Abstract
Dupilumab is a monoclonal antibody that represents the first approved targeted biological therapy for adults, adolescents, and children older than 6 years with moderate-to-severe atopic dermatitis (AD). Dupilumab binds the shared chain of the interleukin-4 and interleukin-13 receptor blocking the downstream signaling of these cytokines. The clinical improvements induced by dupilumab were associated with remission of the dysregulated immune mechanisms linked with AD. Dupilumab reversed the epidermal barrier defects and improved the global molecular signature of AD. This review seeks to provide an overview on the development of dupilumab as the first target-specific biological treatment for AD, with a description of the clinical trials that have been performed in different age groups, their outcomes, and reported adverse effects. Novel aspects of dupilumab treatment, as well as the current knowledge on the molecular and cellular mechanisms underlying the treatment of AD with dupilumab, are summarized and discussed.
Allergic diseases affect approximately 1 billion patients, and this prevalence is steadily increasing worldwide. The pathophysiology of allergic diseases is complex, and numerous different factors are involved in its molecular bases. Recent advances in the immunologic and genetic mechanisms underlying allergic diseases are allowing the stratification of their heterogeneous pathophysiology in specific endophenotypes.1 However, up to now, most of the treatments for allergic diseases are not tailored and not biomarker specific. In that regard, the understanding of the molecular basis that defines different clinical subtypes is opening a new era for personalized, target-specific treatments for allergic diseases.2 Up to 20% of children and 10% of adults worldwide experience atopic dermatitis (AD), an itchy and stigmatizing chronic inflammatory skin disease.3 In the cutaneous inflammation involved in AD, the T helper type 2 (TH2) axis is not only important in the acute phase, but also it has a substantial contribution to the chronicity of the disease in which an intensified inflammation occurs.4,5 In this context, increased levels of the TH2 cytokines interleukin (IL)-4 and IL-13 have been associated with an augmented risk of AD with an important function in its pathophysiology. Both cytokines contribute to the suppression of filaggrin and other terminal differentiation proteins. Interleukin 4 and IL-13 have important effects in the downregulation of antimicrobial peptides, in the induction of epidermal hyperplasia, spongiosis, and suppression of synthesis of lipids, which translate into deleterious skin barrier–modulating effects.6 Targeting TH2-related cytokine receptors would be beneficial for endophenotypes with a strong implication of the TH2 inflammatory axis.5,7,8 Dupilumab is the first Food and Drug Administration (FDA)- and European Medicines Agency (EMA)-approved targeted biological therapy for adults, adolescents, and children older than 6 years with moderate-to-severe AD. Dupilumab is a human monoclonal antibody that binds to the shared chain of the IL-4 and IL-13 receptor blocking downstream signaling of these 2 cytokines.9,10 Early phase clinical trials of dupilumab demonstrated evidence of clinical improvement in adults with moderate-to-severe AD. Afterward, several large, randomized, double-blind, placebo-controlled phase III trials demonstrated that dupilumab improved the signs and symptoms of AD in adults, adolescents, and children older than 6 years. This article provides an updated overview on the development of dupilumab as a target-specific biological treatment for AD in the context of personalized medicine with a focus in the clinical trials performed over the last years, novel aspects of dupilumab treatment, and the molecular and cellular mechanisms that underlie the treatment of AD with dupilumab.
