Abstract
Many dyspneic patients who undergo computerized tomographic pulmonary angiography (CTPA) for presumed acute pulmonary embolism (PE) have no identified cause for their dyspnea yet have persistent symptoms, leading to more CTPA scanning. Right ventricular (RV) dysfunction or overload can signal treatable causes of dyspnea. We report the rate of isolated RV dysfunction or overload after negative CTPA and derive a clinical decision rule (CDR). We performed secondary analysis of a multicenter study of diagnostic accuracy for PE. Inclusion required persistent dyspnea and no PE. Echocardiography was ordered at clinician discretion. A characterization of isolated RV dysfunction or overload required normal left ventricular function and RV hypokinesis, or estimated RV systolic pressure of at least 40 mmHg. The CDR was derived from bivariate analysis of 97 candidate variables, followed by multivariate logistic regression. Of 647 patients, 431 had no PE and persistent dyspnea, and 184 (43%) of these 431 had echocardiography ordered. Of these, 64 patients (35% [95% confidence interval (CI): 28%–42%]) had isolated RV dysfunction or overload, and these patients were significantly more likely to have a repeat CTPA within 90 days (P = .02, χ 2 test). From univariate analysis, 4 variables predicted isolated RV dysfunction: complete right bundle branch block, normal CTPA scan, active malignancy, and CTPA with infiltrate, the last negatively. Logistic regression found only normal CTPA scanning significant. The final rule (persistent dyspnea + normal CTPA scan) had a positive predictive value of 53% (95% CI: 37%–69%). We conclude that a simple CDR consisting of persistent dyspnea plus a normal CTPA scan predicts a high probability of isolated RV dysfunction or overload on echocardiography.
Keywords
In the past decade, multidetector-row computerized tomographic pulmonary angiography (CTPA) has become the imaging mainstay in the diagnosis and exclusion of acute pulmonary embolism (PE). CTPA can also disclose clinically important alternative diagnoses, most often pulmonary infiltrates suggestive of pneumonia.1,2 However, CTPA is not without harm and has been known to cause adverse effects, including radiation exposure, contrast nephropathy, and false positive diagnoses.3–10 Despite these risks, the rate of CTPA recidivism has increased in recent years. For example, in one study, almost 40% of patients who had CTPA in the years 2000–2001 underwent a second CTPA scan that was negative for PE within the next few years. 11
Approximately 70% of patients who undergo CTPA scanning have dyspnea as a chief complaint, and many of these patients continue to have dyspnea despite normal imaging studies and a negative emergency department (ED) evaluation. 12 Persistent dyspnea is a major reason that many patients return to an ED and often receive a repeat CTPA scan. 11 These data suggest that pathologies other than PE are responsible for persistent/recurrent symptoms and recurrent use of health care resources such as CTPA.
Pulmonary hypertension (PH) is a proliferative and sometimes obstructive vasculopathy defined by a mean pulmonary arterial pressure higher than 25 mmHg with a normal pulmonary arterial capillary wedge pressure. If left untreated, PH can cause right ventricular (RV) failure and death. PH has multiple etiologies, including idiopathic, hereditary, connective-tissue disease, HIV, congenital heart disease, and portal hypertension (collectively termed “pulmonary arterial hypertension” [PAH]), or it may occur as a secondary complication of treatable diseases, including left heart disease, chronic lung disease, and sleep-disordered breathing.13–17 However, although PH has major implications on morbidity and mortality, recent studies suggest that it is frequently overlooked and underrecognized.18,19 Early diagnosis and treatment improve outcomes. Since RV function determines functional status, exercise capacity, and outcomes in PH, and since RV dysfunction is commonly observed in PH patients, we hypothesized that a substantial portion of patients with persistent dyspnea, despite negative CTPA for PE, have unrecognized RV dysfunction or overload on echocardiography that contributes to persistent symptoms and CTPA recidivism.20,21 The identification of such patients would allow for the diagnosis and treatment of correctable etiologies (e.g., PAH, chronic heart or lung disease, chronic sleep-disordered breathing). In chronic obstructive pulmonary disease (COPD), RV dysfunction may precede the development of frank PH. 22
One prior study reported on a subset of ED patients with dyspnea in the PRIDE study who underwent echocardiography as part of standard care. 23 In this sample of 134 patients, the authors found that 20% had RV hypokinesis and 30% had moderate or severe tricuspid regurgitation. However, to our knowledge, no prior literature has examined the frequency of RV dysfunction or overload after negative CTPA. We therefore performed this study on patients with negative CTPA scanning and persistent dyspnea to derive a simple clinical decision rule (CDR) to predict a high probability of RV dysfunction or overload on echocardiography and to test whether RV dysfunction was associated with CTPA recidivism.
METHODS
Study design
This was a secondary analysis of a 4-center prospective, noninterventional study of diagnostic accuracy. The details of the methods have been published previously. 12 For the main analysis, we included only patients who had a CTPA negative for PE, had persistent dyspnea, and had transthoracic echocardiography performed within 1 week of enrollment.
Study setting and population
Patients undergoing CTPA scanning for suspected PE were prospectively enrolled at 4 academic medical centers in the United States: Carolinas Medical Center, Charlotte, North Carolina; Northwestern Memorial Hospital, Chicago, Illinois; Wake Forest University Baptist Hospital, Winston-Salem, North Carolina; and Baystate Hospital, Springfield, Massachusetts. All patients provided written informed consent. The study was approved by the institutional review boards of the 4 participating centers. The study protocol was registered on ClinicalTrials.gov before enrollment (identifier NCT00368836).
Experienced research coordinators initiated the screening process with discovery of an order entry for a CTPA scan from anywhere within the hospital, 6 days per week, 12 hours per day. Based on preliminary work that showed that CTPA scans were ordered in equal proportions for inpatients and ED patients, the screening process was designed to produce equal proportions of enrolled inpatients and outpatients. 24 Inclusion criteria required that patients had 1 of 15 signs or symptoms of PE and 1 of 21 known risk factors for PE (Table 1) and had a CTPA scan ordered. 6 Patients were excluded if they were unlikely to provide follow-up (e.g., imprisonment, homelessness, no telephone, history of noncompli-ance) or if they were hemodynamically unstable or in-tubated, had prior fibrinolytic treatment within 48 hours, had PE diagnosed within the last 6 months and were currently receiving systemic anticoagulation, had known active tuberculosis, or refused to give consent. Patients were enrolled within 24 hours of CTPA imaging.
