Abstract
Pregnancy outcomes in patients with pulmonary hypertension remain poor despite advanced therapies. Although consensus guidelines recommend against pregnancy in pulmonary hypertension, it may nonetheless occasionally occur. This guideline document sought to discuss the state of knowledge of pregnancy effects on pulmonary vascular disease and to define usual practice in avoidance of pregnancy and pregnancy management. This guideline is based on systematic review of peer-reviewed, published literature identified with MEDLINE. The strength of the literature was graded, and when it was inadequate to support high-level recommendations, consensus-based recommendations were formed according to prespecified criteria. There was no literature that met standards for high-level recommendations for pregnancy management in pulmonary hypertension. We drafted 38 consensus-based recommendations on pregnancy avoidance and management. Further, we identified the current state of knowledge on the effects of sex hormones during pregnancy on the pulmonary vasculature and right heart and suggested areas for future study. There is currently limited evidence-based knowledge about both the basic molecular effects of sex hormones and pregnancy on the pulmonary vasculature and the best practices in contraception and pregnancy management in pulmonary hypertension. We have drafted 38 consensus-based recommendations to guide clinicians in these challenging topics, but further research is needed in this area to define best practices and improve patient outcomes.
SUMMARY OF RECOMMENDATIONS
From “Basic biology of PH and pregnancy”
When counseling pregnant women or women considering pregnancy, healthcare practitioners should inform them regarding the risks, including possibly persistent pulmonary hypertension of the newborn (PPHN), and potential benefits of selective serotonin-reuptake inhibitors (consensus based [CB]).
From “Clinical research on pregnancy outcomes in PH”
Although recent studies suggest improved outcomes in the modern era, maternal morbidity and mortality clearly remain high (CB).
In general, patients with pulmonary hypertension (PH), particularly pulmonary arterial hypertension (PAH), should be counseled to avoid pregnancy (CB).
Given the high mortality rate, rapidly evolving treatment practices, and the need to make complex decisions, early referral to an experienced PH center, ideally one with experience in the management of PAH in pregnancy, is an essential part of care of the pregnant patient with PAH (CB).
From “Prepregnancy counseling and contraception”
All patients should be counseled to avoid pregnancy, with a thorough explanation of risks to both mother and fetus (CB).
Permanent contraception should be strongly considered in PAH patients, with recognition that the field of PAH therapy is advancing and that future outcomes may be improved. Therefore, we recognize that permanent contraception is neither available to all patients nor acceptable to all patients (CB).
Of permanent contraception methods, hysteroscopic sterilization is preferred because of the potential for lower procedural risks (CB).
If tubal ligation is planned, a laparoscopic approach is relatively contraindicated because of procedural risks. A minilaparotomy may be a safer method but still requires general anesthesia (CB).
Estrogen-containing contraception is not recommended because of the increased risk of venous thromboembolic disease (VTE) and the possible deleterious effects of estrogen on the pulmonary vasculature (CB).
Progestin-only pills may be used, but often a second method is required because of the relatively high “typical-use” failure rate (CB).
Progestin-only intrauterine devices (IUDs) and implants are acceptable nonpermanent contraception (CB).
Injected progestins (depo-provera) are relatively contraindicated because of a likely increased risk of thrombotic events (although risk is likely to be mitigated if anticoagulants are used in this population). However, this method may be appropriate in patients without a history of VTE, because of its superior efficacy to oral progestin-only pills, when the other, more effective methods listed above are not available (CB).
Barrier and fertility awareness–based methods are not recommended to prevent pregnancy in PH (CB).
Centers managing patients with PH should have access to family-planning services able to give specialist advice (CB).
From “Genetic counseling in PH”
Genetic counseling should be offered to patients with idiopathic PAH or heritable PAH (CB).
Genetic testing should not be performed in the absence of genetic counseling (CB).
As genetic mutations underlying World Health Organization (WHO) groups 2, 3, 4, and 5 PH and PAH not due to idiopathic or heritable disease have not been described, genetic testing and/or counseling is not required at present for these conditions (CB).
From “Pregnancy management”
Pregnancy in PAH patients is associated with a high maternal mortality rate. The highest-risk periods are the peripartum period and the immediate postpartum period (up to 2 months) (CB).
A multidisciplinary approach with high-risk obstetricians, PH physicians, cardiologists, anesthesiologists, and neonatologists is recommended (CB).
Close clinical monitoring, including monthly follow-up visits in the first and second trimesters and weekly clinic visits in the third trimester, is recommended (CB).
All pregnant PH patients should be offered pregnancy termination. In particular, patients with worsening right heart failure, especially early in their pregnancy, and patients with other high-risk features should be given the option to have their pregnancy terminated. If patients want to continue pregnancy, augmentation of PAH therapy should be considered to improve the chances of a favorable outcome (CB).
Cesarean section is the preferred mode of delivery and should be used unless not available or in cases of emergencies (CB).
Epidural or spinal-epidural anesthesia is recommended rather than general anesthesia (CB).
Because of the potential for catastrophic consequences, pregnant PH patients and their providers should be counseled on inducers and symptoms of vasovagal syncope. Triggers of vasovagal syncope should be avoided (CB).
From “Role for PAH-directed therapy in pregnancy and delivery”
In patients who are in WHO functional class (FC) IV or have evidence of severe right ventricular (RV) impairment, parenteral prostaglandins are recommended. Most published experience in parenteral prostaglandins is with intravenous (iv) epoprostenol (CB).
In select patients with more preserved RV function who are in WHO FC III, inhaled prostaglandins may be considered. Most published experience in inhaled prostaglandins is with iloprost. An appropriate treatment response needs to be verified (CB).
Oral phosphodiesterase 5 inhibitors may be considered in patients who are in WHO FC I or II and who have normal RV function. Most published experience is with sildenafil. Close follow-up for deterioration is highly recommended for patients treated with phosphodiesterase 5 inhibitor monotherapy (CB).
Parenteral prostaglandins can be combined with oral phosphodiesterase inhibitors in pregnancy. Successful outcomes have been reported with this regimen (CB).
For patients meeting strict criteria for vasodilator-responsive PAH who are not in WHO FC IV and who do not have RV dysfunction, calcium channel blocker therapy may be continued in pregnancy, with close follow-up for deterioration (CB).
The currently available endothelin receptor blockers and soluble guanylate cyclase stimulator are pregnancy category X and should not be used in pregnancy. If a PAH patient who is taking one or more of these medications becomes pregnant, their use should be immediately discontinued (CB).
At the time of delivery, iv prostaglandins may be considered in patients not already treated with this class of medication (CB).
At the time of delivery, PAH patients require close monitoring, with a central venous catheter, an arterial line, and careful attention to volume status (CB).
Close monitoring is recommended for all pregnant PAH patients, with routine evaluation by history, physical examination, imaging (echocardiography) and laboratory testing as appropriate for deterioration (CB).
Routine monitoring with Swan-Ganz catheterization is not recommended in the peripartum period because of the risk of complications. Other techniques to noninvasively assess cardiac output (CO) in the peripartum period, such as lithium dilution, can be considered (CB).
From “Role for anticoagulation in the pregnant PH patient”
If a PAH patient has been receiving anticoagulation therapy before pregnancy, the risks and benefits of this therapy should be reevaluated and discussed at the time of pregnancy (CB).
Warfarin should be stopped and either unfractionated or low-molecular-weight heparins used if anticoagulation is continued (CB).
Prophylactic heparin is recommended in the peripartum period (CB).
New oral anticoagulants are currently not recommended for PAH or chronic thromboembolic PH (CB).
INTRODUCTION
Pulmonary hypertension (PH) has long been recognized to affect women in substantially greater numbers than men. Epidemiologic data on pulmonary arterial hypertension (PAH), as classified by the fifth World Symposium on Pulmonary Hypertension, 1 specifically shows that women are affected 3–4 times more commonly than men.2,3 Progressive pulmonary vascular obliteration in all forms of PH frequently leads to right heart failure and often to death. Increasing data now suggest a mechanistic role for sex hormones in pulmonary vascular disease. Moreover, women affected by PH are often young and of childbearing age.2,3 How pregnancy-induced shifts in sex hormones may affect pregnant PH patients is currently an area of intense debate. Further, the physiologic burden of pregnancy in PH patients is an area of major concern, as it has been linked to unacceptably high maternal and fetal mortality. 4
While expert guidelines on the management of PH rightly caution that pregnancy is contraindicated in PH, 5 occasionally PH is diagnosed during pregnancy or a PH patient becomes pregnant despite appropriate warning. Data on the management of the pregnant PH patient are limited, and thus recommendations for management of these situations would serve as helpful guides to clinicians. We sought to perform a rigorous and extensive literature review and also convened experts with experience in this topic to generate a statement regarding the current knowledge of and treatment recommendations surrounding pregnancy in PH. Although it is clear that there are limited data on this relatively rare yet important clinical challenge, our document attempts to address current understanding of sex hormones in pulmonary vascular disease, physiologic effects of pregnancy in the PH patient, recommendations for pregnancy avoidance, and management of the pregnant PH patient. Where evidence is lacking and expert opinion is used, we have noted this accordingly, in an attempt to help clinicians as they face challenging PH cases. In addition, we recognize that there may be socioeconomic, religious, or cultural differences that drive management of pregnancy in PH. Through the Pulmonary Vascular Research Institute (PVRI), we have convened an international collaboration of experts to address pregnancy in PH. We have collectively attempted to be sensitive to these issues in crafting our document and to reach consensus in drafting the recommendations, but the reader is cautioned to implement recommendations appropriate for the culture of the patient. Finally, because most data are focused on the subset of PH patients with PAH, this is a focus of our data review, but much of the statement is applicable to non–WHO group 1 PH.
We have attempted to comprehensively cover the known and unknown scientific points as well as clinical management questions, but we recognize that not all literature on this topic can be discussed and that the topic is rapidly evolving. The reader is recommended to consult both primary sources and reviews. In future years, we plan to update this statement, and we also plan to address other issues related to gender and PH. Our document may point out areas of great interest for researchers in the field. Future studies will hopefully address unanswered questions in the management of pregnancy in PAH and other forms of PH.
METHODS
Recognizing that, despite recommendations to the contrary, pregnancy occurs in our female patients with PH, our goal was to produce a summary statement on pregnancy in PH with a focus on (1) current knowledge of basic science on sex hormones in PH, (2) physiology specific to PH and pregnancy, (3) recommendations for pregnancy avoidance, and (4) management of the PH patient during pregnancy. Healthcare providers may use these recommendations to optimize care of the female PH patient, both those who chose to avoid pregnancy and those who present with known or newly diagnosed PH and are pregnant.
Our group convened in September 2013 and through conference calls drafted an outline of this document, focusing on the areas of interest outlined above. Each author of the document was assigned a major heading to review literature and draft recommendations. After an initial draft of the literature was assembled, we met to review findings and generate recommendations at the PVRI meeting in Giessen, Germany, in January 2014. After this meeting, each author composed a set of recommendations for his or her major heading. Although recommendations exist for drafting guidelines, 6 our group recognized that the field of pregnancy and PH had insufficient evidence to use the recommended methods for diseases of greater prevalence and with a larger body of evidence. Instead, we first performed a rigorous literature search within each topic, using PubMed, then met both via teleconference and in person at the PVRI meeting. Each author summarized recommendations for clinical care as either evidence based or consensus based (CB).
After an initial draft of recommendations, all CB recommendations were voted on by the members of the writing committee via an internet-based polling system (REDCap). To be included in the final document, recommendations required agreement of 7 of 8 authors. Recommendations not meeting this requirement were reworded until this threshold was reached.
REVIEW AND RECOMMENDATIONS
Overview of PH classification and pathophysiology
The group of pulmonary hypertensive disorders encompasses a heterogeneous entity of diseases that are characterized by a mean pulmonary artery pressure (mPAP) of ≥25 mmHg. 7 The increase in mPAP may or may not be accompanied by a significant increase in pulmonary vascular resistance (PVR), even though marked increases in PVR are typically observed in more severe precapillary forms of the disease.8–12 The WHO differentiates 5 major groups of pulmonary hypertensive disorders that differ in their pathogenesis, severity, structural abnormalities, prognosis, and treatment strategies (Table 1). 1
Updated clinical classification of pulmonary hypertension (PH)

Note: Adapted from Simonneau et al. 1
PAH (WHO group I PH) is one of the most severe types of pulmonary hypertensive disorder. PAH is a progressive and devastating disease of multifactorial etiology that eventually results in significant hemodynamic alterations, severe pulmonary vascular remodeling, increased PVR, RV failure, and death.8–10 Despite the development of new pharmacological treatments over the past 2 decades, no cure exists, and 3-year mortality remains unacceptably high, at 55%. 13 The hallmark of PAH is an abnormal and progressive hyperproliferative, antiapoptotic pulmonary vascular remodeling process that involves all cell types of the pulmonary artery (PA) wall.8–10 Although PAH was initially thought to be a disease primarily characterized by vasoconstriction and abnormal shear stress, it is now established that the pathogenesis of PA remodeling in PAH is much more complex and includes genetic abnormalities, epigenetic phenomena, altered microRNA function, enhanced extracellular matrix deposition and degradation, perivascular inflammation, mitochondrial dysfunction, and recruitment and activation of circulating and resident progenitor cells, leading to vascular obstruction with the formation of plexiform and occlusive lesions.8–10 These alterations work in concert to generate a profound pulmonary vascular remodeling process that shares similarities with malignant cell growth.8–10 Despite the use of advanced therapies, PAH remains a debilitating and deadly disease, 13 and the effectiveness of currently available PAH treatments is limited because these drugs primarily target the vasoconstrictive aspect of the disease.14,15 However, while it is generally accepted that vasoconstriction is important in early stages and/or milder forms of the disease, the major factor responsible for the high PVR in severe, established PAH is the formation of occlusive neointimal and plexiform lesions in small, peripheral PAs.8–10 In addition, while PAH is primarily a disease of the small-resistance PAs of the pulmonary vascular compartment, recent research established that the larger-conductance vessels of the pulmonary vascular system are affected by PAH as well and contribute to disease pathogenesis and increased PVR by exhibiting altered mechanical properties (e.g., increased vascular stiffness).8,16–18 Ultimately, the increase in PVR results in RV stress, maladaptive RV remodeling, RV failure, and death.8–10
WHO group 2 and group 3 PH, while much more common than PAH, typically is characterized by less severe hemodynamic alterations and PA remodeling.11,12 WHO group 4 and group 5 PH, on the other hand, may be as severe as PAH and may exhibit structural and pathogenetic features that overlap with WHO group 1 disease.19,20
Pregnancy in patients with any type of PH is associated with significant morbidity and mortality.21–23 Because of the relatively high prevalence of PAH in women of childbearing age,13,24 pregnancy is a particular challenge in patients with this type of PH. In particular, pregnancy in women with severe PAH, compared with that in women with mild PAH, is associated with higher maternal morbidity and adverse fetal outcomes. 25 A detailed understanding of the effects of pregnancy on the pulmonary vasculature and RV is therefore of critical importance. The remainder of this section therefore reviews the currently available data on how pregnancy affects the pulmonary vasculature, followed by a review of pregnancy's effects on the RV.
Physiologic changes of pregnancy/concerns in PH
The mechanical and hormonal changes of pregnancy affect each major organ system. These changes are primarily due to (1) mechanical compression of the surrounding structures caused by the enlarging uterus, (2) increases in the levels of sex hormones, and (3) increases in the circulatory volume. The effects on the major organ systems most relevant to PH (cardiovascular, respiratory, and hematologic systems) are reviewed here.
Mechanical and hormonal changes in pregnancy
Mechanical changes in pregnancy. Mechanical changes in pregnancy are primarily due to hormone-mediated changes in maternal chest wall diameter and enlargement of the uterus, thereby affecting cardiac and pulmonary function. In addition, in the supine position, blood flow in the inferior vena cava may be compromised because of uterine compression.
