Abstract
Andeans and Tibetans have less altitude reduction in birth weight than do shorter-resident groups, but only Tibetans are protected from pulmonary hypertension and chronic mountain sickness (CMS). We hypothesized that differences in neonatal oxygenation were involved, with arterial O2 saturation (SaO2) being highest in Tibetans, intermediate in Andeans, and lowest in Han or Europeans, and that improved oxygenation in Andeans relative to Europeans was accompanied by a greater postnatal decline in systolic pulmonary arterial pressures (
Studies at high altitude have long provided a useful model for persons interested in the pulmonary circulation.1–4 While pulmonary arterial pressures (Ppa's) are generally elevated at high altitude, there is considerable population variation in both Ppa and pulmonary circulation–related disorders such as chronic mountain sickness (CMS; Fig. 1A)11,36 and subacute infantile mountain sickness. 37 Specifically, directly measured Ppa and hypoxic pulmonary vasoconstrictor responses are lowest in Tibetans, intermediate in Andeans, and highest in European-derived residents of Colorado (Fig. 1B). Recent estimates using echocardiography also suggest intermediate values in Ethiopians. 15 While multiple factors contribute to the etiology of CMS and no well-controlled comparative epidemiological studies exist, a reduced prevalence among Tibetans is consistent with the lower hemoglobin levels seen in Tibetans, relative to Andeans, in multiple reports.38–40 Tibetans are also protected, relative to Han, from subacute infantile mountain sickness, a syndrome characterized by severe pulmonary hypertension in 4–6-month-old infants that has also been reported in European-derived populations in Colorado.37,41,42 Whether subacute infantile mountain sickness is less common in Andeans than in Europeans is unknown, but lower Ppa's have been reported in Andean than in European children. 43
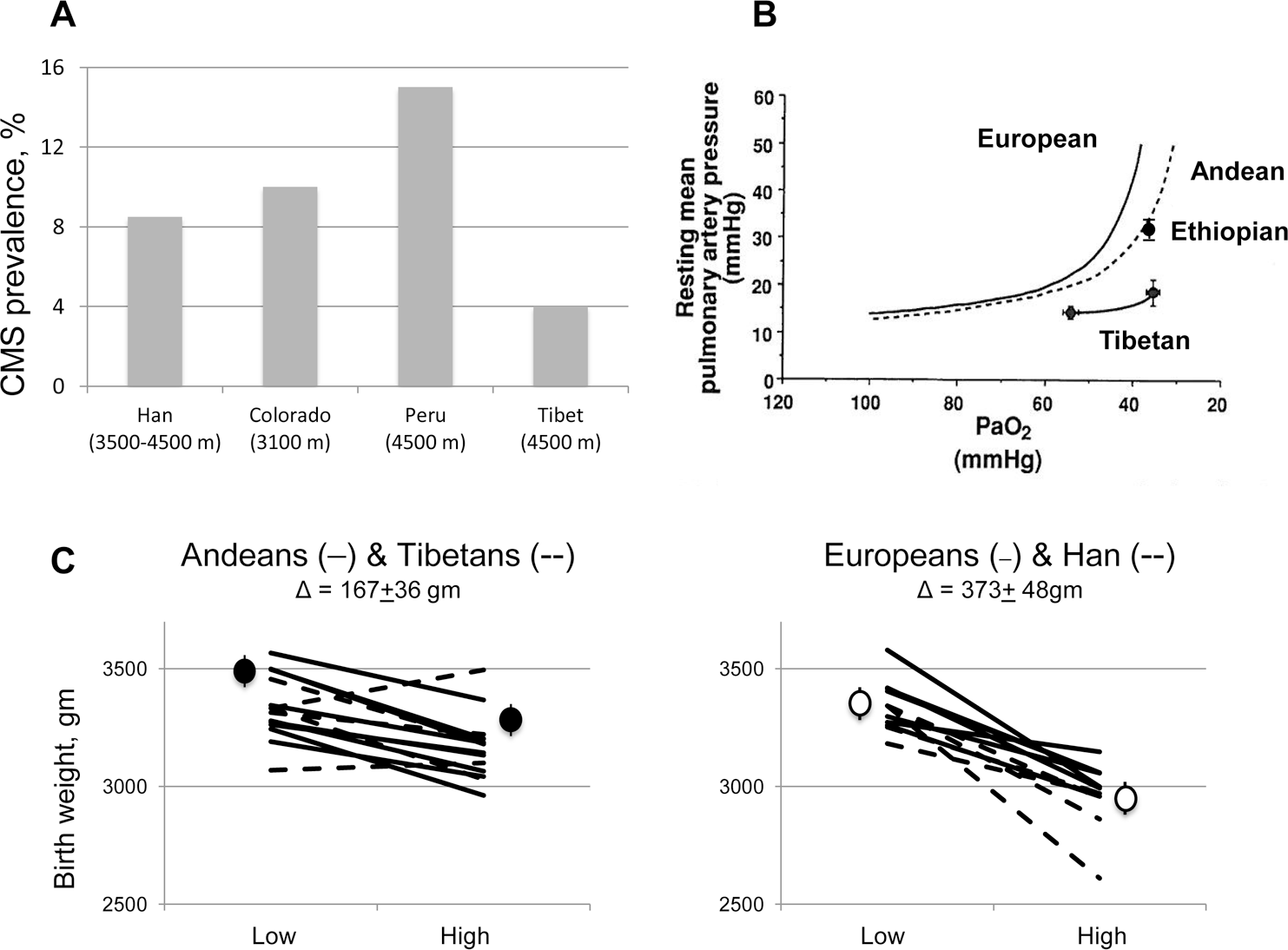
A, Chronic mountain sickness (CMS) is a debilitating condition occurring chiefly in men and characterized by excessive polycythemia in the absence of chronic obstructive pulmonary disease or other known causes and by symptoms of dyspnea, palpitations, sleep disturbance, cyanosis, dilitation of veins, paresthesia, headache, and/or tinnitus. 5 Studies in a total of 14,643 male high-altitude residents show a much lower prevalence of chronic mountain sickness (CMS) in Tibetans than in Han, Coloradans, or Andeans, especially when the altitude of residence is taken into account. Data are from 13,233 Han in Qinghai Province, China, 6 110 European-derived residents of Leadville, Colorado (I. Asmus, personal communication); 213 Andeans in Peru,7,8 and 1,087 Tibetans in the Tibet Autonomous Region; 9 plot adapted from Moore et al. 10 B, In healthy adult males, the PaO2-induced rise in resting pulmonary artery