Abstract
Objectives:
The study aims to evaluate the therapeutic efficacy and safety of Chinese herbal medicine (
Design:
A randomized, controlled, multicenter study from December 2013 to August 2015.
Settings/Location:
All patients are from China.
Subjects:
One hundred forty patients with either NSCLC or gastric cancer were enrolled in this trial.
Interventions:
The intervention group (
Outcome measures:
Platelet (PLT) count was tested at day 0, 7, 14, and 21 as the primary outcome for evaluation. Safety measurements, including red blood cells (RBC), hemoglobin (HBG), white blood cells (WBC), neutrophil (NE)#, aspartate aminotransferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), creatine kinase (CK), creatinine (Cr), and blood urea nitrogen (BUN) were tested at day 0 and 21 as the secondary outcomes.
Results:
(1) Two patients in the intervention group and four patients in the control group were lost upon follow-up. (2) PLT count: there was no significant difference in PLT count between the two groups from baseline (day 0), day 7, and day 14. At day 21, the intervention group indicated an upward trend of PLT count with a statistically significant difference than that of the control group (
Conclusions:
In conclusion,
Introduction
Cancer is the leading cause of death in China and a major public health concern, increasing in rate of both incidence and mortality. 1 –3 Lung cancer (incidence 733.3/100,000 and mortality 610.2/100,000) and gastric cancer (incidence 679.1/100,000 and mortality 498.0/100,000) are the first and second most prominent cancers in China, respectively, in terms of incidence and mortality rates. 4
Chemotherapy is the predominant treatment method for cancer with substantial progress being made in the last 50 years. Platinum-based chemotherapy is used significantly, with benefits, including increasing overall survival rates, enhancing quality of life (QoL), and increasing survival in advanced-stage disease. 5 Unfortunately, thrombocytopenia is a common side effect in cancer patients resulting from cytotoxicity. 6 Development of chemotherapy-induced thrombocytopenia (CIT) may cause a delay or reduction to the course of chemotherapy, subsequently affecting its effectiveness. 7 Moreover, CIT is the main dose-limiting toxicity factor of platinum-based chemotherapy. 8 CIT is treated with platelet (PLT) transfusion, recombinant human thrombopoietin (rhTPO), and recombinant human interleukin-11 (rhIL-11), 9 all of which are approved by the China Food and Drug Agency (CFDA). There are, however, present risks associated with transfusion in terms of transfusion-related acute lung injuries. 10 Several side effects of rhTPO and rhIL-11 have been documented, including fever, chills, fatigue, rash, edema, dyspnea, pleural effusion, atrial arrhythmia, and headache. 11,12
Traditional Chinese Medicine (TCM) is a complementary and alternative medicine that is used not only in the treatment of cancer and cancer-related conditions, but also to improve the side effects of anticancer therapies. 13 A meta-analysis of clinical studies has found that TCM may improve cancer-related symptoms, QoL, enhance immune function and reduce the side effects of chemotherapy. 14
TCM theory dictates the pathology of CIT as a heat toxicity that damages the
Materials and Methods
Study design
The study is a randomized, controlled, multicenter trial. It is registered in the Chinese Clinical Trial Registry (ChiCTR) under registration no.: ChiCTR-TRC-13003888.
A study sample size of 120 patients was calculated based upon a prior study. 21 The allocation ratio was 1:1, with 60 patients in the intervention group and 60 patients in the control group. A sample size of 144 patients was deemed sufficient to compensate for a 20% attrition rate. 21
Allocation: independent third-party researchers used opaque envelopes to seal distribution cards containing computer-generated random numbers that were then sent to researchers at collaborating centers. Once a patient was deemed eligible, they were randomized into a group according to the provision on the distribution card. Patients and researchers were not blinded to the type of treatment, and the participants were informed of the study design when recruited.
Participants
The participants were recruited from six collaborating medical hospitals in China.
Inclusion criteria
Inclusion criteria: (1) diagnosed with NSCLC or gastric cancer by imaging and/or histopathology or cytology; (2) no age limit; (3) Karnofsky Performance Status score ≥60; (4) Expected survival time ≥6 months; (5) indicated to receive initial platinum-based chemotherapy only, without receiving radiotherapy and/or chemotherapy within 2 months of trial commencement; (6) no diagnosis of comorbid disease (heart, liver, kidney, blood disease); and (7) willingness to participate in clinical research and to sign informed consent.
