathogen is not eliminated, and symptomatic infection occurs when cellular immunodeficiency is caused by HIV infection or the use of immunosuppressive medicines such as corticosteroids. PCP has a gradual onset and is characterized by difficulty breathing. 7 This respiratory disorder may be accompanied by fever, dry cough, and weight loss. The severity of PCP depends largely on host immune status and ability to combat the pathogen rather than on the pathogen itself. 8
In AIDS patients, the introduction of antiretroviral therapy (ART) in the late 1990s has dramatically reduced the incidence of PCP. However, even now, PCP is recognized as an opportunistic infection (OI) in AIDS patients and the most common indicator of AIDS. The latest PCP incidence rate among AIDS patients in Western Europe and the United States was ∼0.9–1.9 cases per 100 person-years, 9,10 although the real number is likely higher due to the presence of some patients who develop PCP without being aware of being infected with HIV. 11 In Japan, PCP accounts for approximately half of the cases of AIDS indicator diseases, 12 and ∼90% of HIV-related PCP (HIV-PCP) cases occur in patients with CD4+ T lymphocyte cell counts <200 cells/μL. 13
In a clinical setting, the timing of the initiation of ART after starting treatment for an OI attributable to HIV infection is still controversial. We often observe clinical deterioration shortly after the initiation of ART in patients with advanced immunodeficiency, 14 known as immune reconstitution inflammatory syndrome (IRIS). In HIV-PCP patients, it is thought that PCP-related IRIS (PCP-IRIS) can be avoided by delaying the initiation of ART until after treatment for PCP has been completed. 15 In addition, because HIV-infected patients who have advanced immunodeficiency and develop OIs have often been excluded from most of the past clinical studies of ART, there is no clinical consensus regarding when to initiate ART in HIV-positive PCP patients. Only two previous reports of randomized control trials discussed the appropriate timing of the initiation of ART in HIV-positive patients with PCP. 16,17
However, as patients with severe HIV-PCP cases were excluded from previous reports, it is unclear whether the early initiation of ART before the completion of treatment for PCP is applicable for all HIV-PCP patients, including those with severe cases. In addition, we need information on applicability and feasibility for patients undertaking the integrase-strand transfer inhibitor (INSTI)-based ART, as the protease inhibitor (PI)-based ART was mainly selected in the two previous studies. In this study, to examine the validity of early ART initiation in all HIV-PCP patients, we extracted the data for 91 patients diagnosed with HIV-PCP who started treatment with ART for the first time in our hospital and analyzed the efficacy and safety of the early initiation of ART in a real-world clinical HIV-PCP population, including patients with severe cases.
Materials and Methods
Subjects
A retrospective cohort study was conducted in HIV-PCP patients who were diagnosed with AIDS from April 2014 to December 2019 and received first-line ART at the National Hospital Organization Nagoya Medical Center. The study was approved by the Ethics Committee at the Nagoya Medical Center (Registration #2020-094) and conducted according to the principles expressed in the Declaration of Helsinki. Both inpatients and outpatients were enrolled.
Of the 99 patients, we divided 91 into 2 groups: the early ART group, which was composed of patients who commenced ART within 21 days after the start of treatment for PCP, and the deferred ART group, which was composed of patients who started ART after 22 days. We excluded eight patients due to no date record of PCP onset (n = 1), an unclear basis for the diagnosis of IRIS (n = 5), or a short follow-up period (n = 2). HIV infection was determined by a rapid screening test, followed by confirmation with Western blot and the molecular detection of the RNA viral load (VL). 18
PCP was comprehensively diagnosed based on clinical symptoms (fever, nonproductive cough, and dyspnea); laboratory values, including β-
Data collection
HIV-PCP patients were evaluated on the basis of baseline characteristics [age, sex, nationality, men who have sex with men (MSM) status, respiratory failure (defined as requiring oxygen therapy), pneumothorax, and comorbidities] in each group. CD4+ and CD8+ cell counts and the VL were examined at the following time points: AIDS diagnosis, the beginning of ART, and 24 weeks after the initiation of ART. The levels of interleukin-2 receptor (IL-2R) and ferritin were measured at one time point between the diagnosis of PCP and the start of ART. White blood cell (WBC) counts, the level of C-reactive protein (CRP), and the level of albumin were assessed within 2 weeks before ART initiation. We examined the intervals between the initiation of treatment for PCP and ART and compared ART regimens and steroid administration for PCP. The ART regimen was selected for each patient in accordance with the anti-HIV treatment guidelines in Japan.
