Abstract
Our objective was to determine whether left ventricular (LV) vorticity (ω), the local spinning motion of a fluid element, correlated with markers of ventricular interdependency in pulmonary hypertension (PH). Maladaptive ventricular interdependency is associated with interventricular septal shift, impaired LV performance, and poor outcomes in PH patients, yet the pathophysiologic mechanisms underlying fluid-structure interactions in ventricular interdependency are incompletely understood. Because conformational changes in chamber geometry affect blood flow formations and dynamics, LV ω may be a marker of LV-RV (right ventricular) interactions in PH. Echocardiography was performed for 13 PH patients and 10 controls for assessment of interdependency markers, including eccentricity index (EI), and biventricular diastolic dysfunction, including mitral valve (MV) and tricuspid valve (TV) early and late velocities (E and A, respectively) as well as MV septal and lateral early tissue Doppler velocities (e′). Same-day 4-dimensional cardiac magnetic resonance was performed for LV E (early)-wave ω measurement. LV E-wave ω was significantly decreased in PH patients (P = 0.008) and correlated with diastolic EI (Rho = −0.53, P = 0.009) as well as with markers of LV diastolic dysfunction, including MV E(Rho = 0.53, P = 0.011), E/A (Rho = 0.56, P = 0.007), septal e′ (Rho = 0.63, P = 0.001), and lateral e′ (Rho = 0.57, P = 0.007). Furthermore, LV E-wave ω was associated with indices of RV diastolic dysfunction, including TV e′ (Rho = 0.52, P = 0.012) and TV E/A (Rho = 0.53, P = 0.009). LV E-wave ω is decreased in PH and correlated with multiple echocardiographic markers of ventricular interdependency. LV ω may be a novel marker for fluid-tissue biomechanical interactions in LV-RV interdependency.
Pulmonary hypertension (PH) is a disease of chronically increased right ventricular (RV) afterload that results in alterations in RV hypertrophy, volume, and contractility. 1 Because the right ventricle (also RV) and left ventricle (LV) share a circulatory system, interventricular septum, and pericardium, the RV pressure and/or volume overload associated with PH can result in RV dilatation, leftward shift of the interventricular septum, and changes in left ventricular (also LV) geometry. 2 These direct interactions between the RV and the LV, referred to as ventricular interdependency, can result in decreased LV distensibility, preload, and elastance, as well as increased LV end-diastolic pressure, leading to LV diastolic dysfunction.3–5 Specifically, PH is associated with echocardiographic markers of diastolic dysfunction, including reduced transmitral early (E-wave) velocity as well as reduced early septal and lateral tissue Doppler velocities (e′).6,7 Ventricular interdependence can be assessed via cardiac imaging using both the eccentricity index (EI), a measurement of septal curvature that directly reflects interventricular pressure gradient and interdependency, and indices of LV diastolic function.8,9 However, prior studies that have validated the use of EI and LV diastology as markers of ventricular interdependence have typically included more advanced forms of PH. Given its significant prognostic value, identifying a novel marker of ventricular interdependence in earlier-stage disease is of clinical relevance. 10
The development of interventricular septal shift in association with LV diastolic dysfunction raises the possibility that alterations in intraventricular complex fluid dynamics may serve as a mediator and/or a marker of ventricular interdependency. Large-scale vortical ring formations develop in the LV during diastole that have altered formation times, position, and circulation in diastolic dysfunction.11–13 Consequently, altered vortex properties may be present before significant changes occur in ventricular morphology or function. 14 Vorticity (ω), the local spinning motion of an element of fluid, represents a parallel quantitative measure of flow and its spatial distribution at the fluid-tissue interface. Four-dimensional (4D) flow cardiac magnetic resonance imaging (CMR) utilizes time-averaged phase-velocity-encoded acquisition in the X-, Y-, and Z-planes and can generate high-fidelity spatial and temporal renderings of these complex flow patterns while offering a novel method for quantifying ω. Using a same-day 4D CMR and echocardiography in a cohort of controls and PH patients, we aimed to determine whether LV ω correlated with echocardiographic indices of ventricular interdependence.
METHODS
Study Population
This prospective study was approved by the National Jewish Health (NJH) Institutional Review Board, and all subjects gave written informed consent. PH patients were consecutively enrolled from the NJH PH clinic. To remove confounding causes of altered loading conditions and biventricular dysfunction, all subjects were 18 years or older and without known cardiomyopathy, atrial fibrillation, coronary artery disease (history of myocardial infarction; prior mechanical revascularization, including percutaneous coronary intervention or coronary artery bypass surgery; or known coronary artery stenosis > 50%), moderate or worse valvular heart disease, or advanced liver disease. In addition, control subjects were asymptomatic, without known cardiac disease, lung disease, or smoking history. Before enrollment, all PH patients had right heart catheterization (RHC)-proven PH, as per World Health Organization criteria. 1 Mean pulmonary artery pressure (mPAP), pulmonary artery wedge pressure (PAWP), and pulmonary vascular resistance (PVR) were measured/calculated during RHC. RHC was not performed on controls because of ethical considerations.
