Abstract
Cardiac catheterization is important in the diagnosis and risk stratification of pulmonary hypertensive vascular disease (PHVD) in children. Acute vasoreactivity testing provides key information about management, prognosis, therapeutic strategies, and efficacy. Data obtained at cardiac catheterization continue to play an important role in determining the surgical options for children with congenital heart disease and clinical evidence of increased pulmonary vascular resistance. The Pediatric and Congenital Heart Disease Task Forces of the Pulmonary Vascular Research Institute met to develop a consensus statement regarding indications for, conduct of, acute vasoreactivity testing with, and pitfalls and risks of cardiac catheterization in children with PHVD. This document contains the essentials of those discussions to provide a rationale for the hemodynamic assessment by cardiac catheterization of children with PHVD.
Cardiac catheterization is important in the diagnosis and risk stratification of pulmonary hypertensive vascular disease (PHVD) in children. Acute vasoreactivity testing provides key information about management, prognosis, therapeutic strategies, and efficacy. Data obtained at cardiac catheterization continue to play an important role in determining the surgical options for children with congenital heart disease and clinical evidence of increased pulmonary vascular resistance. Hemodynamics (pressures, systemic and pulmonary blood flow, and vascular resistance index) obtained at cardiac catheterization relate directly to outcome. 1 Cardiac catheterization can be applied uniformly across all age groups and neurodevelopmental stages to demonstrate disease progression or regression and response to therapy. 2 Current guidelines for evaluation of pulmonary hypertension in adults 3 include recommendations for cardiac catheterization, with specific definitions, and discussion of prognostic and therapeutic implications of a positive acute vasoreactivity test. The functional classification, diagnostic spectrum, phenotype, and progression of PHVD in children differ from those in adults and have been discussed by Pulmonary Vascular Research Institute (PVRI) task forces.2,4,5 These differences directly affect the conduct of, data required for, and value of cardiac catheterization undertaken in children with disorders of the pulmonary circulation.
The aim of this document is to provide a comprehensive, globally applicable guideline for cardiac catheterization in pediatric PHVD. It reflects the ideas and experience of the Pediatric Task Forces at the sixth and seventh PVRI meetings held in Cape Town, South Africa (February 2012) and Istanbul, Turkey (January 2013). Members of the Pediatric Task Forces included pediatric specialists from cardiology, pulmonology, anesthesiology, cardiac critical care, cardiac surgery, and neonatology, with global representation from 12 countries.
DIAGNOSTIC EVALUATION BEFORE CARDIAC CATHETERIZATION
Invasive hemodynamic evaluation should, in general, be the last diagnostic procedure, and all the results of the previous tests should be integrated and reviewed when deciding the best approach to cardiac catheterization for every patient. The diagnostic algorithm for pediatric PHVD differs from that used in adult patients, and specific tests (e.g., for congenital metabolic diseases, gastroesophageal reflux disease) may be needed in the evaluation of children. At the PVRI Pediatric Task Force meeting there was general agreement that a contrast-enhanced computed tomography (CT) scan (preferably high-resolution) be undertaken whenever possible before cardiac catheterization, as it will permit a focused approach to cardiac catheterization, shorten fluoroscopy time, and limit the radiopaque contrast required. CT scanning is extremely helpful to evaluate suspected pulmonary veno-occlusive disease, pulmonary capillary hemangiomatosis, or interstitial lung disease, the exclusion or evaluation of congenital lung or heart malformations (e.g., anomalous systemic or pulmonary drainage, pulmonary sequestration, aortopulmonary collaterals, lung hypoplasia, pulmonary vein stenosis, airway malformations), and the extent of parenchymal lung disease in bronchopulmonary dysplasia (BPD). The new 320 multislice CT scanners minimize the radiation dose and the acquisition time, making sedation unnecessary. Magnetic resonance imaging (MRI) is a useful tool not only to study anatomy in congenital heart disease but also to evaluate right ventricular size and function, and it may have a role in the measurement of cardiac output, quantification of flow to each lung, or flow derived from collaterals. 6 Newer real-time imaging sequences may make it feasible in even young children without the need for sedation or general anesthetic. 7 However, it is not considered a replacement for cardiac catheterization but an extremely useful adjunct, especially in follow-up evaluations of response to treatment. 8 Needless to say, MRI with specialized evaluations for PHVD remains limited by the expense of the equipment and the scarcity of qualified persons to perform the scans and to interpret the findings.