Study end points
We investigated outcomes on the day of the patient's visit that was closest to ∼24 weeks after the initiation of ART; we were unable to ensure that follow-up occurred at precisely 24 weeks because this was a retrospective analysis.
The primary end point of this study was AIDS progression or death within 24 weeks after ART initiation. AIDS progression was defined as any new or relapsing HIV-related OI, which was listed in the Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV. 19 The secondary end points were the virological suppression rate (VL <50 copies/mL) and changes in CD4+ cell counts between the initiation of ART and the baseline at 24 weeks after ART initiation. Safety outcomes were defined as the incidence of IRIS and grade 3 or 4 laboratory and clinical adverse events within 24 weeks after the initiation of ART according to the Division of AIDS Table for Grading the Severity of Adult and Pediatric Adverse Events (corrected version 2.1). 20 Of note, IRIS was defined based on: (i) evidence of an increase in CD4+ cell counts and/or a decrease in plasma VL in response to ART and (ii) symptoms consistent with an infectious/inflammatory condition, temporally related to the initiation of ART, that could not be explained by the expected clinical course of previously/newly diagnosed OI or side effects of ART itself. 21
Statistical analysis
Differences in baseline characteristics, laboratory values, ART regimens, study end points, and safety outcomes were evaluated between the two groups with Fisher's exact test for categorical variables and Student's t-test or the Mann–Whitney U test for continuous variables. A propensity score was calculated for each patient, and propensity score matching was performed to account for the selection bias inherent in a retrospective study. The covariates used in constructing the propensity score were age, sex, ethnicity, respiratory failure, pneumothorax, CD4+ cell count, and VL before ART. One-to-one matching was completed using the nearest neighbor matching on the logit of the propensity score between the two groups. All analyses were performed using R (R statistical software, version 4.0.0). All p values were two sided, and p ≤ .05 was considered statistically significant.
Results
Evaluation of baseline characteristics before ART
During this study period, 133 patients were diagnosed with AIDS and received first-line ART. Ninety-one patients included in this study had HIV-PCP and had developed PCP before the initiation of ART, although no additional patients developed PCP due to unmasking IRIS. Of the 91 patients, 36 and 55 patients were allocated to the early ART and deferred ART groups, respectively (Fig. 1). The baseline characteristics of the two groups are shown in Table 1. Before matching, the mean age of the early ART group (46.8 years) was significantly older compared with the deferred ART group (41.0 years) (p = .02). Eighty-nine patients were male, 84 were Japanese, and 52 were MSM; the two groups were similar in terms of these characteristics. Thirty-five patients had respiratory failure, and four had pneumothorax combined with PCP. The deferred ART group had slightly larger proportions of patients with these two characteristics, due to the severity of PCP in that group, although there were no significant differences.