pressure, as measured by cardiac catheterization, is least in 5 Tibetans at 3,600 m, 11 intermediate in 58 Andeans at 3,700–4,500 m,12,13 and greatest in 28 European-derived residents at 3,100 m in Colorado; 14 plot adapted from Groves et al. 11 Measurements obtained by echocardiography in 76 Amhara males at 3,700 m in Ethiopia 15 at a PaO2 estimated from the reported SaO2 are similar to Andean values. Circles and lines represent mean ± SEM values. C, Lines show the altitude-associated decline in birth weight in a total of 305,935 infants from 15 studies in which birth weights were collected by the same investigator at both low (sea level) and high altitude (˜3,600 m) and 5 in which birth weights collected at one altitude are compared to the group value at the other altitude;16,17–33 plots adapted from Moore. 34 Circles depict mean ± SEM values for the ancestry groups shown. The 167 ± 36-g birth-weight reduction in Andeans or Tibetans (left; solid and dashed lines, respectively) is half (45%) the 373 ± 48-g reduction seen in European or Han high-altitude residents (right; solid and dashed lines, respectively). No differences in altitude-associated birth-weight reductions are apparent between Andeans and Tibetans when ancestry is controlled using surnames or gene markers, unlike a previous summary based on literature data in which population admixture was likely present. 35
One factor influencing Ppa during adulthood is perinatal hypoxia, as shown by human and experimental animal studies in which greater hypoxia in utero or during the neonatal period increased the reactivity of the pulmonary vasculature to hypoxia later in life.44,45 Perinatal hypoxia is greater at high than at low altitudes, as demonstrated by the known reduction in birth weight resulting from slowed fetal growth, 46 Doppler indices of fetal hypoxia, 47 and lower postnatal arterial O2 saturations (SaO2's). 48 Perinatal hypoxia also varies among populations; the altitude-associated reductions in fetal growth are only half (45%) as great in Tibetans or Andeans as in Han or Europeans residing at the same altitudes (Fig. 1C), and Tibetans have considerably higher SaO2's than Han over the first 4 months of postnatal life. 49
In line with the concept of developmental origins of adult disease 50 and observations that perinatal hypoxia influences Ppa later in life, we considered that altitude-associated disorders may be interrelated. That is, babies who were growth restricted in utero or otherwise hypoxic during the perinatal period may have lower SaO2's as a result of impaired lung development or altered respiratory control. Such lower SaO2's could then lead to higher Ppa as the result of delayed or absent regression of muscularized pulmonary arterioles that, together with the factors contributing to lower SaO2, could render persons susceptible to developing CMS later in life.