INDICATIONS FOR CARDIAC CATHETERIZATION IN PEDIATRIC PHVD
In general, the diagnosis of PHVD will have been made or suggested on the basis of a review of the clinical and family history, electrocardiography, chest x-ray, and echocardiographic assessment. Physicians who treat children with PHVD agree that cardiac catheterization is required to confirm the diagnosis and evaluate severity and prognosis.1,9 It may be a useful adjunct to guide and assess the effects of therapy that can be applied across all ages and neurodevelopmental stages. 2 In addition, in PHVD associated with unrepaired congenital heart disease, hemodynamic evaluation by cardiac catheterization plays a key role in the assessment of suitability for shunt closure, especially in patients who present late or without clinical signs of excessive pulmonary blood flow despite an anatomically large defect or signs of right-to-left shunting. 10
There is a broad consensus that a diagnostic cardiac catheterization with acute vasoreactivity testing be undertaken at least once in any patient with important pulmonary hypertension, especially if targeted pulmonary hypertensive therapy is indicated. Exceptions to this rule include transient forms of PHVD (e.g., persistent pulmonary hypertension of the newborn), patients receiving inhaled nitric oxide (NO) therapy in the intensive care unit, and those in severe right heart failure. In very sick children or babies (Panama functional class IV, significant right ventricular dysfunction, extremely low weight, or hemodynamic instability), it may be prudent to start therapy and postpone the catheterization until the patient's clinical condition improves. 11 Risks and benefits of catheterization should be assessed in each case, and it may be appropriate in some children to treat without catheterization. In addition, catheterization may be forgone in patients with established Eisenmenger syndrome, with appropriate clinical and noninvasive diagnostic evaluations.
In general, we recommend repeating cardiac catheterization 6-12 months after the initiation of targeted PHVD therapy, before a change in therapy, and if there is any change in clinical status that requires further clarification. However, each individual case deserves careful scrutiny, as well as consideration of the institutional experience, to deal with complications and adverse events. Parental attitude, the assent of the child or adolescent, and the financial burden of repeat cardiac catheterization should be taken into account in different settings and countries.
GENERAL ANESTHESIA VERSUS SEDATION
Optimally, catheterization to obtain basal measurements of pulmonary pressures, resistances, and cardiac output would be performed in an awake, but relaxed, cooperative child, using only local anesthesia. Unfortunately, this is not possible in the majority of patients under 12 years of age, and in older patients the neurodevelopmental maturity of the child will determine the degree of cooperation. Therefore, most catheterizations are performed either under general anesthesia with endotracheal intubation (or laryngeal mask airway) and mechanical ventilation or with sedation, allowing the patient to breathe spontaneously. For patients being submitted for their first diagnostic catheterization, the decision to withdraw pulmonary hypertension-specific therapy before the catheterization in order to obtain “basal measurements” should be taken on a case-by-case basis. In patients undergoing follow-up cardiac catheterization to assess the effects of therapy or the need for additional therapy, we recommend that specific drug therapy should be continued according to the outpatient schedule, even during the period of fasting before the procedure. 11
We recommend the presence of an anesthesiologist familiar with the management of neonatal and childhood PHVD when either sedation or general anesthesia is used. Spontaneous breathing with sedation has the potential risk of respiratory depression and hypercapnia, which may alter pulmonary artery pressures and resistances. End-tidal CO2 or partial pressure of CO2 in arterial blood by blood gas analysis should be monitored closely, and an attempt should be made to mirror the normal cardiopulmonary state of the patient. This is particularly important in children with BPD who may have chronic CO2 retention.