DUPILUMAB: CHARACTERISTICS AND PHARMACOKINETICS
Dupilumab (Dupixent; Sanofi and Regeneron Pharmaceuticals, Inc) is a human immunoglobulin G4 monoclonal antibody that binds to the IL-4Rα chain that is shared by the receptor type I IL-4R for IL-4 and the receptor type II IL-4R for both IL-4 and IL-13, blocking downstream signaling of both IL-4 and IL-13 (Fig. 1). Dupilumab has a molecular weight of 147 kDa, and it has been produced in Chinese hamster ovary cells by recombinant DNA.11
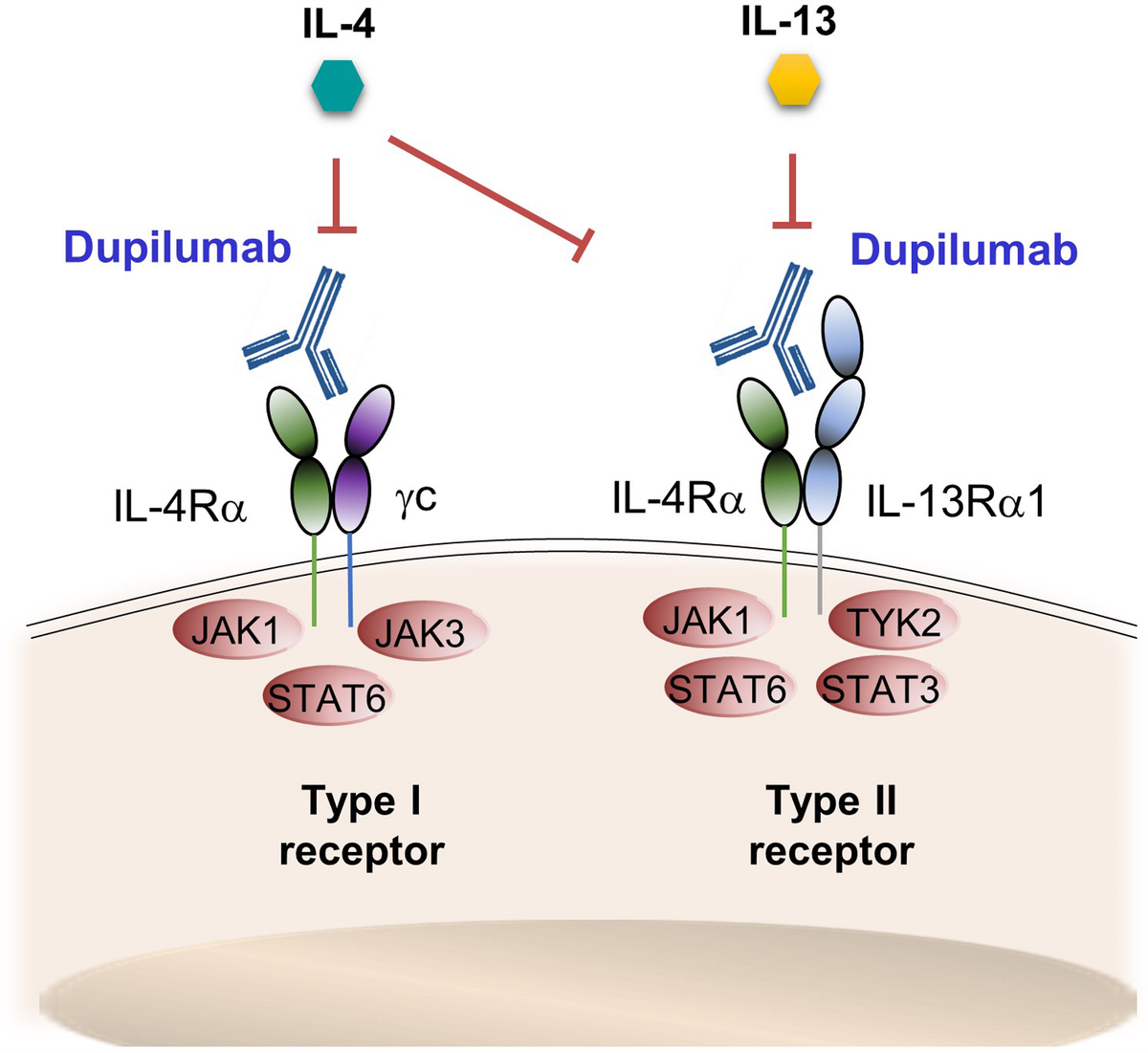
Dupilumab binds to the chain IL-4Rα, which is shared by type I receptor (IL-4 signaling) and type II receptor (IL-4 and IL-13 signaling), thus blocking the downstream signaling of IL-4 and IL-13.
