Abstract
HIV/hepatitis C virus (HCV) patients have a 3-fold increased fracture incidence compared to uninfected patients. The impact of HCV therapy on bone health is unclear. We evaluated bone turnover markers (BTM) in well-controlled (HIV RNA <50 copies/ml) HIV/HCV-coinfected patients who received pegylated interferon-α and ribavirin (PEG-IFN/RBV) in ACTG trial A5178. Early virologic responders (EVR: ≥2 log HCV RNA drop at week 12) continued PEG-IFN/RBV and non-EVRs were randomized to continuation of PEG-IFN alone or observation. We assessed changes in C-terminal telopeptide of type 1 collagen (CTX; bone resorption marker) and procollagen type I intact N-terminal propeptide (P1NP; bone formation marker), and whether BTM changes were associated with EVR, complete early virologic response (cEVR: HCV RNA <600 IU/ml at week 12), or PEG-IFN treatment. A total of 192 subjects were included. After 12 weeks of PEG-IFN/RBV, CTX and P1NP decreased: −120 pg/ml and −8.48 μg/liter, respectively (both p < 0.0001). CTX declines were greater in cEVR (N = 91; vs. non-cEVR (N = 101; p = 0.003). From week 12 to 24, CTX declines were sustained among EVR patients who continued PEG-IFN/RBV (p = 0.027 vs. non-EVR) and among non-EVR patients who continued PEG-IFN alone (p = 0.022 vs. Observation). Median decreases of P1NP in EVR vs. non-EVR were similar at weeks 12 and 24. PEG-IFN-based therapy for chronic HCV markedly reduces bone turnover. It is unclear whether this is a direct IFN effect or a result of HCV viral clearance, or whether they will result in improved bone mineral density. Further studies with IFN-free regimens should explore these questions.
Introduction
H
Bone homeostasis is a dynamic process of ongoing resorption and formation. Bone turnover markers (BTM) have been used extensively to provide a physiologic window into the effects of multiple disease states and interventions on bone health. 10,11 In this study, we measured specific BTMs: collagen breakdown products, such as C-terminal telopeptide of type 1 collagen (CTX) to indicate osteoclast activity (bone resorption), and procollagen type I intact N-terminal propeptide (P1NP) to measure osteoblast activity (bone formation).
Most, 12 but not all 5,13 previous cross-sectional studies have demonstrated abnormalities in BTM in HCV-infected patients without cirrhosis. Limited data from HCV-monoinfected patients suggest that HCV therapy improves BMD and markers of inflammation. Successful HCV clearance in response to interferon (IFN) therapy was associated with a two-thirds reduction in the risk of bone fracture in postmenopausal women with osteoporosis and chronic HCV-induced liver disease. 14 In another cohort of HCV-monoinfected patients, antiviral therapy with pegylated interferon-α (PEG-IFN) led to significant on-treatment increases in lumbar spine and hip BMD and a reduction in serologic markers of bone resorption irrespective of subsequent treatment response. 15 Nishida et al. 16 also showed that IFN-α and ribavirin therapy during HCV monoinfection was associated with a decrease in bone resorption and increased BMD.
The effects of HCV treatment on bone turnover and BMD in HIV/HCV-coinfected patients are unknown. To address this, we investigated the impact of anti-HCV therapy with pegylated IFN-α and ribavirin (PEG-IFN/RBV) on bone turnover among HIV/HCV-coinfected patients, and explored the relative impact of HCV virologic control and the use of PEG-IFN. Our primary hypothesis was that treatment of HIV/HCV-coinfected patients with PEG-IFN/RBV will result in significant decreases in BTMs.
Materials and Methods
We carried out a retrospective study utilizing available plasma samples from the AIDS Clinical Trial Group (ACTG) study A5178 in which HIV/HCV-coinfected patients received HCV treatment with PEG-IFN/RBV. 17 Patients who achieved an early virologic response (EVR; ≥2 log HCV RNA drop at week 12) continued PEG-IFN/RBV while those without EVR were randomized to continuation of PEG-IFN alone or observation off therapy. Among those with fully suppressed HIV viremia (HIV RNA <50 copies/ml) at entry into A5178, CTX and P1NP were measured at baseline, at week 12 (during PEG-IFN/RBV therapy), and at week 24 (while on PEG-IFN/RBV, PEG-IFN alone, or observation).