But if lower birth weights predispose persons to pulmonary hypertension and CMS, what then accounts for the apparent Andean-Tibetan differences in Ppa and CMS prevalence, given that both groups are similarly protected from altitude-associated reductions in birth weight (Fig. 1)? We considered that differences in neonatal oxygenation could be involved. Specifically, we hypothesized (1) that SaO2 was highest in Tibetans, intermediate in Andeans, and lowest in Han or Europeans and (2) that improved oxygenation in Andeans, relative to that in Europeans, was accompanied by a greater postnatal decline in Ppa. While the long period over which CMS develops precluded assessing these infants’ later-in-life susceptibility to CMS, consistent with such a possibility are our observations that persons with CMS weighed less at birth than healthy controls
51
and that young men with a preclinical form of CMS have more sleep-disordered breathing, nocturnal hypoxemia, and markers of oxidative stress consistent with impaired ventilatory control.
52
To test the first hypothesis, we measured SaO2 from 6–24 hours to 4–6 months of postnatal age in healthy Andeans born at 3,600 m and compared their data with those acquired previously in healthy Tibetan and Han infants at the same altitude. For the second hypothesis, we estimated Ppa as the systolic Ppa (
MATERIAL AND METHODS
Ethical approval
All participants in the Bolivian studies gave their written informed consent to study procedures, which had been approved by the human subject review committees of the Colorado Multiple Institutional Review Board of the University of Colorado Denver (COMIRB) and the Colegio Médico, its Bolivian counterpart. The studies conducted in Lhasa, Tibet Autonomous Region, China, were approved by the Tibet Institute of Medical Sciences and COMIRB. 49
Subjects
A total of 50 women and their infants were recruited in La Paz (3,600 m) and El Alto (4,100 m), Bolivia, from June 2001 to May 2003. They comprised (1) five (5) healthy European and 15 healthy Andean infants studied longitudinally from 1 week to 4–6 months of age, (2) 21 healthy Andean infants studied cross-sectionally from 6–24 hours to 1 month of age, and (3) nine (9) sick infants studied longitudinally from 1 week to 4–6 months of age. The healthy, longitudinal Andean mother-infant pairs were a subset of a larger group described previously. 53 Healthy babies were considered those born at term to Andean women without pregnancy complications (i.e., preeclampsia, gestational hypertension, or gestational or other forms of diabetes) and without a diagnosis of fetal distress, newborn respiratory distress, or neonatal pulmonary hypertension. The same criteria were applied to the healthy cross-sectionally studied babies, except that prenatal records were not available, and hence they were judged healthy on the basis of not requiring resuscitation at birth or not having respiratory distress, as judged by treatment with O2. Sick babies were born to mothers of European, Andean, or mixed (mestiza) ancestry and had received a diagnosis of fetal distress as documented by meconium-stained amniotic fluid, newborn respiratory distress, or respiratory depression as evidenced by requiring O2 treatment at the time of birth and the first days of life or pulmonary hypertension documented by echocardiography.