Exclusion criteria
Exclusion criteria: (1) did not meet the inclusion criteria; (2) mental disorders (cancer combined with moderate depression or above) as assessed by previous medical history or referring clinician (to exclude patients unable to correctly describe the subjective symptoms); (3) patients who had serious uncontrolled comorbid disease or acute infection; (4) pregnant or lactating women; (5) previous record of poor drug compliance by medical treatment history.
Treatment and follow-up
Patients in both groups received conventional chemotherapy. The intervention group received a daily
Outcome measures
Primary outcomes measurements
Primary outcomes: effectiveness was measured with PLT count of both groups. PLT count was assessed at the baseline and at three time points: day 7, 14, and 21. The data of each time point was analyzed and compared between intervention and control groups, and for both NSCLC and gastric cancer subgroup patients.
Secondary outcome measurements
Secondary outcomes: safety measurements included count of white blood cells (WBC), red blood cells (RBC), hemoglobin (HGB), alanine aminotransferase (ALT), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), creatine kinase (CK), blood urea nitrogen (BUN), and creatinine (Cr). The above figures were assessed at baseline and day 21. The data from each time point was analyzed between the two groups.
Adverse events
All participants were asked about any adverse events during their visits and were also instructed to record any unexpected events throughout the trial in a diary.
Statistics methods
The analyses were performed using SPSS software (IBM, Version 21.0) by a third-party clinical evaluation center. Frequencies and descriptive statistics were used for patients' demographic presentation, mean and standard deviations were calculated for the continuous variables, and group differences were analyzed using Pearson chi-square test for categorical variables. A Kolmogorov–Smirnov test was used to check the distribution of data. Independent
Results
Characteristics of the patients
From December 2013 to August 2015, 144 patients were monitored in total, with 140 patients allocated evenly between an intervention group (
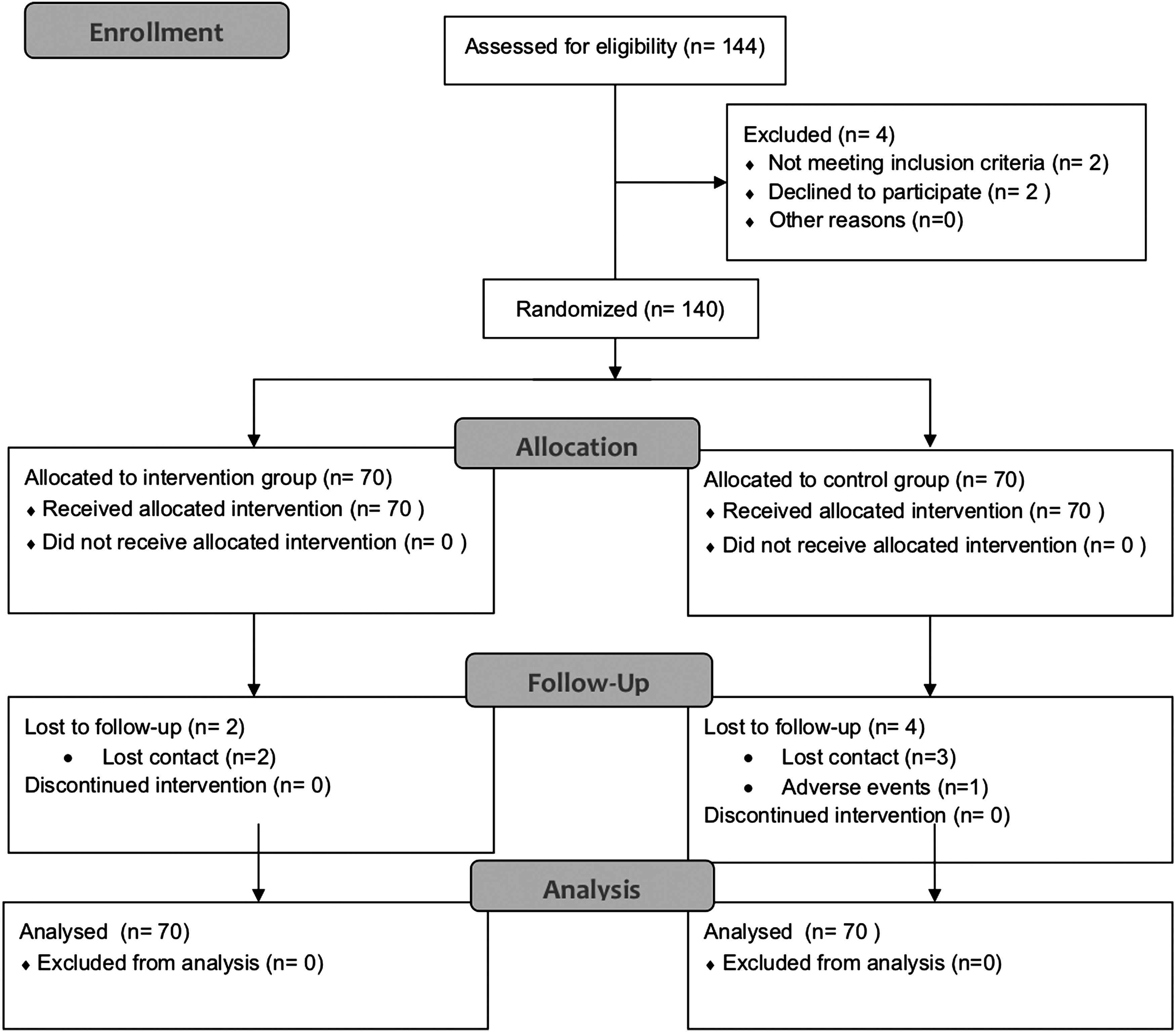
Flow diagram of enrolment.
There were no significant differences at the baseline between intervention and control groups' patient characteristics, in terms of age, gender ratio, and cancer type ratio (Table 1).
Comparison of Baseline Characteristics Between the Two Groups
CI, confidence interval; SD, standard deviation.
Primary outcomes