Predictor variables: demographics and symptoms and comorbidit
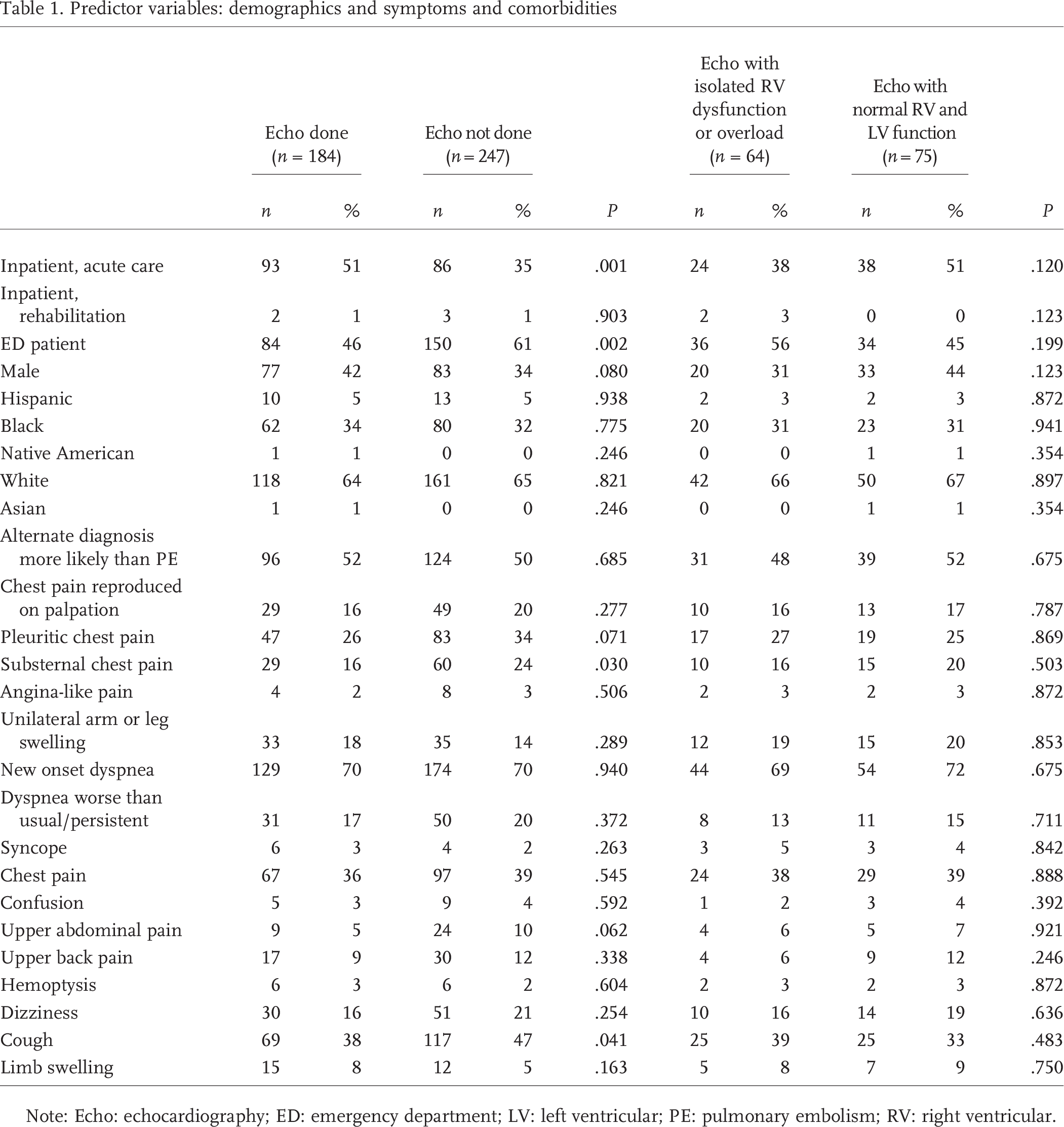
Note: Echo: echocardiography; ED: emergency department; LV: left ventricular; PE: pulmonary embolism; RV: right ventricular.
Study protocol
CTPA images were obtained at each site as part of standard care with 64-channel multidetector equipment, capable of ≤2.5-mm collimation. 6 Intravenous contrast was given to all patients according to local protocol; a computer-controlled mechanized timing injector was used in all cases. Images were obtained with energy, pitch, and rotation settings as required for the patient's body habitus.
Contiguous 1.25-mm-thick images were routinely reconstructed at mediastinal (width: 400 Hounsfield units [HU]; center: 0 HU) and lung (width: 1,600 HU; center: 600 HU) window settings. All patients had reconstructions that included transverse, coronal, and sagittal views. Images were converted to a digital file with a Digital Imaging and Communications in Medicine format, devoid of any annotations or protected health information, and were transferred to an independent reference reading (IRR) laboratory (Medical Metrics, Houston). Radiologists read the examinations on a standard picture-archiving and communication workstation and were free to modify their window settings as needed.