2-dimensional (2D) and Doppler echocardiography
Data acquisition was performed with a Vivid 7 ultrasound system (General Electric Medical Systems, Milwaukee) equipped with a 3-MHz transducer. Pulsed tissue Doppler imaging (TDI) recordings were acquired from an apical 4-chamber view during a short end-expiration pause. Pulsed-wave Doppler echocardiography was used to obtain biventricular diastolic parameters, including mitral valve (MV) and tricuspid valve (TV) early and late filling peak velocities (E and A, respectively), E/A, and E-wave deceleration time (DT). Diastolic e′ and a′ (late) velocities were obtained with pulsed TDI for both the MV (septal and lateral annulus) and the TV (lateral only). RV isovolumic relaxation time (IVRT) was also measured via tissue Doppler tracings. LV and RV diastolic dysfunction was staged as per American Society of Echocardiography guidelines.15,16 RV systolic pressure (RVSP) was estimated with the tricuspid regurgitant envelope method. 17
2D CMR
CMR was performed on the same day as echocardiography. Subjects underwent CMR imaging in the supine position with a 1.5-T clinical magnetic resonance imaging system (MAGNETOM Avanto, Siemens Healthcare Sector, Erlangen, Germany) and an 8-channel phased array cardiac coil. A cine steady-state free-precession technique with retrospective gating was used to image from the base to apex during brief end-expiratory breath-holds with contiguous short-axis slices in 8-mm increments. Ventricular volumetric and functional analyses were performed off-line by a blinded reader using commercially available software (Syngo Argus, MR B17, Siemens). LV and RV end-diastolic volumes (LVEDV and RVEDV, respectively), as well as end-systolic volumes (LVESV and RVESV, respectively), were determined according to the modified Simpson rule. LV and RV ejection fraction (LVEF and RVEF, respectively) and LV stroke volume (LV SV), cardiac output (LV CO), and cardiac index (LV CI) were also calculated. LV EI was measured during maximum ventricular diastole and systole as described previously. 8 Left atrial (LA) volume, right atrial volume, and LA EF were calculated with previously described methods.18,19
4D flow CMR visualization and ω analysis
The 4D flow CMR was performed with interleaved 3-directional velocity encoding (voxel volume: 11.7–20.3 mm3, α: 15°, echo time: 2.85 ms, repetition time: 48.56 ms, velocity encoding: 100 cm/s, temporal resolution: 50 ms). The 4D flow images were acquired with a radio frequency–spoiled gradient echo pulse sequence, prospective electrocardiographic gating, and respiratory navigators, using bellows as previously described. 20 Raw 4D CMR phase-contrast data sets were filtered for noise with a magnitude mask, antialiased with a custom MATLAB program, and converted to Ensight data format with the Velomap software package. 21 ParaView (Kitware, Clifton, NY) was utilized for visualization and ω quantification. LV ω vectors were thresholded to remove low- and high-magnitude regions (0.03–0.7 s−1). The LV region of interest was selected by super-imposition of 2D CMR images on ω maps, and ω was then calculated for the entire-LV volumes for all subjects at each diastolic time step. LV E- and A (late)-wave ω were computed at the time steps corresponding to maximal MV E- and A-wave velocity. Blood flow topologies were analyzed visually with ω-vector glyphs, velocity vector glyphs, and streamlines and assessed for the presence of vortical motion in the LV. An LV vortex was defined as rotational blood flow where closed streamlines were observed. 22 Measurements of ω were obtained by two independent readers in blinded fashion.
Laboratory diagnostics
Serum was collected from all subjects via phlebotomy for brain natriuretic peptide (BNP) and creatinine measurements immediately before echocardiography and CMR. After collection and centrifugation, samples were aliquotted and stored at −20°C, as per manufacturer's instructions for use. Samples were sent to a core laboratory facility for analysis, run in triplicate, and averaged by a single blinded operator. Glomerular filtration rate (GFR) was calculated with the Modification of Diet in Renal Disease Study equation. 23
Statistical analysis
Where appropriate, either a 2-tailed Student t test or the Wilcoxon rank-sums method was used for intervariable comparisons PH and control groups. Values are reported as either mean ± 1 standard deviation or median with 25% and 75% interquartile ranges. Spearman Rho correlation coefficients were used to test the relationship between ω and continuous variables in JMP, version 9.0 (SAS Institute, Cary, NC). Statistical significance was defined as P < .05. The Shrout-Fleiss intraclass correlation coefficient (ICC) was utilized to assess for interobserver variability.24(p7)
RESULTS
Characterization of LV and RV morphology and function
A total of 13 PH patients were enrolled, including 9 idiopathic PH and 4 scleroderma-associated PH patients. Ten age-matched controls were enrolled. There were no significant differences in age, body mass index, or sex between the control and PH cohorts (see Table 1). In addition, there were also no differences in LV volumes, EF, CI, or SV As anticipated, the PH cohort had significantly increased RVSP, RVEDV, and RVESV and decreased RVEF and LA EF compared to controls. Pharmacologic therapies in the PH cohort at the time of protocol performance included sildenafil (N = 6), tadalafil (N = 1), and ambrisentan (N = 1). The remaining 5 PH patients were diagnosed with PH at the time of RHC and were not treated with PH pharmacologic therapy at the time of enrollment. RHC was performed on all PH patients within 152 ± 223 days before enrollment for clinical indications. Right heart pressures in the PH cohort were moderately elevated, with a mean mPAP of 43.4 ± 14.5 mmHg and mean PVR of 9.1 ± 5.8 Wood units. BNP was significantly elevated in the PH cohort relative to that in controls, but there was no difference in GFR.
Demographic, biometric, and chamber size/function for control and PH cohorts

Note: Unless otherwise noted, data are presented as median (interquartile range). BMI: body mass index; HR: heart rate; SBP: systolic blood pressure; SD: standard deviation; DBP: diastolic blood pressure; GFR: glomerular filtration rate; BNP: brain natriuretic peptide; mPAP: mean pulmonary artery pressure; PAWP: pulmonary artery wedge pressure; PVR: pulmonary vascular resistance; RVSP: right ventricular systolic pressure; RVEF: right ventricular ejection fraction; RVEDV: right ventricular end-diastolic volume; RVESV: right ventricular end-systolic volume; RA: right atrium; LVEF: left ventricular ejection fraction; LVEDV: left ventricular end-diastolic volume; LVESV: left ventricular end-systolic volume; LV SV: left ventricular stroke volume; LV CI: left ventricular cardiac index; LA: left atrium; EF: ejection fraction; NS: not significant.
Although transmitral and tissue Doppler parameters demonstrated normal LV and RV diastolic function in all controls, most PH patients had either stage I LV diastolic dysfunction (N = 10) or stage II LV diastolic dysfunction (N = 2), with only 1 subject having normal LV diastolic function (N = 1). Consistent with the presence of LV diastolic dysfunction, the PH cohort had significantly increased A-wave velocity and decreased E/A compared to controls (see Table 2). Accordingly, the PH cohort demonstrated significantly reduced MV septal and lateral e′, reduced MV lateral a′ velocity, and increased MV E/e′. There was no difference in MV E, septal a′, or DT. All PH subjects had either stage I RV diastolic dysfunction (N = 6) or stage II RV diastolic dysfunction (N = 7). As expected, the PH cohort demonstrated significantly decreased TV A, TV E/A, TV e′, and RV IVRT and increased TV E/e′. There was no difference noted in TV E, TV a′, or TV DT.