During the period of treatment and subsequent follow-up, there was no significant difference in PLT count between the two groups at baseline, day 7, and 14. There was significant downward trend in PLT count from baseline to day 21 in both groups. At day 21 the control group showed a continuing downward trend of PLT count, while the intervention group showed an upward trend of PLT count; there was a statistically significant difference between the two groups at this time (
Primary Outcomes of Platelet Count in Two Groups
CI, confidence interval; SD, standard deviation.
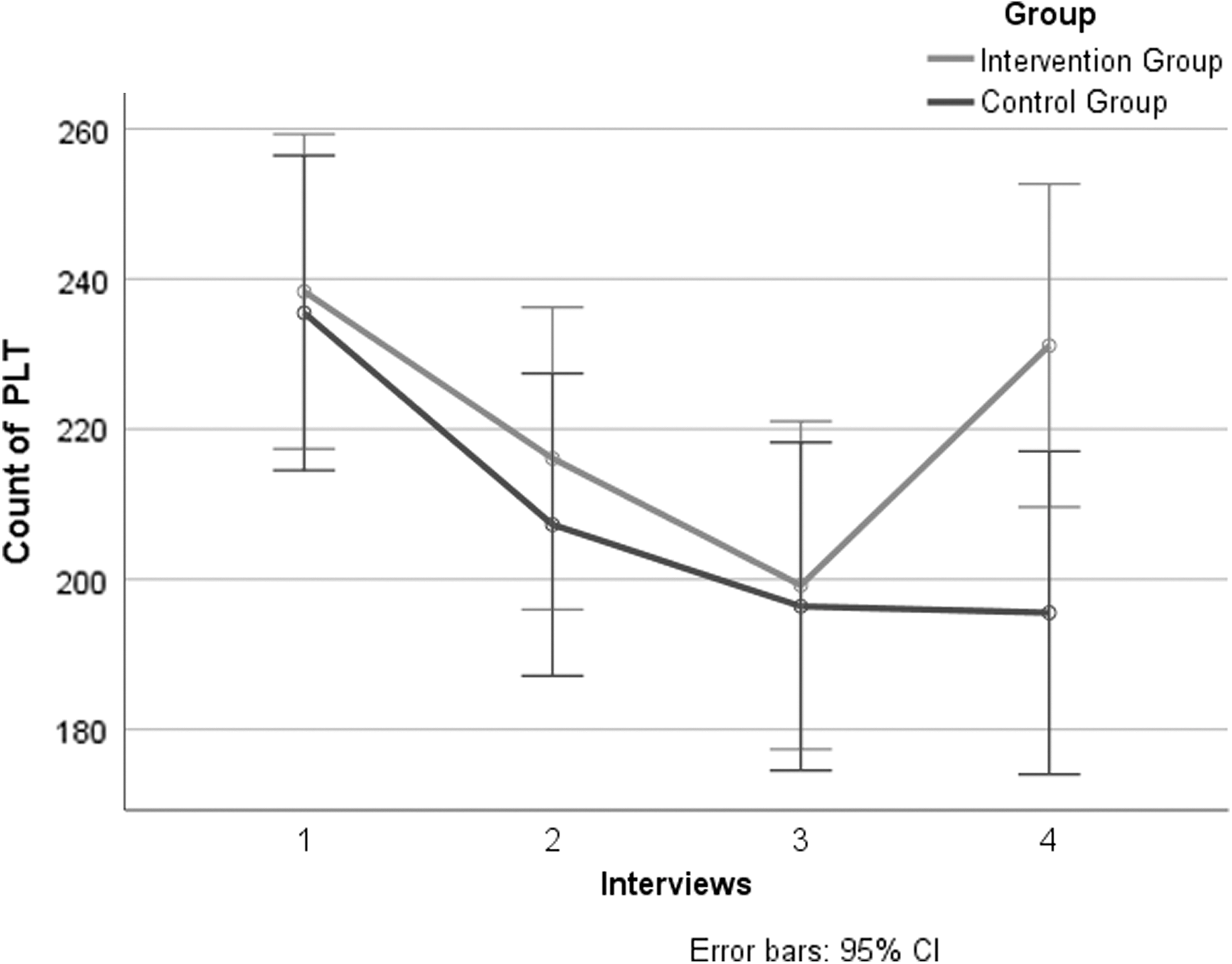
Primary outcomes of PLT count in the two groups of four interviews. CI, confidence interval; PLT, platelet.
Further subgroup analysis of NSCLC patients: PLT count in NSCLC patients in the two groups decreased from baseline to day 21. A significant difference existed between the two groups (
Primary Outcomes: Subgroup Analysis of Platelet Count in Nonsmall Cell Lung Cancer Patients
CI, confidence interval; SD, standard deviation.
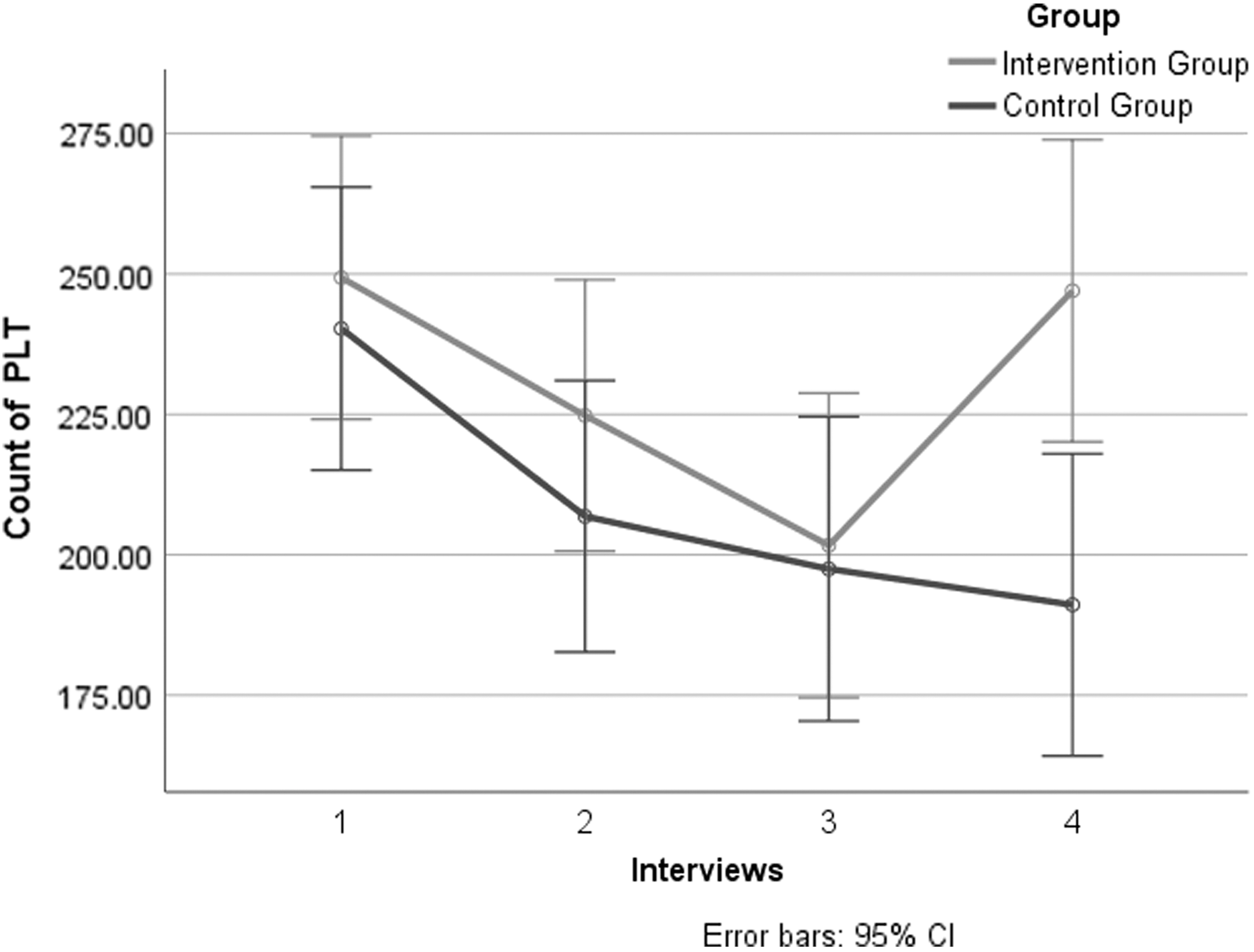
Primary outcomes: subgroup analysis of PLT count in NSCLC patients of four interviews. CI, confidence interval; NSCLC, nonsmall cell lung cancer; PLT, platelet.
Further subgroup analysis of gastric cancer patients: PLT count decreased in gastric cancer patients and there was no significant difference between the two groups (Table 4; Fig. 4).
Primary Outcomes: Subgroup Analysis of Platelet Count in Gastric Cancer Patients
CI, confidence interval; SD, standard deviation.
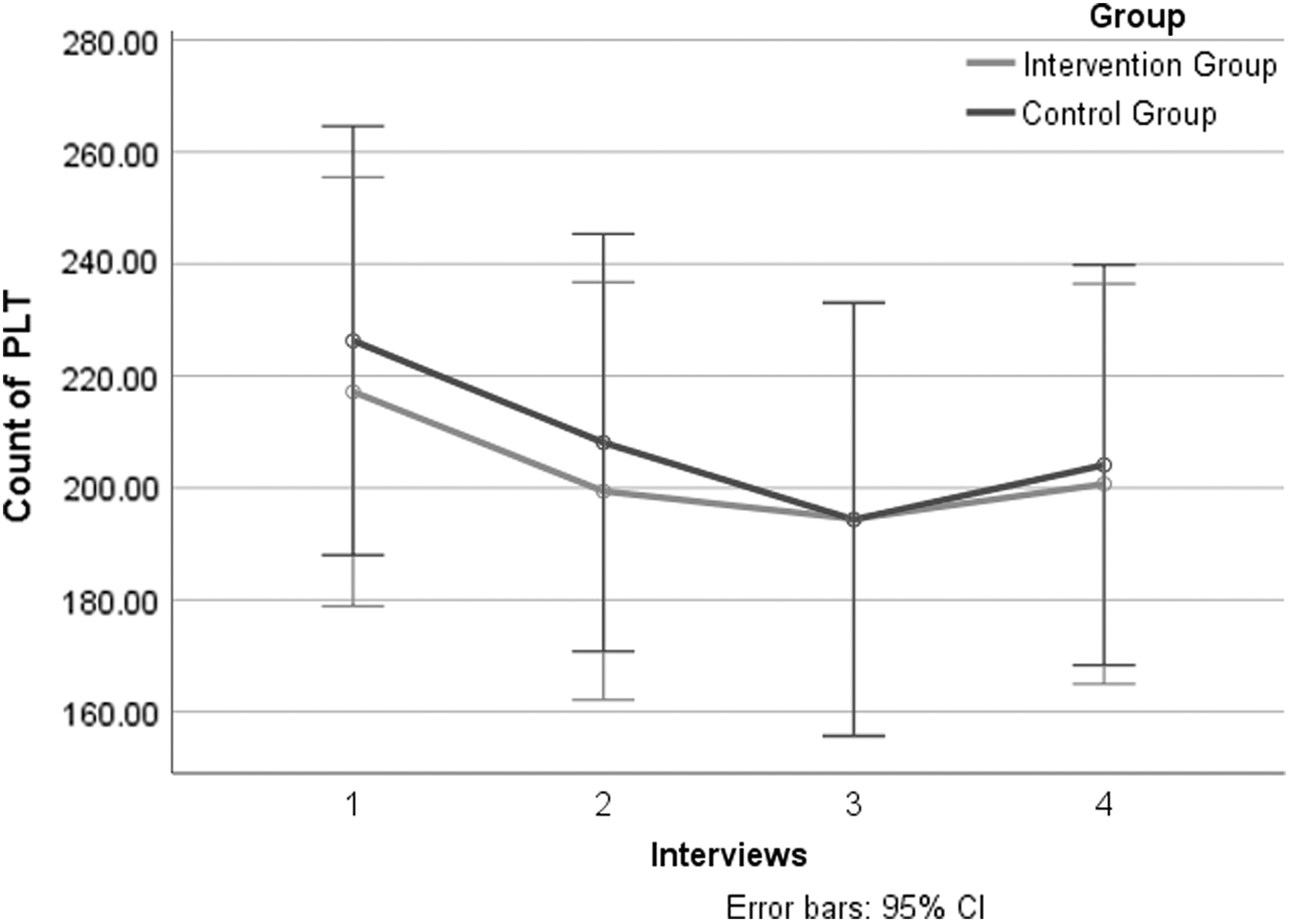