General anesthesia and mechanical ventilation provide stable conditions and adequate analgesia and avoid hypercarbia and hypoxia but require the availability of a trained pediatric anesthesiologist with experience in the management of children with cardiopulmonary disease. Nevertheless, it also has its disadvantages: general anesthesia may modify myocardial performance and lower systemic arterial pressure and vascular resistances. The serious-complication rates of cardiac catheterization in children with pulmonary hypertension range from 2% to 6%, and mortality ranges from 0% to 1.4%.9,11–18 Complications are related to hypoxemia, hypercapnia, acidemia, hypothermia, tachycardia, or systemic hypotension. Children under 2 years of age may have a 3-fold increased risk of the complications from cardiac catheterization. 17
The risks of general anesthesia, sedation, and cardiac catheterization are predictably increased in certain groups. A profile of a patient who requires careful evaluation by the team would include new diagnosis of PHVD, treatment naivete, advanced functional class (III/IV), right ventricular dysfunction, suprasytemic pulmonary artery (PA) pressure, age younger than 3 years, left ventricular systolic dysfunction, or restrictive cardiomyopathy. 11
We recommend highly that there be discussion between the pediatric anesthesiologist, the pediatric pulmonary hypertension expert, and the cardiac catheterization team before cardiac catheterization to carefully evaluate the best approach to obtain the required data. Specific recommendations for anesthetizing patients with pulmonary hypertension have been published but will have to be modified, depending on the child, the local experience, and the resources available. 19 Clearly vulnerable moments for the patient are induction and emergence from anesthesia and procedural events associated with systemic hypotension that render the right ventricle performing at suprasystemic pressures especially prone to ischemia. 20 Careful postcatheterization monitoring is important, and consideration of overnight admission to an intensive care unit may be prudent for patients with severe right ventricular or biventricular dysfunction.
CONDUCT OF THE CARDIAC CATHETERIZATION
In general, the conduct of the cardiac catheterization will depend on whether the child has a shunt, which will determine the information required to calculate flow and resistance. In addition, there is usually a requirement for hemodynamic evaluation under basal conditions (normal systemic arterial pH and CO2, normoxia) and assessment of the hemodynamic change after administration of a pulmonary vasodilator or temporary occlusion of a shunt.
The Fick principle is beguilingly simple and states that blood flow is proportional to the oxygen consumption divided by the extraction of oxygen across the same vascular bed. However, obtaining the information required to calculate flow is subject to multiple difficulties and errors and if incompletely understood leads to unreliable data, and therefore calculations of flow and resistance always should be interpreted as estimations within the context of the procedure. 21
MEASUREMENT OF OXYGEN CONSUMPTION
There have been many publications attesting to the unreliability of predicting oxygen consumption with tables and formulas.22–28 However, there is no efficient, accurate, inexpensive, reliable, and user-friendly method to measure oxygen consumption (
Often,
The expression of the flows and resistances as a ratio is a mathematical ploy to minimize the effect of oxygen consumption. However, one should be cognizant that interventions such as hyperoxia may increase systemic vascular resistance and decrease the pulmonary vascular-to-systemic resistance ratio (PVR : SVR, or Rp: Rs), without necessarily affecting PVRI (PVR index) or pulmonary resistance.32–34
BLOOD SAMPLING TO MEASURE OXYGEN SATURATIONS
In general, blood sampling to measure the oxygen saturation for the calculation of flow should include samples proximal and distal to the shunt. Formulas for the calculation of a mixed venous sample from samples in the superior vena cava (SVC) and inferior vena cava in subjects with a shunt are available. 21 In general, to save time and blood, a sample in the SVC can be compared with one from the PA to detect the presence of a shunt and to calculate systemic and pulmonary blood flow. In practice, a step-up in oxygen saturation from the SVC to the PA of 3% or greater is likely to indicate the presence of a shunt. However, it should be remembered that a 6% saturation difference between the SVC and the PA or a 4% saturation difference between the right atrium or right ventricle and the PA may be a false-positive step up (i.e., a shunt is thought to be present when none in fact exists) in 5% of patients. 35 Most sources of error are related to sampling before complete mixing has occurred and to changes in physiologic state between sampling in different sites (Table 1).