The inclusion criterion for all Bolivian subjects was current residence at high altitude (La Paz, 3,600 m, or the neighboring city of El Alto, 4,100 m). The exclusion criterion was a lack of birth-weight or gestational-age data. Additional inclusion criteria for the healthy mother-infant pairs were the absence of maternal smoking, absence of known risk factors for maternal complications of pregnancy (chronic hypertension, gestational diabetes, diabetes), and absence of hypertensive complications in the current pregnancy (gestational hypertension, preeclampsia, eclampsia). All but one of the Bolivian women had lived at high altitude throughout the current pregnancy; that woman had moved there in her seventh month, but neither her nor her infant's characteristics differed from those of the other subjects, and hence their data are included here. The women were classified as Andean, European, or mestiza on the basis of self-identification, and classification was verified by reference to parental and grandparental surnames and, in the case of the 20 longitudinally studied healthy Andeans or Europeans, a panel of 100 ancestry-informative genetic markers (AIMs), as described previously.16,54 A woman was classified as Andean if she had 3 or more Andean parental surnames and/or at least 76% AIMs of indigenous American origin. European ancestry was assigned if 3 or more parental surnames were of non-Hispanic European origin, she was of European nationality, or the majority of her AIMs were of European or other low-altitude-population origin. Women not meeting these criteria were considered mestiza. Inclusion criteria for the 15 Tibetan and 15 Han infants studied in Lhasa, Tibet Autonomous Region, China (3,600 m), were that the births occurred consecutively in June 1991 and that the babies were healthy at the time of enrollment. 49
Study design
This was a prospective, observational cohort study in which pulse oximetry and echocardiographic measurements (with Doppler) were carried out at high altitude in Bolivia in healthy or sick babies born to women classified as Andean, European, or mestiza. Longitudinal studies in healthy European and Andean babies were carried out at 1 week and 1, 2–3, and 4–6 months of postnatal age, with the actual infant ages at the time of study being 13 ± 2 and 12 ± 1 days (1 week), 39 ± 4 and 40 ± 2 days (1 month), 99 ± 4 and 97 ± 2 days (2–3 months), and 184 ± 20 and 183 ± 5 days (4–6 months), respectively. Echocardiographic studies in the same infants were conducted at the same time points, with the actual infant ages at the time of study being 9 ± 2 and 15 ± 2 days (1 week), 39 ± 4 and 44 ± 4 days (1 month), 97 ± 2 and 103 ± 4 days (2–3 months), and 186 ± 16 and 187 ± 5 days (4–6 months), respectively. The actual infant ages for the longitudinal oxygenation and echocardiography studies of sick babies were 13 ± 2 and 5 ± 4 days (1 week), 43 ± 6 and 39 ± 7 days (1 month), 97 ± 6 and 101 ± 3 days (2–3 months), and 174 ± 17 and 187 ± 12 days (4–6 months), respectively. For purposes of comparing data for the Andeans to previously acquired Tibetan and Han data at earlier time points (6–24 and 24–48 hours), we conducted additional cross-sectional respiratory studies in healthy Andean babies at 0.5 ± 0.3 days (6–24 hours, n = 4), 2.2 ± 0.2 days (24–48 hours, n = 4), 8 ± 1 days (1 week, n = 10), and 34 ± 2 days (1 month, n = 4). The Tibetan and Han data were also acquired with a prospective, observational cohort design in which studies had been conducted at the same time points as in Bolivia, namely, 0.6 ± 0.2 and 0.7 ± 0.2 days (6–24 hours), 1.6 ± 0.3 and 1.6 ± 0.3 days (24–48 hours), 7 ± 1 and 7 ± 1 days (1 week), 30 ± 6 and 30 ± 3 days (1 month), 55 ± 4 and 59 ± 53 days (2–3 months), and 120 ± 7 and 123 ± 4 days (4–6 months), respectively. 49
Variables and instrumentation
Information on maternal age, self-identified ancestry, and residential and reproductive history was collected by questionnaire or from medical records. Infant birth weight, gestational age, sex, whether the babies were small for gestational age as judged by being less than the tenth centile of sea-level values for a given gestational age and sex, 55 Apgar scores, and postnatal complications were obtained from delivery and newborn records. Babies born before 37 weeks gestation were considered preterm.
Arterial oxygenation studies were conducted in Bolivia over a 2–3-hour period that included quiet and active sleep, as well as periods of wakefulness and feeding, and used pulse oximetry (Nellcor N-200, Hayward, CA) in the clinic, the hospital, or the subject's home, with the probe placed on the lateral aspect of the foot. The SaO2 was recorded in beat-to-beat mode and reported as the average of the stable values recorded. To simplify reporting and group comparisons, the pulse oximetry data from the cross-sectionally studied Andeans were combined with those from the longitudinally studied Andean infants. The same protocol was followed in Tibet, except that a Biox 3740 oximeter (Ohmeda, Louisville, CO) was employed.
