Abstract
With obesity on the rise among people living with HIV (PLWH), there is growing concern that weight gain may result as an undesired effect of antiretroviral therapy (ART). This analysis sought to assess the association between ART regimens and changes in body mass index (BMI) among ART-experienced, virologically suppressed PLWH. ART-experienced, virologically suppressed PLWH ≥18 years of age in the Observational Pharmacoepidemiology Research and Analysis (OPERA) cohort were included for analysis if prescribed a new regimen containing one of the following core agents: dolutegravir (DTG), elvitegravir/cobicistat (EVG/c), raltegravir (RAL), rilpivirine (RPV), or boosted darunavir (bDRV), for the first time between August 1, 2013 and December 31, 2017. Multivariable linear regression was used to assess the association between regimen and mean changes in BMI at 6, 12, and 24 months after switch. In unadjusted analyses, BMI increases ranged from 0.30 kg/m2 (bDRV) to 0.83 kg/m2 (RPV) at 24 months following switch, but gains were observed with every regimen. In adjusted analyses, compared to DTG, only bDRV was associated with a smaller increase in BMI at all time points, while EVG/c and RAL were associated with smaller increases in BMI at 6 months only. Overall, results were consistent in analyses stratified by baseline BMI category. BMI increases were relatively small but followed an upward trend over time in this cohort of treatment-experienced, suppressed PLWH. Gains were attenuated with a longer period of follow-up. BMI gains did not differ by regimens, except for bDRV regimens, which were consistently associated with smaller BMI increases than DTG.
Introduction
There is growing evidence showing that weight gain, notably obesity, is on the rise among people living with HIV (PLWH). 1,2 In Canada and the United States, among 14,084 PLWH in NA-ACCORD, the median body mass index (BMI) at antiretroviral therapy (ART) initiation increased from 23.8 kg/m2 (9% obese, BMI ≥30 kg/m2) in 1998 to 24.8 kg/m2 (18% obese) in 2010. 2 Among 1,682 PLWH in the United States Military Natural History Study, the proportion of overweight/obese PLWH nearly doubled, from 28% in 1985–1990 to 53% in 1996–2004. 1 Weight gain among PLWH was initially attributed to an improvement in health, especially among those who were severely immunocompromised. 3 –6 However, with earlier ART initiation and more effective medications, there is now growing concern that obesity and weight gain may be an undesired effect of ART.
Several studies have reported weight gain when ART-naive PLWH start ART, with BMI increases ranging from 0.4 to 1.3 kg/m2 over 12 to 24 months, and body weight increases of 2.7 to 4.4 kg over 12 to 24 months. 7 –14 Among ART-naive PLWH, the use of integrase strand transfer inhibitors (INSTI) has been associated with greater weight gain than other existing classes. 7,15 However, among ART-experienced PLWH, BMI changes associated with different classes have been mixed. Some studies suggest greater weight gain when switching to an INSTI-based regimen, whether virologically suppressed at switch 16 –19 or not. 20,21
Other studies conducted among virologically suppressed ART-experienced PLWH switching to an INSTI found no difference in weight gain across different core agents [dolutegravir (DTG), elvitegravir/cobicistat (EVG/c), raltegravir (RAL)]. 22,23 In ART-naive PLWH only, greater weight gains have been reported with RAL compared to core agents other than DTG, 10,11,24,25 and with rilpivirine (RPV) compared to efavirenz (EFV). 15 Recent reports have shown that DTG- and bictegravir (BIC)-based regimens have been associated with greater weight gain than other core agents in ART-naive 11,15,26 and ART-experienced PLWH. 18,21 Weight gain has also been reported with tenofovir alafenamide (TAF) use in ART-naive 15 and ART-experienced PLWH switching from tenofovir disoproxil fumarate (TDF) to TAF. 27,28
Disentangling whether weight gain is attributed to an improvement in health status or an undesired effect of therapy is of paramount importance. Analyses of a large number of PLWH who are virologically suppressed and in whom return to health has been achieved and maintained may help in this endeavor. The extent to which weight gain is associated with specific ART agents among ART-experienced, virologically suppressed PLWH remains unclear. Thus, this observational study sought to assess changes in BMI following a regimen switch among ART-experienced, virologically suppressed PLWH in a U.S. population-based cohort; evaluating the first exposure to commonly used core agents: DTG, EVG/c, RAL, RPV, or boosted darunavir (bDRV).
Methods
Study population and design
The Observational Pharmacoepidemiology Research and Analysis (OPERA®) cohort is a database of prospectively collected data from electronic medical records (EMRs). At the time of this study, there were over 101,000 PLWH in the OPERA cohort receiving routine clinical care at out-patient clinics across 19 U.S. states (∼8% of the total population of PLWHV in the United States). Approximately 21% of PLWH in OPERA receive care through the Ryan White HIV/AIDS Program. All data reflect routine medical care, with visits and testing scheduled at the discretion of the treating providers. Information captured in the EMRs system at each site is retrieved, cleaned, aggregated, and anonymized to maintain patient confidentiality. OPERA complies with all HIPAA and HITECH requirements and receives annual Institutional Review Board approval by Advarra IRB, including a waiver of informed consent and authorization for use of protected health information.