Causes of inaccuracy in flow and resistance calculations

Note: If assumptions are made, always consider best and worst scenario in calculations.
Less common sources of error related to blood sampling may include failure to detect an anomalous pulmonary vein to the SVC or an innominate vein or differential saturations in the right and left PA in the presence of PDA or aortopulmonary collaterals, or failure to sample before and after the PDA in the aorta. Errors may be incurred by failure to recognize pulmonary vein desaturation or to assume that aortic desaturation is due to a right-to-left intracardiac shunt.
When there are multiple sources of pulmonary blood flow, for example, aortopulmonary collaterals after the Fontan operation, the collection of truly mixed PA saturation is difficult because the collaterals enter the pulmonary circulation distally. MRI may provide a better way of quantifying pulmonary and systemic blood flow in the evaluation of patients after the Fontan or cavopulmonary anastomoses. 6
HEMOGLOBIN
Changes in hemoglobin may affect pulmonary vascular resistance, not only through changes in viscosity but also because hemoglobin is integral to the Fick equation. 36 The shunt across a ventricular septal defect (VSD) is increased if the hemoglobin is low and decreased if, in the same patient, the hemoglobin is increased. 37
In patients without cardiac or systemic-to-pulmonary shunts, the pulmonary and systemic blood flows are considered equivalent. Thermodilution catheters in the PA simplify the measurement of blood flow and calculation of resistance.
38
If thermodilution catheters are unavailable, then clearly only pulmonary and systemic artery saturations, hemoglobin, and
ANATOMICAL CONSIDERATIONS IN THE CALCULATION OF FLOW AND RESISTANCE
In some situations, especially if blood flow and pressure are unequally distributed between the lungs, it may be difficult to estimate the total PVRI in a meaningful way. Examples of these situations include unequal distribution of pulmonary blood flow (e.g., hypoplasia of one lung) and unequal distribution of flow with different pressures in multiple lung segments (e.g., peripheral PA stenosis or after PA unifocalization for pulmonary atresia with VSD and major aortopulmonary collaterals). In severe pulmonary valve regurgitation after the insertion of a right ventricle-to-PA conduit, it may not be possible to calculate the PVRI accurately because the diastolic pressure in the PA approximates the right ventricular end-diastolic pressure. Thus, the mean PA pressure will be decreased and lead to calculation of a PVRI lower than would be found if the pulmonary valve were competent. In these cases, careful evaluation of the individual case and integration of the information provided by angiography, MRI, or CT and lung perfusion scan are helpful. Furthermore, surgical decision making in these cases, especially if there is a shunt in the presence of unilateral lung hypoplasia, may be quite complex. 39
MEASUREMENT OF THE PA CAPACITANCE INDEX
The PA capacitance index (PACi) can be derived from routine information collected at cardiac catheterization and may offer insights into static right ventricle-to-PA coupling, which has been reported to be an important determinant of outcome in children with pulmonary vascular disease.40,41 The PACi can be calculated with the following equation:
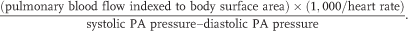
PACi values less than 0.7–0.9 mL/mmHg/m2 have been suggested as predictors of poorer prognosis in children with PHVD.
PA distensibility 42 and studies of pulmonary flow and flow reserve may be included if an intravascular Doppler catheter is available or transthoracic echocardiography is available for use in the cardiac catheterization laboratory. 43 Pulmonary flow reserve of less than 1.4 is highly predictive of adverse events in children with idiopathic pulmonary arterial hypertension (IPAH). 43
ACUTE VASOREACTIVITY TESTING
In many institutions around the world, out of necessity, breathing 100% oxygen is used for pulmonary vasoreactivity testing.