Echocardiography was performed with an ATL HDI 3000 equipped with a 5–7.5-MHz sectorial transducer, continuous and pulsed-wave Doppler, and color Doppler flow (Royal Philips Electronics, Amsterdam). All studies were carried out in the Clínica del Sur (Southern Clinic) in La Paz, Bolivia. The measurements were made in a warm environment in the quiet, awake state without the use of sedation and with Doppler measurements across the pulmonary valve being made early in the study to avoid elevation of estimated pressures because of agitation. The initial study included detailed documentation of anatomy, namely, great-vessel relationships, pulmonary venous return, interatrial and interventricular communications, and the presence of a patent ductus arteriosus. All studies included a review of cardiac anatomy; morphometric measurements (ventricular dimensions, septal and free-wall thicknesses, valve dimensions); ventricular shortening and ejection fractions; Doppler flow measurements across the aortic, mitral, tricuspid, and pulmonary valves; and presence of patent foramen ovale (PFO). Right ventricular systolic intervals (pre-ejection period [PEP], acceleration time [AT], and ejection time [ET]) were measured, as well as the R-R interval for calculating heart rate (HR). Measurements were made on a minimum of 3 complete waveforms; the maximal and minimal values (reflecting respiratory variation) were then averaged for the reported result. In healthy infants, systolic time intervals were incorporated into the regression equation of Li
56
to estimate
Statistics
Data are reported as the mean ± standard error of the mean in the text, tables, and figures or as the mean and 95% confidence intervals for proportions, except when indicated otherwise. Comparisons between groups at a single time were carried out with one-way analysis of variance (ANOVA), with Sidak multiple comparisons to identify the source of the differences observed. The effects of time, ancestry, and their interaction on SaO2 or echocardiographic characteristics were assessed by means of 2-way ANOVA with Tukey's multiple comparisons. Differences in proportions between groups were tested with χ 2 . All statistics were conducted in GraphPad software Prism 6.0 (GraphPad, La Jolla, CA). Comparisons are reported as significant when the 2-tailed P < 0.05 and as trends when 0.05 < P < 0.10.
RESULTS
Maternal and infant characteristics
By study design, the mothers of the healthy Andean and Tibetan babies had more often been born at high altitude and had lived there longer than the healthy Han or European women (Table 1). Values were intermediate in the mothers of sick babies, with fewer being born at high altitude than in the healthy Andean group but more than was the case for healthy Europeans. The mothers of the sick babies had lived at high altitude for as long as the healthy Andeans and longer than the healthy Europeans. Maternal age was similar in all groups, as was parity, with the exception of higher parity in the mothers of the longitudinally studied Andean babies. By study design, none of the healthy mothers experienced pregnancy complications, but most of the sick babies were born to women with complications of pregnancy or labor (Table 2). Neither parity nor maternal age was related to infant birth weight, SaO2, or
Characteristics of Han, Tibetan, Andean, and European healthy and sick mother-infant pairs studied at high altitude (3,600 m)

Note: Data are counts or mean ± SEM. HA: high altitude (>2,500 m); NS: not significant. Different letters signify differences between groups at P < 0.05.