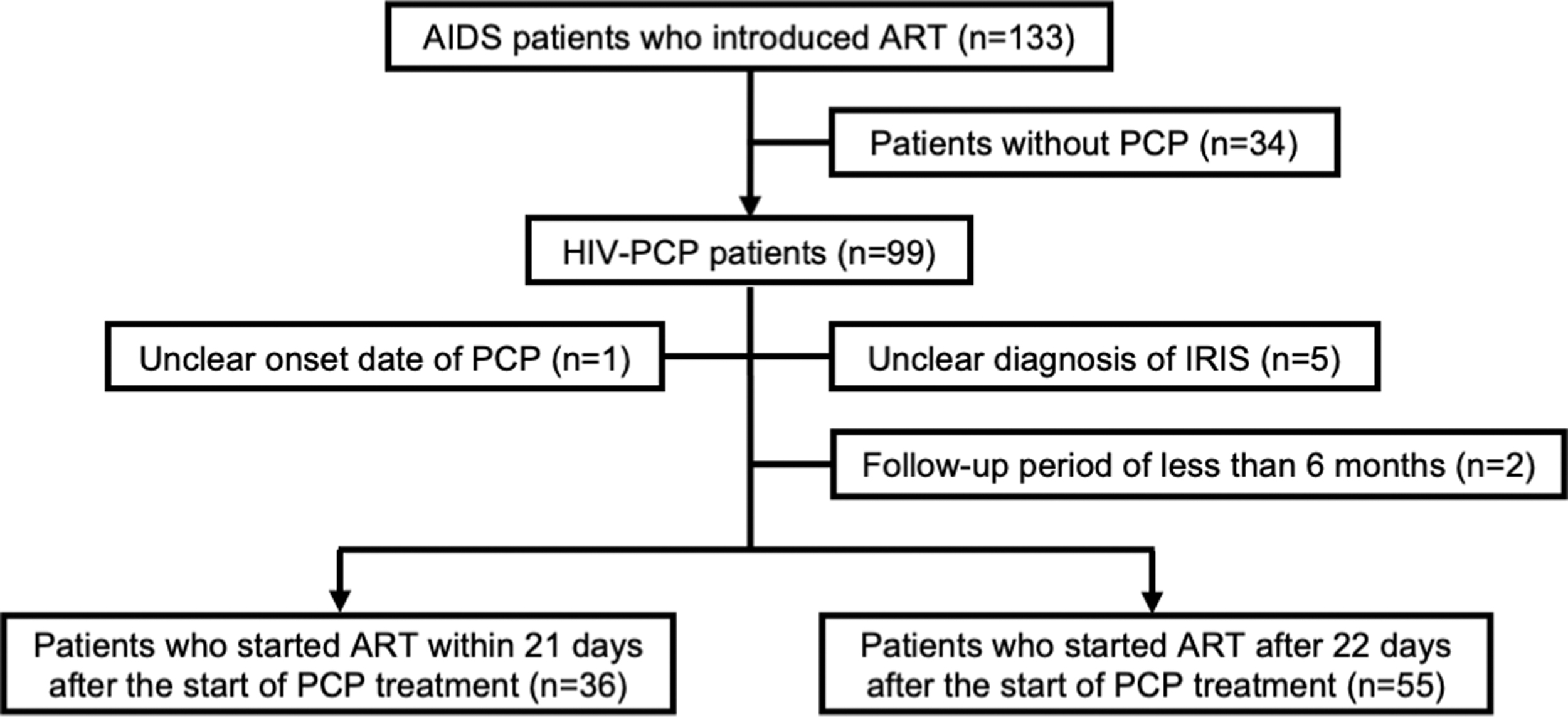
Inclusion and exclusion of HIV-PCP patients in this study. Ninety-one patients were divided into two groups: the early ART group (n = 36) and the deferred ART group (n = 55). ART, antiretroviral therapy; PCP, Pneumocystis pneumonia.
Baseline Characteristics
Student's t-test p value.
Fisher's exact test p value.
Positive for the HBs antigen.
Positive for the anti-HCV antibody.
p < .05.
ART, antiretroviral therapy; HBs, hepatitis B surface; MSM, men who have sex with men; PML, progressive multifocal leukoencephalopathy; SD, standard deviation.
With regard to comorbidities, the two groups had similar incidences: esophageal candidiasis (p = 1.00), extrapulmonary cryptococcosis (p = .52), and cytomegalovirus (CMV) infection except in the liver, spleen, or lymph nodes (p = 1.00), progressive multifocal leukoencephalopathy (PML) (p = .15), Kaposi's sarcoma (p = .56), non-Hodgkin lymphoma (p = .56), untreated syphilis (p = .74), and hepatitis B virus (HBV) infection (positive for hepatitis B surface antigen) (p = .24). These baseline characteristics were not significantly different between the two groups, except for age.