Clinical, breath, and blood data collection
To minimize missing or biased data and to simulate the knowledge held by decider clinicians, clinical data were obtained in real time at the bedside, as opposed to chart review, including all data in Tables 1 and 2. Each data field had an explicit definition, and coordinators were trained by the principal investigator (JK) according to an explicit set of standard operating procedures on how to collect them. Vital signs recorded were those closest in time to when the patient was enrolled, and all pulse oximetry readings were obtained with the patient breathing room air. Blood samples were drawn by qualified phlebotomists within 12 hours of CTPA scanning, and the D-dimer assay and fibrinogen concentrations were analyzed on instruments cleared by the US Food and Drug Administration. 12 For collection of breath data, including end-tidal CO2 and O2, and respiratory mechanical values (tidal volume, minute ventilation, inspiratory and expiratory flow rates and times, airway dead space volume, and oxygen and CO2 volume), patients breathed room air, wore a nose clip, and breathed into a duckbill mouthpiece for 1–2 minutes, as previously described. 24
Predictor variables: comorbidities
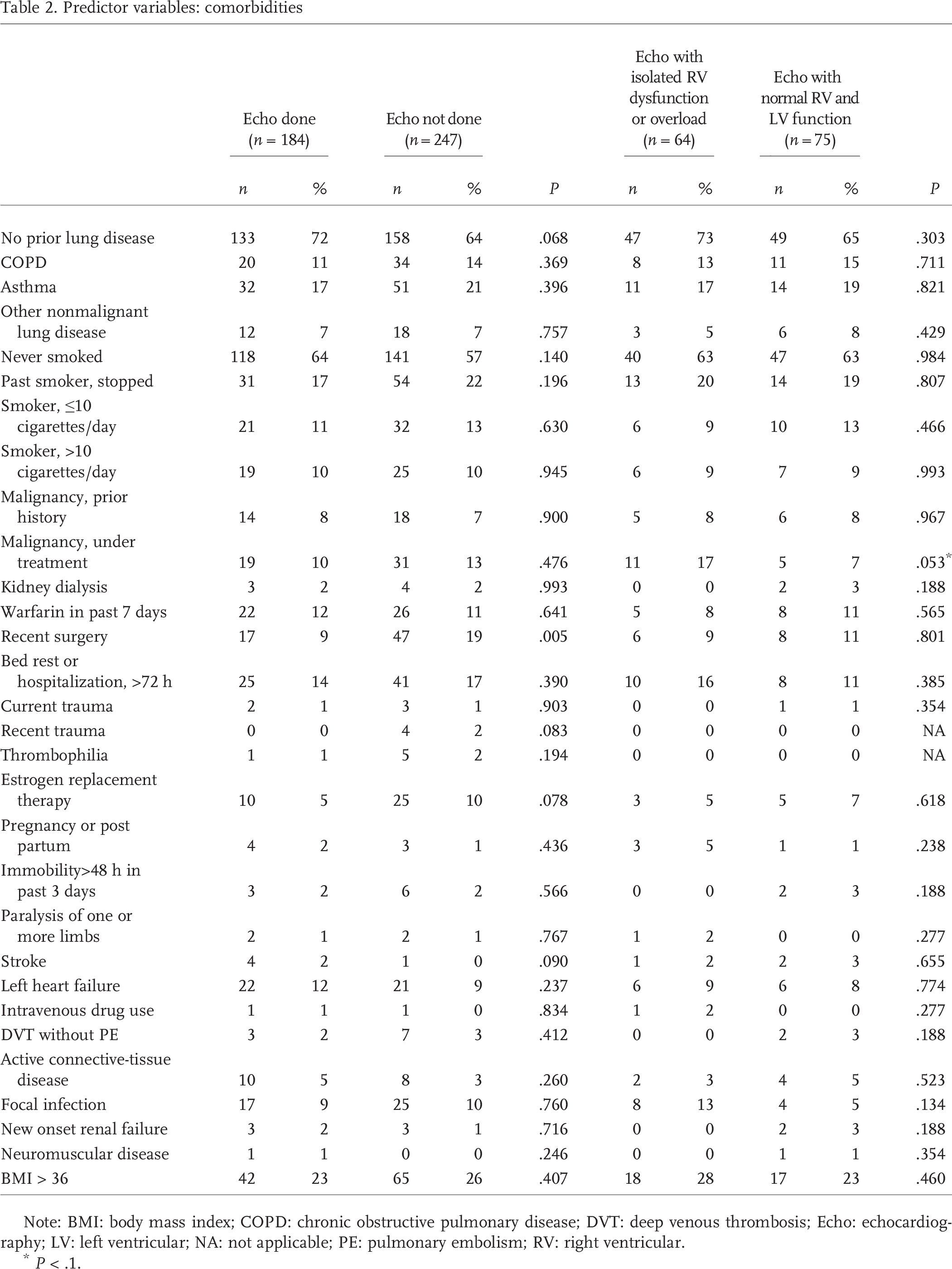
Note: BMI: body mass index; COPD: chronic obstructive pulmonary disease; DVT: deep venous thrombosis; Echo: echocardiography; LV: left ventricular; NA: not applicable; PE: pulmonary embolism; RV: right ventricular.
P < .1.
CTPA interpretation
Interpretations by the IRR laboratory (Medical Metrics), as well as the site interpretation, were integrated into this analysis. At the site hospitals, images were interpreted by board-certified hospital radiologists who had completed a fellowship in emergency radiology or body imaging. Later, at the IRR laboratory, images that were stripped of protected health information and any added comments or markings were interpreted by 1 of 2 board-certified radiologists who had completed fellowship training in body imaging. The IRR radiologists interpreted images as “no PE,” “positive for acute PE”, or “positive for chronic PE.” Chronic PE was characterized by 3 criteria: indistinct or fuzzy margins of the filling defect, adherence of clot to the vessel wall, and a webbed or “plexiform” appearance of the filling defect.25,26 “Positive for other finding” was further graded, including a specific code for pulmonary infiltrate suggestive of pneumonia (which had to be read both at the site and by the IRR) or parenchymal lung disease. Parenchymal lung disease was considered present if the IRR indicated findings of emphysema, fibrosis, scarring, or diffuse ground-glass appearance. Radiologists were not asked to systemically grade the severity of these findings. They could, however, make a comment about the finding, and many used the words “mild,” “moderate,” or “severe.” All CTPA scans were also evaluated for evidence of RV dilation, defined as a ratio of the RV diameter to the left ventricular (LV) diameter greater than 0.9 in transverse plane.27,28 The IRR radiologist also dichotomously recorded a field designated as “signs of pulmonary hypertension,” based on findings of enlarged diameter (RV/LV diameter ratio >0.9) with or without contrast reflux into the vena cava, dilated central pulmonary arteries (e.g., diameter of main pulmonary artery greater than that of ascending aorta), tortuous pulmonary arteries, vascular pruning, and/or a mosaic perfusion pattern, defined as sharply demarcated regions of variable attenuation without evidence of destruction or displacement of pulmonary vessels. 29 All parameters were weighed equally, and radiologists were allowed autonomy to put these findings together in making a dichotomous assessment. A normal CTPA scan was defined as having no significant pathological finding. Interpretations of CTPAs at each site were performed as part of standard care, and the final, written interpretation entered into the medical record was abstracted with a standardized approach to determine the site reading.
Echocardiography
Transthoracic echocardiograms were performed within 7 days after CTPA scanning at the discretion of the clinical care team. All echocardiograms were performed in IAC echocardiography-approved laboratories, using contemporaneous equipment, and were interpreted by board-certified cardiologists with specialty training in echocardiography. We included only results of echocardiograms that had adequate acoustic imaging to evaluate RV morphology and function. We abstracted data from the standard echocardiographic clinical report form. RV dysfunction or overload was defined as present with any one of the following: cardiologist qualitative interpretation of RV hypokinesis, dilation (defined as an end-diastolic diameter >35 mm in the apical 4-chamber view), or evidence of PH (peak tricuspid regurgitant jet velocity >2.7 m/s, obtained by continuous-wave spectral sampling from reliable waves). 30 Normal LV function required an ejection fraction greater than 45% and no evidence of diastolic dysfunction.