This analysis included PLWH who were at least 18 years of age, ART-experienced, and virologically suppressed (VL <200 copies/mL) at or before switching regimen between August 1, 2013 and December 31, 2017. Eligible PLWH had to have switched to a three-drug ART regimen containing one and only one of the following core agents: DTG, EVG/c, RAL, RPV, or bDRV, without prior exposure to any of these. No restriction was made on the dosing schedule of core agents. In addition, included PLWH had to have a plausible weight measurement (BMI ≥10 and ≤50) at or within 3 months before switch. PLWH were observed from baseline until the first of the following censoring events: (1) discontinuation/addition of a core agent, (2) 12 months after the last clinical contact, (3) 3 months before the first pregnancy-related diagnosis, (4) death, or (5) study end (December 31, 2018).
Measurements
The primary outcome of this study consisted of changes in BMI since regimen switch, over the follow-up period. BMI was defined as weight (kg)/height (m) 2 and assessed at switch and during follow-up (6, 12, and 24 months ±3 months from each time point). Secondary outcomes included an assessment of the proportion of PLWH with >5% or >10% increase in weight (kg) and the proportion of PLWH transitioning from their baseline BMI category to another (underweight: BMI <18.5, normal: ≥18.5 to <25.0, overweight: ≥25.0 to <30, obese: ≥30) over each follow-up period.
Comorbidities and concurrent medications were obtained from EMRs. Endocrine disorders consisted of diagnoses of type 1 or type 2 diabetes mellitus, hyperlipidemia, hyperthyroidism, hypothyroidism, or thyroiditis. Substance abuse consisted of a diagnosis of alcohol dependence or drug abuse. Lipodystrophy was identified in the EMR by searching for diagnosis codes and text consistent with a diagnosis of lipodystrophy, lipohypertrophy, lipoaccumulation, hyperadioposity, or lipoatrophy within 12 months before or on index date.
Concomitant medications associated with weight gain were defined as prescriptions of antipsychotics/mood stabilizers (chlorpromazine, clozapine, haloperidol, lithium, olanzapine, valproic acid, risperidone, and quetiapine), antidepressants (phenelzine, tranylcypromine, citalopram, amitriptyline, nortriptyline, mirtazapine, and paroxetine), antihyperglycemics (insulin, chlorpropamide, gliclazide, glipizide, glyburide, pioglitazone, rosiglitazone, repaglinide, tolbutamide, and rosiglitazone), antihypertensives (atenolol and metoprolol), corticosteroids (cortisone, prednisolone, prednisone, methylprednisolone, and hydrocortisone), hormones (human growth hormone/somatropin and medroxyprogesterone), anticonvulsants (gabapentin, valproic acid/sodium valproate, carbamazepine), and antihistamines (cyproheptadine). 29,30
Medications associated with weight loss were defined as prescriptions of anti-infectives (metronidazole, amphotericin, atovaquone, pyrimethamine, and ethionamide), antineoplastics (aldesleukin and interleukin-2, capecitabine, carboplatin, cytarabine, dacarbazine, fluorouracil, hydroxyurea, imatinib, irinotecan, methotrexate, vinblastine sulfate, and vinorelbine tartrate), bronchodilators (albuterol and theophylline), cardiovascular drugs (amiodarone, acetazolamide, hydralazine HCl, and quinidine), stimulants (methylphenidate HCl and phentermine), antidepressants (fluoxetine and bupropion), anticonvulsants (topiramate), antihyperglycemics (exenatide, liraglutide, and semaglutide), anti-inflammatories (sulfasalazine), weight loss medications (bupropion-naltrexone, naltrexone, liraglutide, lorcaserin, orlistat, and phentermine-topiramate), dementia treatment (galantamine and rivastigmine). 29,30
Statistical analyses
Descriptive analyses were performed to compare baseline demographic and clinical characteristics, mean changes in BMI with 95% confidence intervals (CI), the proportion of PLWH with >5% or >10% increase in weight, and the proportion of PLWH changing a BMI category. Variables were summarized using medians with interquartile ranges (IQRs) or proportions by specific core agent group. PLWH without BMI measurements within a time window of interest were excluded from evaluation at that time point.
Multivariable linear regression was used to assess the association between core agent group and mean changes in BMI at 6, 12, and 24 months ±3 months following switch, as a difference in mean BMI change. Baseline variables included in the adjusted models were selected a priori, based on the literature, including baseline age, sex, race/ethnicity, BMI, lipodystrophy, endocrine disorders, hypertension, concomitant medications associated with weight gain and weight loss, viral load (i.e., variation from undetectable to <200 copies/mL), CD4 cell count, substance abuse (i.e., diagnosis of alcohol dependence or drug abuse), and TAF use. Continuous variables were modeled using restricted cubic splines. All analyses were performed overall and stratified by baseline BMI category.
Results from these analyses can be interpreted as the difference in mean BMI change between PLWH in each of the core agent groups compared to those on DTG. For example, a 12-month point estimate of 0.2 kg/m2 comparing X to DTG would be interpreted as “PLWH on X gained on average 0.2 kg/m2 more than PLWH on DTG after 12 months of use.”
Because PLWH without BMI measurements at specific time points were excluded from the corresponding analysis, a sensitivity analysis was conducted to assess the robustness of results using inverse probability-of-censoring weights (IPCW). IPCW weights were constructed using a logistic regression model and accounted for patients who discontinued treatment, were lost to follow-up, died, became pregnant, or did not have an available BMI measurement during the time windows of interest.