Next, we performed a comparative analysis of the laboratory values before ART (Table 2). The median CD4+ cell counts in the early ART group (26 cells/μL) were significantly higher than those in the deferred ART group (17 cells/μL) (p = .03), although the majority of patients in each group had advanced immunodeficiency. Similarly, the CD4/CD8 ratio and the WBC counts were also significantly higher in the early ART group (p < .01 and p = .02, respectively). The VL tended to be higher in the early ART group, but the difference was not significant (p = .05). Furthermore, the inflammatory markers of IL-2R, ferritin, and CRP were not significantly different between the two groups. The median intervals between the initiation of treatment for PCP and ART were 15 days in the early ART group and 31 days in the deferred ART group (p < .01). These data suggest that the progression of PCP driven by HIV-1 differed between the two groups, whereas the inflammation levels were normal and equivalent in the two groups.
Laboratory Values Before Art
Mann–Whitney U test p value.
From diagnosis of AIDS to the beginning of ART.
Within 2 weeks before the beginning of ART.
Interval (days) between the initiation of treatment for PCP and ART.
p < .05.
ALB, albumin; CRP, C-reactive protein; IL-2R, interleukin-2 receptor; IQR, interquartile range; PCP, Pneumocystis pneumonia; VL, viral load; WBC, white blood cell.
Comparison of ART management
The ART regimen and administration of steroids with ART were retrospectively analyzed (Table 3). All patients received ART containing an INSTI plus two nucleos(-t)ide reverse transcriptase inhibitors (NRTIs) (referred as backbone drugs), except for one patient (one boosted PI and two NRTIs). With regard to the NRTIs, a combination of emtricitabine (FTC)/tenofovir alafenamide (TAF) was administered more often in the early ART group (53%), while another combination of FTC/tenofovir disoproxil fumarate (TDF) was used more often in the deferred ART group (49%) (p = .03). This is most likely because the early ART group included more patients in whom ART was initiated after 2017, when TDF/FTC was switched to TAF/FTC as the main NRTI regimen.
Art Regimens and Steroids
Fisher's exact test p value.
Other: Raltegravir (n = 4 and 3 in early ART and deferred ART, respectively), darunavir/cobicistat (n = 1 in early ART), or elvitegravir/cobicistat (n = 1 in deferred ART).
p < .05.
3TC, lamivudine; ABC, abacavir; BIC, bictegravir; DTG, dolutegravir; FTC, emtricitabine; INSTI, integrase-strand transfer inhibitor; NRTI, nucleos(-t)ide reverse transcriptase inhibitor; PI, protease inhibitor; TAF, tenofovir alafenamide; TDF, tenofovir disoproxil fumarate.
The administration of steroids with ART was more common in the early ART group (p < .01). All patients in the early ART group started ART while being treated for PCP. Steroids were used for PCP treatment in the early ART group, whereas they were used in the case of persistent inflammation even after PCP treatment in the deferred group (Table 3).
Analysis of study outcomes
To assess the effect of early ART on HIV-PCP, we further extracted and compared the primary end point data for 34 patients in the early ART group and 54 patients in the deferred ART group (Table 4). The median evaluation time was 24.3 weeks [IQR: 23.0–25.9 weeks]. There were four patients who had experienced AIDS progression/death (12%) in the early ART group and five in the deferred ART group (9%) within the time of the evaluation. Statistical analysis showed no significant difference in the incidence of AIDS progression or death between the two groups (p = .73). One patient in the early ART group died, although the cause of death was not related to disease progression or treatment.
Study End Points and Safety Outcomes in all Evaluable Cases
Fisher's exact test p value.
Mann–Whitney U test p value.