LV diastolic parameters for control and PH cohorts
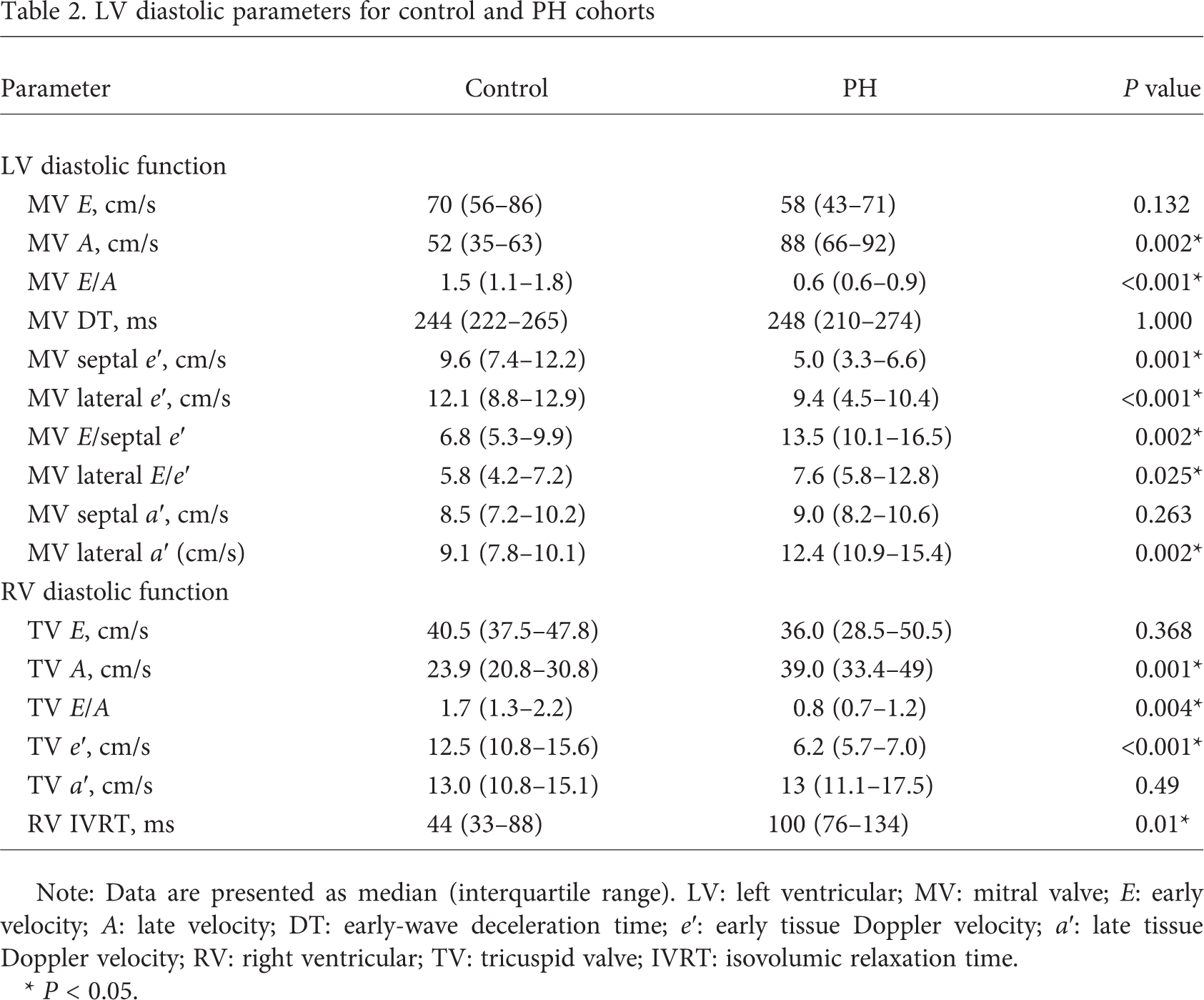
Note: Data are presented as median (interquartile range). LV: left ventricular; MV: mitral valve; E: early velocity; A: late velocity; DT: early-wave deceleration time; e′: early tissue Doppler velocity; a′: late tissue Doppler velocity; RV: right ventricular; TV: tricuspid valve; IVRT: isovolumic relaxation time.
P < 0.05.
Vorticity analysis
Temporal resolution for each 4D CMR acquisition was 50 ms. The number of acquired frames per cardiac cycle was dependent on heart rate and varied from 10 to 30 frames, with an average of 17 ± 5 frames. Average voxel volume for the cohort was 18.0 ± 3.0 mm3.
We first assessed for qualitative differences in large-scale intracavitary blood formations between controls and PH patients. Mapping of E-wave diastolic MV inflow using velocity glyphs revealed a clockwise recirculation immediately distal to the anterior MV leaflet (AMVL) near the septal wall that was visualized in all controls but not in PH patients (see Fig. 1). Streamline visualization of this AMVL recirculation revealed a clockwise vortex that was present in all controls (N = 10) and absent in all but 1 PH patient (N = 12; see Fig. 2). We then sought to characterize ω patterns in the region of the observed AMVL vortex, using ω-glyph mapping. During the E-wave phase of diastole, a region of high ω was visualized forming a complete loop around MV inflow at the level of the MV leaflets (see Fig. 3). However, PH subjects demonstrated a less concentrated region of ω glyphs around the MV inflow. Conversely, PH patients demonstrated a region of increased peak A-wave ω that circumscribed the MV leaflets but was absent in controls.