Hormonal changes in pregnancy
Sex hormone levels increase dramatically during pregnancy and have a major effect on the body.26–30 The fetoplacental unit formed by the fetus and the placenta produces all the hormones necessary for a successful pregnancy. Steroid precursors are delivered from both the fetus and the mother.
One of the first hormones to peak during pregnancy is plasma human choriogonadotropin (hCG). This peak occurs in the tenth week of pregnancy. One of the many effects of hCG is the stimulation of the hormone relaxin, which, like estrogens and progesterone, mediates vasodilatory responses and facilitates blood flow to critical organs. 31
Progesterone and estrogens are the major sex hormones for the remainder of the gestation period. Plasma progesterone is initially produced by the corpus luteum, but its production is taken over by the placenta during the transition to the second trimester. Progesterone exhibits a first peak in the first month of pregnancy, followed by a sharp and continuous increase throughout the remainder of the pregnancy, ending with a peak concentration at around 340 pg/mL immediately before delivery.27,30 The major role of progesterone is to create a protective environment for the fetus in the uterine cavity by stimulating endometrial glands to nourish the zygote and by maintaining the homeostasis of decidual cells. Furthermore, progesterone inhibits the occurrence of uterine contractions by decreasing prostaglandin synthesis and by attenuating sensitivity to oxytocin. Progesterone also mediates major cardiovascular and respiratory adaptations in pregnancy (see below).
Levels of the three major estrogens (estrone [E1], 17β-estradiol [E2], and estrione [E3]) rise steadily throughout pregnancy. E2 levels reach values of up to 7,200 pg/mL.27,30 Major effects of estrogens include stimulation of the growth of the myometrium and of the ductal system of the breast. Like progesterone, estrogens also have significant effects on the cardiopulmonary system (reviewed below).
Other sex hormones increased during pregnancy include prolactin, sex hormone–binding globulin, dehydroepiandrosterone (DHEA), and testosterone.26–29 Both progesterone and estrogen levels drop sharply immediately after delivery.
Pregnancy effects on the cardiovascular system
Plasma volume and dilutional anemia. One of the most significant changes in the cardiovascular system is a significant increase in plasma volume. Plasma volume starts to increase early in pregnancy and peaks shortly before delivery at values that are 50%–70% above the patient's prepregnancy values.32–34 The major cause of the pregnancy-induced increase in plasma volume is hormone-mediated vasodilation, which leads to underfilling of the vascular system and atria, resulting in activation of the renin-angiotensin-aldosterone system and a decrease in natriuretic peptide release, allowing for significant retention of sodium and volume (6–8 L).35–38 The latter is mainly contained in the uterus, breast, muscle, and skin. Red cell mass increases above the prepregnancy baseline, also as a consequence of increased erythropoietin production, but only by 20%–30% (and even less so in patients not taking iron supplements).32,39,40 Since red cell mass rises less than the plasma volume, a significant dilutional anemia occurs, with normal hemoglobin values as low as 10.5 g/dL. 41 The resultant decrease in blood viscosity facilitates perfusion of the fetoplacental unit and may also lower cardiac work.
Cardiac axis, heart size, and heart rhythm. Because of mechanical forces from the enlarging uterus and a resulting upward shift of the diaphragm, a leftward shift of the cardiac axis occurs. Consequentially, nonspecific ST and T wave changes can be seen on an electrocardiogram. 42 This is accompanied by mild dilation and stretching of all cardiac chambers, as well as mild tricuspid regurgitation,43,44 changes that are due to the increase in plasma volume. Left ventricular (LV) mass increases slightly;44,45 however, the ratio of wall thickness to ventricular radius does not change, indicating an eccentric hypertrophy similar to what is seen with athletic training. 46 These shifts in cardiac axis and anatomy, in combination with hemodynamic, hormonal, and autonomic changes, represent the major causes of the frequently observed benign supraventricular arrhythmias during pregnancy. 47 Similarly, increases in heart rate by 15–20 beats per minute are common.48,49
CO, systemic vascular resistance, and mean arterial pressure (Fig. 1). CO rises sharply in the first trimester and then increases more gradually to peak values of around 30%–50% above the prepregnancy baseline.49–52 Initially, this is mediated by an up to 35% increase in stroke volume (SV). The reason for the increase in SV is likely multifactorial and due to the increased plasma volume, decreased afterload, increased contractility, and eccentric hypertrophy associated with increased LV compliance.46,53 The increase in contractility likely results from a Frank-Starling response triggered by increased plasma volume and possibly also from direct inotropic effects of sex hormones (e.g., estrogens and prolactin). However, increases in contractility have not been uniformly described, and transient decreases in systolic function have been reported as well.54,55 Later in pregnancy, an additional increase in heart rate contributes to the increases in CO as well. CO values of up to 8.7 L/min at week 36–38 may be seen.45,56–58 LV ejection fraction remains unchanged.43,44 B-type natriuretic levels increase slightly but remain within normal limits. 59
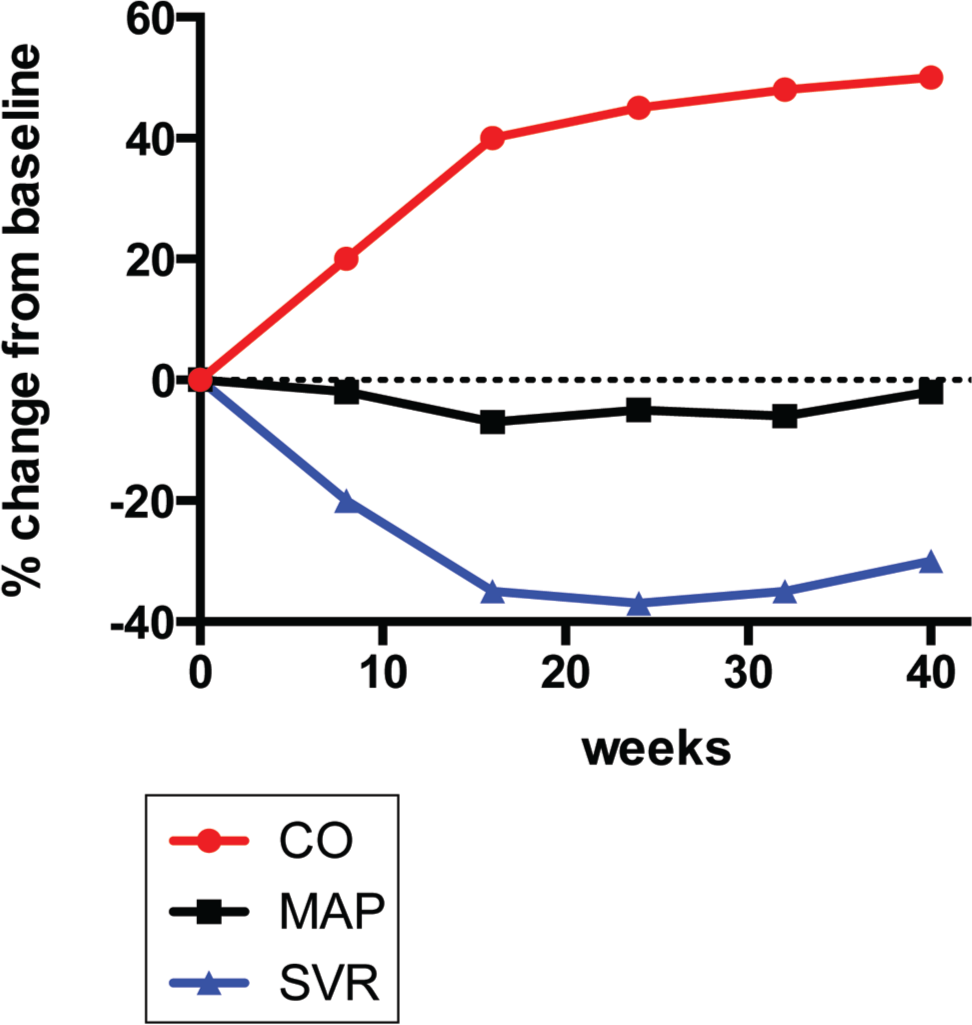
Changes in cardiac output (CO), mean arterial pressure (MAP), and systemic vascular resistance (SVR) during pregnancy. Values above dotted line represent an increase from baseline, while values below dotted line depict a decrease.
Systemic vascular resistance (SVR), on the other hand, decreases by up to 40% as a consequence of progesterone- and estrogen-mediated vasodilation as well as the generation of a high-flow and low-resistance circuit in the uteroplacental circulation; this results in a significant drop in diastolic blood pressure.49,60–64 The mechanisms of hormone-mediated vasodilation are not completely understood but likely include increased endothelial prostacyclin and nitric oxide production and decreased responsiveness to the vasoconstrictive effects of angiotensin II and norepinephrine, as well as reduced aortic stiffness.65–70 The latter allows for increased compliance of the vascular system. The structural correlates of these changes include fragmentation of reticular fibers and changes in the structure of elastic fibers, as well as hypertrophy and hyperplasia of smooth muscle cells. 71
Because of the opposing effects of increased CO and decreased SVR on mean arterial pressure (MAP), the latter decreases only slightly. Systolic and diastolic blood pressure may decrease slightly in the first 2 trimesters (to a statistical mean of about 105/60 mmHg); however, in the third trimester these values usually increase back to prepregnancy baselines, as does MAP.49,60–64
Pulmonary vasculature (Fig. 2). Changes similar to those in the systemic vasculature are observed in the pulmonary vascular system, where the increase in CO and the decrease in PVR result in a grossly unchanged mPAP. As in the systemic vasculature, the decrease in PVR is primarily hormone mediated; recruitment of previously nonperfused pulmonary vessels (similar to what is seen during exercise) has been suggested as well. A normal pulmonary vascular system can therefore easily accommodate the increased CO and increased pulmonary blood flow that occur during pregnancy (Fig. 2; upper-right corner). However, in the setting of pulmonary vascular disease (e.g., PAH or pulmonary embolism), these compensatory mechanisms are significantly impaired, resulting in a significant afterload stress on the RV (Fig. 2; lower-right corner). The inability of the pulmonary vasculature to accommodate the increased CO therefore explains the frequent worsening of PH and the frequent occurrence of RV failure during pregnancy or in the postpartum period, where CO rises even more (see below).21–23
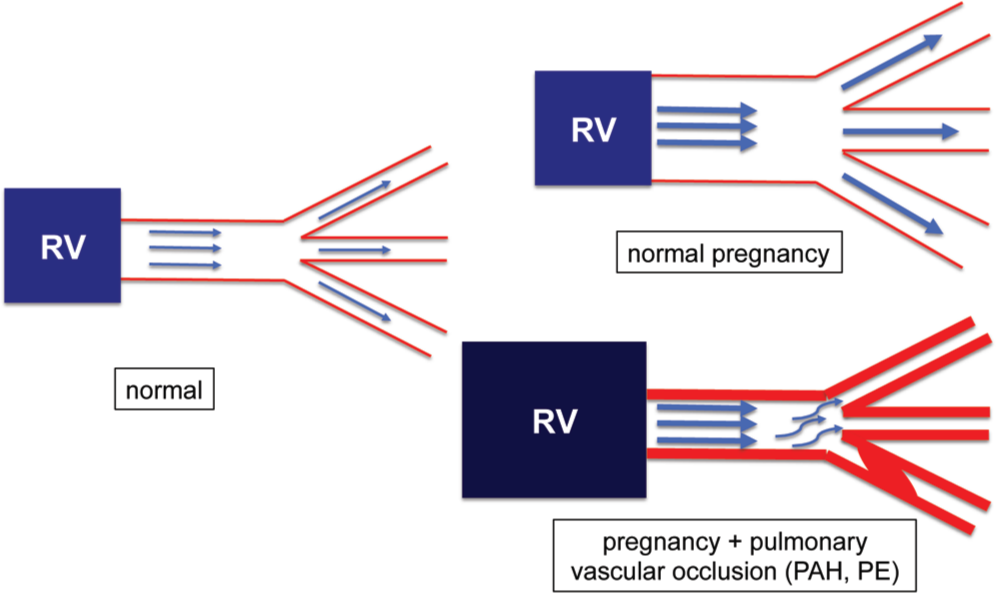
Adaptation of the pulmonary vascular system and the right ventricle (RV) to increased pulmonary blood flow during pregnancy in a healthy patient and in pulmonary vascular disease. Note that the diseased pulmonary vasculature in pulmonary arterial hypertension (PAH; characterized by vasoconstriction, pulmonary vascular remodeling with lumen obliteration, and in situ thrombosis) is unable to accommodate the increased cardiac output, thus leading to RV strain, dilation, and eventually decompensation. PE: pulmonary embolism.
Effects of labor on the cardiovascular system. While the first 9 months of pregnancy are characterized by marked but gradual changes in the cardiovascular system, labor and delivery are associated with even more rapid and dramatic changes. In particular, the intrathoracic pressure swings and volume shifts associated with labor and delivery have significant effects on cardiac preload and afterload. For example, the pronounced respiratory efforts associated with labor and delivery cause significant changes in intrathoracic pressure, thus leading to marked pressure swings in the central venous system as well as in the arterial system. 72 Furthermore, large volume shifts occur during and after delivery through the release of vena caval obstruction, as well as through autotransfusion of 300–500 mL of blood from uterine contractions.39,73 A significant additional increase in blood volume is therefore seen in the peripartum and postpartum periods, accompanied by increases in CO (15%–25% above prelabor values) and SVR.39,73 On the other hand, postpartum hemorrhage may lead to marked volume shifts in the other direction, highlighting the complexities of labor and delivery in patients with pulmonary vascular disease.
Pregnancy effects on the respiratory system
Lung mechanics and gas exchange. Early in pregnancy, structural changes occur in the thorax to accommodate the enlarging uterus. The circumference of the lower chest wall increases, and the subcostal angle may increase from 68° to 100° or more in the first trimester. The chest diameter increases by a minimum of 2 cm, resulting in a “barrel chest” appearance of the thorax.74–76 These changes are due to the relaxing effects of the hormone relaxin on the ligamentous attachments of the lower ribs. 77 By term, the diaphragm rises up to 4 cm. Interestingly, diaphragmatic excursion is not limited by the enlarging uterus and increases by up to 2 cm.
Pregnancy is a hypermetabolic state, and oxygen consumption increases by up to 20%. 78 Respiratory adaptations to the increased oxygen demand are primarily mediated via progesterone-mediated increases in tidal volume, while respiratory rate does not change significantly.79,80 The underlying mechanisms of progesterone-mediated hyperventilation are not completely understood; increased sensitivity of the medulla to carbon dioxide has been suggested, as well as direct stimulatory effects of progesterone on the respiratory center.79,81,82 Minute ventilation increases by approximately 50%, resulting in a respiratory alkalosis that is accompanied by a compensatory metabolic acidosis.79,83–85 The consequence of the increased alveolar ventilation is a significant rise in alveolar and arterial oxygen tension84,86 and also a sense of dyspnea. In fact, up to 70% of normal pregnant women experience dyspnea during pregnancy. 87 The changes in maternal arterial oxygen and carbon dioxide tensions (Pao2 and Paco2, respectively) facilitate oxygen and carbon dioxide transfer across the placenta. Normal values for pH, Pao2, and Paco2 in pregnancy are provided in Table 2.