CMV, cytomegalovirus; IRIS, immune reconstitution inflammatory syndrome.
As a secondary end point, an undetectable HIV-1 RNA VL (<50 copies/mL) at 24 weeks was achieved in 32 (94%) patients in the early ART group and 43 (80%) patients in the deferred ART group, suggesting that virological outcome appeared to be better in the early ART group, although there was no statistically significant difference (p = .07). As an additional secondary end point, the increases in CD4+ cell counts at 24 weeks were comparable between the two groups.
With regard to safety outcomes, 9 (27%) patients in the early ART group and 11 (20%) in the deferred ART group developed IRIS (p = .60). Notably, the incidence of PCP-IRIS was similar between the two groups (p = .73): four (12%) and five patients (9%) in the early and deferred ART groups, respectively. The median interval from ART initiation to development of PCP-IRIS was 11 days. Because all cases were mild, the patients recovered from PCP-IRIS after the administration of increased steroid doses. Four cases of HBV-related IRIS were observed in the deferred ART group, which is largely because the deferred ART group had more HIV/HBV coinfected patients at baseline. In addition, IRIS due to mycobacterium infection, CMV-related retinitis, varicella-zoster virus infection, herpes simplex virus infection, PML, and Kaposi's sarcoma were observed. Nineteen grade 3 or 4 laboratory adverse events and five grade 3 or 4 clinical adverse events occurred, and the distributions in the two groups were not significantly different (p = .60 and p = 1.00, respectively). These results suggest that the timing of ART introduction in patients with HIV-PCP does not affect the safety of the treatment.
Moreover, we performed propensity score matching analyses with one-to-one matching to investigate the end points and outcomes in cohorts with matched baseline characteristics. After the one-to-one matching, 24 patients were extracted in each group, and the 2 groups were well balanced in terms of the baseline characteristics (Supplementary Table S1). Regarding the NRTIs, a combination of FTC/TAF was administered to 9 patients in the early ART group and 6 patients in the deferred ART group, while another combination of FTC/TDF was used for 9 patients in the early ART group and 10 patients in the deferred ART group. In the analysis of the propensity-score matched cohort, we found no significant differences in AIDS progression/death, VL <50 copies/mL at 24 weeks, changes in CD4+ cell counts at 24 weeks, incidence of PCP-IRIS, or grade 3 or 4 adverse events (Table 5). These study end points and safety outcomes in the propensity-matched patients also suggest that early ART introduction is well tolerated and safe in untreated HIV-positive patients with PCP.
Study End Points and Safety Outcomes in Propensity-Score Matched Patients
Fisher's exact test p value.
Mann–Whitney U test p value.
Discussion
In this study, we retrospectively analyzed the efficacy and safety of early ART introduction in a real-world HIV-PCP population, including those with severe cases. Of the 91 patients, 36 patients (early ART group) started ART within 21 days after the initiation of PCP treatment, and 55 (deferred ART group) started ART after 22 days. We found no statistically significant difference in the incidence of AIDS progression/death within 24 weeks after the initiation of ART between the two groups. In addition, the data showed that the increase in CD4+ cell counts after 24 weeks and the frequency of grade 3 or 4 laboratory or clinical adverse events were similar between the two groups, which were also supported by our analyses of the propensity-score matched cohort. In contrast, the virological outcome tended to be better in the early ART group, although there was no significant difference. These results suggest that early ART introduction is well tolerated and safe, even for patients with severe HIV-PCP.