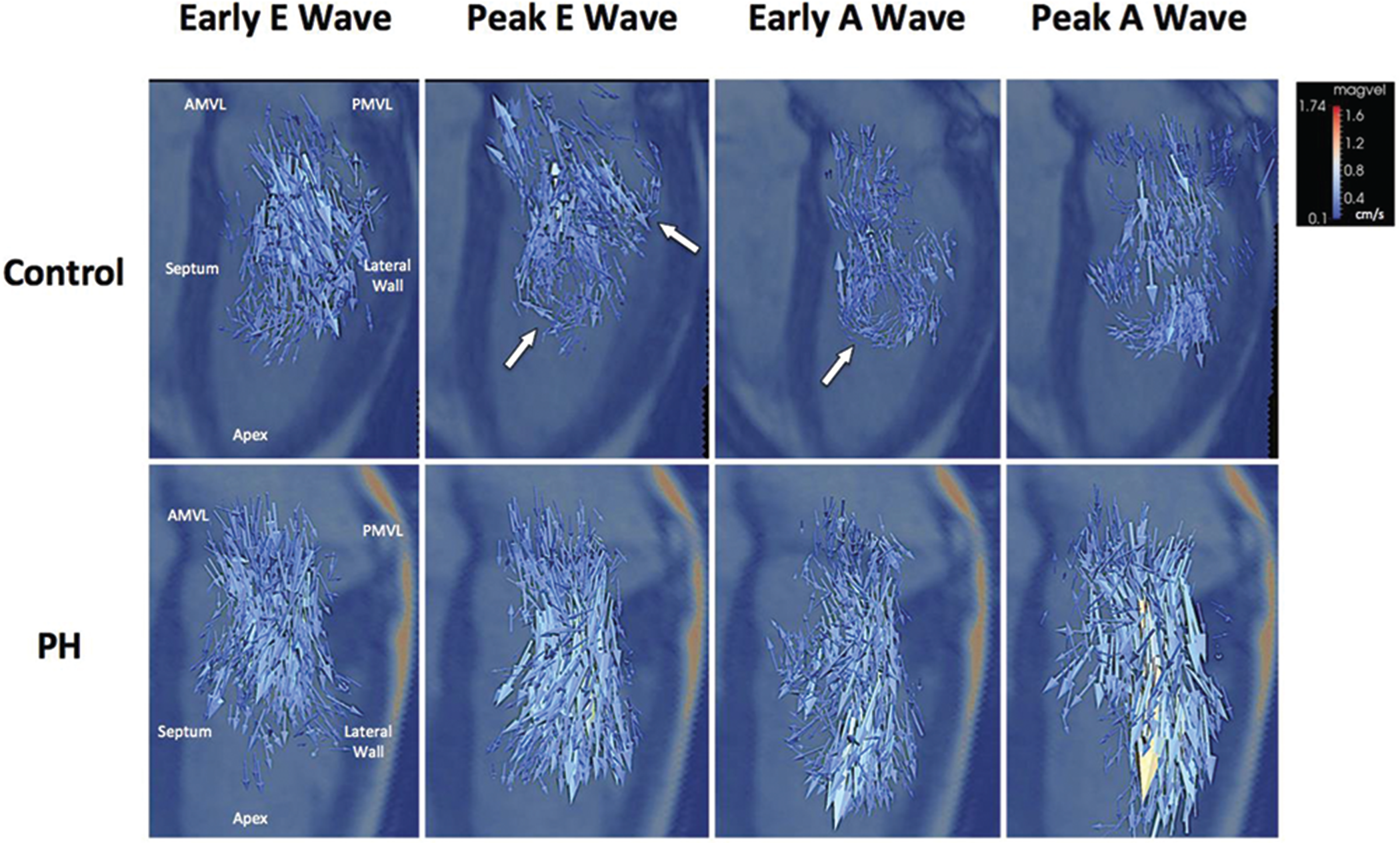
In situ visualization of the left ventricular (LV) diastolic flow using velocity-vector glyphs in controls (top) and pulmonary hypertension (PH) patients (bottom; LV base at top of image, LV apex at bottom). Note the recirculation (arrows) around the anterior mitral valve leaflet (AMVL; left) and the posterior mitral valve leaflet (PMVL; right) during peak E-wave filling/early A-wave filling in the control subject that is absent in the PH patient. Glyph size and color (see key) are proportional to velocity magnitude.
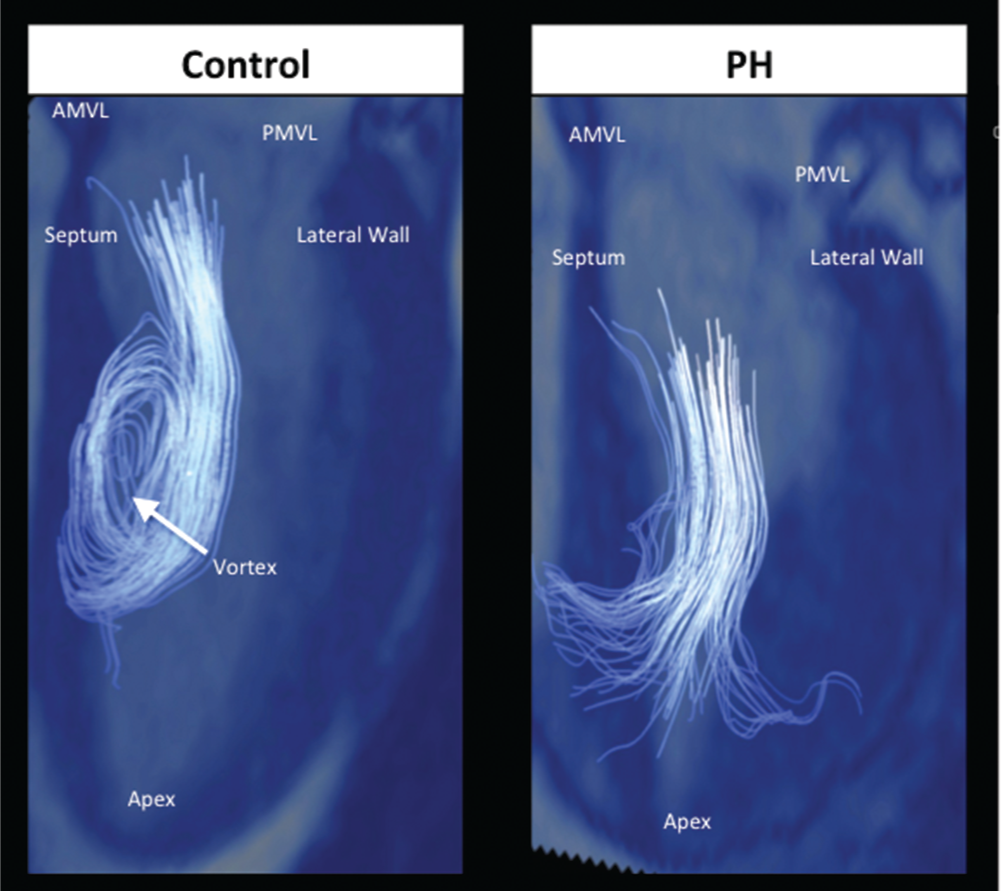
Streamline visualization of flow emanating from the mitral valve into the left ventricle during early diastole superimposed on 4-chamber steady-state free-precessing image. Streamlines are color coded (blue to red) according to velocity magnitude (see key in Fig. 1).