Normal values for pH and arterial oxygen and carbon dioxide tensions (Pao2 and Paco2, respectively) in pregnancy
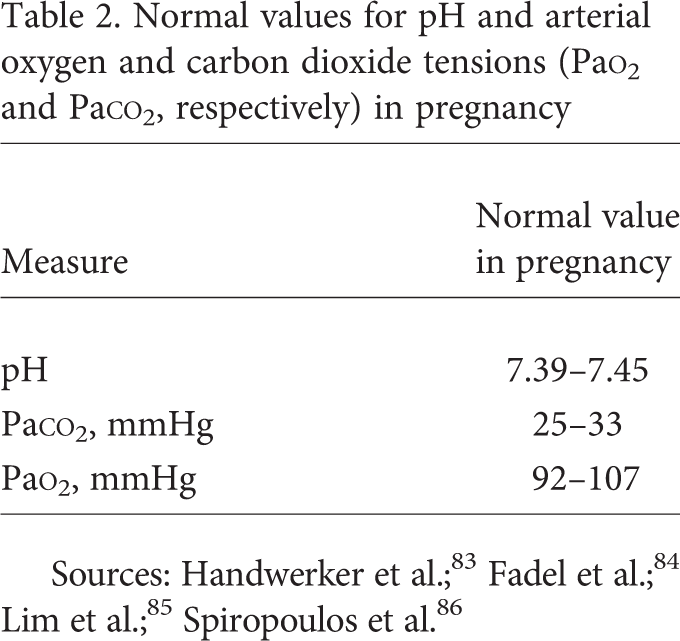
Functional residual capacity (FRC) decreases by 20% as a consequence of displacement by the enlarging uterus. 88 This is the correlate for the rapid desaturations that occur in pregnant patients despite the slightly increased Pao2.
Airway anatomy. Upper-airway anatomy is affected by pregnancy-induced weight gain and by pregnancy-induced fluid retention and associated oropharyngeal edema; this has significant implications for airway management in pregnant patients, as a difficult airway situation may arise. This is exemplified by a study demonstrating an increase by 34% in the number of patients with a Mallampati class 4 airway from week 12 to week 38. 89 In addition, the process of labor may significantly affect airway anatomy. Specifically, a recent study demonstrated that the modified Mallampati classification increased by 1 grade in 33% of parturients and by 2 grades in 5% of parturients. 90 Interestingly, airway edema does not correlate with the duration of labor or the amount of fluid administered and may still be present, at least in part, at 48 hours postpartum. 90 The exact cause of this phenomenon is unclear. The combination of a difficult airway and decreased FRC can make endotracheal intubation extremely challenging, a scenario that is especially precarious if the patient has an already compromised pulmonary vascular bed, as is the case in PH.
Physiologic changes in the postpartum period
The postpartum period is characterized by a rapid drop in estrogen and progesterone levels, followed by gradual normalization over 6 weeks. Blood volume and hematological changes normalize within 6–8 weeks, 91 while CO and SVR may take up to 3 months, or even longer, to reach prepregnancy values. 92
Summary
Major physiologic changes of pregnancy in the cardiovascular system include volume expansion and increased CO, as well as major volume and pressure swings during and after labor.
Pregnancy-induced changes in the respiratory system include decreased FRC, respiratory alkalosis, and airway edema.
Basic biology of PH and pregnancy
Pregnancy effects on the pulmonary vasculature in PAH
As reviewed in “Overview of PH classification and pathophysiology,” the normal pulmonary vasculature responds to pregnancy by dilating and by recruiting previously nonperfused vessels, resulting in decreased PVR. Along those lines, the impact of vasoconstrictor stimuli is attenuated, as evidenced by the observation that pregnancy is characterized by attenuated hypoxic vasoconstriction.93,94 Women have a more distensible pulmonary circulation than men; 95 however, whether these changes are even more pronounced during pregnancy is unknown.
While it is unclear how many of the changes observed in the pulmonary vasculature during pregnancy are directly attributable to increases in sex hormone levels, there is irrefutable evidence that sex hormones have many pharmacological influences on the pulmonary circulation. For example, E2, testosterone, and progesterone all attenuate PA vasoconstriction.96–100 Underlying mechanisms include increases in endothelial nitric oxide synthase activity, increased prostacyclin synthase activity, and decreased endothelin 1 activation.96,101–106 Interestingly, progesterone receptors have been identified in myofibroblasts resident in plexiform lesions. 107 Progesterone causes pulmonary vasodilation 99 and may inhibit vascular remodeling and attenuate experimental PH. 108
The effects of endogenous E2 in PH are largely unknown but likely to be complex. For example, women are more prone to developing PAH,13,24 but E2 levels and female sex correlate with higher RV ejection fraction,109–111 and female sex is associated with better PAH survival.13,24 E2 has several interesting properties relevant to PAH pathogenesis. Particularly, in addition to vasodilator effects, it has proangiogenic and anti-inflammatory properties. 112 Interestingly, pulmonary microvascular density, markers of angiogenesis, and diffusion capacity in women vary over the menstrual cycle, and estrogen replacement in ovariectomized animals increases lung capillarization, thus suggesting that angiogenesis and diffusion characteristics in pregnant patients may be influenced by E2 and other sex hormones as well. 113 Depending on the context, E2 or its metabolites may exert antiproliferative or proproliferative effects on vascular endothelial and smooth muscle cells.114–120 The antiproliferative mechanisms have been implicated in protective E2 effects in animal models of PAH;105,106,112,115,116 whether they play a role in the adaptation of the pulmonary vasculature to pregnancy, however, remains unknown.
DHEA exerts a variety of effects on the pulmonary vasculature that include vasodilation and stimulation of antiproliferative pathways;121–126 increases in this hormone thus would be expected to facilitate the adaptation of the pulmonary vascular system to pregnancy-induced hypervolemia. Major effects of sex hormones on the pulmonary vasculature are depicted in Figure 3.
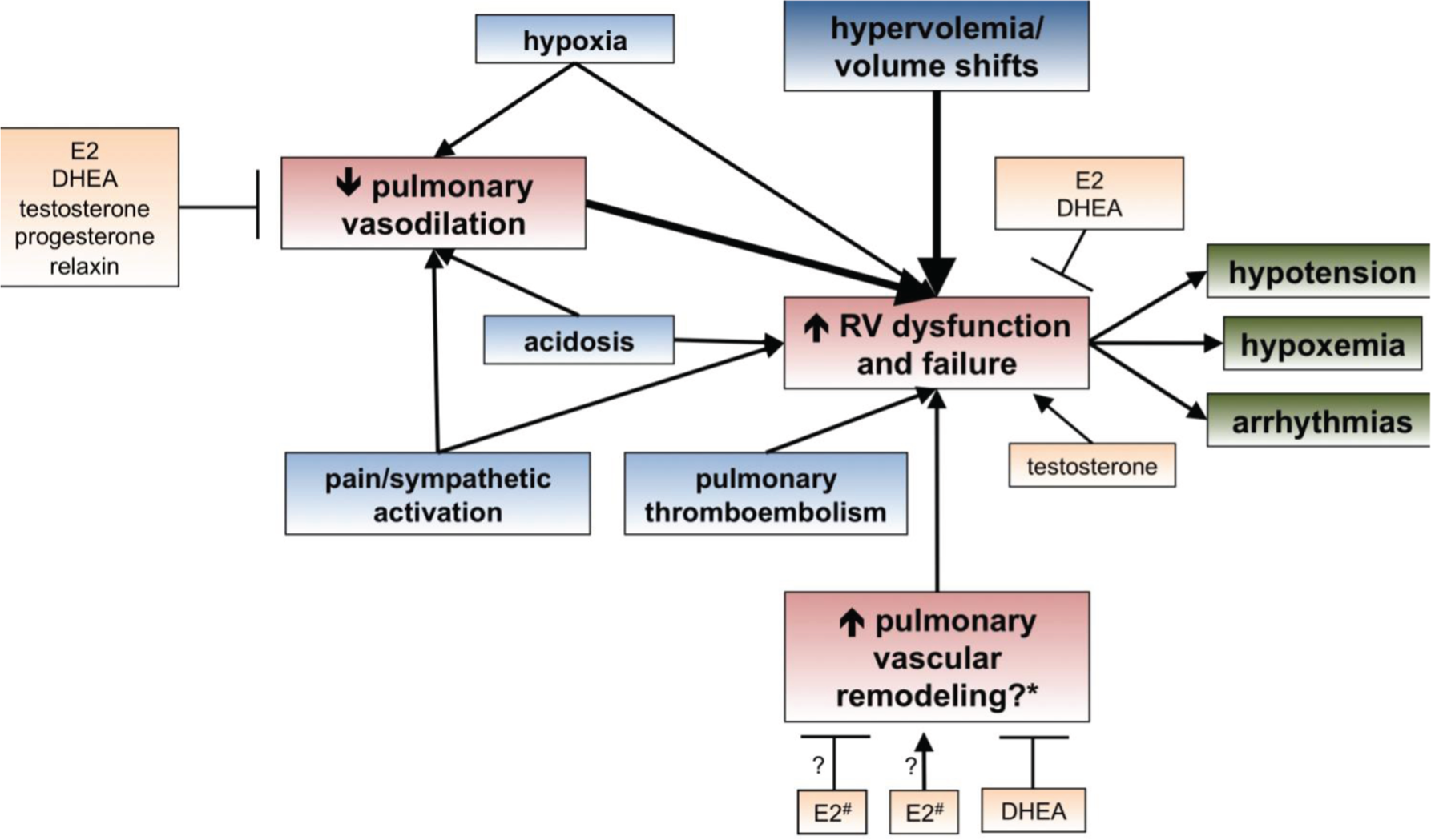
Pathophysiology of pulmonary hypertension (PH) and right ventricular (RV) dysfunction in pregnancy. Items in red represent the underlying preexisting alterations in PH; items in blue represent pregnancy-related modifiers that may aggravate these alterations. Potential contributions of sex hormones are shown in yellow (derived from studies in normal animals as well as PH models). Decreased pulmonary vasodilation and vascular recruitment, as well as hypervolemia and volume shifts, are the major contributors to RV dysfunction and failure in pregnant PH patients. Volume shifts are primarily due to intermittent mechanical compression of the inferior vena cava, effects of respiration-induced changes in intrathoracic pressure on venous return, autotransfusion during and after delivery, and postpartum blood loss. Hypotension, hypoxemia, and arrhythmias may further worsen RV failure and initiate a vicious cycle. DHEA: dehydroepiandrosterone. *Whether worsening pulmonary vascular remodeling contributes to worsening of PH and RV function during pregnancy remains unknown. #The contribution of 17β-estradiol (E2) to pulmonary vascular remodeling in PAH is unknown, and inhibitory as well as stimulatory effects may exist, depending on the clinical context.
Relaxin has vasodilator properties in the systemic vasculature31,127 and appears to act as a pulmonary vasodilator as well.127–129 It is unknown whether the structural changes that are reported in the systemic vasculature of pregnant subjects (e.g., remodeling of the extracellular matrix, with changes in the structure of reticular and elastic fibers 71 ) are also present in the pulmonary vascular compartment, but a report of relaxin-induced inhibition of collagen accumulation in the main PAs in a PH model suggests that processes similar to those in the systemic vasculature may also occur in the pulmonary vascular component. 129
Several other hormones implicated in the pathogenesis of PAH are further increased by pregnancy. For example, increased prolactin may play a role in precapillary PAH. 130 Specifically, this hormone is already significantly increased in PAH patients and correlates inversely with the 6-minute-walk distance and peak oxygen uptake. Serum parathyroid hormone can also be elevated in PH. 131 Elevated plasma aldosterone levels were reported in PAH patients. 132 Finally, pregnancy increases glucocorticoid levels and may lead to hyperglycemia. In addition, maternal diabetes is a risk factor for PPHN. 133 Hence, pregnancy can upregulate many hormones already pathogenically elevated in PAH. It is possible that increases in these pharmacologically active hormones may exacerbate their deleterious effects on the pulmonary vasculature.
The overarching problem in the pregnant PH patient is that the physiologic compensatory vasodilator response of the pulmonary vasculature is decreased or absent, thus leading to a significant increase in mPAP and PVR.8–10 The inability of the pulmonary vascular bed to accommodate the increased CO results in significant RV afterload stress, and RV strain and RV failure may occur (Fig. 3). The resulting hypotension and hypoxemia (the latter potentially exacerbated by increased right-to-left shunting through a patent foramen ovale) represent major health risks for both the mother and the fetus; this is reflected by the dramatic morbidity and mortality associated with pregnancy in PAH.21–23 Deterioration is most frequent between weeks 20 and 24, during labor and delivery, or in the postpartum period,21–23 as these time points represent the stages with the most significant changes in hemodynamics and volume status. Deterioration around week 24 reflects an inability of the cardiopulmonary system to accommodate the increased CO and meet cardiovascular demands, while deterioration during labor and delivery or in the postpartum phase frequently is triggered by volume shifts and intravascular pressure swings, as well as the negative effects of pain, Valsalva maneuvers, hypoxia, and acidosis on pulmonary vascular tone and/or RV function (Fig. 3). Thromboembolic phenomena may contribute to deterioration in the peripartum period as well. A recent review of the literature demonstrated that the majority of maternal deaths occurred in the peripartum period, mainly within the first month of delivery, with RV failure and circulatory collapse being the main causes of death. 21
Since the pulmonary vasculature in PAH is characterized by nitric oxide and prostacyclin deficiency as well as by increased endothelin 1 activation,8,10 there appears to be a lack of substrate for sex hormones to initiate their vasodilator properties. Furthermore, there is concern that prosurvival and proproliferative effects of estrogens may worsen the pulmonary vascular remodeling in pregnant PAH patients. For example, several studies have demonstrated proproliferative effects of E2 on PA smooth muscle cells (PASMCs) in animal models of PH and in human disease.134–137
In premenopausal women, E2 synthesis occurs mainly in the ovarian follicles and corpus luteum; synthesis also occurs to a lesser extent in nonglandular tissues such as adipose tissue, liver, skin, muscle, and brain. In men and postmenopausal women, adipose tissue is a major source of estrogen synthesis. 138 Aromatase (CYP19A1) is a member of the cytochrome P450 (CYP) superfamily and synthesizes E2 through the aromatization of androgens, specifically testosterone and androstenedione. Further metabolism is mediated by several CYP enzymes. In particular, CYP1B1 catalyzes the oxidation of E1/E2 to 2- and 4-OHE2 (2- and 4-hydroxy estradiol, respectively). E2 hydroxylation can occur by CYP enzymes (including CYP1B1), resulting in 6, 7, 15, and 16 (α- and β-) hydroxyestradiol. E2 can also be converted to (1) E1 by 17β-hydroxysteroid dehydrogenase and hence via oxidation to 16α-OHE1, which is mitogenic, or (2) 2-OHE2 by CYP1A1/2, CYP1B1, and CYP3A4. Catechol-O-methyltransferase (COMT) catalyzes the methylation of catechol-E2s to methoxy-E2s, which simultaneously lowers the potential for DNA damage and increases the concentration of 2 methoxyestradiol, an antiproliferative metabolite. Both aromatase and CYP1B1 are expressed by human PASMCs and by the medial layer of human PAs; notably, expression of aromatase is increased in female PASMCs.139,140 Hence, there is local synthesis and metabolism of E2 in human PAs. It has also recently been shown that endogenous E2 contributes to the development of PH in the female hypoxic mouse and the sugen/hypoxic rat, in part by restoring impaired bone morphogenetic protein type 2 receptor (BMPR2) signaling in the female lung. 139 This is significant, as the primary genetic defect of heritable PAH (HPAH), present in at least 70% of cases of HPAH, is a mutation in the gene encoding BMPR2, coupled with dysfunctional Smad signaling that leads to abnormal cell proliferation associated with pulmonary vascular disease. 141 Studies in human PASMCs have also demonstrated that E2 enhances other mediators of proproliferative signaling, such as the mitogenic transcription factors CEBPβ and FOS, 137 the calcium-binding protein S100A4/Mts1, 135 and several components of the serotonin production and transport axis (all of which have been implicated in PAH pathogenesis). 136 A contribution of proproliferative estrogen metabolites (e.g., 16α-OHE1) to PAH development has recently been recognized,140,142 and it is possible that excess production of such metabolites during pregnancy may contribute to worsening pulmonary vascular remodeling and hemodynamics in PAH patients. Pregnancy exerts complex effects on the various CYP isoforms; 143 however, whether and how pregnancy affects CYP1B1 expression and activity remain unknown. In addition to genetic alterations in CYP1B1, two promoter single-nucleotide polymorphisms in the gene coding for aromatase that result in elevated E2 production have been associated with an increased risk of portopulmonary hypertension. 144
Knowledge gaps. While it is plausible that estrogens and other sex hormones promote pulmonary vascular remodeling in PAH, it is unknown whether the frequently observed worsening of PAH in pregnancy is indeed due to direct effects of sex hormones on the pulmonary vasculature or merely reflects an effect of the hypervolemia and volume shifts that are associated with the pregnant state. Studies investigating whether pulmonary vascular remodeling indeed progresses in pregnant PAH animals are therefore needed. Along those lines, the effects of sex hormones on pulmonary vascular remodeling during pregnancy require further investigation. Similarly, the contribution of estrogen metabolites to PAH development in pregnancy requires further study. Since worsening of PAH frequently occurs in the postpartum period21–23 and therefore at a time point at which sex hormone levels decrease dramatically, it is possible that a “sex hormone withdrawal phenomenon” results in PA vasoconstriction in the postpartum state. However, this has not yet been investigated. Studies investigating cyclical changes in vasomotor tone related to the menstrual cycle demonstrate worsening vasoconstriction during periods of low estrogen levels,100,145–147 suggesting that such a phenomenon may contribute at least in part to the frequent PAH decompensation observed in the postpartum stage.
