Abstract
Two new definitions of exercise-induced pulmonary hypertension (EIPH) have emerged. Both rely on measuring cardiac output (CO), yet this remains unstandardized. In our cohort of patients undergoing invasive cardiopulmonary exercise testing, we found that using thermodilution CO rather than direct Fick CO led to a significant excess of EIPH diagnoses.
Letter to the Editor
Since exercise-induced pulmonary hypertension (EIPH) was removed from the guidelines in 2008, 1 considerable progress has been made in redefining the boundary between normal and pathologic exercise hemodynamic responses.2–5 Currently, two leading candidates for a new EIPH definition exist. Lewis et al. 3 and Naeije et al. 4 have proposed the criterion of an abnormal pressure-flow relationship—or mPAP to cardiac output (CO) slope—of >3 mmHg min L–1 during exercise defines EIPH, while Herve et al. 5 proposed the combination of a peak exercise mPAP >30 mmHg and total pulmonary resistance (TPR) of >3 Wood units (WU) for the diagnosis. Despite dependence of both on accurate CO measurements, there remains significant variability in the CO method used in both research and practice. This is due to convenience, since thermodilution CO (TDCO) is more readily attainable, as well as the assumption that direct Fick CO (DFCO) and TDCO measurements are interchangeable, which is based on historical studies showing good agreement at rest. 6 However, comparison studies during exercise in both healthy and diseased states remain scant.7,8
We hypothesized that TDCO and DFCO measurements during exercise would not necessarily agree and tested whether use of TDCO rather than DFCO would affect the accuracy of the proposed EIPH diagnostic criteria. To investigate this, we performed, with institutional review board approval, a retrospective single-center study of participants that underwent invasive cardiopulmonary exercise testing (iCPET) with paired TDCO/DFCO measurements. TDCO and mixed venous oxygen saturations were simultaneously measured at rest and at the end of each 2-min stage of graded supine bicycle ergometry (stage 1, 15 W; increases of 10 W/stage). Systemic arterial oxygen saturations were obtained by pulse oximetry, while VO2 gas exchange was measured by metabolic cart and averaged over 1 min at rest and 30 s at the end of each stage. Hemodynamics were measured at end-expiration at rest and averaged over the respiratory cycle during exercise. 3 TDCO and DFCO were compared using regression, Bland–Altman analysis, and appropriate paired statistical comparisons, while the significance of discordant EIPH diagnoses was tested using Fisher’s exact test. Values were reported as mean ± SD, or median [25%ile, 75%ile] when appropriate.
From 2014 to 2016, 29 participants referred for exertional dyspnea had both TDCO and DFCO measured during iCPET. Nine participants with resting mPAP ≥ 25 mmHg were excluded, leaving 20 participants free of resting PH (15 women, 5 men; age, 56 ± 16 years; weight, 74 ± 21 kg). Regression comparison of all TDCO and DFCO values, adjusted for participant repeated measures, showed excellent correlation (R2 = 0.93, P < 0.001). However, TDCO progressively underestimated DFCO as CO increased (β-coefficient = 0.80) (Fig. 1a). Accordingly, peak exercise CO values were significantly lower when measured by thermodilution versus direct Fick (9.0 ± 3.9 L/min versus 11.33 ± 4.3 L/min, P < 0.001). Bland–Altman analysis comparing TDCO to DFCO revealed a significant overall negative bias of −1.81 L/min with TDCO, with unacceptably broad limits of agreement (Fig. 1b). Furthermore, TDCO underestimated DFCO more so at higher cardiac outputs (Fig. 1b, superimposed regression), with a −0.6 L/min bias at mean outputs <5 L/min but −2.2 L/min bias at outputs >10 L/min.
(a) Paired thermodilution cardiac output (TDCO) and direct Fick cardiac output (DFCO) measurements for 20 patients without resting pulmonary hypertension (PH). Linear regression showed excellent agreement between the two methods (R2 = 0.93, P < 0.001), but TDCO underestimated the gold standard DFCO as outputs increased (β-coefficient = 0.80). (b) Bland–Altman plot comparing the difference between TDCO and DFCO in the same cohort. Use of TDCO versus DFCO led to a mean bias of −1.81 L/min and unacceptably broad limits of agreement. Bias and lack of agreement were worse as output increased, as illustrated by the superimposed regression.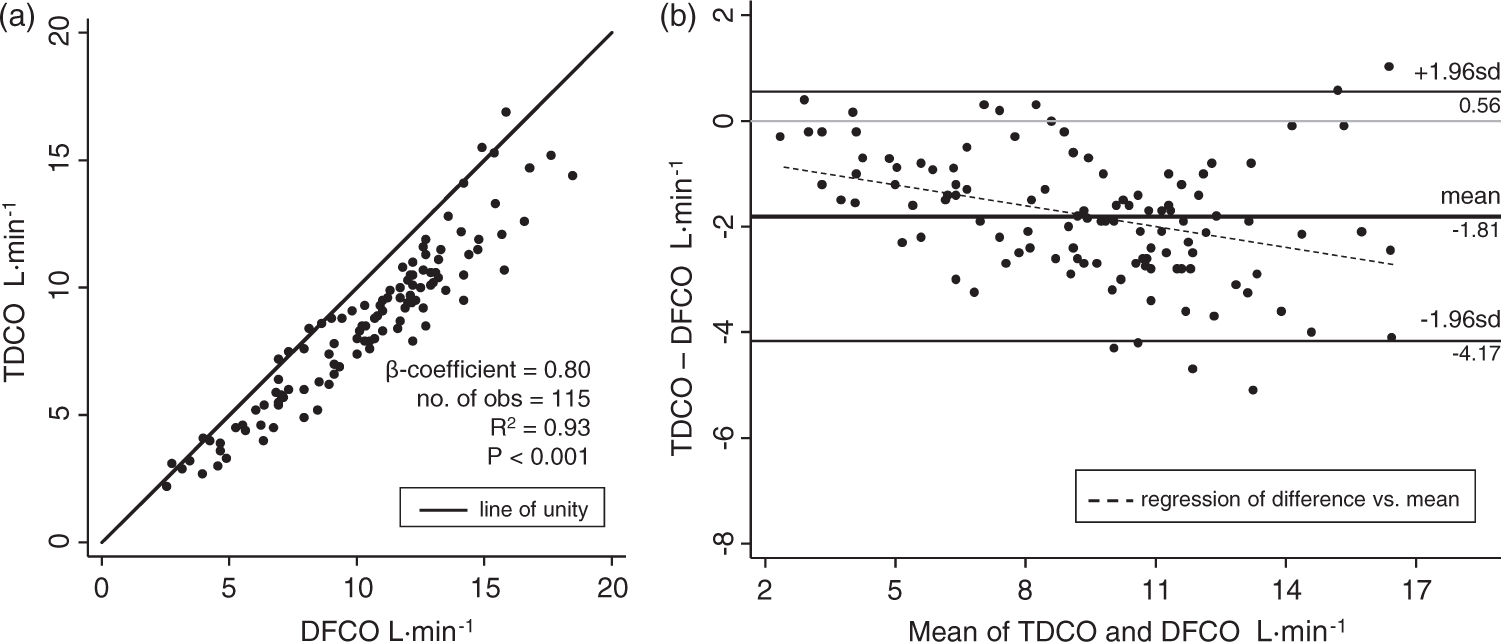