Clinical follow-up
All patients had telephone follow-up at 90 days. A scripted query was used, and a specific data collection template was designed to discover interval adverse events; diagnostic testing, including CTPA scanning with the results; and any change in treatment status.
Data analysis
The primary patient sample consisted of those with persistent dyspnea and no PE on CTPA. Persistent dyspnea was defined as the patient's statement that he or she had the sensation of shortness of breath while breathing room air at rest.
It could be expected that clinicians selected patients with more severe symptoms for echocardiography; therefore, the first analysis compares frequency and parametric clinical variables with a χ 2 test and an unpaired t test, respectively, between patients with and without echocardiography. Specifically, we compared the 97 clinical variables in Tables 1–4. To create the prediction rule, all candidate predictor variables were included and screened with univariate analysis that used a χ 2 test for frequency data and an unpaired t test for parametric data. Dichotomous variables with P < .1 were entered into multivariate logistic regression analysis. Parametric values with P < .1 were subjected to receiver operating characteristic (ROC) analysis to determine whether they had significant predictive value (i.e., lower limit of 95% confidence interval [CI] above 0.5). Logistic model fit was assessed by P values from the Pearson's goodness-of-fit χ 2 , the Hosmer-Lemeshow test, the pseudo-R 2 (McFadden), and the area under the ROC curve in the derivation data set (C statistic).31,32
Predictor variables measured on day of enrollment (continuous data)
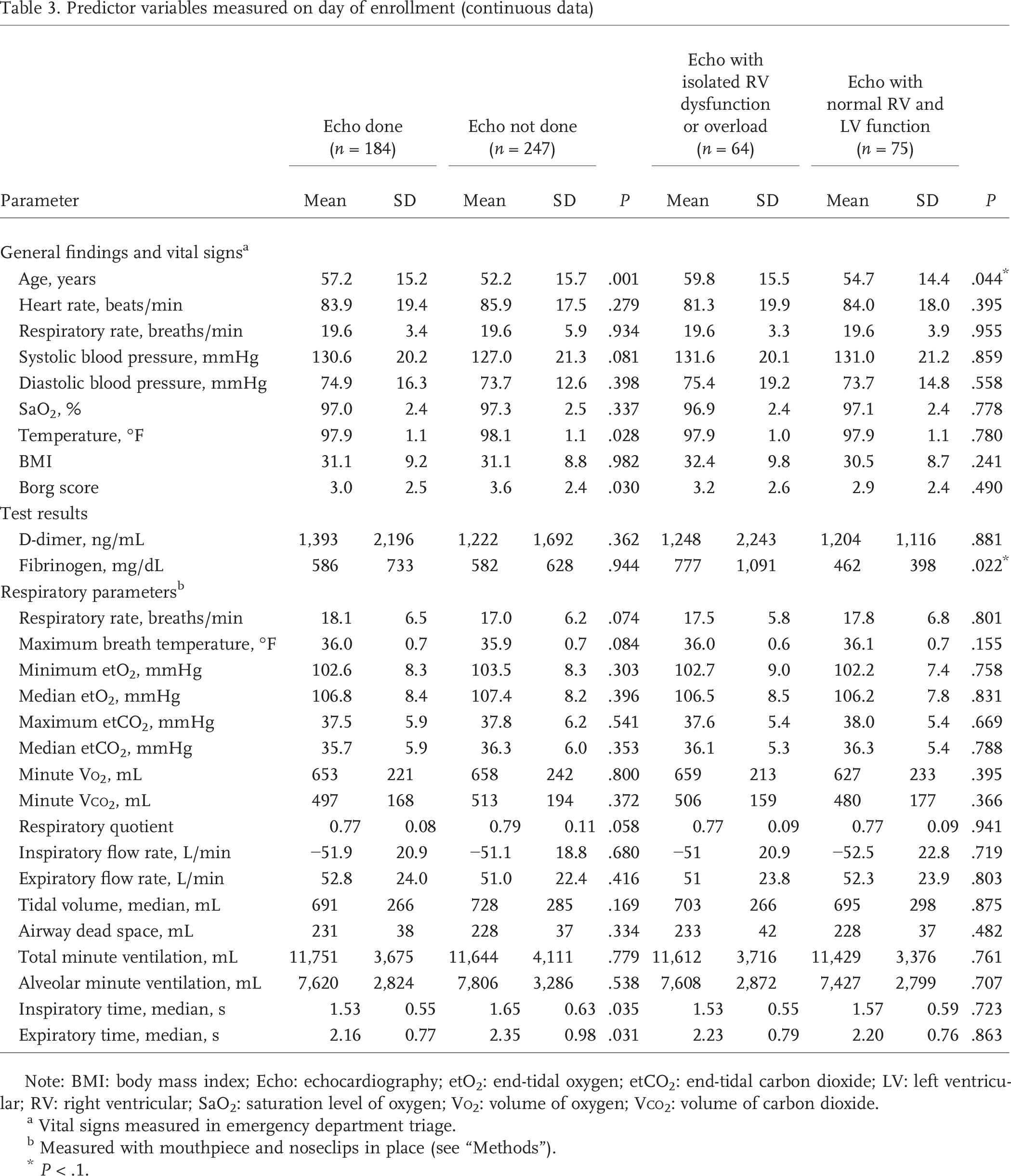
Note: BMI: body mass index; Echo: echocardiography; etO2: end-tidal oxygen; etCO2: end-tidal carbon dioxide; LV: left ventricular; RV: right ventricular; SaO2: saturation level of oxygen; Vo2: volume of oxygen; Vco2: volume of carbon dioxide.
Vital signs measured in emergency department triage.
Measured with mouthpiece and noseclips in place (see “Methods”).
P < .1.