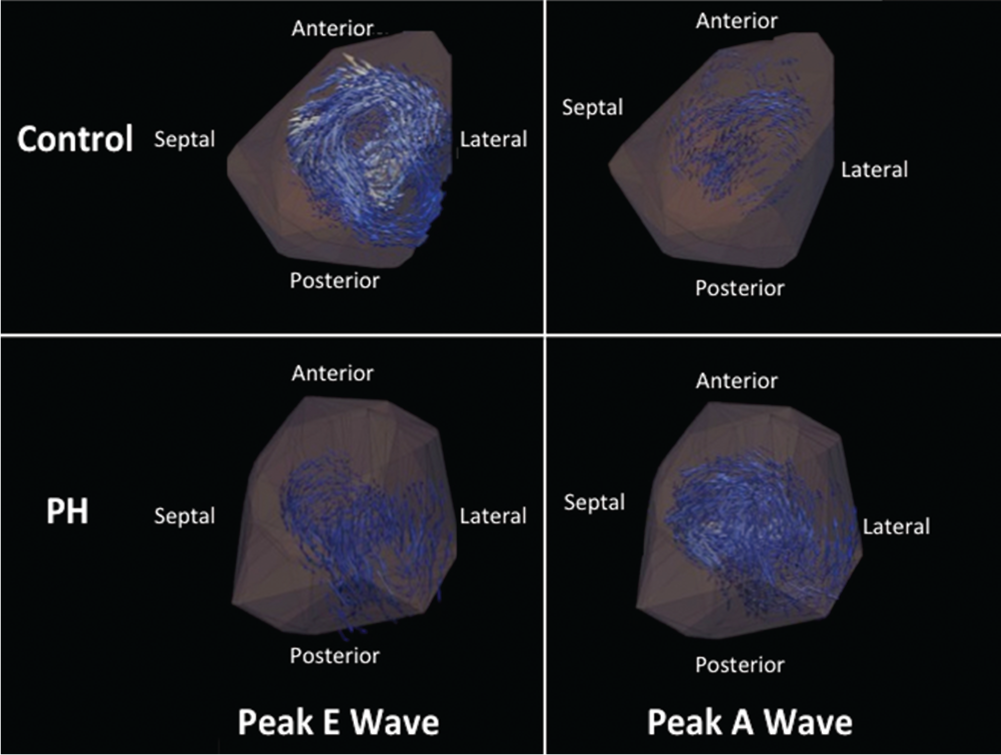
Representative images of vorticity vectors (blue/white glyphs) in the left ventricle (LV; gray mask) of a control subject (top) and a pulmonary hypertension (PH) patient (bottom) during peak E- and A-wave phases of diastole, viewed from a short-axis orientation (LV apex in foreground, mitral valve [MV] annulus in background). Depicted vorticity glyphs represent values between 0.03 and 0.7 s−1. Note the region of high vorticity forming a complete loop around the MV inflow in the control during peak E-wave filling that is not evident in the PH patient during the same time period.
On the basis of these qualitative differences, we then sought to quantitate diastolic-phase-specific ω. LV E-wave ω was found to be significantly reduced in PH subjects compared to controls (577 [168–769] vs. 988 [717–1,264] s−1, P = 0.008; see Fig. 4). However, there was no difference in LV A-wave ω between PH and controls cohorts (1,031 [619–1,401] vs. 640 [120–1,484] s−1, P = 0.227). Interobserver-agreement analysis for ω calculation used the Fleiss ICC method and demonstrated an ICC value of 0.94 for LV E-wave ω and 0.93 for LV A-wave ω, both of which are considered excellent agreement.
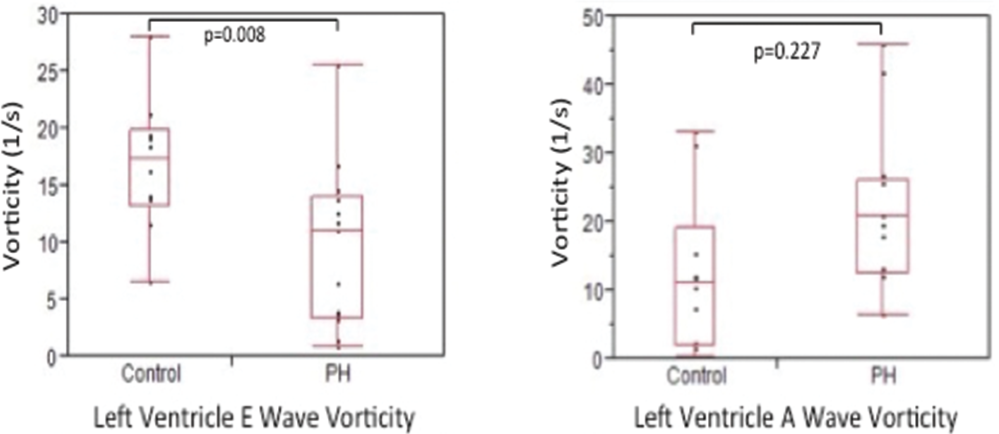
Left ventricular E- and A-wave vorticity in controls and pulmonary hypertension (PH) patients. Box plots display the distribution of data including the minimum (bottom whisker), first quartile (bottom of rectangle), median (horizontal line within rectangle), third quartile (top of rectangle), and maximum (top whisker).
Because ω generation is dependent on surrounding surface area, we tested the relationship between left heart ω and chamber volume. No significant relationship was identified between any diastolic phase of LV ω and LV chamber size, either LVEDV or LVESV. Furthermore, there was no correlation between LV ω and LA volume. Because MV inflow rate was anticipated to affect ω, correlations were performed between ω and CO as well as between ω and CI. However, no significant relationship was identified between any measure of LV ω and either CO or CI. LV SV demonstrated a significant positive correlation with LV E-wave ω (Rho = 0.53, P = 0.01) but not with LV A-wave ω. Finally, since ω should be a function of LV relaxation properties, we tested the relationship between ω and parameters of LV diastolic function. LV E-wave ω correlated with septal and lateral e′, MV E, and MV E/A (see Table 3). Similarly, LV A-wave ω was significantly associated with both septal and lateral a′ as well as with MV A.