Importantly, use of TDCO led to a significant excess of EIPH diagnoses regardless of diagnostic criteria used. Exercise mPAP/CO slopes calculated using TDCO were significantly greater than those using DFCO (median 3.0 [1.0, 6.7] versus 1.9 [0.7, 3.1] mmHg·min·L–1, P = 0.04). This led to significantly more EIPH diagnoses (mPAP/CO > 3 mmHg min L–1) when TDCO was used (10/20 versus 6/20 participants, or 20% of the cohort, P = 0.01). Similarly, TPR was significantly greater with TDCO versus DFCO (median 3.7 [2.0, 5.4] versus 3.0 [1.7, 3.4] WU, P < 0.001), leading to a similar excess of EIPH diagnoses using the Herve criteria (10/20 versus 6/20 participants, P = 0.01).
Exercise testing has long been used to test deficiencies in cardiopulmonary reserve. The removal of EIPH hemodynamic criteria in 2008 was due to concerns more about methodology and false positivity rather than questions about the entity of EIPH itself. 9 Fortunately, recent insights have identified the slope of the exercise pressure-flow relationship as a superior determinant of pathology, which has led to two leading proposed diagnostic criteria for EIPH.4,5 The current study does not challenge the potential of these proposals—rather, it seeks to address methodological concerns. By showing that exercise measurements of TDCO underestimate DFCO at higher cardiac outputs, we demonstrate that using TDCO leads to an increase in false-positive EIPH diagnoses regardless of the criteria used. An important study by Hoeper et al. compared resting DFCO and TDCO in a population of 35 PH patients. 10 In agreement with the Hoeper study, our cohort demonstrated only minimal bias and good agreement at outputs <5 L/min. Marked disagreement between DFCO and TDCO in our study became apparent only under exercise conditions. Although data comparing TDCO and DFCO during stress conditions remain scant, others have demonstrated diminished agreement at high outputs in disease states such as a hypertension/heart failure population 8 and a critically ill cohort. 11 Along similar lines, our study adds to the literature by demonstrating DFCO and TDCO disagreement during stress exercise in a cohort of unexplained dyspnea, which is precisely the cohort in which EIPH is entertained.
One limitation of our study is the size and single-centered nature of this cohort. However, the implications of these data motivated us to report our findings early in the ongoing discussion of the new EIPH criteria. Second, although we maintained good quality pulse oximetry waveforms during exercise, use of pulse oximetry for DFCO calculations may underestimate directly measured arterial saturations, which could falsely elevate DFCO if arterial saturations fell markedly during exercise. Our population was free of resting hypoxemia, however, and most participants in fact had only minimal decrease (≤3% decrease) in pulse oximetry saturation during exercise. Still, we performed sensitivity analysis using values of 100% for all systemic arterial oxygen measurements (assuming that pulse oximetry if anything would underestimate true arterial saturation during exercise). This led to only one reclassification of a discordant EIPH diagnosis as concordant; furthermore, this did not change our overall statistical comparisons. Conversely, we also entertained the possibility that pulse oximetry overestimated true arterial saturation, but this would have only contributed to a greater discordance between DFCO and TDCO. Lastly, tricuspid regurgitation (TR) could lead to falsely low TDCO measures. In our cohort, however, only two participants had more than mild TR; both had concordant pressure-flow relations by both CO estimations.
To summarize, there is growing enthusiasm for two new diagnostic criteria for EIPH. The current study demonstrates that poor agreement between TDCO and DFCO at high outputs significantly affects calculations of both criteria, resulting in significantly more false positives when using TDCO. Further work in standardizing CO measurements is needed before either criterion can form the basis of a new definition of EIPH.
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
Financial support for this study comes from the National Heart, Lung, and Blood Institute (grant nos. L30 HL110304 (RJT) and T32-HL007227-40 (SH)).