Clinical characteristics of sick babies
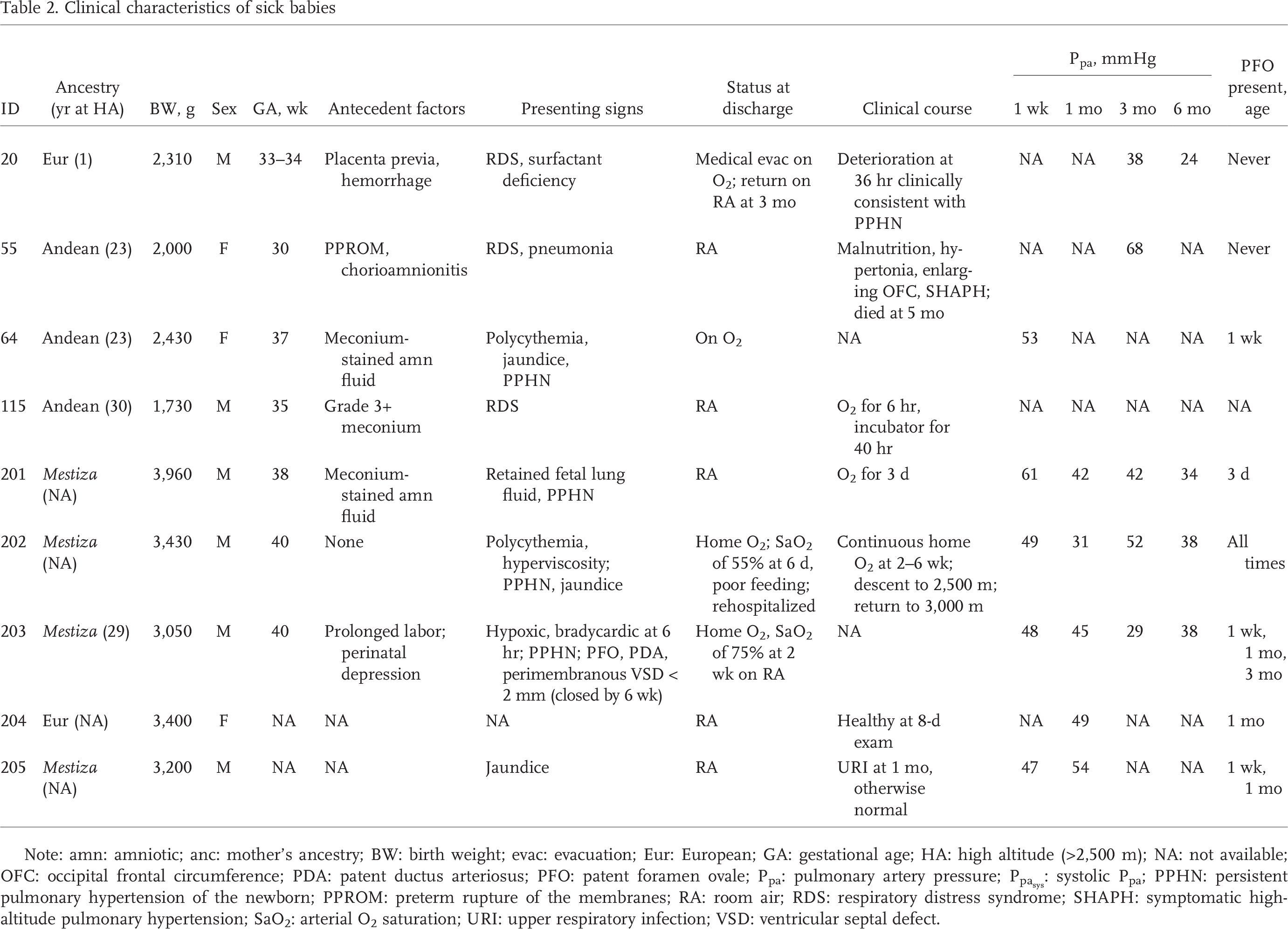
Note: amn: amniotic; anc: mother's ancestry; BW: birth weight; evac: evacuation; Eur: European; GA: gestational age; HA: high altitude (>2,500 m); NA: not available; OFC: occipital frontal circumference; PDA: patent ductus arteriosus; PFO: patent foramen ovale; Ppa: pulmonary artery pressure;
Birth weights and gestational ages were similar in all the healthy groups, although absolute birth weights were somewhat lower in the Han (Table 1). Sick babies were more often born prematurely. Approximately equal sex ratios were seen in Tibetans and Han, but there were more male than female babies in the healthy European and sick-baby groups, and more females in the healthy, longitudinal, and cross-sectional Andean groups. Apgar scores were similar among the various groups, with the exception of the sick babies, whose values were somewhat lower. Approximately half the sick babies required supplemental O2 (Table 2).
Pulse oximetry studies
The SaO2 showed consistent variations according to infant age and activity, and values were below sea-level norms at all study times (Figs. 2–4). Healthy Tibetans, Han, and Andeans had their highest SaO2's 6–24 hours after birth (Fig. 2). Across all study times and healthy groups, SaO2 was highest in the awake state (88.7% ± 1.8% [standard deviation]), intermediate while feeding and during active sleep (86.4% ± 2.8% and 86.4% ± 3.5%), and lowest during quiet sleep (85.9% ± 3.5%).