Dupilumab administered subcutaneously has a bioavailability of 64%, and the estimated total volume distribution is approximately 4.8 ± 1.3 L (mean ± SD). Dupilumab is characterized by nonlinear pharmacokinetics reaching concentrations of 70.1 ± 24.1 μg/mL after 1 week of an initial subcutaneous dose of 600 mg.11,12 After this starting dose, dupilumab 300 mg administered weekly or in 2-week intervals produces steady-state concentrations at week 16 from 73.3 ± 24.1 to 79.9 ± 41.4 μg/mL for the dose of 300 mg in 2-week intervals and from 173 ± 75.9 to 193 ± 77 μg/mL for the dose of 300 mg administered weekly. Once the steady-state dose is reached, the median of nondetectable concentration (<78 ng/mL) is 10 weeks for the dose of 300 mg in 2-week intervals and 13 weeks for the dose of 300 mg administered weekly.11 The concentration-time profile of dupilumab is similar to other monoclonal antibodies with target-mediated clearance.12 Dupilumab, as a monoclonal immunoglobulin G4 antibody, is expected to be degraded through catabolic pathways into small peptides and amino acids.11
Interleukin 4 AND IL-13 FUNCTIONS
Interleukin 4 and IL-13 are central cytokines in atopic diseases. They are produced by different cell types, such as TH2 cells, basophils, mast cells, or eosinophils, among others. Interleukin 4 is the major stimulus for the differentiation of TH2 cells, with a suppressive effect over the development of TH1 cells and a cross-regulation to other TH pathways. Interleukin 13 shares effector functions with IL-4, such as induction of immunoglobulin E class switching in B cells, upregulation of B cell receptors, regulation of dendritic cells, macrophages' function, and regulative effects over T cells.13–15 Both cytokines favor the recruitment of eosinophils and promote their survival.6,16 As it has been indicated, there are 2 receptors for IL-4: type I IL-4R, which binds only IL-4, and type II IL-4R, which binds both IL-4 and IL-13. Both receptors have 2 chains, which consist of IL-4Rα chain (CD124, dupilumab target) for type I and type II receptors, chain γc (CD132), which is specific for type I receptor, and IL-13Rα1 chain, which is specific for type II receptor (Fig. 1).16
Biologics that specifically target the IL-13 molecule or IL-13 receptor (IL-13Rα1 chain) have also demonstrated to be effective in the treatment of AD. In that respect, tralokinumab, a monoclonal antibody that blocks the IL-13 molecule, has recently received approval from the EMA and FDA for the treatment of moderate-to-severe AD.17 The biological ASLAN004, which is a monoclonal antibody that binds the IL-13Rα1 chain, is currently under evaluation in a phase IIb study for the treatment of AD.18
DUPILUMAB FOR THE TREATMENT OF ADULTS, ADOLESCENTS, AND CHILDREN WITH AD
Phase III Trials in Adult Patients
Three large, randomized, double-blind, placebo-controlled phase III trials evaluated the effect of dupilumab on AD over a period of 16 weeks (SOLO 1 and SOLO 2 trials) or dupilumab in combination with topical corticosteroids (TCSs) over a period of 52 weeks (LIBERTY AD CHRONOS trial).19,20 In the 3 trials, adults (≥18 years of age) experiencing moderate-to-severe AD inadequately controlled by topical treatments were included. Patients were randomly assigned (SOLO 1 and SOLO 2: 1:1:1; LIBERTY AD CHRONOS: 3:1:3) to 300-mg dupilumab administered subcutaneously weekly or in 2-week intervals or placebo. On day 1, a dose of 600-mg dupilumab or placebo was administered as a loading dose in the 3 trials. The primary end point in the 3 studies was the proportion of patients with Investigator’s Global Assessment (IGA) score of 0/1 (clear/almost clear) together to a reduction of 2 points or higher from baseline on this score at week 16. An improvement in Eczema Area and Severity Index (EASI) score of at least 75% from baseline at week 16 was a coprimary end point in LIBERTY AD CHRONOS and key secondary end point in SOLO 1 and SOLO 2. The results of the 3 studies showed that a higher proportion of patients who received dupilumab versus those who received placebo achieved the primary end point at week 16. In LIBERTY AD CHRONOS, the same efficacy of dupilumab was also seen at week 52. The most common adverse effects in dupilumab groups in all the trials were conjunctivitis and reactions in the site of injection (complete details can be checked in the studies by Simpson