44
However, there remains controversy about how well the response to breathing Fio2 of 1.0 predicts operability.44,45 The disadvantages of 100% oxygen are that it may decrease the accuracy of flows calculated by the Fick equation because the small arteriovenous O2 difference magnifies any saturation error, and calculated shunts will tend to be larger and resistances lower as a result. The
The combination of oxygen and inhaled NO12,46,47 may identify patients with significant pulmonary vasoreactivity who might not be recognized if O2 or NO were used separately. A combination of 40% O2 with 20–40 ppm inhaled NO seems to work well, as
Other agents that may be used for acute vasoreactivity testing include intravenous epoprostenol48,49 and adenosine,50,51 but they may result in systemic hypotension and have not been described well in children. Adenosine frequently causes side effects in comparison with inhaled NO. 51 Delivering pulmonary vasodilator drugs by inhalation or nebulization may promote pulmonary vascular selectivity and minimize adverse systemic reactions. Inhaled iloprost,52,53 treprostinil, 54 nitroglycerin, and milrinone 55 have all been reported in children undergoing cardiac catheterization. Nevertheless, NO, if available was considered the current standard for acute vasoreactivity testing by the task force. Table 2 lists the drug doses recommended for acute vasoreactivity testing.
Agents used for acute pulmonary vasoreactivity testing in children

Both inhaled nitroglycerine and milrinone are prepared by mixing the drug with normal saline to make a volume of 3 mL and nebulized over 10 minutes with a jet nebulizer using 8 L/min of 50% air-oxygen mixture.
Adenosine use is controversial, and there is a lack of information reporting its use in children.
Acute vasoreactivity testing in patients with systemic-to-pulmonary shunt lesions
In patients with pre- and posttricuspid systemic-to-pulmonary shunts, there is a general consensus that a decrease in PVRI below 4 Wood units (WU)·m2 and/or in the ratio of pulmonary:systemic vascular resistance (PVRI:SVRI [SVR index]) below 0.3 indicates that shunt closure may be undertaken safely with a good long-term prognosis. If the PVRI is between 4 and 8 WU·m2, temporary balloon occlusion of the shunt, if possible, may provide additional useful information. 56 In some centers, a decrease in PVRI to less than 6–8 WU·m2 may be used to undertake shunt closure, but it is emphasized that the individual center experience and resources for postoperative care, both short and long term, are important considerations.57–59 The decision to close a shunt is made using all the available information and is not solely dependent on the hemodynamics obtained at cardiac catheterization.10,56,59 Different criteria may be used, especially in older patients, to decide whether atrial septal defects ought to be closed, because a number of different factors affect atrial-level shunting and streaming that may be unrelated to pulmonary vascular resistance.60–62
Acute vasoreactivity testing in patients without significant systemic-to-pulmonary shunts
In this subset of patients (usually with idiopathic PHVD, following repair of congenital heart disease, or with pulmonary hypertension due to left heart disease), cardiac output may be measured with thermodilution or the Fick principle. After a baseline evaluation of hemodynamics, acute vasoreactivity testing is suggested in patients with presumed idiopathic PHVD to identify those patients older than 1 year of age who may benefit from treatment with a calcium channel blocker. 1 The Sitbon 63 criterion for a positive response (decrease in mean pulmonary arterial pressure of >10 mmHg reaching <40 mmHg, with an increase or no change in cardiac output) is not always applicable to children, as some will have mean pulmonary arterial pressure lower than 40 mmHg even in the presence of severe PHVD. Barst64,65 proposed a specific definition of a positive test for children: decrease in mean PA pressure of >20%, an increase or no change in cardiac index, and a decrease or no change in PVRI:SVRI. This definition of acute vasoreactivity correctly identified children in the REVEAL registry who reacted well to long-term calcium channel blocker therapy, and none of these 22 children treated with calcium channel blocker therapy, as either mono or combination therapy, died or underwent transplantation within 5 years of follow-up. 65 However, the Barst criteria were not associated with prolonged survival or continued response to calcium channel blockers in the pediatric IPAH UK registry. The authors of the pediatric IPAH UK registry suggested that, provided that the PVRI decreased to 4.5 WU·m2 or less, the long-term response to calcium channel blockers was good. They noted that the condition of 2 of 5 children with a PVRI > 5 WU·m2 deteriorated and suggested that stricter acute vasoreactivity criteria be used to predict long-term response with near normalization of PA pressures and resistance. 