Pregnancy effects on the RV in PAH
During pregnancy, both the LV and the RV exhibit hypertrophic growth in order to compensate for the increase in blood volume and CO. 54 In particular, these chambers undergo eccentric hypertrophy, a finding that is characterized by a proportional change in wall thickness. 148 Even though diastolic function may be impaired,44,54 the hypertrophy is considered to be physiological (or adaptive), similar to the cardiac hypertrophy observed in athletes.148,149 While the SV increases, LV systolic function is maintained,43,44 although transient decreases in systolic function have been described.54,55 On the other hand, other studies found no change in LV ejection fraction.43,44 The histological correlate of the increase in heart size and weight is cardiomyocyte hypertrophy, with an increase in cardiomyocyte length. 148 Pregnancy-induced cardiac hypertrophy is reversible and starts to regress soon after delivery.50,54
Cardiac function and remodeling during pregnancy. While the effects of sex hormones and pregnancy on the LV have been relatively well characterized, these data cannot necessarily be extrapolated to the RV, as the two chambers are embryologically, anatomically, physiologically, biochemically, and electrophysiologically different.150,151 However, studies specifically investigating the impact of sex hormones and pregnancy on the RV are sparse, and investigations are needed that specifically focus on the effect of these factors on RV structure and function. Studies from the LV have demonstrated that both mechanical stress associated with pregnancy-induced hypervolemia and direct effects of sex hormones cause a wide range of changes in the myocardium. 152 These include the activation of second messengers such as protein kinase C, mitogen-activated protein kinase, and Src tyrosine kinase. 148 It has been proposed that activation of the latter represents one of the mechanisms of estrogen-mediated heart hypertrophy in late pregnancy. 148 In addition, E2 has been shown to down-regulate cardiac Kv4.3 gene expression, an effect that has been linked to the longer QT interval observed in pregnancy. 148
Interestingly, alterations in markers of pathological cardiac hypertrophy—such as α- and β-MHC (myosin heavy chain) switch or changes in atrial natriuretic peptide, phospholamban, or SERCA (sarco/endoplasmic reticulum Ca2+-ATPase), all of which have been implicated in the pathophysiology of RV failure in PAH153,154—have not been detected in pregnancy. 148 Additional studies that further characterized cardiac hypertrophy in late pregnancy in mice demonstrated increased cardiac angiogenesis, absence of fibrosis, and decreased expression of matrix metalloproteinase 2, 15, and 17. 155 Despite an increase in cardiac contractility, LV pressures are increased, suggesting a component of cardiac dysfunction. 155 These changes were reversed in the postpartum period. 155 Furthermore, the myocardium of late pregnancy exhibits decreased proteolytic activity of the ubiquitin-proteasome system, as well as decreased oxidative stress. 156 The decrease in proteasome activity appears to be independent of E2 levels. 156
Sex hormone effects on cardiac function. A significant body of work has demonstrated protective effects of E2 and/or female sex in various forms of LV injury, all of which would expected to benefit RV function in pregnant PH patients. In particular, several studies in various types of LV injury (e.g., chronic pressure overload or myocardial ischemia) have demonstrated protective effects of E2 and/or female sex on cardiac function and mortality.157–165 These improved outcomes are accompanied by favorable estrogen receptor–mediated E2 effects on myocardial metabolism, inflammation, microRNA signaling, fibrosis, matrix remodeling, and apoptosis.157–166 E2 mediates these favorable adaptations to LV injury by targeting multiple cardiac cell types, such as cardiomyocytes, endothelial cells, fibrocytes, progenitor cells, and inflammatory cells (reviewed by Murphy 167 ). Interestingly, E2 has consistently been shown to decrease cardiac hypertrophy in LV injury models and in in vitro studies,160,164,165,168–170 a finding that seems to contradict findings of E2-mediated increases in cardiac hypertrophy during pregnancy. 148 Differences in the type and duration of cardiac hypertrophy (adaptive versus maladaptive) may explain this discrepancy, at least in part.
Despite a significant number of studies investigating effects of E2 and sex on LV function in pregnancy and disease, how pregnancy and sex hormones affect the function and molecular makeup of the normal RV remains relatively unexplored. Furthermore, the effects of sex hormones and pregnancy on an already diseased RV have not been well characterized. In particular, it is unknown whether and how pregnancy and sex hormones affect physiological or biochemical parameters in the RV once these are already altered by chronic pressure overload. What is currently known is that both healthy women and women with PAH exhibit RV function superior to that of their male counterparts.109–111,171 Furthermore, in female hormone-replacement therapy users, RV ejection fraction correlates with E2 levels, suggesting either a direct effect of E2 on RV function or a more indirect effect via modification of RV afterload. 110 Along those lines, basic-science investigations in nonpregnant rodents with PH demonstrated protective estrogen receptor–mediated E2 effects on RV function that are associated with decreased RV stress signaling, increased RV capillary density, and decreased fibrosis as well as with decreased expression of the matrix-remodeling enzymes ADAM15, ADAM17, and osteopontin.112,115,172 Taken together, these data suggest salutary E2 effects on RV function in PH. Whether these animal findings translate to human disease is currently unknown.
Similarly, DHEA has been shown to exert RV-protective effects in rodent models of PAH, where its administration was associated with decreased oxidative stress, decreased apoptosis, and decreased activation of cardiac-remodeling mediators (e.g., Rho kinase, STAT3, and NFATc3), 173 as well as with improved mitochondrial bioenergetics and antioxidant mechanisms. 174 In humans, DHEA levels are associated with higher mass and higher RV SVs in healthy women. 110 Testosterone, on the other hand, is associated with lower RV ejection fraction and higher RV mass in healthy subjects, 110 and testosterone increases RV hypertrophy and fibrosis in male mice undergoing PA banding. 175 Sex hormone effects on RV function in PAH are summarized in Figure 3.
Hormone-mediated cardioprotection and the peripartum period. While these studies suggest beneficial effects of major pregnancy hormones on RV function, they do not, however, explain the high prevalence of pregnancy-associated RV failure in PAH. One potential explanation is that despite potential beneficial effects of E2 and DHEA on RV function, increased testosterone levels during pregnancy176,177 negatively affect RV performance. Furthermore, rapid decreases of E2 and DHEA levels after delivery may contribute to RV failure in the postpartum stage. In this scenario, the negative effects on RV function, occurring simultaneously with worsening pulmonary vasoconstriction, would provide a “perfect storm” and cause the RV to decompensate. Alternatively (or in addition), the RV stress associated with the hemodynamic changes and volume shifts during pregnancy, and particularly the peripartum period, may “overwhelm” E2- and DHEA-mediated protection and thus induce RV failure, especially in light of the known transient decrease in cardiac function that normally occurs during pregnancy.54,55,156
In sum, pregnancy is associated with a number of adaptive changes in the cardiopulmonary compartment that serve to accommodate the increased CO and to meet the increased cardiovascular demands of the pregnant body. In PAH, however, several of these compensatory mechanisms are attenuated or simply overwhelmed. Pregnant PAH patients usually die of RV failure and cardiovascular collapse, with the majority of deaths occurring in the early postpartum period. The changes in hemodynamics and volume status observed during pregnancy appear to be a major cause of the worse outcomes in pregnant PAH patients. The role of sex hormones is not yet clear, as some studies show beneficial effects (e.g., vasodilation) while others suggest that they exert harmful effects (e.g., proliferation of PASMCs) on the pulmonary vasculature. Data for cardiac function are more clear-cut, as there is a solid body of evidence that supports beneficial effects of E2 and DHEA on cardiac function, even though studies specifically focusing on the RV are sparse and primarily in animals. In addition, studies investigating the effects of sex hormones specifically on RV function in the setting of pregnancy do not exist; this represents a significant knowledge gap in the field. The contribution of the abrupt fall in sex hormones after delivery to the high prevalence of cardiac decompensation in the postpartum period requires further investigation.
Effects of pregnancy on non–group 1 PH
The great bulk of current literature on sex hormone effects on the pulmonary vasculature and the potential effects of pregnancy are focused on PAH (WHO group 1 disease) or on hypoxia-associated PH (group 3). The effects of sex hormones on WHO group 2, 3, 4, and 5 PH and their potential effects on the pulmonary vasculature in pregnancy are essentially unstudied and are a major knowledge gap at present.
PPHN: potential maternal influences
Studies suggest a possible association between maternal use of the selective serotonin-reuptake inhibitor (SSRI) fluoxetine late in the third trimester of pregnancy and the risk of PPHN in the infant.178,179 In further studies it was shown that an increased risk for PPHN was indicated for high maternal age, first parity, high maternal body mass index (BMI), and possibly maternal smoking. Adjusting for these variables and year of birth, an association between maternal use of SSRIs and PPHN in births after 34 completed weeks was still identified, with a risk ratio of 2.4 (95% confidence interval [95% CI]: 1.2–4.3) when based on women who reported the drug use in early pregnancy. When a subgroup of the women who had prescriptions for SSRIs from the antenatal care later in pregnancy were studied, the risk ratio estimate was 3.6 (95% CI: 1.2–8.3). 180 However, these findings remain controversial, and some conclude that, because of sample sizes and quality issues in studies, the absolute risk cannot be determined. 181 If there is a risk, the mechanism behind the association between SSRIs and PPHN is unclear, but an increased risk for respiratory problems after maternal use of SSRIs is well known, and PPHN could be a rare component of this association. Possible etiological mechanisms are polymorphisms in the promoter alleles of the serotonin transporter gene 182 and in enzymes involved in catecholamine metabolism, 183 which may predispose to neonatal adverse effects, including respiratory distress. 184 Experimentally, it has been suggested that prolonged exposure to SSRIs can induce PPHN through direct effects on the fetal pulmonary circulation via serotonin. Serotonin causes pulmonary vasoconstriction, contributes to maintenance of high PVR in the normal fetus, and hence may contribute to the hypertensive effects of SSRIs.185,186 A recent review concluded that the risk of PPHN is increased for infants exposed to SSRIs in late pregnancy, independent of the potential moderator variables examined. Although the statistical association was significant, clinically the absolute risk of PPHN remained low even in the context of late exposure to SSRIs. 187
Summary
The overriding problem in PH during pregnancy is the decreased ability of the pulmonary vasculature to accommodate pregnancy-induced increases in CO and circulating blood volume. This represents a particular problem during labor and delivery, when additional hemodynamic changes and volume shifts occur and when other stressors (e.g., catecholamine release, acidosis, hypoxemia, thromboemboli) may further compromise the pulmonary vascular bed.
Consequentially, the postpartum period represents the most vulnerable time point in pregnant PH patients, with RV failure representing the most common PH-related complication. An increased risk for clinical deteriorations also exists in the late first trimester and between weeks 20 and 24.
The effects of pregnancy-induced increases in sex hormones on the pulmonary vasculature in PH are complex and incompletely understood. While estrogens may facilitate the adaptation of the pulmonary vasculature to the increased CO via vasodilation and may favorably affect RV function, theoretical concern exists that proproliferative estrogen effects may worsen pulmonary vascular remodeling.
The incomplete understanding of the effects of sex hormones and their metabolites on pulmonary vascular remodeling and RV function during pregnancy and immediately after delivery represents a significant knowledge gap in the field. This should be addressed in future basic-science and clinical investigations.
High maternal age, first parity, increased maternal BMI, and possibly maternal smoking represent risk factors for PPHN. In addition, available studies suggest a possible association between maternal use of SSRIs in the third trimester of pregnancy and an increased risk of PPHN in the infant.
Summary of recommendations
When counseling pregnant women or women considering pregnancy, healthcare practitioners should inform them regarding the risks, including possibly PPHN, and potential benefits of SSRIs (CB).
Clinical research on pregnancy outcomes in PH
PAH has been associated with poor outcomes in nonpregnant patients. In the era before the availability of PAH-specific treatments, the National Institutes of Health (NIH) registry for PAH, which included mostly prevalent patients, reported a 50% survival rate of 2.8 years, 188 while the most recent large national registries in the era of available multiple therapies report average survival rates of 50% at 7 years.189–191 It is therefore not surprising to see a dismal outcome in PAH patients who become pregnant. Deterioration in pregnancy is reported to occur between the twentieth and twenty-fourth weeks of gestation, early in third trimester, and in the postpartum period. 22 Sudden hemodynamic instability is associated with a high maternal mortality. In such cases, RV failure is the most common cause of death (“Physiologic changes of pregnancy/concerns in PH” and “Basic biology of PH and pregnancy” above).
The currently available PAH-specific therapies have affected survival in nonpregnant PAH patients. Therefore, it is important, when analyzing the data, to take into account the availability of specific drug therapies and the multidisciplinary approach adopted by most large PH centers. In addition, it is crucial to identify the experience accumulated in various PH centers around the world, taking into account the availability of PAH-specific therapies in different parts of the world. A summary of selected studies reporting outcomes in the mother and offspring is outlined in Table 3. Given that in 1996 prostacyclins became available for PAH treatment, we examine the published experience by separating reports temporally into pre- and postprostacyclin eras.