et al,19 Blauvelt et al,20 and Thaçi et al21). Additional phase III trials confirmed the obtained results.22,23 Post hoc analyses of the results of SOLO 1 and SOLO 2 and LIBERTY AD CHRONOS trials showed that dupilumab produced an improvement from baseline in Peak Pruritus Numerical Rating Scale as early as day 2 of treatment and that improvement was sustained up to 1 year of treatment.24 Dupilumab equally improved the severity of AD in all the anatomical regions included in EASI, that is, head, neck, trunk, and upper/lower extremities over 1 year of treatment.25,26 Based on the results of the phase III trials: SOLO 1 and 2 and LIBERTY AD CHRONOS, dupilumab received FDA approval in the United States in March 2017 for its use in the treatment of inadequately controlled moderate-to-severe AD in adult patients. Dupilumab treatment was recommended to be used with or without TCS. Marketing authorization throughout the European Union was granted by the EMA in September 2017, which followed further authorizations in other regions of the world.23
DUPILUMAB AFTER FDA AND EMA APPROVAL IN ADULT PATIENTS
After FDA and EMA approval, the efficacy and safety of dupilumab in long-term maintaining response were studied in a phase III, randomized, double-blind trial (LIBERTY AD SOLO-CONTINUE). In this study, patients cataloged as high responders in the trials SOLO 1 and 2 at week 16 (IGA, 0/1; EASI, 75) were randomized to continue dupilumab monotherapy at the original regimes (300 mg weekly or in 2-week intervals), regimes with longer intervals (300 mg in 4- or 8-week intervals), or placebo for 36 weeks. Dupilumab administered weekly or in 2-week intervals for 36 weeks maintained the best clinical efficacy reached in SOLO 1 and 2 in contrast to the regimes with longer intervals (every 4 or 8 weeks). Therefore, dupilumab 300 mg, administered weekly or in 2-week intervals, was effective for long-term maintenance treatments.27 After dupilumab approval, real-world evidence of the effectiveness and safety of dupilumab was analyzed by multinational registries from daily practice settings that included adult patients with moderate-to-severe AD. Although the studies varied in their design or the specific measured outcomes, the vast majority showed a significant improvement in signs and symptoms of AD because of the treatment with dupilumab, supporting in this way the results obtained in the phase III trials. In these cases, and similar to the clinical trials, the most frequent adverse effects were conjunctivitis and eye irritation.28–35
Regarding the role of dupilumab on allergic contact dermatitis (ACD), which is characterized by a predominance of TH1 immune response (although other TH pathways, such as TH2, TH17, and TH22, can also be involved), some studies have demonstrated that patch tests results are not affected by dupilumab.36–38 In that sense, Raffi et al36 found in a retrospective study that included 48 patients with AD that dupilumab did not produce major inhibiting effects in patch test results and that most of the reactions were maintained. However, others have found that dupilumab induced variable effects on ACD lesions in AD patients with comorbid ACD.39 A recent systemic review, analyzing 19 studies on the effect of dupilumab in ACD, found that specific responses associated with ACD may be lost, remain unaffected, or worsened because of dupilumab treatment.40
The differential effect of dupilumab in ACD could be attributed to the specific immune pathway involved in ACD that mainly depends on the allergen that triggers the reaction. In that sense, it has been demonstrated that nickel induces TH1 and TH17 immune responses, whereas rubber or fragrances induces the TH2 pathway,41 in which dupilumab could be relevant because of its immunosuppressive effect on this pathway. However, clinical trials are necessary to establish the efficacy of dupilumab in ACD induced by certain allergens, such as fragrances and other allergens triggering TH2 immune responses.
DUPILUMAB FOR THE TREATMENT OF ADOLESCENTS AND CHILDREN
The FDA (United States) and EMA (European Union) extended the approval of dupilumab in 2019 for adolescents (ages 12–17 years) who have inadequately controlled moderate-to-severe AD.42 Moreover, in 2020, the FDA and EMA approved dupilumab as the first biologic medicine for children aged 6 to 11 years with moderate-to-severe AD.