Correlations between LV and LA E-wave vorticity (ω) and individual parameters of LV and RV diastolic function
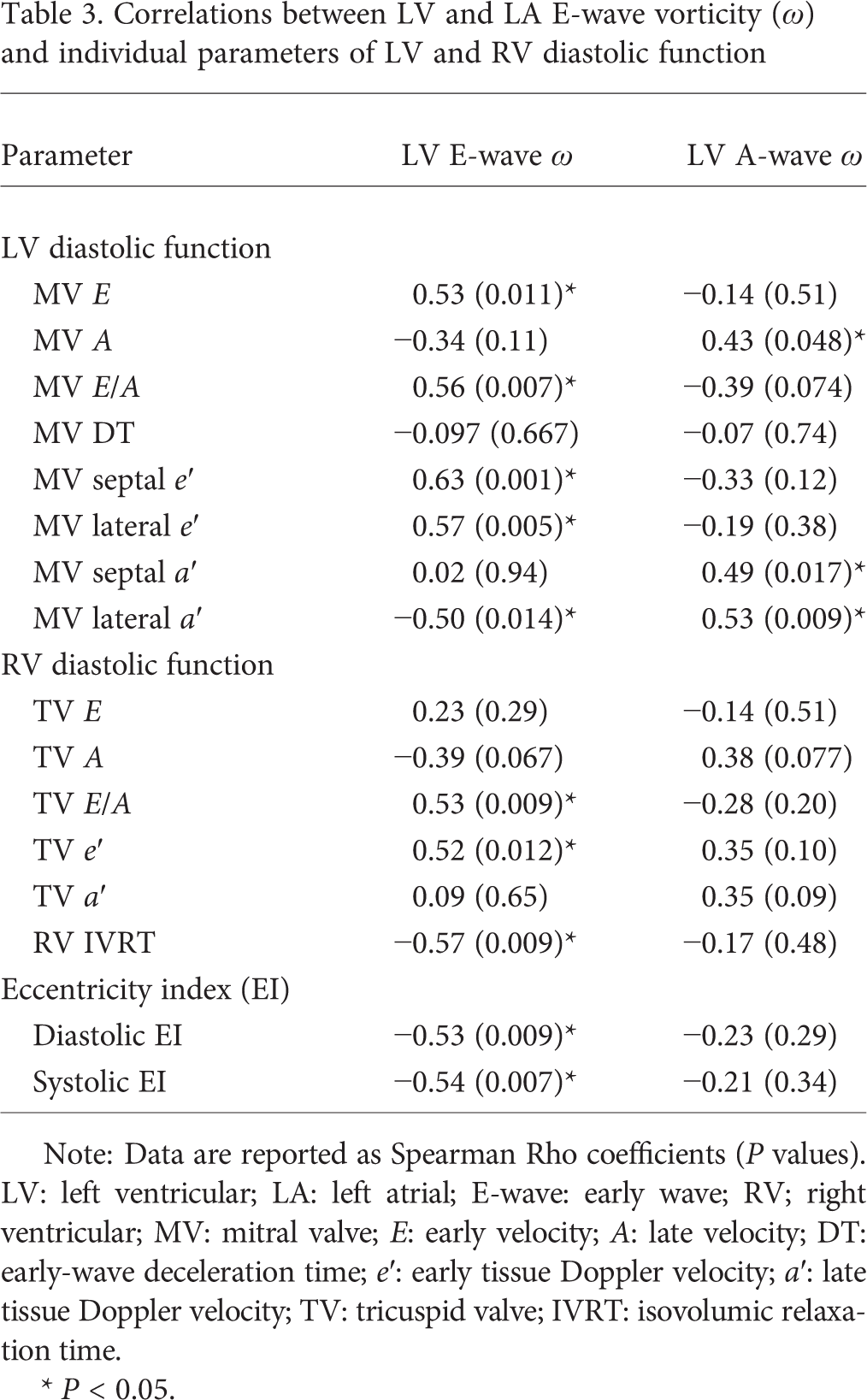
Note: Data are reported as Spearman Rho coefficients (P values). LV: left ventricular; LA: left atrial; E-wave: early wave; RV; right ventricular; MV: mitral valve; E: early velocity; A: late velocity; DT: early-wave deceleration time; e′: early tissue Doppler velocity; a′: late tissue Doppler velocity; TV: tricuspid valve; IVRT: isovolumic relaxation time.
P < 0.05.
Interdependency assessment
In order to assess the relationship between diastolic ω and diastolic ventricular interdependency, we first characterized diastolic EI in both groups, using short-axis CMR images. Consistent with the presence of PH, diastolic EI was significantly increased in PH subjects compared to controls (1.2 [1.07–1.28] vs. 1.09 [1.03–1.16], P = 0.004; see Fig. 5). Similarly, systolic EI was also increased in PH when compared to controls (1.39 [1.06–1.63] vs. 1.15 [1–1.19], P = 0.019). On the basis of these findings, we then correlated LV E- and A-wave ω with markers of diastolic ventricular interdependency, specifically EI and RV diastolic dysfunction. LV E-wave ω showed a significant inverse correlation with both diastolic and systolic EI (see Table 3). LV E-wave ω also demonstrated significant associations with several indices of RV diastolic dysfunction, including with TV e′, TV E/A, and RV IVRT (see Fig. 6). However, LV A-wave ω failed to show significant correlations with EI or any RV diastolic parameter.
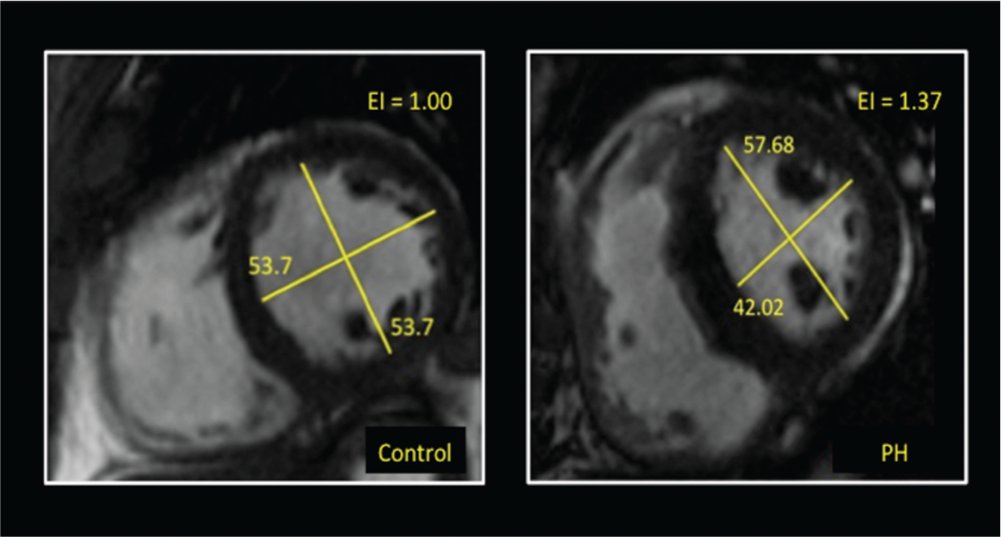
Representative short-axis left ventricular cardiac magnetic resonance images with associated horizontal and vertical diameter measurements for calculation of diastolic eccentricity index (EI). Note the increased EI in a pulmonary hypertension (PH) patient (right) when compared to that in a control subject (left), indicating decreased septal curvature in PH. Diameter measurements are presented in millimeters.