There are two previous reports of randomized control trials investigating the timing of ART in HIV-positive patients with PCP. One was the ACTG A5164 trial conducted by the AIDS Clinical Trial Group, 16 in which 282 randomized subjects were enrolled in a comparison of ART introduction within 14 days of starting acute OI treatment or after the completion of OI therapy. In that study, early ART initiation resulted in a lower incidence of AIDS progression/death with no increase in adverse events or loss of virological response compared with deferred ART. However, focusing on only the subgroup of HIV-PCP subjects, which accounted for ∼60% of the study population, the analysis lacked the appropriate power to determine statistical significance. In addition, since the proportion of patients with respiratory failure was unknown, the severity of PCP could not be determined. As an ART regimen, a boosted PI (mainly lopinavir/ritonavir) with two NRTIs was used in over 85% of subjects, while non-nucleoside reverse transcriptase inhibitor (NNTRI)-based regimens with two NRTIs were used in the others. We needed to obtain clinical evidence that the early initiation of ART with our current INSTI-based regimen in patients with HIV-PCP is safe.
The other previous study was the IDEAL study performed by Schafer et al., 17 which randomized 61 patients with PCP or toxoplasma encephalitis to groups with the initiation of ART within 7 days after the start of OI treatment or after the completion of OI therapy. Their study supported the idea that immediate ART, receiving atazanavir/ritonavir and TDF/FTC, was safe and had no negative effects on the incidence of disease progression or IRIS, immunological and virological outcomes, or quality of life. Notably, because only 24 patients with PCP were randomized to the immediate arm and 26 to the deferred arm, minor differences in the incidence rates of severe adverse event (SAE) might not have been detectable. In addition, there was no information on whether the patients with severe cases were excluded or not in that study.
Therefore, we retrospectively analyzed the efficacy and safety of the early initiation of INSTI-based ART in a representative, biologically relevant HIV-PCP population, including those with severe cases. Our conclusions differed from those of the ACTG A5164 trial regarding clinical progression. This discrepancy may be partly due to differences in patient populations and group definitions between the previous report and our study. In previous studies, there were no instances in which ART was introduced between day 15 and 21, and PCP patients with respiratory failure were not examined, so the patient population was different from that seen in actual clinical practice. In contrast, in the present study, the early group was defined as patients with the initiation of ART within 21 days, so that the subjects differed with respect to the timing of the initiation of ART. ART was introduced within 14 days in 16 of the 36 patients, and there was a difference in the patient background. Moreover, because ∼38% of the subjects had severe PCP with respiratory failure, it is representative of actual clinical practice.
Dutertre et al. reported that the risk of severe IRIS requiring hospitalization seems greater in severely immunocompromised HIV-infected patients receiving INSTI-based regimens as first-line ART than in those not receiving an INSTI. 22 In addition, Wijting et al. reported that INSTI-containing ART was independently associated with an increased incidence of IRIS but was not associated with mortality or the risk of hospital (re) admission. 23 In our study, all PCP-IRIS cases were mild, and the patients recovered after receiving increased doses of steroids. This observation also supports the idea that early ART introduction is well-tolerated and safe in untreated HIV-positive patients with PCP. Nevertheless, because this retrospective study was conducted in a single center, further investigations may be needed to support the generalization of our conclusions.
Conclusion
We retrospectively analyzed 91 HIV-PCP patients and compared the ART introduction timing between the early ART group and the deferred ART group. We found no significant difference in the incidence of AIDS progression/death between the two groups. Although the virological outcome tended to be better in the early ART group, the increases in CD4+ cell counts after 24 weeks were equivalent in the two groups. Analysis of the propensity score-matched cohort, which adjusted for selection bias, showed no significant difference in any outcome. These results suggest that early INSTI-based ART introduction can be considered for untreated HIV-positive patients with PCP.
Footnotes
Acknowledgments
The authors thank Dr. Junji Imamura and Ms. Yumiko Obayashi for technical assistance.
Authors' Contributions
Conceived and designed the study: T.K., Y.Y., and Y.I. Analyzed data: T.K., M.I., C.H., M.M., A.K., Y.Y., N.H., Y.I., and Y.H. Wrote the article: T.K., Y.I., and Y.H.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was supported, in part, by JSPS KAKENHI (JP19H03482) (to Yasumasa Iwatani) and by Health and Labour Sciences Research Grants (20HB2001) (to Yoshiyuki Yokomaku).
Supplementary Material
Supplementary Table S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