In the case of adolescents, the approval was supported by the results obtained in a phase III trial (LIBERTY AD ADOL) and its extension (LIBERTY AD PED-OLE). In the former, adolescents with moderate-to-severe AD were randomized (1:1:1) to 200- or 300-mg dupilumab administered in 2-week intervals, 300-mg dupilumab in 4-week intervals, or placebo. The coprimary end points were the proportion of patients with IGA score of 0 or 1, an improvement in IGA score of 2 points or more from baseline, or an improvement of 75% or more in the EASI scale from baseline at week 16. The results showed that both coprimary end points were achieved in patients receiving dupilumab as opposed to the placebo group (P < 0.001 for all comparisons vs placebo). Numerically, the 2-week interval regime was superior to the 4-week interval regime.43 The open-label extension study, LIBERTY AD PED-OLE, reinforced the results obtained in the trial LIBERTY AD ADOL, providing evidence on the safety and efficacy of dupilumab in long-term treatment (52 weeks) in adolescents. The pharmacokinetic profile of dupilumab on this patient population was found similar than that in adults.44 A post hoc study of the trial LIBERTY AD ADOL showed that dupilumab produced a clinically meaningful improvement in symptoms, signs, and quality of life versus placebo.45 The improvement from baseline in the Peak Pruritus Numerical Rating Scale score was significant by day 5 of treatment with dupilumab.24 In the case of children aged 6 to 11 years, the approval was based on a phase III trial (LIBERTY AD PEDS), which demonstrated that dupilumab (administered 300 mg every 4 weeks or as a weight-related regimen every 2 weeks) with concomitant medium-potency TCS was efficacious in children with severe AD.46 Currently, the efficacy of dupilumab administered with standard-of-care low-potency TCS in children aged 6 months to 5 years with inadequately controlled moderate-to-severe AD is being evaluated in a 2-part phase II/III trial (NCT03346434: LIBERTY AD PRESCHOOL).
There are relevant aspects in the treatment of AD in adults, adolescents, and children with dupilumab who have not been addressed and analyzed in depth. These imply questions on the effective duration of dupilumab treatment over time, when would be reasonable to wean or stop the treatment, and how it should be done. These constitute practical issues that physicians face in their clinical practice and are associated with the treatment cost, the fact that AD symptoms wax and wane over time, or the specific circumstances of the patients that may prevent them from maintaining a relatively demanding dosing regimen (300 mg of dupilumab in 2-week intervals) over long periods.47 Long-term observational studies have analyzed dupilumab treatment administered under the approved regimen together with topical and initial concomitant systemic treatment for 84 weeks. Treatment improved clinical signs, symptoms, and quality of life up to 84 weeks.48 However, little information exists on the effective duration of dupilumab treatment, the possibilities and circumstances to reduce the regimen, or whether, on the contrary, dupilumab should be considered as an uninterrupted treatment. In other inflammatory skin diseases, such as psoriasis, the analysis of the use of biologicals, such as anti–tumor necrosis factor agents, anti–IL-12, and anti–IL-23, has demonstrated that an uninterrupted treatment was necessary to maintain the efficacy of the agents over the disease. The option of decreasing the dose was found to reduce the efficacy of the biological treatment.49
In the case of dupilumab, a study found that longer intervals of dupilumab administration (300 mg in 4- or 8-week intervals) than the approved dose (300 mg in 2-week intervals) were less efficient in disease control, especially patients receiving dupilumab every 8 weeks experienced more flares, had a higher risk of skin infections, and used more rescue medications than patients under the approved dose. In that way, the authorized regimen of dupilumab administered in 2-week intervals was the recommended regimen for long-duration treatment.27 It is also important to consider that decreasing the frequency of the dose can potentially lead to the induction of antibodies against the biological treatment, because it was suggested for other treatments with biologicals.50
Although, currently, the 300-mg dose every 2 weeks is the recommended regimen for long-duration treatment with dupilumab,27 more studies are needed to establish treatment options for longer periods than those studied to date, as well as the clinical characteristics that must be fulfilled for a potential dose reduction, for the establishment of longer intervals or even for a total cessation of the treatment and the different options to carry it out.