Results
This study included a total of 10,653 ART-experienced, suppressed PLWH who switched to DTG (n = 3,478), EVG/c (n = 3,721), RAL (n = 545), RPV (n = 1,959), or bDRV (n = 950). However, the analytical sample size decreased at each time point for all groups: DTG (6-month: n = 3,273; 12-month: n = 2,757; 24-month: n = 1,548), EVG/c (6-month: 3,484, 12-month: n = 2,774, 24-month: n = 1,506), RAL (6-month: n = 513, 12-month: n = 402, 24-month: n = 217), RPV (6-month: n = 1,830, 12-month: n = 1,513, 24-month: n = 795), and bDRV (6-month: n = 880, 12-month: n = 697, 24-month: n = 370).
Baseline demographic and clinical characteristics are shown in Table 1. Compared to DTG users, EVG/c users were younger, less likely to have comorbid conditions, but more likely to use TAF; RAL users were older, more likely to have hypertension and prescribed medications associated with weight gain, but less likely to use TAF; RPV users were younger, more likely to be female and non-Hispanic black, more likely to be on TAF, but less likely to have comorbid conditions; bDRV users were also more likely to be female and non-Hispanic black, and less likely to have comorbid conditions. At the time of switch, treatment groups were well balanced in terms of body weight (median 79.4 to 80.9 kg) and BMI (median 26.1 to 26.6 kg/m2).
Demographic and Clinical Characteristics at Regimen Initiation
p Value <.05 for the comparison with DTG.
ART, antiretroviral therapy; bDRV, boosted darunavir; BMI, body mass index; DTG, dolutegravir; EVG/c, elvitegravir/cobicistat; IQR, interquartile range; PI, protease inhibitor; RAL, raltegravir; RPV, rilpivirine; TAF, tenofovir alafenamide.