Predictor variables measured on day of enrollment (bivariate data)
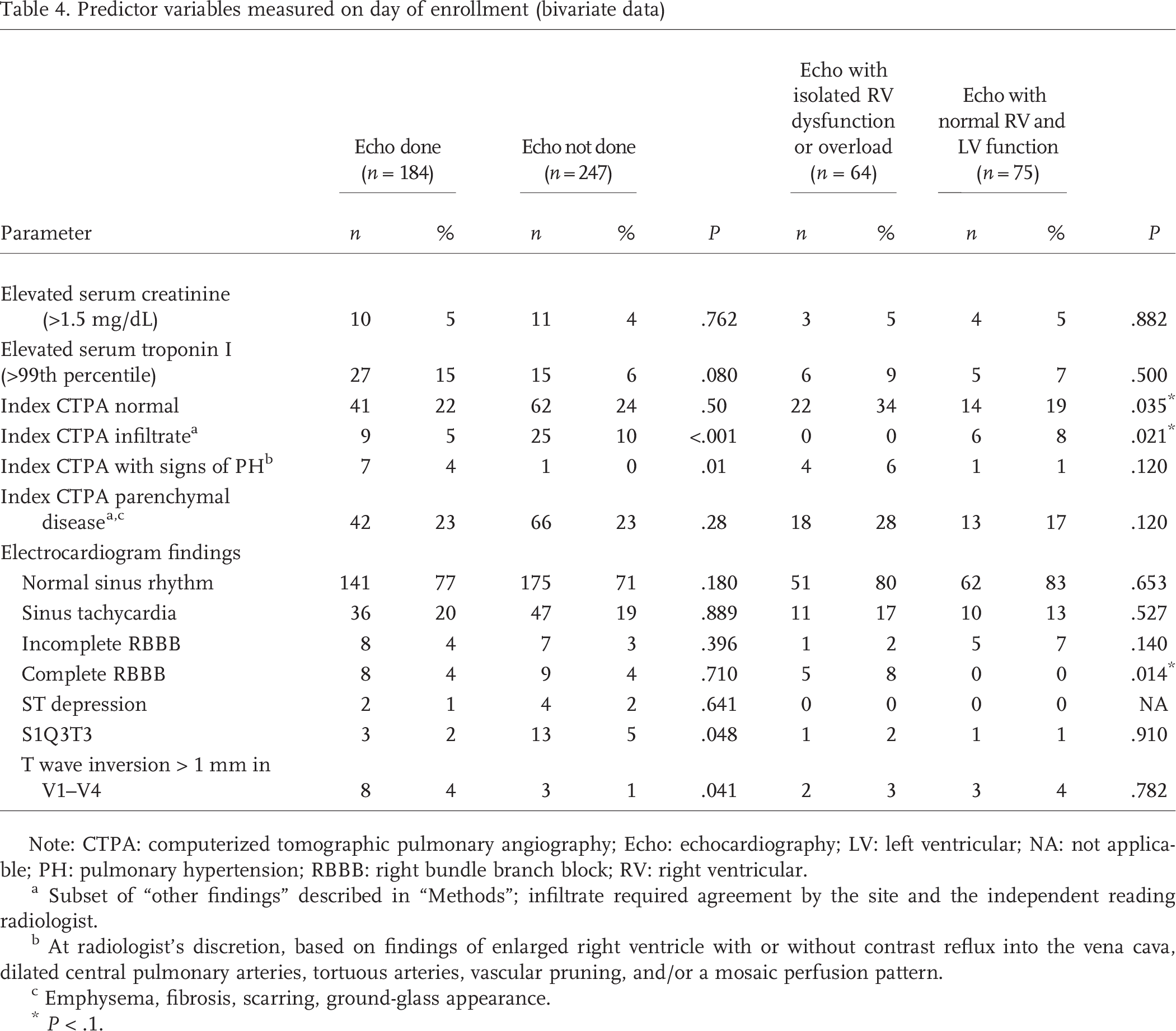
Note: CTPA: computerized tomographic pulmonary angiography; Echo: echocardiography; LV: left ventricular; NA: not applicable; PH: pulmonary hypertension; RBBB: right bundle branch block; RV: right ventricular.
Subset of “other findings” described in “Methods”; infiltrate required agreement by the site and the independent reading radiologist.
At radiologist's discretion, based on findings of enlarged right ventricle with or without contrast reflux into the vena cava, dilated central pulmonary arteries, tortuous arteries, vascular pruning, and/or a mosaic perfusion pattern.
Emphysema, fibrosis, scarring, ground-glass appearance.
P < .1.
RESULTS
Characteristics of the study population and prevalence of RV dysfunction
Patients were enrolled from January 30, 2007, to April 27, 2008. Figure 1 shows a flow diagram of patients, starting with subjects screened for inclusion and ending with repeated CTPA instances. Six hundred seventy-eight patients signed a consent form and underwent CTPA, and 647 had complete data, including a CTPA and clinical, blood, and breath sample data. The criterion standard for any filling defect consistent with PE was found in 120 of the 647 patients (18% [95% CI: 15%–22%]). Ninety-six patients did not have persistent dyspnea, leaving 431 patients with persistent dyspnea. As part of usual care, clinicians ordered transthoracic echocardiography that yielded adequate acoustic images for 186 of these 431 patients (42%), with 2 having images inadequate to assess RV function, leaving 247 who did not undergo echocardiography. Of the 184 patients with usable echocardiography, 64 (35% [95% CI: 28%–42%]) had isolated RV dysfunction or overload, 45 (24%) had LV dysfunction, and 75 (41%) had normal RV and LV function (Fig. 1). Of the 64 patients with an abnormal RV on echo, 20 had RV dilation or hypokinesis, together with an RV systolic pressure higher than 35 mmHg; 24 had only dilation or hypokinesis; and 20 had RV systolic pressure higher than 35 mmHg with normal RV size and contraction. Patients with LV dysfunction were not included in the analysis. On follow-up, 54 of the 647 patients (8%) had a repeat CTPA for recurrent dyspnea.