ADVERSE EFFECTS OF DUPILUMAB
Conjunctivitis was the most frequent adverse effect reported in adults, adolescents, and children with AD treated with dupilumab in phase II and phase III trials and real-life settings studies. The severity of conjunctivitis was, in most cases, mild or moderate, and it rarely conducted into the discontinuation of the treatment. The rate of dupilumab-associated ocular disease has been estimated in 32% to 36%,51,52 and a previous history of conjunctivitis and higher AD severity at baseline have been linked to an increase in conjunctivitis risk during dupilumab treatment.53,54
The pathomechanism of conjunctivitis induced by dupilumab treatment has been associated with a decrease in conjunctival goblet cells, which are cells specialized in the secretion of mucus essential for the correct function of the ocular surface.55 A local infiltration of highly activated immune cells and an increase in the local production of TH1-related cytokines have also been observed in patients with conjunctivitis induced by dupilumab treatment.56 The target receptor of dupilumab, IL-4Rα, is expressed in epithelial cells of the conjunctiva, and IL-13 expression has been associated with goblet cell proliferation and stimulation of mucus production in animal models. In that respect, blocking IL-13 function due to the binding of dupilumab to IL-4Rα would decrease mucus secretion and promote conjunctival inflammation, which would be reinforced by a local increase in TH1-related cytokines induced by the inhibition of IL-4/IL-13 signaling.55,56
Besides conjunctivitis, patients treated with dupilumab had a higher risk of reactions at the site of injection and a higher incidence of headache, as shown in a meta-analysis.57 Cases of hair loss during dupilumab treatment that clinically resembled alopecia areata (AA) have been described in some studies58–60; however, others have reported improvement of preexisting AA during treatment with dupilumab.60–62 In that sense, the role of dupilumab on AA warrants further study.
DUPILUMAB AND INFECTIONS INCLUDING SARS-CoV-2 (COVID-19)
The first meta-analysis that analyzed adverse events associated with dupilumab in 8 randomized controlled trials showed that adult patients with moderate-to-severe AD treated with dupilumab had a decreased risk of cutaneous infections. However, the risk of other infections, such as the ones produced by herpes virus and infections that affect the upper respiratory tract or the urinary tract, was unaffected by dupilumab.57 Further studies confirmed these observations, although they found that besides skin infections, certain serious and severe infections were lower in the dupilumab-treated group.63
Regarding coronavirus disease 2019 (COVID-19) produced by SARS-CoV-2, the initial studies published in the first period of the pandemic showed that dupilumab did not increase the rate of infection by SARS-CoV-2.64–66 Dupilumab treatment was demonstrated to be safe and well tolerated in AD patients with COVID-19, and it was recommended that the treatment should be continued during the COVID-19 pandemic.67,68 The first population-based estimate of the incidence and consequences of COVID-19 in AD patients under dupilumab treatment showed that dupilumab was not associated with a higher risk of COVID-19, hospitalizations, or mortality associated with COVID-19 compared with AD patients under other treatments (systemic corticosteroids, azathioprine/mycophenolate mofetil, phototherapy).69 In line with these observations, a recent large-scale study involving 1237 patients with moderate-to-severe AD analyzed the impact of dupilumab treatment over COVID-19 symptoms compared with other therapeutic treatments (phototherapy, oral