Large proportions of PLWH had gained ≥5% of their body weight at 6 months (DTG: 19.6%, EVG/c: 19.5%, RAL: 17.2%, bDRV: 16.9%, RPV: 20.1%), 12 months (DTG: 26.4%, EVG/c: 28.0%, RAL: 23.9%, bDRV: 21.8%, RPV: 29.6%), and 24 months (DTG: 32.2%, EVG/c: 34.0%, RAL: 28.6%, bDRV: 25.7%, RPV: 35.8%). Fewer had gained >10% of their body weight at 6 months (DTG: 5.3%, EVG/c: 5.1%, RAL: 3.1%, bDRV: 4.2%, RPV: 5.4%), 12 months (DTG: 8.7%, EVG/c: 10.1%, RAL: 7.0%, bDRV: 7.6%, RPV: 10.4%), and 24 months (DTG: 14.9%, EVG/c: 13.9%, RAL: 11.5%, bDRV: 11.6%, RPV: 15.2%) [Supplementary Table S1].
Unadjusted changes in BMI since switch are shown in Figure 1. There was an upward trend in weight gain over time across all groups. BMI increases were lowest with bDRV and RAL, and highest with RPV, EVG/c, and DTG at all time points (Fig. 1). Over time, the proportion of PLWH within the normal BMI range decreased, while the proportion of PLWH categorized as overweight or obese increased with all core agents (Fig. 2). Across all groups, among those who had a normal BMI at baseline, although the majority (77%–79%) remained in the normal category, many transitioned from normal to overweight (18%–21%), while ≤1% transitioned from normal to obese at 12 months. The proportion transitioning from the normal BMI range to underweight ranged from 1% to 3%. However, among those who were overweight at baseline, 9%–15% experienced weight loss resulting in a normal BMI at 12 months, and 13%–15% gained more weight and transitioned into the obese category at 12 months.
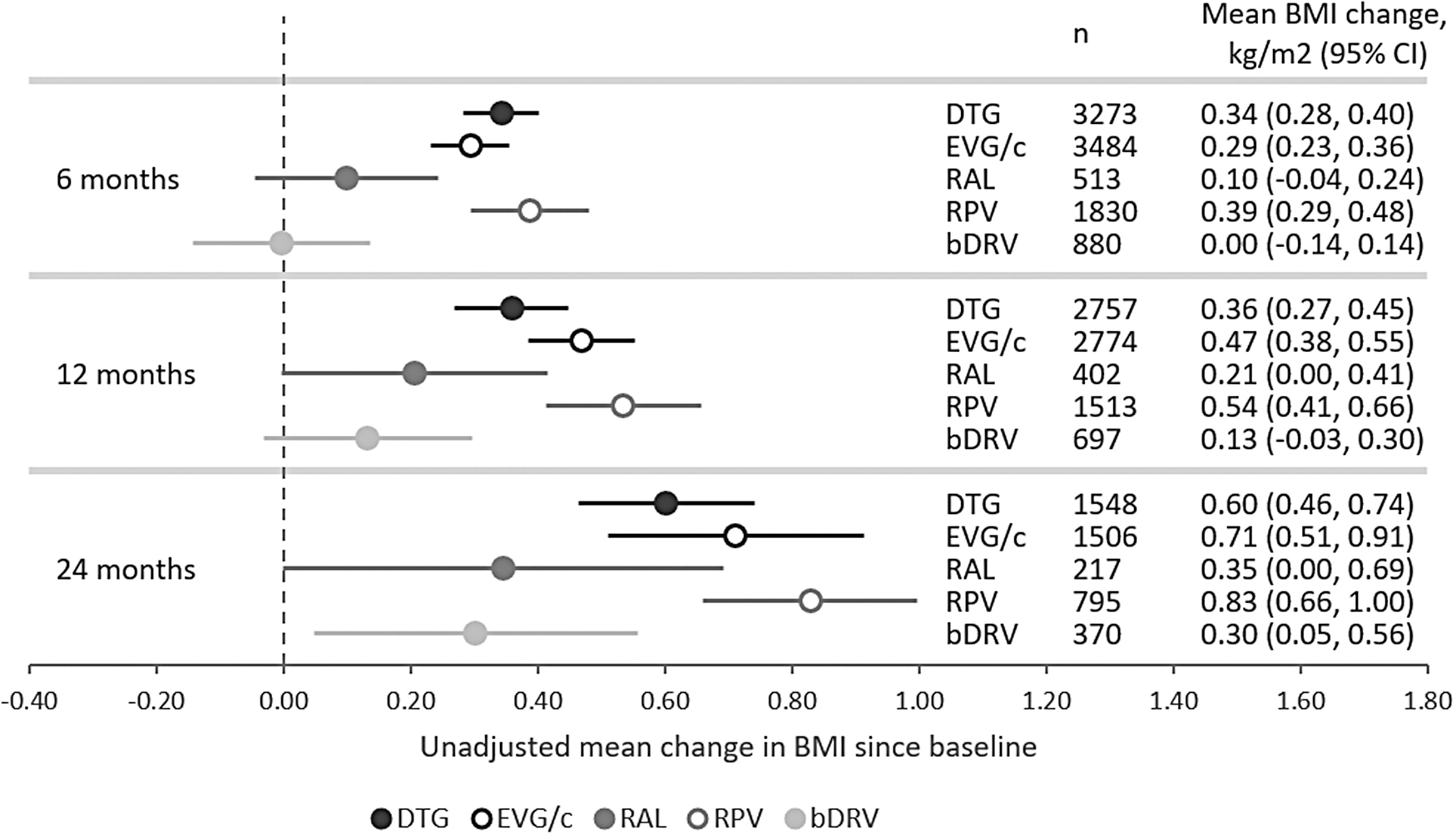
Unadjusted mean BMI change from baseline at 6, 12, and 24 months following initiation. BMI, body mass index.
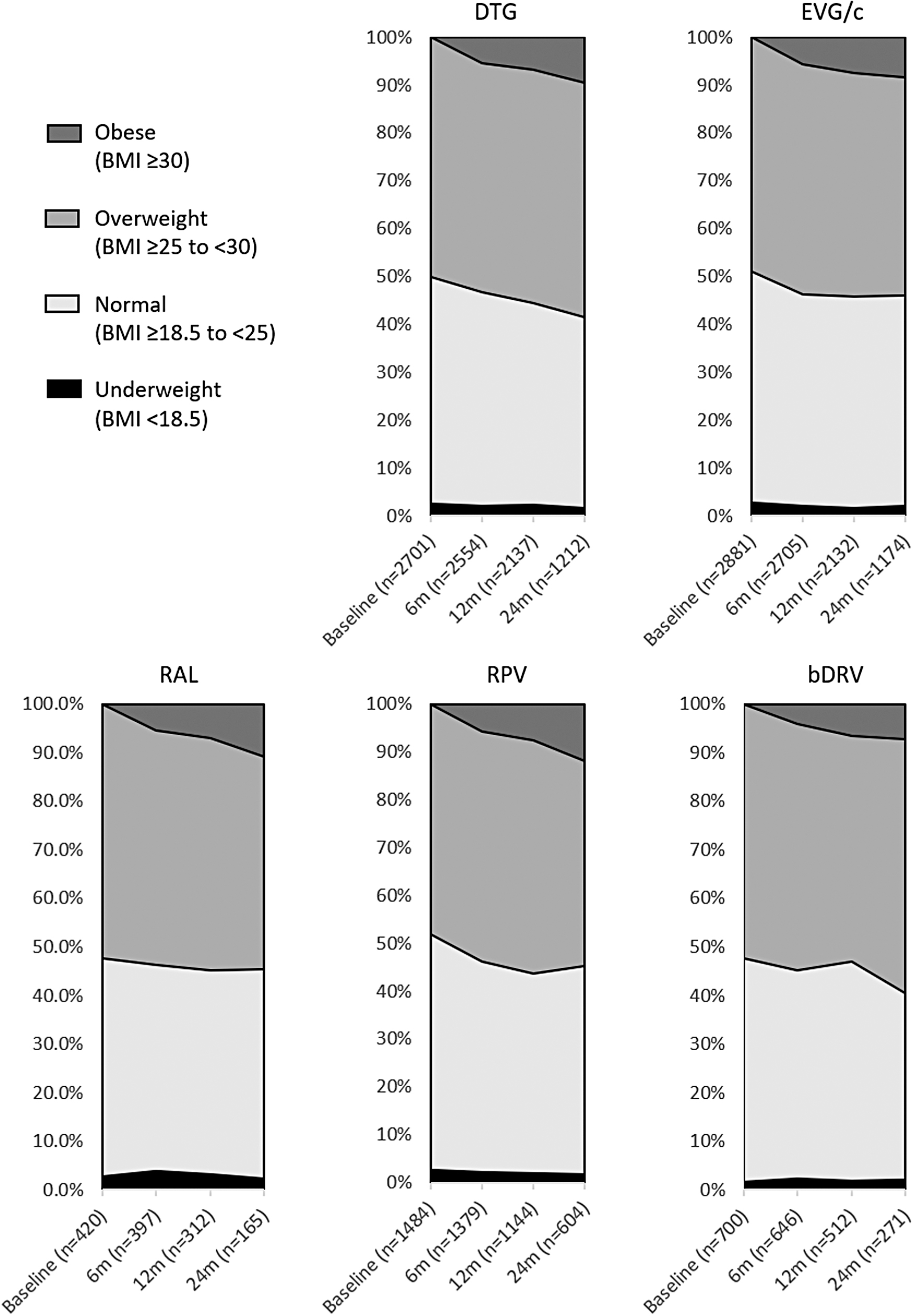
Distribution of BMI categories at baseline and 6, 12, or 24 months after regimen initiation, among those with a baseline BMI <30 kg/m2.