We assessed changes in CTX and P1NP from week 0 to week 12, and evaluated the associations of these changes with EVR or complete EVR (cEVR: HCV RNA <600 IU/ml at week 12) status. We also assessed whether week 12 to week 24 changes in BTMs differed between EVR and non-EVR patients and by treatment allocation among non-EVRs (continuation of PEG-IFN alone or observation off therapy). CTX was measured using a luminometric assay on Elecsys 2010 (Hoffmann-La Roche); the reference range for men was 158–584 pg/ml, for premenopausal women was 162–573 pg/ml, and for postmenopausal women was 330–1,008 pg/ml. P1NP was measured with radioimmunoassay (UniQ P1NP RIA kit; Orion Diagnostica) and the reference range was 25.91–132.5 μg/liter.
BTM changes within groups were assessed with Wilcoxon signed rank tests and between groups with rank-sum and Van Elteren's tests stratified by potential confounders: race (white or nonwhite) and cirrhosis (defined as Metavir score = 4 or Ishak score ≥5 on liver biopsy). Other covariates considered in the analysis were age (<50 years or ≥50 years), gender, body mass index, prior tenofovir disoproxil fumarate (TDF) exposure (none, ≤48 weeks, or >48 weeks), HCV viral load, and CD4 cell count.
Results
Figure 1 shows the ACTG Study 5178 schema and the samples included in this analysis. A total of 192 HIV/HCV patients with well-controlled HIV infection contributed samples. They were predominantly male (84%) and white (57%). The median age (Q1, Q3) was 48 years (42, 52). At baseline, the median HCV RNA (log10 IU/ml) was 6.60 (6.27, 6.92),13.5% had cirrhosis, and 32% and 20% reported prior TDF use for >48 weeks and ≤48 weeks, respectively.
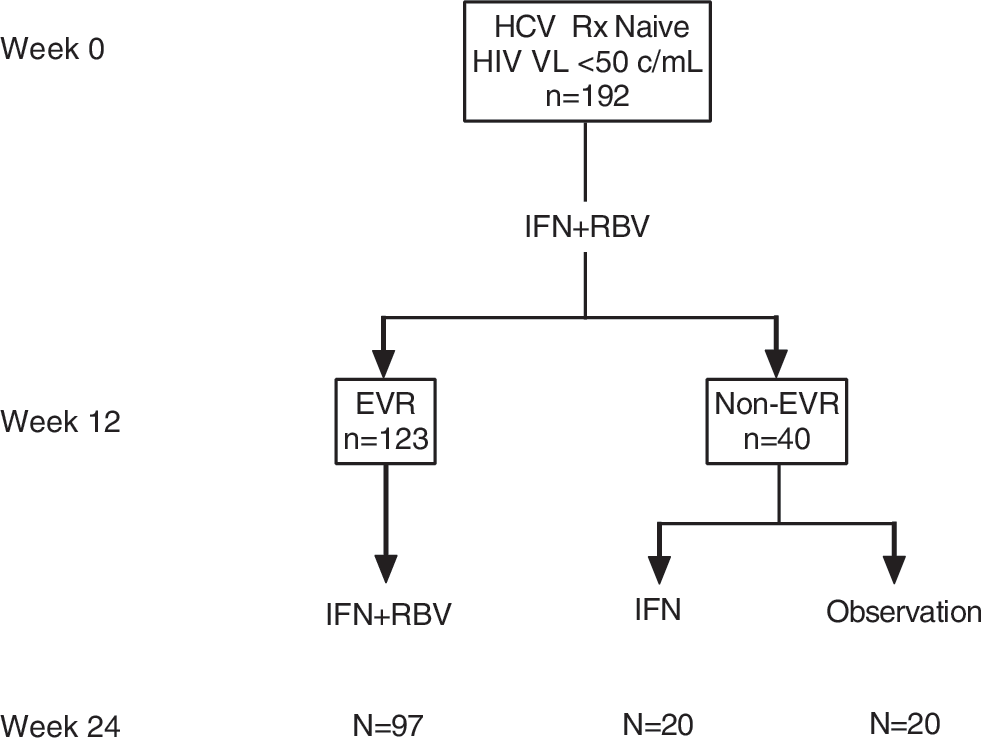
Schema of the AIDS Clinical Trial Group (ACTG) study A5178 and samples included in our analysis. HIV/HCV-coinfected patients received HCV treatment with IFN/RBV. Those who achieved an EVR (≥2 log HCV RNA drop at week 12) continued IFN/RBV while those without EVR were randomized to continuation of IFN alone or observation off therapy. We used samples of patients who had fully suppressed HIV viremia (HIV RNA <50 copies/ml) at entry into A5178. CTX and P1NP were measured at baseline, at week 12 (during PEG-IFN/RBV therapy), and at week 24 (while on PEG-IFN/RBV, PEG-IFN alone, or observation). IFN, pegylated interferon; RBV, ribavirin; EVR, early virologic response [≥2 log hepatitis C virus (HCV) RNA drop at week 12].