66 In other published registries or cohorts, the reported proportion of children with IPAH who were acute responders has been quite variable: 17%–20% in the Van Loon et al. 67 series, 11% in the French registry, 68 6% in the Spanish registry, 69 and 9% in the UK registry. 66 Differences in the patient population included in these series are probably responsible for the variations in the proportion of responders. Thus, for children with idiopathic PHVD, the task force recommends that strict criteria with near-normalization of PA pressure, as suggested by Sitbon, 63 and PVRI < 4.5 WU·m2, as suggested by Moledina, 66 be used to define an acute responder. It is prudent to exclude, as far as possible, the diagnosis of pulmonary veno-occlusive disease before undertaking acute pulmonary vasoreactivity trials, because these patients have a variable response to pulmonary vasodilation but may respond with severe, sometimes fatal, pulmonary edema.
The evaluation of patients with systolic dysfunction of the left ventricle may include an infusion of dobutamine, milrinone, or sodium nitroprusside to decrease the LV end-diastolic pressure and to increase the cardiac output; this may result in a decrease in PVRI below 4 WU·m2; if PVRI remains higher than 4–6 WU·m2 and the transpulmonary gradient greater than 15 mmHg, vasoreactivity testing with inhaled NO may be used, but with caution, because of the risk of inducing pulmonary edema. However, in our experience, the use of inhaled NO in patients with cardiomyopathy is generally well tolerated even if there is an increase in left ventricular end-diastolic or PA wedge pressure. There is considerable interest in using the diastolic pressure gradient between the PA pressure and the LV end-diastolic pressure or the wedge pressure to assess the variable contribution of pulmonary arterial vascular changes in left heart dysfunction.70,71
In patients with postoperative PHVD or Eisenmenger syndrome, the role of vasoreactivity testing is less clear, because very few patients have been treated with calcium channel blockers. 72 However, response to inhaled NO suggests a better prognosis and response to therapy in established Eisenmenger syndrome.73–75 If patients with repaired congenital heart disease undergo cardiac catheterization to evaluate pulmonary hypertension, then it is important to assess for residual shunts, diastolic dysfunction, and pericardial disease, as they may contribute to pulmonary hypertension.
ANGIOGRAPHY
Contrast angiography is not without risk in children with PHVD. We suggest that all data be reviewed before angiography. Evaluation by echocardiography (transthoracic and transesophageal), contrast high-resolution CT, and MRI has significantly reduced the need for routine angiography. Angiography should be performed judiciously and is necessary only to answer specific questions or if there is doubt about interpretation of other imaging modalities.
Although not performed routinely, balloon occlusion pulmonary wedge angiography may provide information about the severity of pulmonary vascular disease76,77 and provides excellent visualization of the pulmonary veins. It also allows for the identification of segmental and subsegmental pulmonary arterial branch stenoses and may be useful in detecting pulmonary arteriovenous malformations. It is usually performed in both lungs, preferably in the posterobasal segment artery of the right or left lower lobe (1 rib space below the takeoff of the right PA or 2 rib spaces below the takeoff of the left PA). Once the balloon is inflated, contrast is injected at 0.3 mL/kg (minimum 2 mL) at a flow rate of 5 mL/s. We use layered contrast with saline to wash the contrast through the vascular bed.
CONCLUSIONS
Cardiac catheterization plays an important role in the diagnosis and evaluation of PHVD in children. However, the need for cardiac catheterization should always be considered carefully, as the risks involved are higher than those in adults.