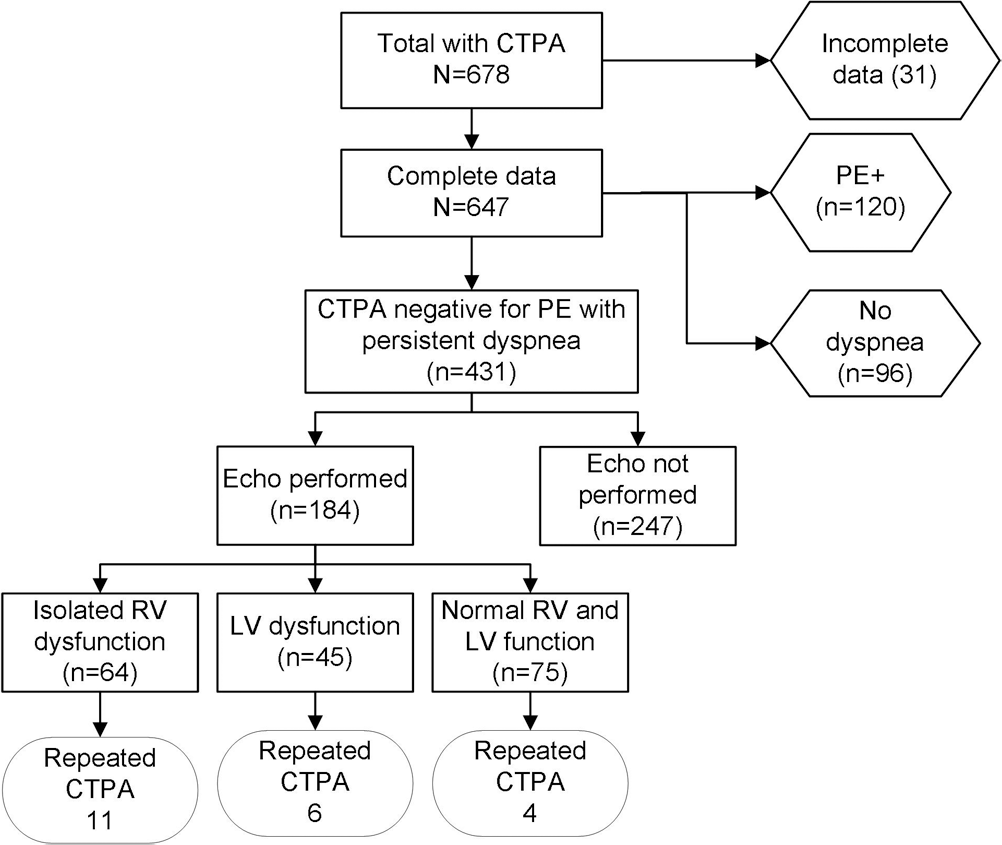
Flow diagram of patients enrolled and results of testing. CTPA: computerized tomographic pulmonary angiography; LV: left ventricular; PE: pulmonary embolism; RV: right ventricular.
Tables 1–4 compare predictor variables between patients with (n = 184) and without (n = 247) echocardiography. These tables then compare patients who had echocardiography between those with isolated RV dysfunction (n = 64) and those with normal RV and LV function (n = 75). Table 1 compares demographic data and symptoms, Table 2 compares comorbid conditions, Table 3 compares measured continuous variables (e.g., vital signs and respiratory data), and Table 4 compares bivariate results (e.g., CTPA and electrocardiographic results). Of note, the prevalence of parenchymal lung disease on CTPA was the same (23%) in the group that had echocardiography and in the group that did not, suggesting that this finding did not drive the decision to order or not order an echocardiogram.
Differences between patients with and without echocardiography
Comparisons of patients with and without echocardiography in Tables 1–4 were performed with a χ 2 or unpaired t test, yielding P < .05 for age, inspiratory and expiratory times, cough, recent surgery, and inpatient status. In particular, patients who underwent echocardiography were more likely to be inpatients or ED patients, were less likely to have substernal chest pain, pleuritic chest pain, or cough, and were less likely to have had recent surgery. Furthermore, they were likely to be older and to have a lower Borg score. They were less likely to have an S1Q3T3 pattern on electrocardiogram, but more likely to have T wave inversions.
Parameters associated with RV dysfunction
In the group undergoing echocardiography (n = 184), 64 (35%) patients exhibited signs of RV dysfunction or overload (Fig. 1). Seventy-five (41%) patients had a normal echocardiogram. Forty-five (24%) patients exhibited isolated LV dysfunction; these patients were not included in the analysis. The probability of isolated RV dysfunction or overload on echocardiography was higher in ED patients (36/86, 42%) than in inpatients/rehabilitation patients (26/98, 27% [95% CI for difference of 15%: 16%–29%], P = .02, exact binomial test). Because radiologists found RV dilation (defined as RV/LV diameter ratio > 0.9) on CTPA in only 7 patients, 4 of whom underwent echocardiography, this variable was not analyzed.
To select candidate variables for entry into multivariate regression analysis, we compared data between the patients with isolated RV dysfunction and the patients with normal echocardiograms. Analysis found that the mean values of the parametric variables age and fibrinogen were elevated with isolated RV dysfunction. However, neither value had a significant area under the ROC curve and were subsequently discarded. Dichotomous variables with P < .1 positively associated with isolated RV dysfunction included complete right bundle branch block, normal index CTPA scan, and malignancy under treatment. CTPA with infiltrate had a negative association.
Outcome data
Table 5 compares outcome data in a fashion similar to that of the previous tables. Patients with isolated RV dysfunction were more likely to be newly diagnosed with COPD or to have a repeat CTPA scan within 90 days. Interestingly, none of the 36 patients in Table 5 with a repeat CTPA scan had a new, acute pathogenic process discovered that was not present on the index CTPA scan.