Summary of selected studies
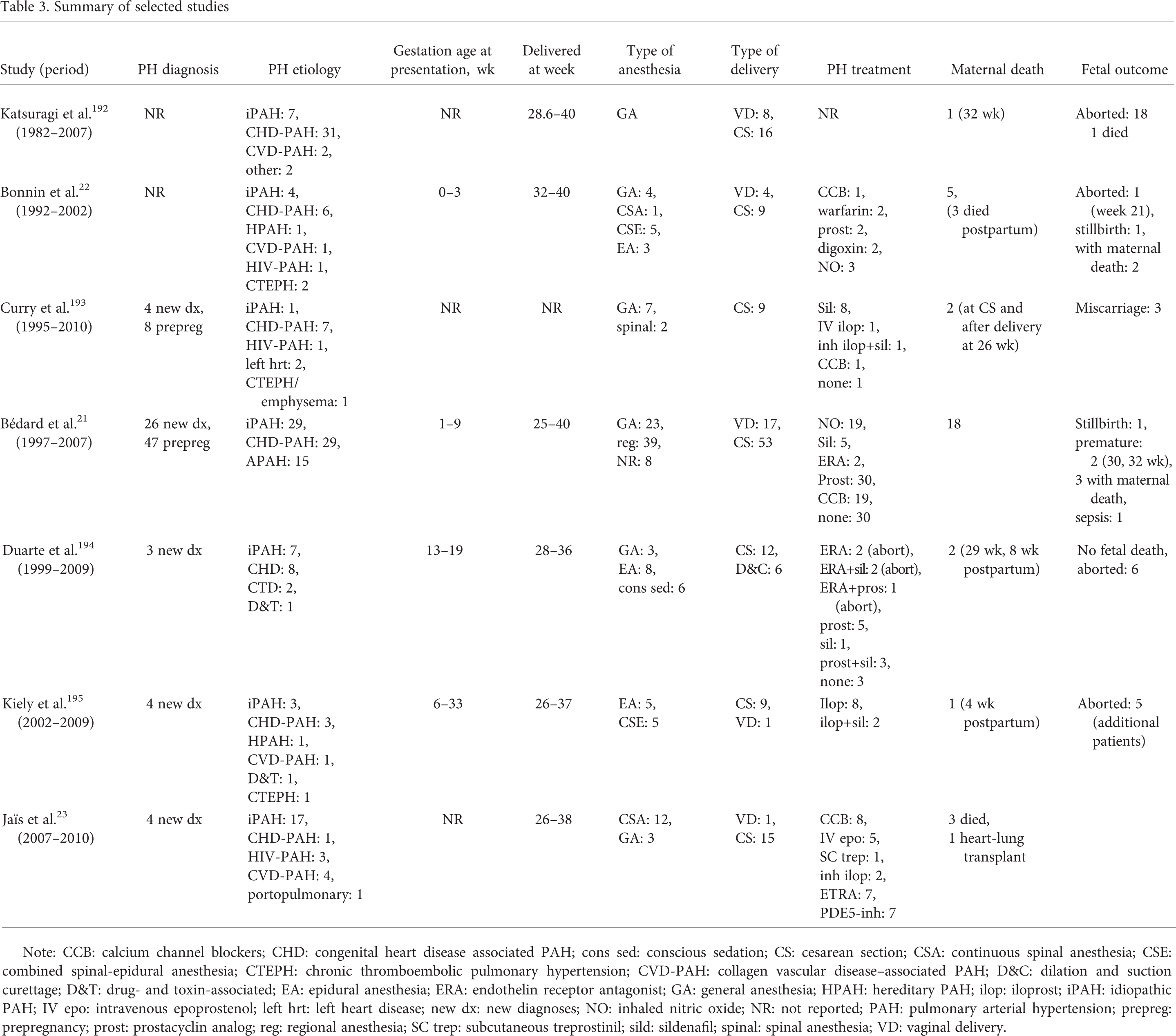
Note: CCB: calcium channel blockers; CHD: congenital heart disease associated PAH; cons sed: conscious sedation; CS: cesarean section; CSA: continuous spinal anesthesia; CSE: combined spinal-epidural anesthesia; CTEPH: chronic thromboembolic pulmonary hypertension; CVD-PAH: collagen vascular disease–associated PAH; D&C: dilation and suction curettage; D&T: drug- and toxin-associated; EA: epidural anesthesia; ERA: endothelin receptor antagonist; GA: general anesthesia; HPAH: hereditary PAH; ilop: iloprost; iPAH: idiopathic PAH; IV epo: intravenous epoprostenol; left hrt: left heart disease; new dx: new diagnoses; NO: inhaled nitric oxide; NR: not reported; PAH: pulmonary arterial hypertension; prepreg: prepregnancy; prost: prostacyclin analog; reg: regional anesthesia; SC trep: subcutaneous treprostinil; sild: sildenafil; spinal: spinal anesthesia; VD: vaginal delivery.
Pregnancy outcomes in the preprostacyclin era
In 1996, epoprostenol was approved for treatment of PAH in the United States. Although not approved by regulatory agencies outside the United States, epoprostenol and its analog iloprost have also been used in Europe and other regions. Historical studies from the preprostacyclin treatment era, including systematic reviews of the published literature, identified a risk of maternal death in the range of 30%–56%. 1 In a review of the published literature between January 1978 and December 1996, Weiss et al. 4 identified 73 patients with Eisenmenger's syndrome, 27 with idiopathic PAH (IPAH, formerly called “primary PH”), and 25 with PAH associated with other conditions (formerly called “secondary PH”). All patients, except for 1, delivered at 32 weeks or later. Pulmonary hypertensive crisis after oxytocin use occurred in 1 patient. Maternal mortality was 36% in Eisenmenger's syndrome, 30% in IPAH, and 56% in associated PAH. All fatalities, except for 3, occurred within 35 days after delivery. Late maternal death several months postpartum was reported in 3 patients with Eisenmenger's syndrome. The causes of death were pulmonary hypertensive crisis with therapy-resistant heart failure (n = 13), sudden death (n = 7), autopsy-confirmed pulmonary thromboembolism (n = 3), cerebral thromboembolism (n = 1), and rupture and dissection of the PA (n = 1). Previous pregnancies, timing of diagnosis and hospital admission, operative delivery, and diastolic PA pressure were significant univariate maternal risk factors. Late diagnosis (P = 0.002, odds ratio: 5.4) and late hospital admission (P = 0.01, odds ratio: 1.1 per week of pregnancy) were independent predictive risk factors of maternal mortality. Twenty-two neonates (weight: 2,372 ± 640 g) survived. One stillbirth and 2 neonatal deaths due to congenital anomalies resulted in a fetal/neonatal mortality rate of 12% (95% CI: 3%–31%). Of note, 1 parturient was treated with heparin and urokinase; operative delivery was assisted by extracorporeal circulation, and postpartum treatment with coumarin, nifedipine, prostaglandins, and diuretics was associated with the patient's survival. Nevertheless, in aggregate, these data suggest that pregnancy in the preprostacyclin era was associated with extremely high maternal and fetal morbidity and mortality.
Pregnancy outcome reports overlapping the pre- and postprostacyclin eras
Mortality and morbidity in pregnant PAH patients improved somewhat over the next years but remained exceedingly high. Bonnin et al. 22 reviewed the charts of 14 pregnant women with 15 pregnancies over a period of 10 years (1992–2002). The overall mortality in this study was 36% (5 of 14 patients). In this series, 3 cases of early deterioration occurred, at weeks 12, 20, and 24 of gestation, in only 1 of which did the patient survive, suggesting that early deterioration should lead to therapeutic abortion. Five of the seven other cases of deterioration occurred during the third trimester.
Experience from Japan was reported in 42 pregnant women who were studied retrospectively from January 1982 to December 2007. 192 Patients were divided into mild-PAH (systolic PAP of 30–50 mmHg on echocardiography or mPAP of 25–40 mmHg by catheterization; n = 14) and severe-PAH (systolic PAP ≥ 50 mmHg on echocardiography or mPAP ≥ 40 mmHg by catheterization; n = 28) groups. The cohort included mostly patients with IPAH or congenital heart disease–associated PAH. All patients with severe PAH before pregnancy had evidence of worsening PA pressures in their last trimester of pregnancy, while most of those with mild PAH had mild or no increase in PA pressures, although this latter group was assessed only by noninvasive methods. Patients with mild PAH delivered mostly at term, whereas those with severe PAH had earlier deliveries. The New York Heart Association (NYHA) functional class worsened in all but 2 patients as pregnancy progressed. There was 1 death immediately after intubation for delivery. Although the authors did not report the use of specific PAH therapies, they mention the use of iv epoprostenol in 3 cases.
Bédard el al. 21 conducted a comparative analysis of articles published between January 1997 and September 2007. Overall mortality was 25%. Parturients who received general anesthesia were 4 times more likely to die than parturients receiving regional anesthesia. Unlike the report from Weiss et al., 4 primigravidae were at higher risk of death than parturients with previous pregnancies. Seventy-eight percent of deaths occurred within 1 month postpartum (15 deaths postpartum vs. 3 deaths during pregnancy). Neonatal/fetal deaths occurred in 10%, 7%, and 13% of patients with IPAH, congenital disease–associated PAH, and patients with associated PAH, respectively.
Pregnancy outcomes in the era of PAH-specific therapies
Over the past decade, there have been several reports of patients with PAH successfully managed during pregnancy, suggesting an improved outcome with current therapies.196–198 This literature, however, is subject to publication bias, may reflect the most successful management of PAH in pregnancy, 199 and should be interpreted with caution.
In a retrospective series conducted between 1999 and 2009 at 5 US medical centers, experience from 18 pregnant PAH patients was reported (50% of patients had congenital heart disease). 194 Six underwent pregnancy termination at a mean gestational age of 13 ± 1 weeks, with no maternal deaths or complications. Twelve elected to continue pregnancy. PAH-specific therapy was administered to 9 (75%) at the time of delivery, consisting of sildenafil, iv prostanoids, or combination therapy. All parturients underwent cesarean section at 34 weeks. There was 1 in-hospital death and 1 additional death 2 months postpartum, responsible for a maternal mortality rate of 16.7%.
Experience from China is evidenced in a retrospective review of 30 consecutive parturients with PAH hospitalized at Peking Union Medical College Hospital from January 1999 to December 2008. 200 The majority of patients were transferred to the PH center at near-term gestation; therefore, they were not followed in a center throughout their pregnancy. Eight patients had IPAH, 7 had congenital heart disease–associated PAH, 10 had rheumatic heart disease–associated PH, and 5 had PH due to other etiologies. PAH-specific therapies were used only in 2 patients with congenital heart disease–associated PAH. The condition of 57% deteriorated to NYHA classes 3 or 4 during pregnancy. Three patients were hospitalized, at 16, 18, and 23 weeks of gestation, for therapeutic abortion because of severe hemodynamic instability, and only 1 survived. The overall maternal mortality rate was 17%, but patients with Eisenmenger's syndrome had 50% mortality. There were 4 fetal/neonatal deaths (13%), and 16 infants were born preterm. All 26 liveborn infants survived.
Published Portuguese experience consists of 3 cases of IPAH from a single center. 201 One patient had a therapeutic abortion, but her PAH deteriorated after abortion, with subsequent demise. The second patient (in whom PAH was well controlled before pregnancy) died 4 months after therapeutic abortion. A third patient (a calcium channel blocker long-term responder) underwent cesarean delivery and was managed intraoperatively with iv prostanoid. Although the patient survived pregnancy and delivery, she ended up requiring long-term PAH-specific therapies.
In 2012, Rosengarten et al. 202 reported their recent experience from 7 patients with PAH who had a total of 9 pregnancies and for whom a careful multidisciplinary approach was implemented. All but 1 patient were treated with iv prostacyclins, and all underwent planned cesarean sections at term. Two patients died within 2 weeks after delivery (22% mortality), and there were no fetal deaths.
In a prospective study from 13 participating centers from Europe, the United States, and Australia, Jaïs and colleagues 23 reported data from 26 pregnancies between July 1, 2007, and June 30, 2010, and their 3-year outcomes. During pregnancy, the patients were regularly seen at their PH centers, usually at 2–4-week intervals. Only one center in France performed right heart catheterizations as part of their regular assessment during pregnancy. A total of 16 pregnancies (62%) were considered successful, defined as survival of the mother and the baby without complications. Six patients underwent induced abortion, and 2 had a spontaneous abortion. Both women with spontaneous abortions died. Three women died in the early postpartum period as a result of right heart failure, whereas 1 required urgent heart-lung transplantation. The outcome of pregnancies was better in patients with lower PVR (500 ± 352 dyn-s/cm 5 ), whereas patients with a very high PVR (1,667 ± 209 dyn-s/cm 5 ) died or required transplantation. Of note, 8 (50%) of the 16 women who had successful pregnancies were so-called vasodilator responders and had nearly normal baseline hemodynamic parameters on calcium channel blocker therapy. The other women with successful pregnancies were calcium channel blocker nonresponders; however, most of them had well-controlled PAH while receiving PAH-specific therapy. Maternal outcome during the year after delivery was analyzed for the 16 women who had successful pregnancies. Two (13%) of them experienced clinical deterioration requiring intensification of PAH therapy. Neither was a long-term responder to calcium channel blockers.
The UK experience is reported in 2 papers. In the earlier report, Kiely et al. 195 analyzed retrospectively the management of 10 pregnancies in 9 women who chose to continue with their pregnancies between 2002 and 2009 in a specialized PH center. All women were treated with 4–7 doses of nebulized iloprost per day, with 2 patients being transitioned to iv prostacyclin and 3 patients additionally receiving sildenafil. Nine patients underwent planned cesarean section. All women received regional anesthesia and were monitored during the peripartum period in a critical care setting. All women survived the pregnancy and postpartum period, and all infants were free from congenital abnormalities. One woman died 4 weeks after delivery, following patient-initiated discontinuation of therapy. Long-term follow-up in the remainder of the patients was a median of 3.2 years (range: 0.8–6.5 years). A second UK report describes retrospectively the outcome of 12 pregnancies in 9 women with PAH between 1995 and 2010 at one center. 193 All women delivered by cesarean section (7 elective and 2 emergency deliveries) under general anesthesia, except for 1 emergency and 1 elective cesarean performed under regional block. There were 2 maternal deaths (in 1995 and 1998), 1 related to preeclampsia and 1 to arrhythmia. Maternal morbidity included postpartum hemorrhage (5 cases) and 1 postcesarean evacuation of a wound hematoma. There were 9 live births, 3 first-trimester miscarriages, and no perinatal deaths.
A most recent prospective international registry including centers from the United States and Europe reported on 26 pregnant PAH patients (17 with IPAH, 9 with associated PAH). Six chose elective abortion, 16 delivered a healthy baby and survived without transplantation, and 4 died or underwent urgent transplantation. 23 Of the 16 patients who successfully delivered, 2 experienced clinical deterioration requiring intensification of PAH therapy.
Taken together, these data suggest that outcomes of pregnant patients have improved with the availability of new PAH therapies, advances in surgical and perioperative management, and use of a team-based, multidisciplinary approach. However, publication bias may exist, and despite the availability of new drugs and technologies, fetal as well as maternal morbidity and mortality remain high.
Summary of recommendations
Although recent studies suggest improved outcomes in the modern era, maternal morbidity and mortality clearly remain high (CB).
In general, patients with PH, particularly PAH, should be counseled to avoid pregnancy (CB).
Given this high mortality rate, rapidly evolving treatment practices, and the need to make complex decisions, early referral to an experienced PH center, ideally one with experience in the management of PAH in pregnancy, is an essential part of care of the pregnant patient with PAH (CB).
Prepregnancy counseling and contraception
Given the risks associated with pregnancy in PH, avoiding pregnancy in the first place is imperative. Contraceptive counseling should begin early after a patient is diagnosed with PH. Depending on the patient's personal wishes and cultural expectations, being informed that pregnancy is contraindicated can be emotionally devastating. The clinician should be ready to provide support. Studies indicate that women are often not well informed about the necessity of contraception and the available options. For example, in a study of patients with congenital heart disease performed in Toronto, Canada, 27 of 80 (34%) of women considered to be at moderate or high risk for maternal complications during pregnancy did not recall being informed of their increased risk. 203 In a larger study of 536 patients with congenital heart disease from 2 tertiary centers in Germany, 20% of women were using a contraceptive method that was contraindicated for their specific cardiac condition, and 28% of patients with high pregnancy-associated risk were not using contraception at all, despite being sexually active. Furthermore, 43% of patients had not been counseled about contraception, and 48% did not recall being informed about the pregnancy-associated risks. 204 A summary of the efficacy of methods of contraception is in Table 4.