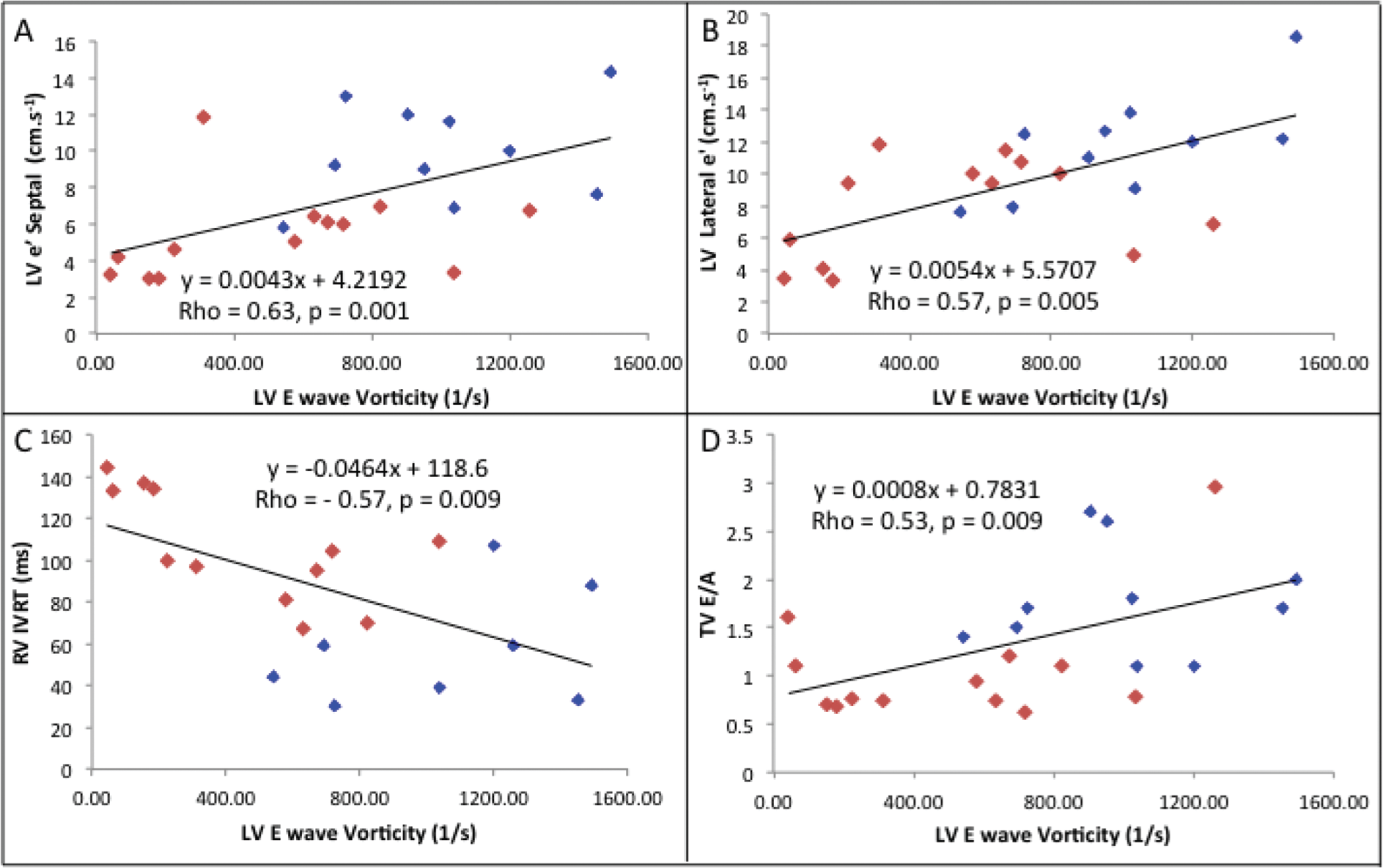
A, B, Linear regression analysis for left ventricular (LV) E-wave vorticity with the LV echo parameters septal LV e′ (early tissue Doppler velocity; A) and lateral LV e′ (B). C, D, Interdependency depicting correlations between LV E-wave vorticity and right ventricular isovolumic relaxation time (RV IVRT; C) and tricuspid valve (TV) E/A (ratio of early to late velocity; D). Blue: control, red: pulmonary arterial hypertension/RV diastolic dysfunction. MV: mitral valve.
DISCUSSION
Using same-day 4D CMR and echocardiographic assessment in a cohort of PH patients and control subjects, we demonstrate that the novel computational hemodynamic parameter intracardiac diastolic ω correlates with multiple indices of ventricular interdependency, including diastolic EI and biventricular diastolic function. The PH cohort had physiologic evidence of interdependency, as demonstrated by increased EI as well as LV diastolic dysfunction. Although LV diastolic dysfunction may reflect other nonpulmonary vascular comorbidities, our study design specifically excluded LV systolic dysfunction, valvular disease, and coronary disease. Furthermore, there was no significant difference in either age or sex between controls and PH patients that would otherwise explain the presence of diastolic dysfunction.