Outcomes based on echocardiography status
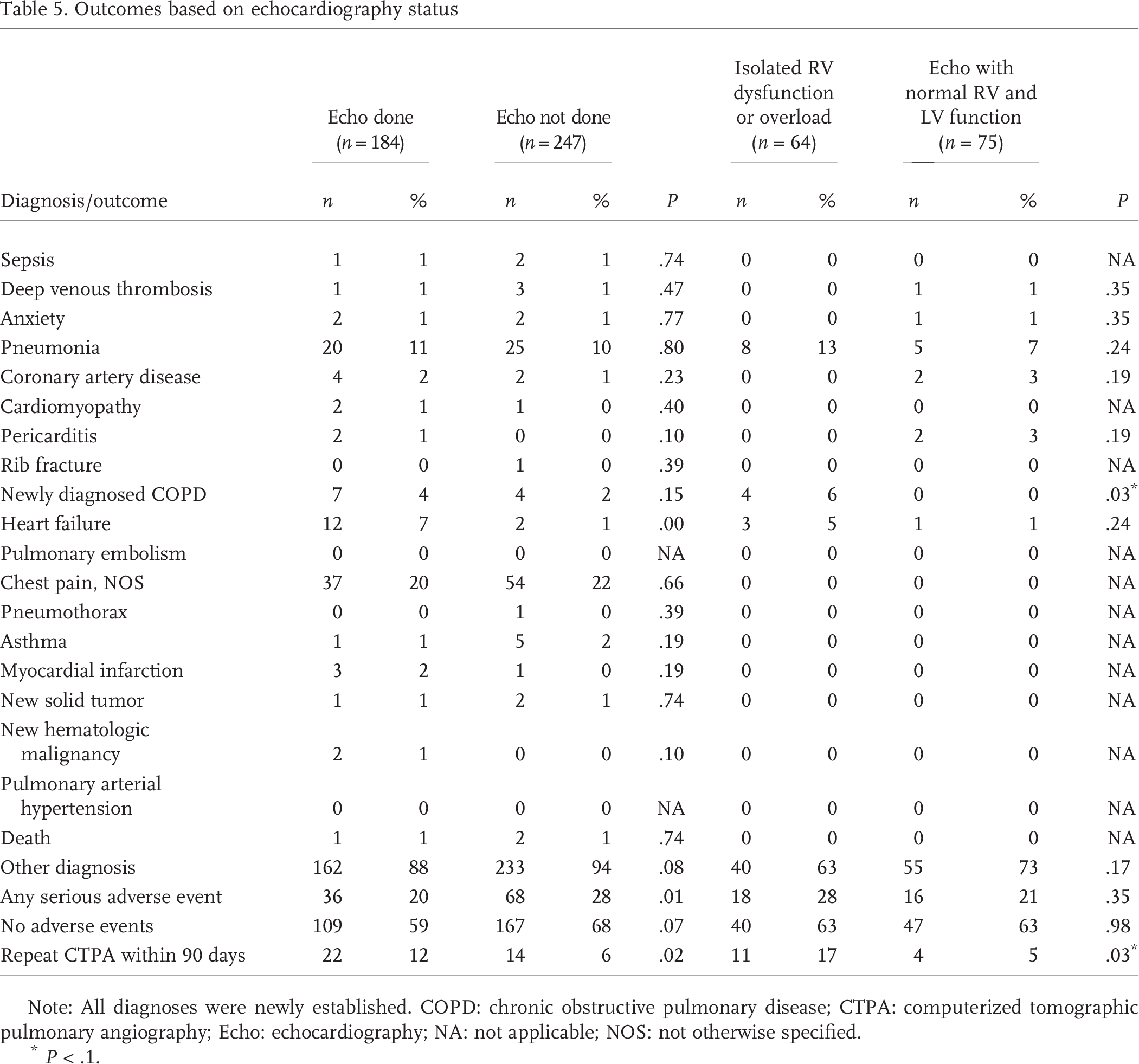
Note: All diagnoses were newly established. COPD: chronic obstructive pulmonary disease; CTPA: computerized tomographic pulmonary angiography; Echo: echocardiography; NA: not applicable; NOS: not otherwise specified.
P < .1.
The 4 predictor variables retained after univariate and ROC analysis (complete right bundle branch block, normal index CTPA scan, malignancy under treatment, and CTPA with infiltrate) were entered into a logistic regression analysis. Table 6 shows the equation coefficients and the corresponding P values for these variables. The model had good fit, demonstrated by a Pearson's goodness-of-fit χ 2 result of P = .52, a McFadden pseudo-R 2 of 0.92, and a Hosmer-Lomeshow test result of P = .95. This analysis showed that only one variable, normal index CTPA scan, had significance. Thus, we constructed a final rule consisting of persistent dyspnea plus a normal CTPA scan as the CDR to predict a high probability of isolated RV dysfunction on echocardiography. This rule was positive in 41 patients who underwent echocardiography, of whom 22 (53%) had isolated RV dysfunction (positive predictive value: 53% [95% CI: 37%–69%]). Five of the 22 rule-positive (CDR+) patients had a repeat CTPA scan within 90 days.
Results of logistic regression
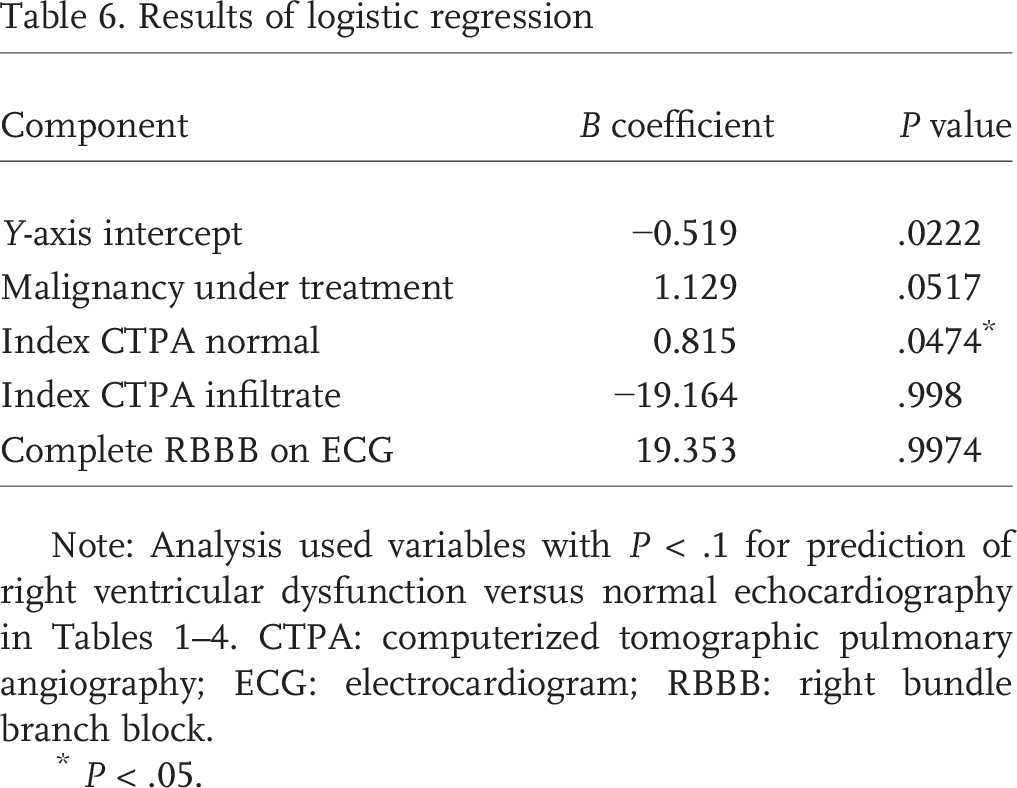
Note: Analysis used variables with P < .1 for prediction of right ventricular dysfunction versus normal echocardiography in Tables 1–4. CTPA: computerized tomographic pulmonary angiography; ECG: electrocardiogram; RBBB: right bundle branch block.
P < .05.