Efficacy of contraception methods suitable for patients with pulmonary hypertension
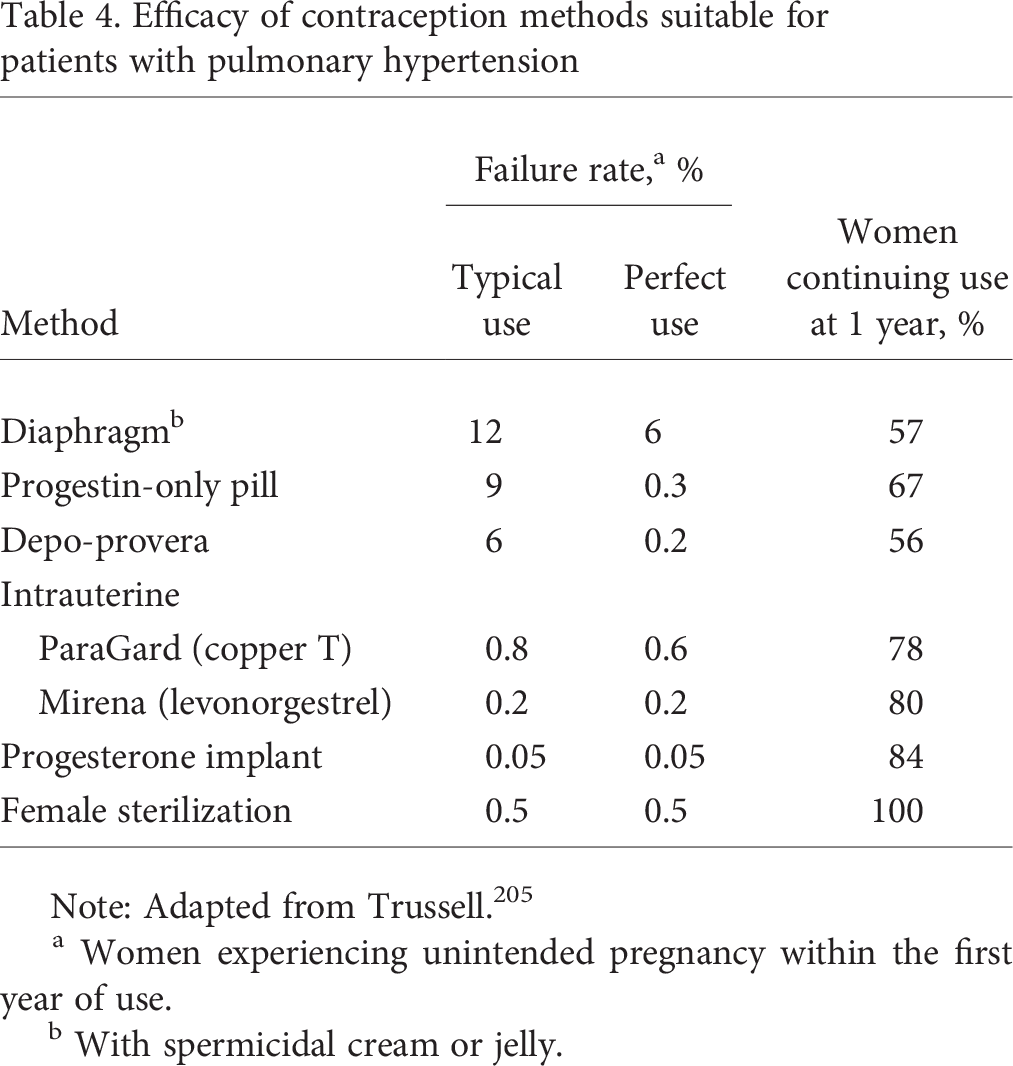
Note: Adapted from Trussell. 205
Women experiencing unintended pregnancy within the first year of use.
With spermicidal cream or jelly.
Progestin-only contraception methods and IUDs are acceptable methods for those patients for whom permanent contraception is either unavailable or unacceptable. Progestin-only contraception methods are available as pills, injections, implants, or IUDs. However, concerns exist regarding an increased risk of VTE with injectable progestins. For example, in an international, multicenter, case-control study conducted by the WHO in 1989–1993, injectable progestin use was associated with an odds ratio for VTE of 2.19; however, because of the wide confidence interval of 0.66–7.26, this did not reach statistical significance. 206 On the other hand, a statistically significant risk was found by Vlieg and colleagues. 207 In this large case-control study of women with a history of VTE, use of injectable progestins was associated with a significantly increased risk of VTE (OR: 3.61 [95% CI: 1.8–7.1]). It is worth noting, however, that this finding was based on a small number of patients: 20 of the 446 of patients who presented with VTE were using injectable progestins, versus 15 of 1,146 controls without VTE. 207 The study was controlled for age and family history of VTE. In a recent meta-analysis, Mantha et al. 208 reviewed 8 studies (either randomized controlled trials or observational case-control or cohort studies) to assess the risk of VTE in women taking progestin-only contraception. Of note, both the WHO study and the Vlieg study discussed above were included in this meta-analysis. While the pooled data indicated no increased risk of VTE associated with either progestin-only pills or progestin-only IUDs, the injectable progestin formulation (depot methoxyprogesterone) was associated with a 2-fold increase risk of VTE. These data thus suggest that the risk of VTE with progestin-only contraception depends, at least in part, on the administration method.
Progestin-only implants are safe and effective methods of contraception. Different formulations are available, with efficacy of 3 years (etonogestrel implant [Nexplanon]), 4 years (levonorgestrel-implant [Sino-implant]), or 5 years (levonorgestrel implant [Jadelle]). Efficacy is similar to that of surgical sterilization, and there is no evidence that these formulations increase thrombotic risk. The implants are placed in the upper arm under local anesthesia.
IUDs are an effective form of contraception for patients with PH. The two commonly used IUDs are the copper IUD and the progestin-releasing IUD (Mirena, Bayer HealthCare Pharmaceuticals). Placement of an IUD can be associated with difficult-to-treat vasovagal reactions when the cervix is manipulated; thus, great caution must be taken for patients with PAH. As mentioned above, there is no increased risk of VTE with the progesterone-releasing IUDs, and the rate of uterine blood loss with menses is lower than that with the copper device. Thus, the progesterone-releasing IUD is favored in women with PAH. Both devices are associated with an increased risk of pelvic infections due to sexually transmitted diseases.
It is hypothesized that the lower serum levels of progestin account for the relative safety of oral and intrauterine progestins, as compared with progestin injections. 208 Peak progestin concentration after injection is 2,500–7,000 pg/mL, and the level then remains >400 pg/mL. 209 On the other hand, plasma progestin concentrations with progestin-releasing IUDs are much lower, with the mean ± SD plasma concentration of levonorgestrel being 260 ± 68 pg/mL during the first 3 months and 129 ± 28 pg/mL after 1 year of treatment. Concentrations associated with oral formulations are even lower. Serum levels with progestin-only implants are similar to those found with progestin-releasing IUDs.
Barrier methods and other methods
Barrier methods of contraception include diaphragms, cervical caps, and sponges. Because barrier methods are event driven and often have a higher failure rate when used alone, they are not recommended as sole forms of contraception in patients with PH.
Fertility-awareness methods, such as “awareness-based” avoidance of intercourse and lactation-induced amenorrhea, are similarly ineffective205,213 and are not recommended to prevent pregnancy in PH patients. Spermicides are among the least effective methods of contraception and are thus not recommended to be used alone as contraception in PH. 214
Permanent sterilization
Because pregnancy will be dangerous for patients with PH for the foreseeable future and because there is no perfect reversible form of birth control for these women, some centers favor permanent contraception. Options include tubal ligation or a device implanted into the fallopian tube (Essure device, Conceptus, Mountain View, CA). Male sterilization (vasectomy) can be combined with a second form of female birth control, alternatively.
Tubal ligation can be performed laparoscopically or via a minilaparotomy. Both procedures are performed with the patient under general anesthesia with positive-pressure ventilation. Although commonly used in healthy women, a laparoscopic approach entails greater risk for the PH patient than for the general population. Patients are positioned with the head down to shift abdominal contents away from the pelvis, resulting in diaphragmatic impingement of the thoracic cavity. Furthermore, manipulation of the cervix is frequently associated with a vasovagal response, and insufflation of carbon dioxide gas into the abdomen causes increased abdominal pressure and may result in a paradoxical CO2 embolism. For these reasons, a minilaparotomy may be a safer approach. Although still requiring general anesthesia, a minilaparotomy is performed in the supine position, and CO2 insufflation is not needed.
Where available, hysteroscopic sterilization is the preferred option for permanent sterilization. This procedure entails the hysteroscopic placement of nickel-titanium coils wound around polyethylene fibers that are inserted into the fallopian tubes. No incision is required. Patients require minimal or no anesthesia, as frequently conscious sedation is adequate to perform the procedure. After placement, the device stimulates an inflammatory response that results in tubal occlusion within 10–12 weeks. The procedure is generally safe and effective, even in high-risk patients. In a retrospective cohort study of hysteroscopic sterilization, 18 women with severe cardiac disease, including 5 with PH, were compared with 157 women without cardiac disease. 215 No intraoperative complications were reported, and follow-up tubal-occlusion rates were 100% in the cardiac group, compared with 98% in the control group. A report of 4 patients with PAH described similar outcomes: no procedure or anesthesia-related complications were encountered. 216
Emergency contraception
In cases where emergency contraception is required, hormonal methods may be considered, with the same caveats discussed above. Alternatively, placement of a copper IUD has also been reported to be a successful method of emergency contraception, with the added benefits of providing a lasting method of contraception going forward and avoidance of exposure to high-dose sex hormones. For patients using the endothelin receptor antagonist bosentan, the dose of emergency contraception will have to be increased because of the presence of drug-drug interactions.
Summary of recommendations
All patients should be counseled to avoid pregnancy, with a thorough explanation of risks to both mother and fetus (CB).
Permanent contraception should be strongly considered in PAH patients, with recognition that the field of PAH therapy is advancing and that future outcomes may be improved. Therefore, we recognize that permanent contraception is neither available to all patients and may not be acceptable to all patients (CB).
Of permanent contraception methods, hysteroscopic sterilization is preferred because of the potential for lower procedural risks (CB).
If tubal ligation is planned, a laparoscopic approach is relatively contraindicated because of procedural risks. A minilaparotomy may be a safer method but still requires general anesthesia (CB).
Estrogen-containing contraception is not recommended because of the increased risk of VTE and the possible deleterious effects of estrogen on the pulmonary vasculature (CB).
Progestin-only pills may be used, but often a second method is required because of the relatively high “typical-use” failure rate (CB).
Progestin-only IUDs and implants are acceptable nonpermanent contraception (CB).
Injected progestins (depo-provera) are relatively contraindicated because of a likely increased risk of thrombotic events (although risk is likely to be mitigated if anticoagulants are used in this population). However, this method may be appropriate in patients without a history of VTE, because of its superior efficacy to oral progestin-only pills, when the other, more effective methods listed above are not available (CB).
Barrier and fertility awareness-based methods are not recommended to prevent pregnancy in PH (CB).
Centers managing patients with PH should have access to family-planning services able to give specialist advice (CB).
Genetic counseling in PH
Genetic counseling in IPAH and HPAH
Mutations in BMPR2 can be found in about 80% of families affected by HPAH.8,2 Other genetic mutations have recently been identified in HPAH, including caveolin 1 (CAV1), 220 KCNK3, 221 and EIF2AK4, 222 that further explain a proportion of the remaining 20% of HPAH. Extensive research in the past decade has focused on the epidemiology of HPAH and downstream signaling associated with BMPR2 mutation, but there is no cure at present for this genetic mutation, and patients with HPAH are treated similarly to patients with IPAH. The BMPR2 mutation is a dominantnegative gene with only weak penetrance, such that approximately only 20% of people with the mutation will develop PAH while the remaining 80% do not develop clinical PAH.218,219
In addition, a small percentage of patients with IPAH also have a BMPR2 mutation but do not have any family members affected by PAH, because of either the low penetrance or de novo mutations. 223 These IPAH patients with BMPR2 mutation can, however, transmit the mutation to their biologic offspring, who are at risk to develop PAH if they receive this allele. The frequencies of the other genes associated with HPAH in the IPAH population are unknown at present. Germ-line genetic mutations have not been described in non–group 1 PH and in PAH that is not idiopathic or heritable.
Clinically available genetic testing can detect most BMPR2 mutations, and some facilities detect CAV1, ALK1, and KCNK5. Others, such as EIF2AK4 and other newly discovered mutations, are screened for only in the research setting at present.
Given the complexity of genetics in HPAH and IPAH, patients wishing to become pregnant should be offered genetic counseling before conception. In the process of genetic counseling, patients will be aided in understanding the risks and benefits of genetic testing and will then be equipped to make an informed decision regarding whether or not they want to undergo genetic testing. They may also consider forms of preimplantation genetic testing or postconception testing for the fetus if they decide to pursue pregnancy. Given the potential psychological and financial risk associated with genetic testing, it is not recommended to undergo genetic testing in the absence of genetic counseling.
Genetic counseling in patients with PAH other than idiopathic or heritable and non–group 1 PH.
Patients with other forms of PAH or non–group 1 PH are not recommended to undergo genetic counseling or testing for BMPR2 mutation unless there are unusual circumstances.
Summary of recommendations
Genetic counseling should be offered to patients with IPAH or HPAH (CB).
Genetic testing should not be performed in the absence of genetic counseling (CB).
As genetic mutations underlying WHO Groups 2, 3, 4, and 5 PH and PAH not due to idiopathic or heritable disease have not been described, genetic testing and/or counseling is not required at present for these conditions (CB).
Pregnancy management
Pregnancy can occur in patients with PH in a number of circumstances. Despite appropriate counseling, the PH patient may desire pregnancy and choose to pursue this. Alternatively, patients may be unaware that they have PH and may present with an initial diagnosis of PH during pregnancy. Below are recommendations for pregnancy management, separated by trimester and delivery, based in part on the European Society of Cardiology guidelines on pregnancy in cardiovascular disease. 224
Therapeutic abortion
Because of the excessive risk to the mother and fetus discussed in “Basic biology of PH and pregnancy,” when pregnancy occurs in PH patients, termination should be offered regardless of WHO FC or other markers of prognosis. 225 Discussion of therapeutic abortion, using a shared-decision model, has been proposed to establish a common set of goals and optimize maternal medical care. 225 The first trimester is the safest time for elective pregnancy termination; however, in the PH patient, pregnancy termination carries greater risk than in the general population and should be performed in an experienced center.224,226 Uterine dilatation and evacuation is the safest procedure. 227 If surgical evacuation is not feasible, medical abortion using prostaglandins E1 or E2 or misoprostol, a synthetic prostaglandin structurally related to prostaglandin E1, can be administered to evacuate the uterus. 227 These drugs are absorbed into the systemic circulation and can lower SVR and blood pressure and increase heart rate; these effects are greater with prostaglandin E2 than with E1. 227
While therapeutic abortion is most commonly performed during the first trimester in the pregnant PH patient, it should also be considered in the second trimester up to the point of fetal viability. After that point, early delivery may be considered if clinically indicated. Different cultural and/or religious preferences and legal obligations are present in the diverse countries of pregnant PH patients. These factors may influence timing, acceptability, and options for therapeutic abortion.
First trimester
Despite strong recommendations for therapeutic abortion, some pregnant PH patients may decide to continue pregnancy. In this case, management should be provided by a multidisciplinary team in a center experienced in the treatment of not just PH patients but also pregnant PH patients. At the minimum, the team should include a PH specialist, a cardiologist, an obstetrician, an anesthetist specialized in managing high-risk pregnancies, and a neonatologist.195,224–226 Pharmacists, intensivists, and social workers are frequently needed as well. In addition, in the case of associated forms of PAH, specialists in the underlying disease should be involved. Patient-specific multidisciplinary management plans should be put in place. 195
A careful baseline assessment of pregnancy risk during this trimester should include history and physical examination, echocardiography, brain natriuretic peptide measurement, and 6-minute-walk testing. A discussion of PAH-directed therapy in pregnancy follows in “Role for PAH-directed therapy in pregnancy and delivery.” Patients should be followed closely, typically monthly, but assessment frequency should be tailored to individual patients and their risks. During the first trimester, the volume changes associated with pregnancy are less pronounced than those in future trimesters; thus, edema and swelling symptoms should trigger an evaluation of PH status. As discussed above in “Physiologic changes of pregnancy/concerns in PH,” hormonal changes associated with early pregnancy may alter pulmonary vascular disease and precipitate worsening. Even in the absence of symptoms, regular assessment of PH status is advisable, using, in addition to physical examination, echocardiography, brain natriuretic peptide, 6-minute-walk testing, or a combination of all at regular intervals during this trimester. Management of PAH-specific therapy is discussed in “Genetic counseling in PH.”