Janus kinase inhibitors, prednisone, methotrexate, cyclosporine, or mycophenolate mofetil) or with limited treatment or no treatment. It was found that dupilumab-treated patients had a reduced incidence of COVID-19 and lower risk of severe symptoms associated with COVID-19 compared with the group of patients under other types of treatments for AD or no treatment. TH2 inflammation, a hallmark of AD, is associated with a decreased protection against viruses by inhibiting TH1 mechanisms that are involved in viral defense. In that sense, targeting TH2 axis by dupilumab would potentially rebalance TH1/TH2 axis and could explain the lower impact that COVID-19 seems to have in AD patients under dupilumab treatment.70 Further studies supported these results.71
With the development of mRNA and DNA vaccines for COVID-19 protection, several questions were raised about their potential effects on AD patients under biological treatments, such as dupilumab. Although vaccines for COVID-19 are safe and it was demonstrated that dupilumab treatment did not attenuate the vaccination response,72,73 different case studies have described eczematous reactions after administration of mRNA vaccines for COVID-19 in patients with AD under dupilumab treatment that quickly resolved after rescued treatments.74,75 A recent large study found that a small percentage of 407 AD patients (2.7%) under dupilumab treatment experienced AD flares after 7.27 ± 3.20 days of vaccination with either mRNA vaccines (BNT162b2, mRNA-1273) or DNA vaccines (AZD1222, Ad26.COV2.S). The flares were easily treated with topical therapies, and there was no discontinuation of dupilumab treatment.76
MOLECULAR AND CELLULAR MECHANISMS OF DUPILUMAB IN AD
The effects of IL-4/IL-13 blockage by dupilumab at a molecular level were initially studied on adult patients with moderate-to-severe AD that enrolled in 2 phase I trials in which dupilumab (150 or 300 mg) or placebo was administered subcutaneously on a weekly basis over a period of 4 weeks.77 In this study, the transcriptomic analyses of pretreatment and posttreatment skin biopsies showed a dose-dependent improvement of the global molecular signature of AD by dupilumab compared with placebo. Suppression of mRNA expression of genes involved in the activation of T cells, dendritic cells, or eosinophils together to an important inhibition in TH2-associated chemokines. The expression of genes related to hyperplasia was also decreased. A significant increase in claudin and lipid products was observed in the group treated with 300-mg dupilumab. Dupilumab reverse the epidermal barrier defects and improve the global molecular signature of AD (Fig. 2).78 A more extensive study involving 54 patients randomized to weekly administration of dupilumab (200 mg) or placebo for 16 weeks confirmed that dupilumab shifted the lesional transcriptome into a healthier one.79 A significant reduction in the cellular infiltrates in AD lesion as well as in the expression of type 2 inflammation genes and type 2 serum biomarkers was initially observed at week 4 and strongly confirmed at week 16. For instance, dupilumab decreased the expression of IL-31 in lesional skin, a cytokine that has been implicated in intense pruritus.79 Interleukin-31 production is predominantly upregulated by TH2 cells in AD, and the receptor A of IL-31 is expressed in keratinocytes and a subgroup of dorsal root ganglion neurons. The activation of sensory nerves in the skin by IL-31 seems to be one of those responsible factors of pruritus in the skin. In that sense, dupilumab might be implicated in the decreased production of IL-31 by TH2 cells with a direct impact on itch.79 The blockade of IL-4 and IL-13 signaling with dupilumab also led