Adjusted changes in BMI relative to the DTG group are presented in Figure 3; these results are to be interpreted as the difference between each group and the DTG group in mean BMI increases over time. After adjustment for confounding factors (i.e., age, sex, race/ethnicity, BMI, lipodystrophy, endocrine disorders, hypertension, medications associated with weight gain and weight loss, viral load, CD4 cell count, substance abuse, and TAF use), at 6 months following switch, most groups had lower increases in BMI than DTG users: EVG/c (−0.15 kg/m2; 95% CI: −0.25 to −0.06), RAL (−0.22 kg/m2; 95% CI: −0.39 to −0.06), and bDRV (−0.30 kg/m2; 95% CI: −0.44 to −0.17). However, at later time points, only bDRV was associated with smaller increases in BMI compared to DTG: 12-month (−0.31 kg/m2; 95% CI: −0.49 to −0.12) and 24-month (−0.29 kg/m2; 95% CI: −0.57 to −0.01]). Results were consistent in sensitivity analyses using IPCW (Supplementary Table S2).
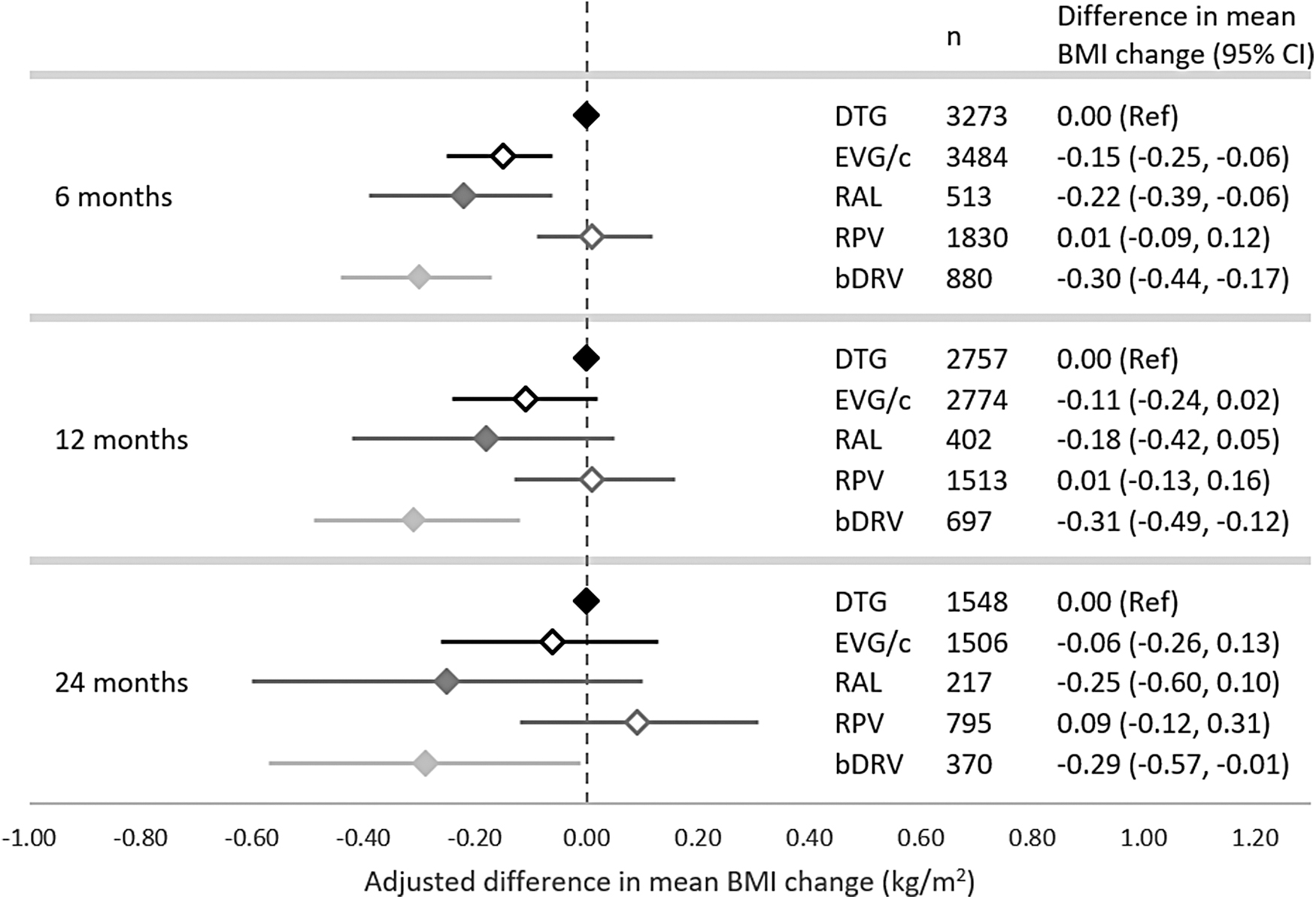
Adjusted* differences in mean change in BMI with each core agent compared to DTG, at 6, 12, and 24 months following regimen initiation.