Second trimester
As discussed in “Physiologic changes of pregnancy/concerns in PH,” a major change in the second trimester is volume expansion and increase in CO, which is achieved in part through vasodilation and recruitment of the pulmonary vascular bed. In most forms of precapillary PH (WHO groups 1, 3, 4, and 5), the diseased pulmonary vasculature cannot accommodate the increased CO. Patients with group 2 PH may also have difficulty accommodating increased flow though the pulmonary vasculature. As a consequence, RV volume loading and subsequent failure may occur. A dilated RV can, in turn, reduce left heart filling, thus further compromising CO. As a function of these volume demands, decompensation often occurs in the second trimester, corresponding to peak blood volume and CO. Vigilant follow-up though the previously discussed multidisciplinary team is essential, and frequency of visits may have to be increased to every 2 weeks, for example. As in the first trimester, follow-up evaluation should include history and physical examination, 6-minute-walk testing, and brain natriuretic peptide measurement. Echocardiography frequency may be increased to every 2 weeks if concerns arise during this trimester of worsening right heart function.
In patients who have developed right heart failure symptoms, standard therapy including diuretics, oxygen therapy if indicated, and, for patients with severe symptoms or failure of outpatient therapy, hospitalization may be considered. In patients requiring diuretics, furosemide is preferred, as spironolactone has antiandrogenic properties and is pregnancy category D. As with all right heart failure, judicious fluid restriction, low-salt diet, and oxygen supplementation are indicated where appropriate. To avoid caval vein compression, the patient should be advised to lie in the lateral position. Management of PAH-specific therapy is discussed below in “Role for PAH-directed therapy in pregnancy and delivery.”
Third trimester
As the major volume expansion has occurred in the second trimester, the third trimester's main concerns are preparation for delivery and ensuring continued safety of the mother and fetus. In the third trimester, weekly follow-up is suggested (Fig. 4). In this trimester, echocardiograms are recommended every 1–2 weeks to monitor the cardiac status and adjust PH medications (Fig. 4). As in the second trimester, important nonpharmacologic therapy involves limiting sodium and fluid intake during pregnancy. Limiting daily fluids to 1.5–2 L, restricting sodium intake, and administering diuretics in the third trimester can reduce RV overdistention and avoid cardiac decompensation. Elective cesarean section is advisable when the fetus becomes viable. A multidisciplinary approach has been reported to lead to a favorable maternal-fetal outcome. 195 Close communication between high-risk obstetrician, anesthesiologist, and PH physician help in the proper and timely management of these high-risk patients. 195
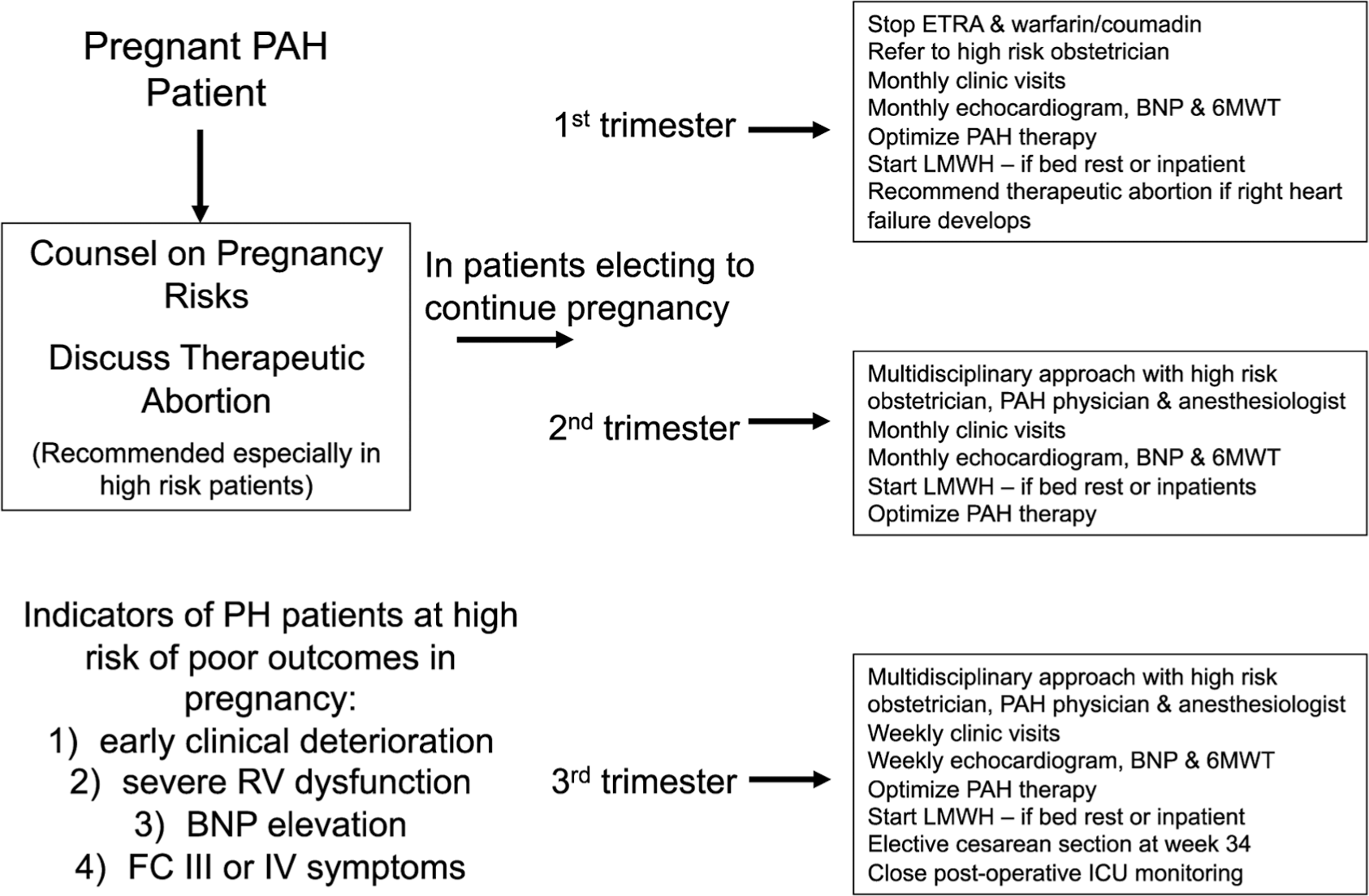
Recommended evaluation of and follow-up for a pregnant patient with pulmonary arterial hypertension (PAH). BNP: brain natriuretic peptide; ETRA: endothelin receptor antagonist; FC: World Health Organization function class; ICU: intensive care unit; LMWH: low-molecular-weight heparin; PH: pulmonary hypertension; RV: right ventricular; 6MWT: 6-minute walk test.
Delivery
The mode of delivery should be carefully selected in the pregnant PH patient, as it may influence the outcome. Cesarean section is the preferred mode of delivery. 224 Although vaginal delivery is usually associated with fewer bleeding complications and infections in the healthy population, 229 the hemodynamic and physiological changes associated may be detrimental to the mother with PH. The advantages and disadvantages of different modes of delivery are discussed below.
Monitoring and general considerations during delivery
During labor and delivery, continuous monitoring of electrocardiogram, pulse oximetry, central venous pressure, and intra-arterial blood pressure should be routine. 224 While PA catheterization may be of benefit in selected cases, there is no consensus regarding its routine use during delivery. 21 Several centers manage patients with central lines and close monitoring of RV function (e.g., by echocardiography). The use of noninvasive hemodynamic monitoring in this setting may be of benefit but has not yet been validated. Close attention must be paid to avoiding conditions that may lead to PA vasoconstriction and worsening RV function; these include hypoxia, hypercarbia, and acidosis. Hemodynamically relevant arrhythmias should be aggressively corrected. Anxiety- and pain-induced catecholamine release may negatively affect hemodynamics; the use of different anesthetic methods is discussed in detail below. In order to optimize oxygen delivery as much as possible, significant anemia should be corrected. The patient should be as euvolemic as possible, and major fluid shifts must be avoided as much as possible. Vasopressors and inotropes should be readily available for hemodynamic support. Intravenous prostacyclins should be readily available; however, the role and management of PAH-specific medications during delivery is discussed in detail in “Role for PAH-directed therapy in pregnancy and delivery.” Individualized management plans for each patient must be discussed and updated before delivery.
Vaginal delivery
This mode of delivery is usually avoided in PAH patients, as it carries a multitude of disadvantages. First, vaginal delivery is associated with frequent use of the Valsalva maneuver, which increases intrathoracic pressure and thus decreases venous return. As the CO in PH patients requires adequate preload, the Valsalva maneuver may critically and rapidly decrease preload and thus precipitate cardiopulmonary collapse. Second, labor-induced vasovagal responses can decrease the venous return and lead to cardiopulmonary collapse. Third, the pain of childbirth may lead to sympathetic nervous system stimulation. As a result, there may be an increase in heart rate and increases in systemic and pulmonary vascular tone, with resultant hemodynamic instability. Labor-induced acidosis, hypercapnea, or hypoxia may further contribute to increases in PA pressures. After delivery of the fetus, an increase in maternal blood volume occurs because of autotransfusion of blood from the contracting uterus and from shifting of peripheral edema from the extravascular compartment into the systemic vasculature. In addition, venous return may increase from the relief of inferior vena cava obstruction by the gravid uterus. Finally, agents used to induce labor may precipitate clinical deterioration. Low-dose oxytocin can be used with caution to induce labor, as it can increase PVR. Because of the potential for hemodynamic instability due to the aforementioned factors, vasopressor or inotropic support may be required. If vaginal delivery is chosen, effective analgesia is imperative. Nitrous oxide should be avoided because of its potential to vasoconstrict the pulmonary vasculature, 230 and epidural analgesia should be considered.
Cesarean section. Cesarean section is the preferred mode of delivery and should be used unless not available or in cases of emergencies.22,195 Cesarean section bypasses the hemodynamic complications associated with labor discussed above and also the autotransfusion associated with vaginal contractions. Elective cesarean section avoids labor and allows for careful, multidisciplinary planning and preparation of anesthesia, optimization of hemodynamics, and development of contingency plans. Moreover, cesarean section does not appear to negatively influence patient outcomes. 197 The choice of anesthesia is important when cesarean section is being considered and should be preplanned during early multidisciplinary meetings with the anesthesiologist, who should be aware of the hemodynamic changes associated with PH (often an obstetric or cardiac anesthesiologist), and with an experienced maternal-fetal medicine specialist. The aim of the anesthesiologist is to achieve an optimum balance between anesthesia and side effects. Most obstetric units would consider regional anesthesia in preference to general anesthesia for this procedure. This will reduce risks associated with general anesthesia (e.g., endotracheal intubation and positive-pressure ventilation, which may increase PVR and RV afterload). However, a regional approach is not always possible, and plans should be made to proceed with general anesthesia if necessary.
Timing of delivery. The optimal timing of planned elective delivery in women with PAH is uncertain and involves an assessment of the maternal risk of continuing pregnancy and risk of neonatal preterm delivery. In stable women, planned delivery around weeks 34–36 is recommended, with delivery before this if there is evidence of symptomatic decline.
Anesthesia in pregnant PH patients
General anesthesia. General anesthesia is known to depress cardiac contractility, increase PVR via positive-pressure ventilation, and increase PAP during laryngoscopy and intubation. 231 Studies by Bédard and others 21 suggest that parturients who receive general anesthesia were more likely to die than parturients receiving regional anesthesia. However, caution should be shown when interpreting these results, as selection bias may be shown toward the sicker parturient receiving general anesthesia. If general anesthesia is required, the patient should be ventilated with the lowest airway pressure and tidal volume possible. A lung-protective ventilation strategy similar to what is used in patients with acute respiratory distress syndrome has been advocated. 232 However, care must be taken to avoid hypercarbia. Catecholamines and iv prostacyclins should be readily available for these patients, and the administration of catecholamines before anesthesia induction should be considered.
Epidural or combined spinal-epidural anesthesia. Epidural anesthesia with incremental doses has traditionally been considered the best approach to regional anesthesia. However, with the advent of modern equipment and techniques, low-dose combined spinal-epidural anesthesia is being increasingly used, because it provides a denser perineal sensory block than epidural anesthesia alone, with little additional risk of hypotension. 233
Spinal anesthesia. Single-dose spinal anesthesia should be avoided in this patient population because of the risk of a rapid rise in block height that may cause uncontrollable hemodynamic instability as sympathetic block rises and systemic hypotension occurs. 22
Postpartum and long-term effects
In the hours and days after childbirth, blood volume is increased by autotransfusion of blood from the contracting uterus and by shifting of peripheral edema from the extravascular compartment into the systemic vasculature. In healthy subjects, these physiologic changes are normally well tolerated, but in the presence of severe pulmonary vascular disease, hemodynamic instability may develop, leading to cardiorespiratory failure and/or sudden death.194,224 Parturition and the first postpartum week have been recognized as particularly vulnerable periods for patients with PAH.23,194 Two systematic literature reviews have described the outcome of pregnancy in women with PH, covering a total of almost 30 years and comprising 198 pregnancies. The mortality of women with Eisenmenger's syndrome (N = 102) was 36% in the first review (1978–1996) 5 and 28% in the second review (1997–2007) 21 . Most of these women died in the first month after delivery, and the main causes of death were heart failure and sudden death, while pulmonary thromboembolism was another frequent cause. In women with IPAH (N = 56), mortality was 30% in the first 4 and 17% in the second 21 review. Similarly, nearly all fatalities in these patients occurred early in the postpartum period, and death was mainly due to heart failure, followed by sudden death and thromboembolism. Thus, the immediate postpartum period is a critical period for acute decompensation in PH patients. Patients should be closely monitored for several days postpartum; monitoring in an intensive care unit in the first few days after delivery is recommended. This is especially pertinent to high-risk patients, particularly those with right heart failure or evidence of hemodyanmic instability during delivery. Prophylactic anticoagulation is important in this period (see “Role for anticoagulation in the pregnant PH patient”). 234 As the volume expansion that occurs in pregnancy is sustained for up to 24 weeks postpartum, improvement in functional status after delivery may take several months.
Detrimental effects of vagal reactions in pregnant patients with PH
Vagal reactions and syncope can have catastrophic consequences in PH patients independent of the presence or absence of pregnancy, because they may be associated with a profound drop in CO. 235 However, in pregnant PH patients, the presence of a vagal reaction is particularly concerning, as the RV is extremely preload dependent and compensatory mechanisms are even more limited than in nonpregnant PH patients. It is therefore of utmost importance that potential inducers of vagal stimuli be recognized and avoided.
Vasovagal syncope is characterized by a temporary reduction of blood flow to the brain that is caused by sudden hypotension, change in heart rate, and/or change in blood volume or its distribution. 236 In general, because of their vasodilatory state, pregnant women are more prone to vasovagal syncope than their nonpregnant counterparts. 237 Since PH patients are more prone to syncope than healthy individuals, 235 the combination of pregnancy and PH therefore creates a “perfect storm,” generating a condition where (1) vasovagal syncope is more prevalent and (2) its consequences are particularly dangerous. Common inducers of vagal reactions during pregnancy include vena cava compression, rapid positional changes (especially getting up too fast), pain, anxiety, straining while pushing during vaginal delivery, hypovolemia, anemia, hyperthermia, and hyperventilation. Iatrogenic causes include administration of vasodilators, interventions that induce vasodilation and decrease venous return (e.g., induction of general or spinal anesthesia), manipulation of the cervix, and any procedure that causes pain and/or anxiety. Common preceding symptoms of vasovagal syncope include nausea, diaphoresis, lightheadedness, and pallor.