to a remission of epidermal abnormalities associated with AD.79 Further studies confirmed these results.80,81 The effects of dupilumab also impacted skin microbiota in the same cohort by increasing microbial diversity and reducing the abundance of Staphylococcus aureus as early as 4 weeks of treatment. These effects correlated with an improvement in clinical symptoms, as well as a reduction in type 2 inflammation.82 Profound changes in skin microbiome due to dupilumab were further confirmed in additional studies, in which together with a decrease in S. aureus, an increase in the relative abundance of Staphylococcus epidermidis and Staphylococcus hominis was also observed. Dupilumab decreases in that way the deleterious effects that S. aureus has in AD skin (Fig. 2).83
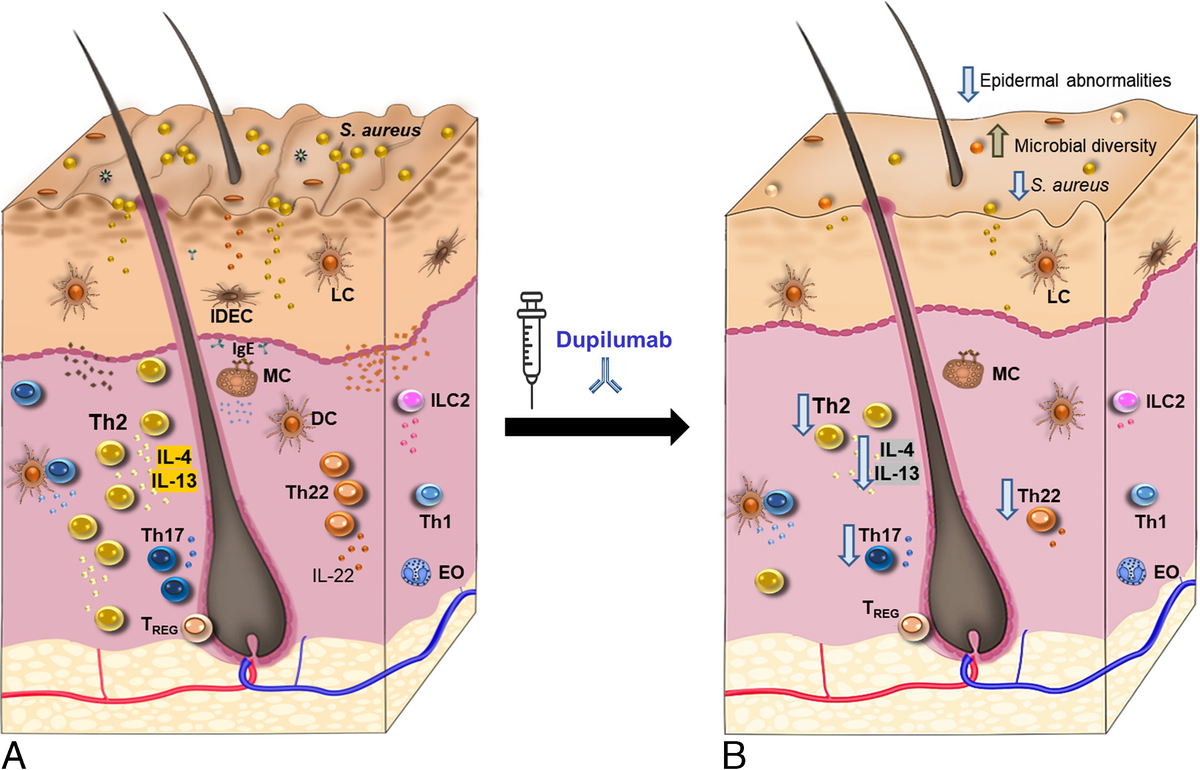
Effects of dupilumab in AD skin (A). Dupilumab reverses epidermal barrier defects and cellular infiltration in AD lesions (B). Dupilumab inhibits markers of inflammation linked to TH2, TH22, and TH17 axes (B). The treatment with dupilumab also impacts the skin microbiome by reducing the abundance of S. aureus and increasing microbial diversity (B). EO, eosinophil; IDEC, inflammatory dendritic epidermal cells; ILC2, type 2 innate lymphoid cells; LC, Langerhans cell; MC, mast cell; TREG, regulatory T cell.
The previously mentioned studies demonstrated that clinical improvements induced by dupilumab are associated with a remission of the dysregulated immune mechanisms linked with AD. However, the effects seem to go beyond inhibition of type 2 inflammation because a suppression of TH17 and TH22 axes has also been observed (Fig. 2).79,84 Other studies have explored the effects of dupilumab administered in the approved dose of 300 mg administered in 2-week intervals. In that respect, using the minimally invasive technique of tape stripping, a study found that dupilumab administered under the approved regimen induced a decrease in markers of inflammation associated with TH2, TH17/TH22 axes, and suppression of proteins associated with atherosclerosis and cardiovascular risk85 previously found to be linked with AD.86,87 However, dupilumab treatment did not affect the TH1 axis.85 It was also found that dupilumab treatment reduced serum AD biomarkers, such as the CCR4 ligand TARC (a chemokine that attracts T cells), the intracellular enzyme lactate dehydrogenase, total immunoglobulin E, and induced a decrease in blood eosinophilia.88 In that respect, a combination of the serum markers TARC, IL-22 (involved in barrier defects), and soluble IL-2 receptor (sIL-2R—an activator of T cells), which reflects different pathways involved in AD, has been found to predict AD severity in dupilumab-treated patients.89
CONCLUSIONS
The extensive knowledge acquired in recent decades in the molecular and cellular mechanisms underlying atopic diseases, such as AD, has allowed the identification of specific biomarkers that are leading to the development of molecule-specific biological treatments, such as dupilumab. The current advances in the molecular and cellular understanding of AD have a pivotal role in the stratification of AD in endophenotypes and in the development of therapies that target-specific molecules in a phenotype-specific way, leaving behind the “one-size-fits-all” therapeutic approach and opening a new era of personalized medicine in AD. Dupilumab treatment of moderate-to-severe AD in adults, adolescents, and children is an example of a promising, effective, and tailored treatment of AD. Further therapeutic strategies aiming to modulate molecular and cellular components involved in the pathophysiology of AD will potentially extend and refine the currently available treatments for AD.