Median baseline (Q1, Q3) CTX and P1NP were 310 (230, 450) pg/ml and 58.24 (44.09, 75.39) μg/liter, respectively. Prior TDF exposure was associated with higher CTX and P1NP in univariate analyses (p = 0.05 and p < 0.01, respectively) and P1NP multivariate analysis adjusting for age, race, and cirrhosis, but not in the CTX multivariate analysis. In multivariate analysis, cirrhosis was independently associated with lower CTX (p < 0.01).
Twelve weeks of PEG-IFN/RBV significantly decreased the median CTX by 38.7% from baseline: −120 (−240, −40) pg/ml, p < 0.0001 (Fig. 2). CTX declines were similar between EVR and non-EVR groups: −140 and −100 pg/ml; p = 0.11, while greater declines were observed between cEVR and non-cEVR categories: −160 and −80 pg/ml; p = 0.003. Whites had greater median decreases in CTX than nonwhites, while cirrhotics had smaller median decreases. After adjusting for race or cirrhosis, we detected no difference in CTX between EVR and non-EVR subjects (p = 0.22 and 0.23, respectively). However, the difference in CTX between cEVR and non-cEVR remained statistically significant after an adjustment for race or cirrhosis (p < 0.01 for both).
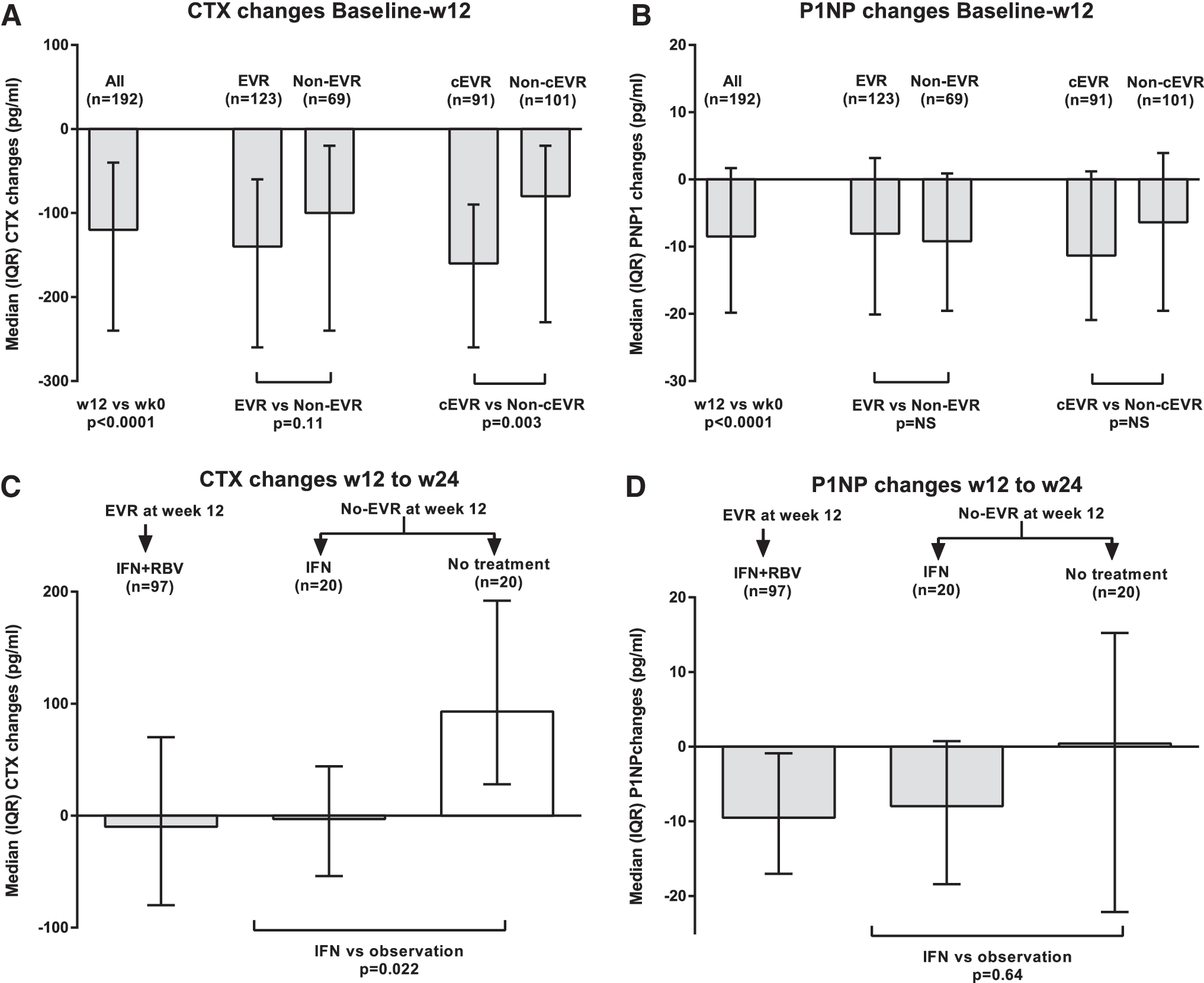
From week 12 to 24, CTX declines were sustained among EVR patients with continued PEG-IFN/RBV (−10 pg/ml median) and non-EVR patients randomized to continue PEG-IFN alone (−3 pg/ml), but rebounded among non-EVR patients who discontinued PEG-IFN/RBV (+93 pg/ml): p = 0.03 for EVR and non-EVR (N = 40, combined) and p = 0.022 for non-EVR on PEG-IFN maintenance vs. observation. The group difference in CTX by EVR status remained significant after adjusting for cirrhosis (p = 0.04) or race (p = 0.03). The difference in CTX declines was also more pronounced among whites (p = 0.05).
The median change in P1NP from baseline to week 12 was −8.48 (−19.81, 1.67) μg/liter (p < 0.001), though of lower magnitude (14.6%) than the decline in CTX. However, the median changes were similar between EVR and non-EVR patients (−8.07 and −9.19 μg/liter; p = 0.82). From week 12 to 24, the overall median P1NP decreased further, to 39.93 (27.82, 57.82) μg/liter. Similarly, there were no statistically significant differences in the median decreases from week 12 to week 24 between EVR and non-EVR patients (−9.5 and −4.6 μg/liter; p = 0.22).
Discussion