In the 12-month analysis stratified by baseline BMI category (Fig. 4), compared to DTG, PLWH with a normal baseline BMI experienced a smaller BMI gain with EVG/c (−0.27 kg/m2; 95% CI: −0.43 to −0.11) and bDRV (−0.26 kg/m2; 95% CI: −0.50 to −0.03), but no statistically significant difference was detected with RAL and RPV. Among those who were overweight at baseline, only bDRV users experienced statistically smaller BMI gains at 12 months compared to DTG users (−0.32 kg/m2; 95% CI: −0.57 to −0.06). In the underweight and obese strata, smaller sample sizes led to wider CI and no statistically significant difference was observed between DTG and other core agents. BMI-stratified results at 6 and 24 months are available in Supplementary Table S3.
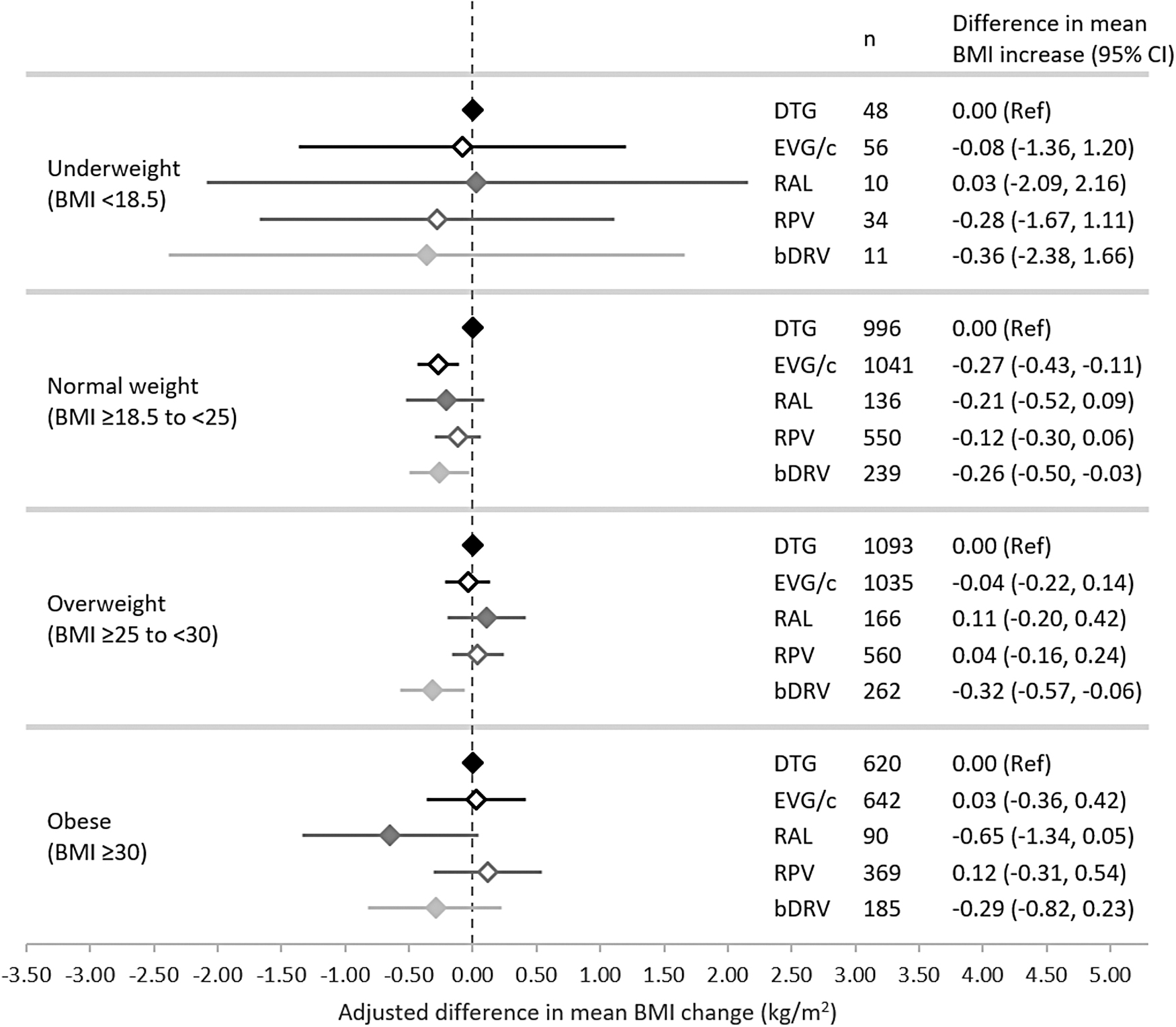
Adjusted* differences in mean change in BMI with each core agent compared to DTG at 12 months following regimen initiation, by baseline BMI category.