DISCUSSION
We found that 64 of 184 (35% [95% CI: 28%–42%]) patients who had persistent dyspnea after CTPA and a clinical picture that caused clinicians to order echocardiography had isolated RV dysfunction or overload. Hypothetically, had echocardiography been ordered for all 431 patients with dyspnea after CTPA and all other patients had a normal echocardiogram, the prevalence of isolated RV dysfunction or overload would still have been 64 of 431, or 15% (95% CI: 12%–18%). However, it is likely that the performance of echocardiographic studies in the patients who did not undergo echocardiography would have revealed at least a few additional cases of RV dysfunction, thus likely increasing that number. Restricting the sample to patients with dyspnea and a normal CTPA scan (i.e., CDR+) increased the prevalence of RV dysfunction or overload to 53% (95% CI: 37%–69%). Had the derived CDR been applied to the entire 647-patient population, the rule would have been positive and therefore suggests that 103 (16%) of patients should have been referred for echocardiography.
Isolated RV dysfunction on echocardiography was defined as a composite of RV hypokinesis and/or evidence of pathological tricuspid regurgitation, and it excluded patients with systolic or diastolic LV dysfunction. Our 35% prevalence of isolated RV dysfunction or overload appears to lie within the 95% CI for RV dysfunction from the PRIDE study (Chen et al. 23 ), which found a 30% (95% CI: 22%–39%) prevalence of moderate or severe tricuspid regurgitation. The two studies cannot be directly compared because our patients all had negative CTPA scanning before echocardiography, and the study by Chen et al. 23 was not intended to discover the rate of isolated RV dysfunction and therefore did not report this variable explicitly. In a large sample of Veterans Affairs patients (n = 10,471, 97% male) who underwent transthoracic echocardiography, Maron and colleagues 18 found that 14% (95% CI: 13.0%–14.4%) had estimated pulmonary artery systolic pressures of at least 40 mmHg. However, this study included any echocardiography that was not done to evaluate a specific cardiovascular disease (e.g., cardiac tamponade).
The prevalence of isolated RV dysfunction in our sample was significantly higher in patients from the ED setting (42% [95% CI 31%–52%]). This is important, because at most hospitals, more than half of all CTPA scans are ordered from the ED, and most show no significant pathology.11,33 At present, no clinical guideline exists to suggest a next step for the persistently dyspneic patient with no diagnosis after CTPA. Patients with isolated RV dysfunction or overload had a significantly higher rate of negative CTPA recidivism, suggesting continued symptoms and unwise use of healthcare resources. This is in line with data from the PAH literature that indicate that patients frequently are undiagnosed for 2 years or more, after being subjected to myriad diagnostic tests, before they are ultimately diagnosed with PAH.34,35 These findings triggered a national early-diagnosis campaign for PH (http://www.sometimesitsph.org).20,36 Our findings suggest that the ED would be an important setting to be included in such efforts. Data in Table 2 indicate that patients with isolated RV dysfunction or overload seldom had other severe associated comorbidities, such as COPD or end-stage cancer. We believe that, taken together, the data suggest that the isolated RV dysfunction or overload had a causative role in persistent symptoms. These data support a theoretical construct to develop an organized clinical protocol that would include echocardiography as an initial follow-up test for selected patients after negative CTPA scanning, followed by referral to a specialty clinic that can carry out a structured algorithm to evaluate for treatable causes of PH and/or RV dysfunction. 37
We therefore derived a CDR as an initial step. Using the univariate-multivariate approach, the final rule consisted of 2 independent criteria: a negative CTPA scan and persistent dyspnea. More than half (53%) of the patients with echocardiography and a positive CDR had isolated RV dysfunction. In a companion paper 38 we found that in a validation sample of persistently dyspneic ED patients with negative CTPA scanning and with echocardiography, 33% (95% CI: 25%–42%) had isolated RV dysfunction. Next steps will be to devise a clinical pathway that refers CDR+ patients for transthoracic echocardiography, followed by appropriate referral. 37 Such a structured diagnostic approach will likely allow for identifying causes of RV dysfunction or overload and for initiating appropriate additional diagnostic and therapeutic measures, with the ultimate goal of improving outcomes and decreasing unnecessary use of healthcare resources.
Limitations
In this derivation, the final rule was restricted to patients with a normal CTPA, which excluded patients with radiographic evidence of chronic diseases, such as cardiomeg-aly, emphysema, or scarring. In a secondary analysis, we created a variable with a broader definition, named “CTPA without acute process” (i.e., no PE, infiltrate, aortic dissection or aneurysm, pneumothorax, mediastinal mass, or lung mass). Substitution of that variable instead of the requirement for a normal CTPA in the CDR would have led to a predictive value positive of 64/179, or 36% (95% CI: 29%–43%). Other limitations to the work include our inability to determine whether patients with our definition of isolated RV dysfunction have treatable causes. For example, the potential contribution of World Health Organization group 3 PH is difficult to assess, because radiologists were not asked to systematically grade the severity of parenchymal abnormalities. However, most of our cases of parenchymal scarring were mild. This does not change our key message, however, that patients with persistent dyspnea and evidence of RV dysfunction need to undergo expert referral for identification of potentially treatable causes of dyspnea. Second, our echocardiographic basis for RV dysfunction includes heterogenous and controversial criteria, and we recognize that tricuspid regurgitation–derived indexes both over- and underestimate pulmonary arterial pressures. 39 We recognize that tricuspid regurgitation may occur with normal RV contractility, but in the setting of PH, moderate-to-severe tricuspid regurgitation worsens prognosis and is therefore considered to define RV dysfunction. 40 We emphasize the intent to use echocardiography as a screening criterion to move the patient on to a specialty clinic for more definitive diagnosis. Third, RV dysfunction can be caused by many pathologies.20,36 The current data do not allow for any conclusions regarding the etiology of RV dysfunction. Fourth, although this CDR is intended to align patients at high risk for repeated CTPAs and repeated ED visits with treatment to reduce symptoms, the proposed protocol could unintentionally increase resource use and cost of care. Finally, the relationship between echocardiographic findings of RV dysfunction or overload and persistent symptoms are only associative; these abnormalities may signify pathology that drove patients back to the ED for repeat testing, or the findings, if known by clinicians, may have lowered their threshold to order a repeat CTPA. We do not have specific data to understand the decision-making process that clinicians used when they ordered repeat CTPA scans.
Conclusions
A simple clinical decision rule, consisting of a normal computerized tomographic pulmonary angiography scan and persistent dyspnea, predicts a high probability of isolated RV dysfunction or overload on echocardiography. Patients with these features may benefit from referral for echocardiography to evaluate for signs of pulmonary hypertension and/or RV dysfunction, with subsequent referral for specialty evaluation for identification of potentially treatable causes.