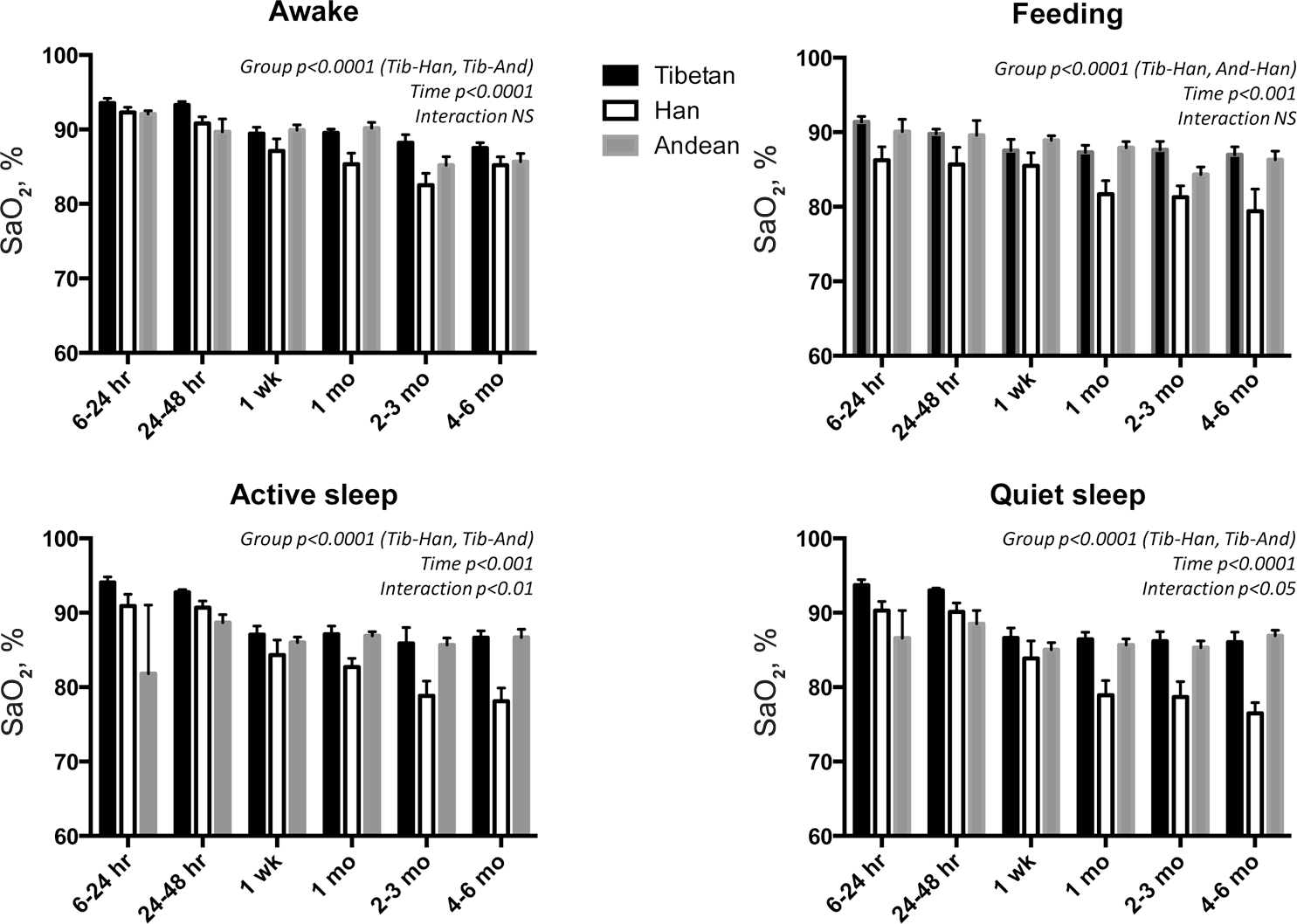
Mean ± SEM arterial O2 saturation (SaO2) in healthy Tibetan (Tib; n = 15), Han (n = 15), and Andean (And; n = 36) babies at 3,600 m declined with advancing age and during quiet sleep relative to that in the other activity states. Across time, values were higher in Tibetan than in Han babies in all states 49 and higher than those in the Andeans in the awake and active- and quiet-sleep states. Tibetan and Andean values during feeding were similar and higher than those observed in Han infants. NS: not significant.