Discussion
In this study of ART-experienced, suppressed PLWH, a large proportion of individuals were considered overweight (38%) and obese (23%) at baseline, which is comparable to the reported 2015–2016 prevalence in the U.S. population for overweight (32%), but much lower for obesity (40%). 31 In OPERA, small absolute increases in BMI from baseline were observed over time with all core agents, reaching statistical significance for DTG, EVG/c, and RPV only. After adjustment for confounding factors, only bDRV users had consistently and statistically significantly lower increases in BMI compared to DTG users. In contrast, although increases in BMI at 6 months were lower for EVG/c and RAL users compared to DTG users, no statistically significant difference could be detected at 12 and 24 months. There was no difference between RPV and DTG in mean BMI increases at any time point. Overall, results were consistent in stratified analyses among PLWH with normal BMI or overweight at baseline, and in sensitivity analyses using IPCW.
There is considerable variability in estimates of BMI or weight gain following a switch of core agent among ART-experienced, suppressed PLWH in the literature. In OPERA, the unadjusted average BMI increase from baseline with DTG was 0.34 kg/m2 at 6 months, 0.36 kg/m2 at 12 months, and 0.60 kg/m2 at 24 months. For context, a person measuring 1.80 m would experience weight gains of 1.1 kg (6 months), 1.2 kg (12 months), and 1.9 kg (24 months).
These results are consistent with results from the SCOLTA study of 1,118 ART-experienced patients, among whom those switching to DTG experienced adjusted BMI increases of 0.28 kg/m2 (p = .006) at 6 months and 0.37 kg/m2 (p = .004) at 12 months after switch. 32 Among 170 PLWH who switched from EFV/TDF/emtricitabine (FTC) to an INSTI or protease inhibitor (PI), those switching to DTG/abacavir (ABC)/lamivudine (3TC) experienced an average weight gain of 5.3 kg at 18 months after switch. 21 Statistically significant weight gains have also been demonstrated in the Swiss HIV Cohort study of virally suppressed, ART-experienced PLWH, which demonstrated accelerated rates of weight gain after a switch to DTG: 0.72 kg/year (95% CI: 0.58 to 0.86) over 18 months of follow-up after switch, compared to 0.47 kg/year (95% CI: 0.34 to 0.61) before switch (p = .029). 33
In this study, PLWH switching to EVG/c experienced BMI increases from baseline of 0.29 kg/m2 at 6 months, 0.47 kg/m2 at 12 months, and 0.71 kg/m2 at 24 months. Similarly, in the SCOLTA cohort, adjusted BMI increases of 0.42 kg/m2 at both 6 and 12 months (p = .003 and p = .004, respectively) were reported with EVG. 32 However, among 368 virologically suppressed PLWH who switched to an INSTI-based regimen in the HIV Outpatient Study, EVG-based regimens were not associated with an increase in BMI. 18
Statistically significant BMI increases from baseline were also observed for RPV users in OPERA: 0.39 kg/m2 at 6 months, 0.54 kg/m2 at 12 months, and 0.83 kg/m2 at 24 months. These estimates align with results from the SCOLTA cohort, which reported that PLWH switching to RPV experienced adjusted BMI increases of 0.30 kg/m2 at both 6 and 12 months (p = .005 and p = .029, respectively). 32 A subanalysis of the DOLBi project also reported a median BMI increase of 0.63 kg/m2 (IQR: −2.9, 3.3; p = .02) and a median weight increase of 1.80 kg (IQR: −1.2, 4.1; p = .03) among 37 experienced, suppressed PLWH following a switch to DTG/RPV dual therapy. 34
In OPERA, no statistically significant increase in BMI from baseline was reported with RAL, while statistically significant BMI gain was only reported at 24 months with bDRV. This is in contrast with the SCOLTA study, where PLWH switching to RAL experienced adjusted BMI increases of 0.26 kg/m2 (p = .001) at 6 months and 0.36 kg/m2 (p = .0004) at 12 months. 32 However, the DOLBi project also reported no statistically significant increases in median BMI (0.29 kg/m2, IQR: 1.6, 3.6; p = .22) or body weight (0.70 kg, IQR: −0.8, 4.0; p = .28) among 17 experienced, suppressed PLWH following a switch to DRV/3TC dual therapy. 34
A previous study found that among 495 virologically suppressed patients on EFV/TDF/FTC from the Vanderbilt Comprehensive Care Clinic, weight change over 18 months was highest among patients switching to an INSTI-based regimen (2.9 kg) compared to switching to a PI-based regimen (0.7 kg) or continuation with EFV/TDF/FTC (0.9 kg). 21 The highest weight increase, 5.3 kg, was observed for patients switching to DTG/ABC/3TC. While this was not statistically different from the 2.8 kg weight gain with a switch to a EVG- or RAL-based regimen, it was statistically significantly greater than with EFV/TDF/FTC continuation. 21 Statistically significantly greater weight gains were also observed over time after switching to DTG compared to RAL and EVG in PLWH virally suppressed for ≥1 year. 18
In addition, ART-experienced PLWH randomized to switch to DTG/ABC/3TC experienced statistically significantly greater weight gains compared to PLWH maintaining a regimen of EFV/TDF/FTC (p = .001). 21 A post hoc analysis of the ADVANCE trial has demonstrated that slower EFV CYP2B6 metabolizer genotypes were associated with lesser weight gain, suggesting that weight differences between the EFV and DTG arms of the trial were driven by impaired weight gain in those with loss-of-function CYB2B6 single-nucleotide polymorphism (SNP). 35
Strengths of this study include the use of the OPERA cohort, which is a large, representative sample of PLWH in the United States, representing ∼8% of all PLWH receiving care in the United States. The sample size for this study was large in each of the core agent groups (ranging from 545 to 3,721 PLWH), for a total of 10,653 PLWH included. The use of electronic health records allowed for a real-world assessment of prescription practices, and the ability to identify and account for a history of disorders. Further, the use of BMI rather than weight as an outcome accounted for variable heights across individuals. The study population was restricted to ART-experienced PLWH with viral load <200 copies/mL at switch, therefore, challenging the hypothesis that weight gain associated to ART is not just the result of a return to health.