Because of the potential catastrophic consequences of vasovagal syncope in the pregnant PH patient, it is of utmost importance that triggers of such events be avoided as much as possible. If the trigger is unavoidable or has already occurred, rapid and focused intervention is critical. Counseling of the pregnant PH patient on inducers and symptoms of vasovagal syncope, as well as on appropriate interventions, is of highest importance. Similarly, medical professionals need to be aware of interventions known to raise vasovagal tone, and they need to be educated that PH patients are particularly susceptible to such reactions. If patients undergo any procedure that may induce vagal reactions, such as placement of an IUD or therapeutic abortion, physicians should place 2 iv lines and have atropine in the procedure room ready to administer.
Summary of recommendations
Pregnancy in PAH patients is associated with a high maternal mortality rate. The highest-risk periods are the peripartum period and the immediate postpartum period (up to 2 months) (CB).
A multidisciplinary approach with high-risk obstetricians, PH physicians, cardiologists, anesthesiologists, and neonatologists is recommended (CB).
Close clinical monitoring, including monthly follow-up visits in the first and second trimesters and weekly clinic visits in the third trimester, is recommended (CB).
All pregnant PH patients should be offered pregnancy termination. In particular, patients with worsening right heart failure, especially early in their pregnancy, and patients with other high-risk features should be given the option to have their pregnancy terminated. If patients want to continue pregnancy, augmentation of PAH therapy should be considered to improve the chances of a favorable outcome (CB).
Cesarean section is the preferred mode of delivery and should be used unless not available or in cases of emergencies (CB).
Epidural or spinal-epidural anesthesia is recommended rather than general anesthesia (CB).
Because of the potential for catastrophic consequences, pregnant PH patients and their providers should be counseled on inducers and symptoms of vasovagal syncope. Triggers of vasovagal syncope should be avoided (CB).
Role for PAH-directed therapy in pregnancy and delivery
Patients may present for the first time in pregnancy with PAH, or women with known PAH may actively plan pregnancy despite appropriate counseling regarding the high risks of maternal death. Finally, PAH patients may accidentally become pregnant. If the woman chooses to continue pregnancy, there is emerging literature reporting on the use of specific PAH therapies in pregnancy.23,194–199 Three classes of drugs are currently available for PAH treatment. Calcium channel blockers have also been used when appropriate.
Prostaglandins
Prostaglandins are potent pulmonary vasodilators that may also act to enhance RV function.238,239 Currently available parenteral prostaglandins—epoprostenol, treprostinil, and iloprost—are not known to be teratogens (Table 5) and are listed as pregnancy category B (epoprostenol and treprostinil) or C (iloprost).
FDA-assigned risk category for PAH medications

Note: FDA: US Food and Drug Administration; PAH: pulmonary arterial hypertension.
B: animal reproduction studies have failed to demonstrate a risk to the fetus, and there are no adequate, well-controlled studies in pregnant women; C: animal reproduction studies have shown an adverse effect on the fetus, and there are no adequate, well-controlled studies in humans, but potential benefits may warrant use of the drug in pregnant women despite potential risks; X: studies in animals or humans have demonstrated fetal abnormalities, and/or there is positive evidence of human fetal risk based on adverse-reaction data from investigational or marketing experience, and the risks involved in use of the drug in pregnant women clearly outweigh potential benefits.
They may thus be continued in patients who receive this class of medication before pregnancy, if treating physicians and patients agree that the benefits outweigh the relatively unknown risks. Prepregnancy use of prostaglandins, however, may be an indicator of a patient with more severe disease. In addition to the usual indications to begin prostaglandin therapy, such as WHO FC III or IV symptoms and reduced RV function,14,240,241 other considerations in pregnancy may compel therapy with parenteral prostaglandins. Examples are worsening symptoms or deteriorating RV function despite therapy with phosphodiesterase inhibitors and nonparenteral prostanoids. It should be recognized, however, that even with parenteral prostanoids, maternal mortality may still be significant. 202 In many centers, iv prostanoids are managed exclusively by the PH specialist; this includes accompanying the patient to the operating room.
In general, parenteral prostaglandins are recommended for pregnant patients with PAH in WHO FC IV or where there is evidence of severe RV impairment. In selected patients in WHO FC III followed in specialized centers, a trial of therapy with nebulized iloprost and addition of sildenafil has been successfully reported. 195 When delivered by the parenteral route, gradual up-titration can be performed as per the usual protocol. Inhaled iloprost or treprostinil may be used in patients with less severe symptoms, and their safe and successful use in pregnancy has been reported.195,198 However, in a patient experiencing deterioration while receiving inhaled-prostaglandin therapy, rapid conversion to parenteral forms (some publications have advocated for iv epoprostenol 242 ) should be considered. If there is no rapid clinical improvement, immediate delivery should be considered because of the high risk of maternal death. In addition, inhaled epoprostenol or nitric oxide 243 has been administered continuously to acutely ill patients as a bridge to parenteral treatment initiation.
Phosphodiesterase 5 inhibitors
Both sildenafil and tadalafil are pregnancy category B (Table 5). There are several reports of successful pregnancies managed with sildenafil either alone or in combination with a prostaglandin.23,193,194,244,245 The use of tadalafil in pregnancy has not been reported. In larger case series of successful pregnancies, sildenafil has generally been used in combination with a prostaglandin.23,194,195 As there is no published experience with tadalafil, we recommend a preference for sildenafil when phosphodiesterase inhibitors are used in pregnancy. Their use, however, should be restricted to either combination therapy with a prostaglandin or, if as a monotherapy, restricted to patients who have normal RV function and near-normal pulmonary hemodynamics or who refuse prostaglandin therapy. If phosphodiesterase inhibitor monotherapy is used, close follow-up for deterioration is indicated.
Calcium channel blockers
PAH patients who have an acute response to inhaled vasodilators such as nitric oxide (defined by a drop in mPAP by 10 mmHg to 40 mmHg or less with preserved CO) are candidates for treatment with calcium channel blocker therapy and have a substantially improved prognosis, compared to patients without an acute vasodilator response. 246 Durable response to calcium channel blocker therapy during pregnancy has been described. 23 In fact, among the patients included in the only prospective registry of pregnant PAH patients, 8 of 16 patients who successfully delivered a baby and survived without transplantation were acute vasodilator responders. 23 Calcium channel blockers are considered safe in pregnancy. 247 For those patients meeting strict criteria for an acute response to inhaled nitric oxide, we recommend therapy with calcium channel antagonists14,246 and close follow-up, as discussed above. In pregnant PAH patients not meeting these strict criteria, there is no role for calcium channel antagonists. In this setting, calcium channel blockers may be detrimental because of their nonselective vasodilatory effects, with a concomitant drop in SVR, which, in the presence of an inadequate CO, may lead to circulatory collapse.
Endothelin receptor antagonists
Endothelin receptor antagonists (ambrisentan, bosentan, macitentan, and sitaxentan) are pregnancy category X (Table 5) and should be discontinued when a pregnancy is discovered in a patient with PAH, because of the known teratogenicity of these medications.
Soluble guanylate cyclase stimulator
Riociguat is pregnancy category X and should be discontinued when a pregnancy is discovered in a PAH or chronic thromboembolic PH (CTEPH) patient.
Summary for PAH-directed therapy during pregnancy
The advent of new therapies for PAH provides the clinician and the patient with a number of choices during pregnancy. These include the timing of treatment initiation in treatment-naive patients, the type of drug to be used, and the mode of drug delivery. Varied approaches in PH centers experienced in the management of PH in pregnancy reflect individual center experience, and where clear evidence of the benefit of one approach over another is lacking, pragmatic approaches will reflect local experience. 195 It should be recognized that, despite improved outcomes and potential theoretical benefits of various treatment approaches, the medical treatment of PAH patients should be carefully chosen and individualized to take into account the severity of illness, comorbidities, social and cultural aspects, patient's wishes, and long-term goals.
Special considerations at delivery
The approach to the use of PAH therapies around the time of delivery will be influenced by the therapies that the patient has been taking during her pregnancy, her stability before planned delivery, and her progress in the peripartum period. When titrating these therapies, one needs to appreciate the physiological changes that are normally occurring in pregnant women and to be receptive to possible worsening of the pulmonary vascular disease and the effects of drugs used in the peripartum period. Oxytocic drugs, for example, can induce hypotension and tachycardia and should always be given by slow infusion.
Key to allowing titration of these therapies is careful monitoring of patients during the peripartum period in a high-acuity environment. Insertion of a central venous catheter to allow measurement of central venous pressure and sampling of blood to assess central venous saturation and insertion of an arterial line for measurement of blood pressure and blood gas sampling are highly recommended. 248 Swan-Ganz catheterization is not routinely used because of the risk of complications, but it can be considered in highly selected cases. Noninvasive measures of CO, such as lithium dilution, can be used to titrate treatment in patients with PAH.195,248,249 This can allow a continuous measure of CO in addition to displaying systemic pressures, heart rate, and other core variables. However, its use has not been validated in prospective studies.
For stable patients receiving iv prostanoids, doses would usually remain unchanged before and during delivery. For patients not receiving parenteral prostanoids, a low-dose infusion of iv prostanoid started several hours before a planned cesarean section would allow the physician to assess the hemodynamic effects of this infusion and avoid problems associated with initiating prostanoids in the immediate peripartum period, when, in an unstable patient, it may be difficult to discern side effects of the drug from pulmonary hemodynamic instability. In these cases, a Swan-Ganz catheter may help assess the therapeutic effects of infused prostacyclins. If patients are receiving oral sildenafil, consideration should be given to using the iv preparation of the drug, particularly if there are any concerns regarding absorption, which can occur postoperatively, for example, following the development of ileus after cesarean section. For surgery under regional anesthesia, patients receiving nebulized iloprost or treprostinil therapy may continue to nebulize during the procedure, in addition to receiving a low-dose iv infusion during the peripartum period. Alternatively, nebulized epoprostenol may be substituted for intermittent inhalation of iloprost or treprostinil.
The treatment goals during delivery are to maintain systemic and right atrial pressures, to monitor fluid balance, and to avoid volume overload, particularly in the first 48 hours. A rising right atrial pressure after delivery may simply reflect fluid overload and can be managed by judicious use of diuretic therapy. If a patient's condition deteriorates with falling central venous saturation, rising right atrial pressure, and other features suggesting worsening of pulmonary vascular status, then increasing the dose beyond the background dose of iv prostanoid is usually appropriate, sometimes coupled with inotropic therapy, such as low-dose dobutamine. For patients developing hypotension, a cause should be sought, such as bleeding, sepsis, or low-output heart failure due to increasing afterload. In this setting, once volume status has been optimized, drugs such as vasopressin or noradrenaline should be considered. 250
While the treatment recommendations above assume access to PAH therapy, in locations where the full complement of PAH-directed therapy is not available, supportive care should be instituted. Such care is discussed in “Pregnancy management” above.
Summary of recommendations
In patients who are in WHO FC IV or have evidence of severe RV impairment, parenteral prostaglandins are recommended. Most published experience in parenteral prostaglandins is with iv epoprostenol (CB).
In select patients with more preserved RV function who are in WHO FC III, inhaled prostaglandins may be considered. Most published experience in inhaled prostaglandins is with iloprost. An appropriate treatment response needs to be verified (CB).
Oral phosphodiesterase 5 inhibitors may be considered in patients who are in WHO FC I or II and who have normal RV function. Most published experience is with sildenafil. Close follow-up for deterioration is highly recommended for patients treated with phosphodiesterase 5 inhibitor monotherapy (CB).
Parenteral prostaglandins can be combined with oral phosphodiesterase inhibitors in pregnancy. Successful outcomes have been reported with this regimen (CB).
For patients meeting strict criteria for vasodilator-responsive PAH who are not in WHO FC IV and who do not have RV dysfunction, calcium channel blocker therapy may be continued in pregnancy, with close follow-up for deterioration (CB).
The currently available endothelin receptor blockers and soluble guanylate cyclase stimulator are pregnancy category X and should not be used in pregnancy. If a PAH patient who is taking one or more of these medications becomes pregnant, their use should be immediately discontinued (CB).
At the time of delivery, iv prostaglandins may be considered in patients not already treated with this class of medication (CB).
At the time of delivery, PAH patients require close monitoring, with a central venous catheter, an arterial line, and careful attention to volume status (CB).
Close monitoring is recommended for all pregnant PAH patients, with routine evaluation by history, physical examination, imaging (echocardiography) and laboratory testing as appropriate for deterioration (CB).
Routine monitoring with Swan-Ganz catheterization is not recommended in the peripartum period because of the risk of complications. Other techniques to noninvasively assess CO in the peripartum period, such as lithium dilution, can be considered (CB).
Role for anticoagulation in the pregnant PH patient
Anticoagulation is recommended for many forms of PAH, including IPAH, HPAH, and some forms of congenital heart diseases–associated PAH,14,224,240,241 in particular for patients with CTEPH (WHO group 4). Low-molecular-weight heparin is usually used in patients who receive chronic anticoagulation, and prophylactic heparin is recommended for all PAH patients in the peripartum period. When low-molecular-weight heparin is used during pregnancy, it may be switched to unfractionated heparin during the preparation for delivery to facilitate rapid discontinuation when indicated. CTEPH patients should continue anticoagulation during pregnancy. Warfarin is a known teratogen and is contraindicated in pregnancy. New oral anticoagulants (e.g., dabigitran, rivaroxaban, and apixaban) are pregnancy category C, and their use in PAH and CTEPH has not been studied; thus, they are not recommended.
Summary of recommendations
If a PAH patient has been receiving anticoagulation therapy before pregnancy, the risks and benefits of this therapy should be reevaluated and discussed at the time of pregnancy (CB).
Warfarin should be stopped and either unfractionated or low-molecular-weight heparins used if anticoagulation is continued (CB).
Prophylactic heparin is recommended in the peripartum period (CB).
New oral anticoagulants are currently not recommended for PAH or CTEPH (CB).
FUTURE DIRECTIONS
There has been tremendous progress in the field of pulmonary vascular disease in the past several decades, which has led to newer therapies, better mechanistic understanding of disease, and improved outcomes. 190 With continued research and experience, there is hope for even greater advances in the future. Major areas of research currently include the effects of sex hormones on PH and the mechanisms of PH in non–group 1 disease. The effects of pregnancy on the pulmonary vasculature and RV, particularly in non–group 1 PH, should be a major focus of research, given the burden of this disease on young women, children, and families across the world. Another area of uncertainty as outcomes improve in this disease is the recommendation for permanent sterilization in PH, as more efficacious therapy may be available within a younger PAH patient's years of potential fertility. Finally, prospective evaluation of management strategies for pregnant PAH patients is needed in order to optimize diagnostic and therapeutic strategies, although such trials are unlikely to occur, given the relative infrequency of pregnancy in this rare condition.
CONCLUSIONS
Despite major advances in the care of PH patients and the advent of several classes of medications to treat this condition, PH in general and PAH in particular remain highly mortal, and pregnancy is poorly tolerated. This document summarizes the current knowledge on pregnancy in PH, with a focus on PAH, as the greatest amount of literature is available for this disease state. While we continue to strongly recommend avoidance of pregnancy in patients with PH, there are situations in which patients may present in pregnancy or become pregnant, and it is our hope that this document will serve as a useful reference to practitioners caring for the pregnant patient with PH. In the future, research into non–group 1 PH, further understanding of the effects of sex hormones on the pulmonary vasculature and the right heart, and more effective treatment of pulmonary vasculature are promising avenues to improve the outcomes for the pregnant patient with PH.