Treatment of HCV with PEG-IFN/RBV in patients coinfected with HIV in ACTG A5178 resulted in an approximately 40% decline in the marker of bone resorption CTX by week 12. Patients who achieved cEVR had a greater CTX decline. These effects were more apparent in whites. In addition, patients who continued PEG-IFN containing HCV therapy had sustained declines in CTX from week 12 to week 24, regardless of HCV virologic suppression. Those who discontinued all anti-HCV therapy after week 12 experienced a return to baseline CTX values. Similarly, median levels of the bone formation marker P1NP decreased significantly with PEG-IFN/RBV therapy, but the changes were not associated with early virologic response status and those individuals who stopped PEG-IFN experienced a rebound in P1NP values.
Our findings suggest that HCV treatment is likely to improve bone health by reducing bone turnover. Independent of its virologic effect, the role of IFN in improved BMD is biologically plausible since interferon types I and II appear to modulate bone turnover by preventing excessive osteoclastogenesis, in part through interactions with the receptor activator of the NF-κB ligand (RANKL). 16,18 Should the observed changes in BTM be a direct IFN effect, current IFN-free therapies might not have the same effect. Indeed, since we also demonstrated a greater decline in bone resorption among persons who achieved cEVR, it is plausible that HCV eradication contributed to the improvements observed. This question will be definitively answered with studies of IFN-free regimens. The contribution of RBV to our findings is unclear. Most preclinical data suggest that RBV would have a negative effect on bone turnover by impairing osteoblast proliferation and enhancing osteoclast formation. 19
A limitation of our study is the lack of posttreatment samples to assess the durability of observed BTM changes and whether they translate into long-term improvements in BMD and decreases in fracture risk. However, the magnitude of reduction in bone resorption is commensurate with that observed with bisphosphonate therapy. 10
In conclusion, our findings suggest that IFN-based HCV therapy has a net beneficial effect on BTMs in HIV/HCV patients. If these findings are confirmed with IFN-free regimens, improvements in bone health may be a secondary goal of HCV therapy.
Footnotes
Acknowledgments
The project described was supported by Award Number U01AI068636 from the National Institute of Allergy and Infectious Diseases and by the National Institute of Mental Health (NIMH) and the National Institute of Dental and Craniofacial Research (NIDCR). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health.
Additional support was provided by a grant from Merck & Co., and by VA MERIT grant I01 CX000418-01A1 to Roger Bedimo.
Author Disclosure Statement
No competing financial interests exist.