Covariates included in the adjustment set were selected with care. Unlike most other studies, the adjustment set included an extensive list of concomitant medications known to be associated with weight gain or loss. This is also one of the first studies to control for TAF use, given recent reports that TAF initiation may be associated with weight gain as evidenced by an average increase of 4.25 kg in body weight over 96 weeks (vs. 2.07 kg for TDF), 15 a 1-year body weight increase of 3.17% (vs. 0.55% for TDF), 27 and a 0.45 kg/m2 adjusted increase in BMI within 6–12 months of switching from TDF to TAF. 28 This is of particular importance given that coadministration of DTG and TAF may lead to even greater weight gains. 26,33
The results of this study were robust to stratification by baseline BMI category and in sensitivity analyses conducted using IPCW. Finally, to the best of our knowledge, no other direct comparison of BMI or weight gain between INSTIs and RPV or bDRV has been published to date.
This study also has limitations. First, PLWH were excluded if they did not have a BMI measurement available during the three specific time windows, which resulted in different populations for each model. This also led to substantial attrition, with only 42% of the baseline population included in the 24-month analysis. In addition, a strong assumption of linearity was made when assessing the association between core agent and changes in BMI across the three subsequent time points in different models.
Despite statistical adjustments, residual confounding cannot be ruled out, given the inclusion of baseline covariates only, and the imbalances observed at baseline (e.g., lower likelihood of comorbidities with EVG/c, RPV, or bDRV than DTG). There is also a potential for unmeasured confounding as these analyses did not adjust for marijuana use, dronabinol use or smoking cessation, calorie consumption, energy expenditure, appetite, or weight distribution (fat vs. lean mass) and did not include time-updated covariates.
Weight and BMI do not accurately estimate visceral and truncal adiposity. Waist circumference and waist-to-hip ratio are considered better surrogates of body fat distribution than BMI, but neither of these measures was available in this cohort nor are they commonly measured in real-world clinical practice, thus limiting their use in EMR-based research. Many studies have assessed changes in body weight rather than BMI as an outcome, which makes any comparisons with these studies more difficult.
Analyses were not adjusted for geographic location, which may have influenced various demographic and sociologic factors associated with weight gain. Moreover, these analyses were restricted to PLWH switching regimen, and thus did not include a control group of PLWH who maintained their regimen. However, selection bias would likely have been introduced by the choice of an arbitrary baseline date and of included regimens for the control group. Finally, weight gain associated with BIC use could not be assessed due to an insufficient number of BIC users at the time this study was conducted, although inclusion of BIC use would have prevented statistical adjustments for TAF use as BIC is always coformulated with TAF.
Conclusion
In summary, in this cohort of ART-experienced, virologically suppressed PLWH switching for the first time to an INSTI, RPV, or bDRV, increases in BMI followed an upward trend over a 24-month window of observation, although these changes in weight gain were relatively small. Compared to DTG, increases in BMI were lower for EVG/c, RAL, and bDRV users at 6 months, but became attenuated with a longer period of follow-up, except for bDRV users who consistently maintained a statistically significantly lower increase in BMI at 12 and 24 months following switch. Large-scale longitudinal studies in ART-experienced, virologically suppressed PLWH are needed to better comprehend the clinical significance of these relatively small gains in BMI over a longer duration and their relationship to cardiometabolic complications. Such studies are also needed to understand the causes and consequences of extreme weight gain associated with ART.
Footnotes
Authors' Contributions
L.B., J.F., V.V., and C.H. share the responsibility for the design of this study. K.M., R.H., J.F., and G.F. contributed to the acquisition of data. L.B. contributed to the analyses. All authors have contributed to the interpretation of results. L.B. drafted the article. All authors have critically reviewed and approved the article.
Acknowledgments
This research would not be possible without the participation of HIV caregivers and their patients. In addition, we are grateful for the following contributions: Terra Fatukasi (study and manuscript support), Robin Beckerman (SAS programming), Jeff Briney (QA/QC), Rodney Mood (site selection and support), Ted Ising (database architecture and support), Bernie Stooks (database support), and Judy Johnson (categorization of clinical terminology).
Author Disclosure Statement
K.M. has received research grants from Gilead Sciences, Merck, Janssen, and GSK/ViiV Health care and honoraria for Speakers Bureau and Advisory Boards from Gilead Sciences, Merck, Janssen and GSK/ViiV Health care; and advisory board participation with Epividian. L.B., J.F., and G.F. are employed by Epividian, Inc.; Epividian has had research funded by ViiV Health care, Merck & Co., Janssen Pharmaceutica, and Gilead Sciences. R.H. has received a research grant from Gilead, speaker honoraria and advisory boards from ViiV Health care, Merck, Gilead Sciences and Janssen, and advisory board participation of Epividian. V.V., C.H., and J.v.W. are employed by ViiV Health care and holds stocks and shares in GSK as part of their employment. M.C. is employed by GlaxoSmithKline and holds stocks and shares in GSK as part of their employment. J.L. has provided consulting services to Gilead Sciences and ViiV Health care.
Funding Information
This work was funded by ViiV Health care.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