Primary outcomes: subgroup analysis of PLT count in gastric cancer patients of four interviews. CI, confidence interval; PLT, platelet.
Secondary outcomes
There was no statistically significant difference between the intervention group and the control group with safety figures, in terms of RBC, HGB, WBC, neutrophil (NE)#, AST, ALT, LDH, CK, Cr, and BUN (
Secondary Outcomes of Safety Figures in Two Groups from Day 0 and 21
ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; CI, confidence interval; CK, creatine kinase; Cr, creatinine; HGB, hemoglobin; LDH, lactate dehydrogenase; NE, neutrophil; RBC, red blood cells; SD, standard deviation; WBC, white blood cells.
Adverse events
During the study, one gastric cancer patient in the control group was diagnosed with gastrointestinal bleeding on day 3 and was withdrawn from the study.
Discussion
CIT is a toxic side effect of platinum-based chemotherapeutics.
23
In the rationale of TCM, chemotherapy drugs generate heat toxins, which directly damage bone marrow and blood collaterals leading to marrow collateral damage and obstructed blood movement. Stagnation of toxic pathogens in bone marrow and weakened generation and transformation of bone marrow cause
TCM is a prominent complementary and alternative medicine therapy used in oncology.
4,13
A systematic review
18
reported that Chinese medicine injections are commonly used to relieve chemotherapy-related symptoms, such as leukopenia, vomiting, and nausea, with
The determination of the safety of Chinese medicine for cancer care remains a priority.
27
RBC, HGB, WBC, NE#, AST, ALT, LDH, CK, Cr, and BUN were tested in this study with no significant differences between the two groups at the baseline and day 21 (
Conclusions
In conclusion,
Footnotes
Acknowledgments
The authors thank the collective trial team and third-party researchers for their assistance in this study. Funding: This study was supported by grants from the PhD Research Program of Beijing University of Chinese Medicine.
Author Contributions
X.L., L.H., and X.C. designed the study. X.L., Q.D., and S.T. collected the data. X.L., Q.D., and S.T. were involved in mortality follow-up and verification. S.Q., X.L., and K.W. analyzed the data. S.Q. drafted the article. S.Q. and X.L. contributed to the interpretation of the results and critical revisions of the article for important intellectual content and approved the final version of the article. All authors have read and approved the final article. S.Q. and X.L. are the study guarantors. D.P. and H.L. contributed to language editing.
Ethics Approval
This study was approved by the Ethics Committee of Dongzhimen Hospital Affiliated to Beijing University of Chinese Medicine (Beijing, China). No:. ECPJ-BDY-2013-36.
Author Disclosure Statement
No competing financial interests exist.