Although multiple significant associations between ω and ventricular interdependency were identified that indicate a potential relationship between intraventricular fluid mechanics and tissue mechanics, our study design precludes identifying specific hemodynamic mechanisms for altered intracardiac ω patterns. However, our findings lead us to speculate that leftward bowing of the interventricular septum due to PH significantly reduces the potential for the shear-layer and fluid-tissue interactions necessary for vorticity generation and possibly vortex formation. Supporting this hypothesis are the findings that LV E-wave ω was significantly reduced in the PH cohort and that the vortex emanating from the AMVL was noticeably absent in almost all PH patients when compared to controls. Consequently, the conditions necessary for vortex generation are altered in PH, resulting in impaired vortex formation (see Fig. 7). This hypothesis is further supported by the finding that LV E-wave ω inversely correlated with EI. Decreased ω in PH during E-wave filling would be anticipated, since septal bowing should significantly diminish a large region of LV relaxation and subsequent generation of the intracavitary pressure gradient that drives passive LV filling. Although there was no difference in MV E velocity or CI between control and PH cohorts to suggest that reduced MV inflow would explain differences in ω, decreased MV inflow as a result of reduced pulmonary venous return in PH would be expected to decrease intracavitary shear and therefore ω as well as vortex formation. Furthermore, because vortices optimize the transfer of energy and momentum, diminished intracavitary ω and vortex formation may lead to reduced cardiac performance, as supported by the finding that LV E-wave ω directly correlated with SV. Although compensatory increases in LA contractility may be expected to overcome the loss in favorable energetics associated with LV vortex dissolution in order to preserve SV, our PH cohort demonstrated reduced LA EF compared to controls. The interventricular septal displacement associated with PH may also serve to alter the orientation and geometry of the MV apparatus relative to MV inflow, altering the fluid-structure interactions that dictate LV diastolic ω. In addition, the increased blood viscosity associated with PH may result in increased blood inertia and reduced microscopic recirculations that determine vorticity. 25
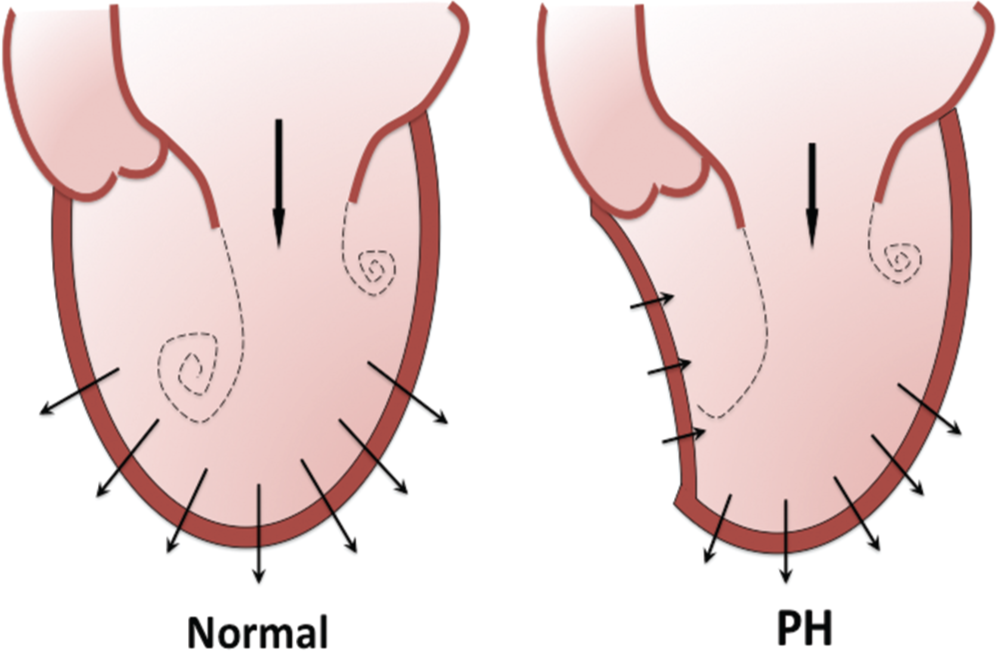
Left ventricular intracardiac inflow patterns in normal (left) and pulmonary hypertension–affected (right) chambers during the E wave. The reduced compliance and ventricular volume diminish the ability to generate vorticity and formation of vortices.
As previous studies have identified changes in intracavitary vortex dynamics that precede alterations in ventricular mechanics, ω analysis may ultimately be a more sensitive noninvasive marker of interdependency than either septal bowing or the development of LV diastolic dysfunction. 14 A clinical tool that could identify interdependency in an earlier disease state may have diagnostic, treatment, and prognostic applications not only for PH but also other cardiac disorders characterized by significant LV-RV interactions, including constrictive pericarditis and pericardial tamponade. In order to more comprehensively assess the relationship between ω, large-scale vortex formation, and interdependency, future studies are required, in larger PH cohorts, that utilize a combination of RHC to characterize interventricular gradient and realtime cardiac imaging to assess interventricular septal conformation, fluid-structure biomechanics, and bioenergetics.
Study limitations
Our study is inherently limited by a small sample size, hindering the ability to derive broad conclusions about the mechanical, hemodynamic, and morphologic correlates of LV ω. Because 4D CMR requires time-averaged X-, Y-, and Z-plane phase-contrast acquisitions, our velocity data are averaged over the duration of the scan (approximately 40–60 minutes). Therefore, our data reflect a range of hemodynamic and respiratory events and may limit the accuracy and strength of the reported correlations. Because our temporal resolution is 50 ms and a normal diastolic period is approximately 120–200 ms, there is potential that hemodynamic events and associated changes in ω were not captured by our acquisition. Although ω and vortex formation represent parallel concepts that describe the microscopic and macroscopic rotation of fluid, respectively, utilizing ω to quantitate vortex phenomena is inherently limited by the potential dissociation between large-scale complex fluid behavior and the behavior of finite elements within those formations. For example, irrotational vortices contain fluid particles that rotate around the center of a vortex without the particle itself rotating around its own axis. The limited spatial resolution of our 4D acquisition limits the ability to measure these smaller-scale fluid dynamics. Furthermore, limited spatial resolution may hinder the accuracy of velocity measurement and therefore of ω measurement. Finally, the PH cohort demonstrated a modestly elevated mean PAWP (11.5 ± 3.4 mmHg), raising the potential for postcapillary PH physiology to confound our interdependency analysis. However, no PH patient had a PAWP (or LV end-diastolic pressure, used in place of PAWP when measured) of >15 mmHg. In addition, mean LVEF was normal in both the PH (71% [66%–79%]) and control (68% [63%–74%]) cohorts. Consequently, our PH cohort represents primarily precapillary PH, as opposed to postcapillary PH.
Conclusions
Using 4D flow CMR in a prospective study comparing a PH cohort to matched controls, quantitative changes in intracavitary LV diastolic vorticity (ω) are associated with large-scale alterations in vortex formation. The ω calculated for early and late LV filling correlated with multiple echocardiographic indices of ventricular interdependency. These results illustrate a potential novel pathophysiologic mechanism for maladaptive fluid-structure interactions in ventricular interdependency in PH.
Footnotes
ACKNOWLEDGEMENTS
We would like to thank Aurelien Stalder, PhD, and Christopher
Glielmi, PhD, for providing the 4D flow acquisition sequence.