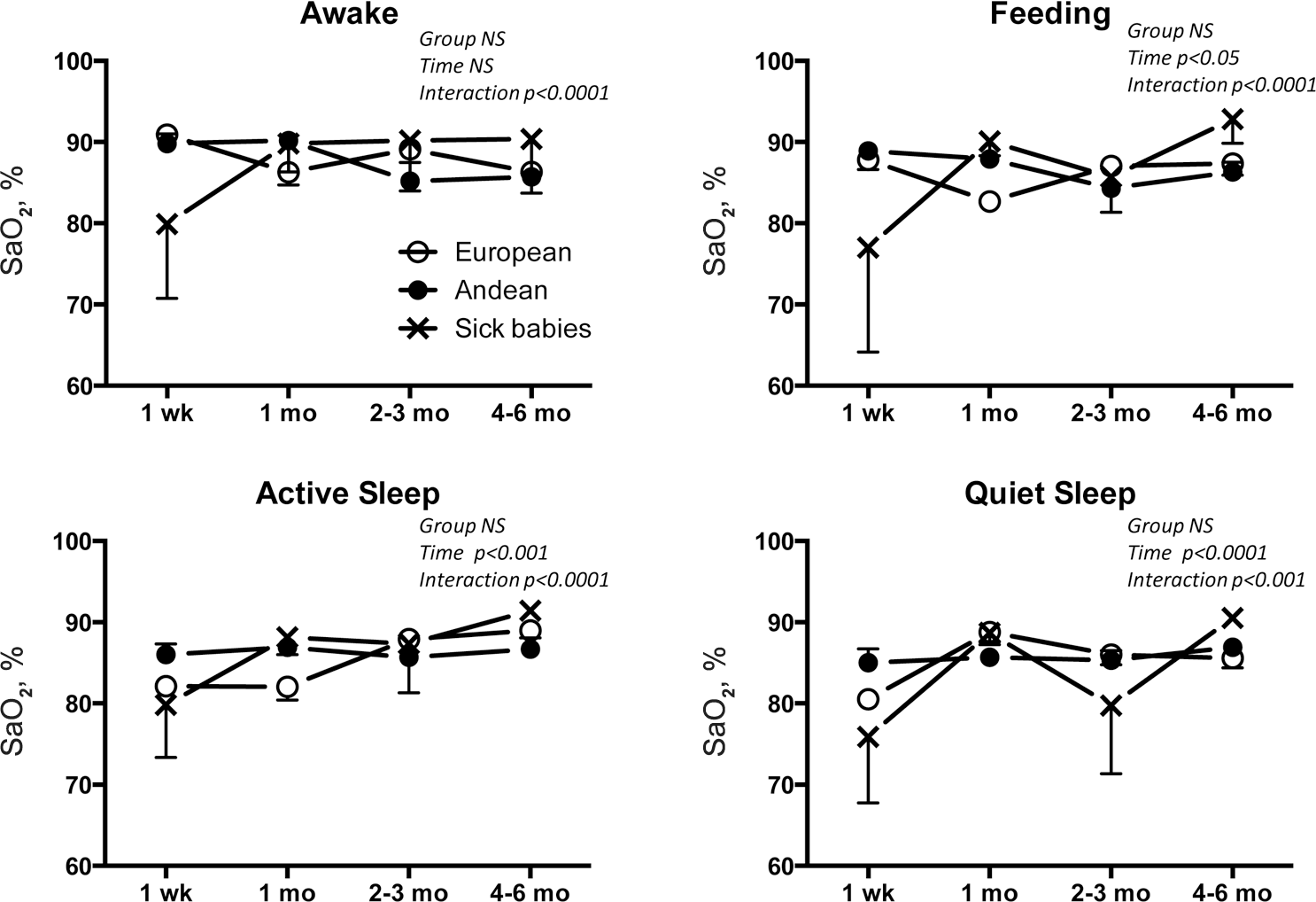
Mean ± SEM arterial O2 saturations (SaO2) are shown for 15 healthy Andeans, 5 healthy Europeans, and 9 babies diagnosed with fetal distress, newborn respiratory distress, or pulmonary hypertension (“sick babies”) of Andean, European, or mestiza descent who were studied longitudinally at 3,600 m. While values did not vary by group, the effects of group interacted with those of time in each activity as a result of greater desaturation in the sick babies, especially at the earliest time point, and the greater age-related rise and more variable values during quiet sleep in European than in Andean babies. NS: not significant.
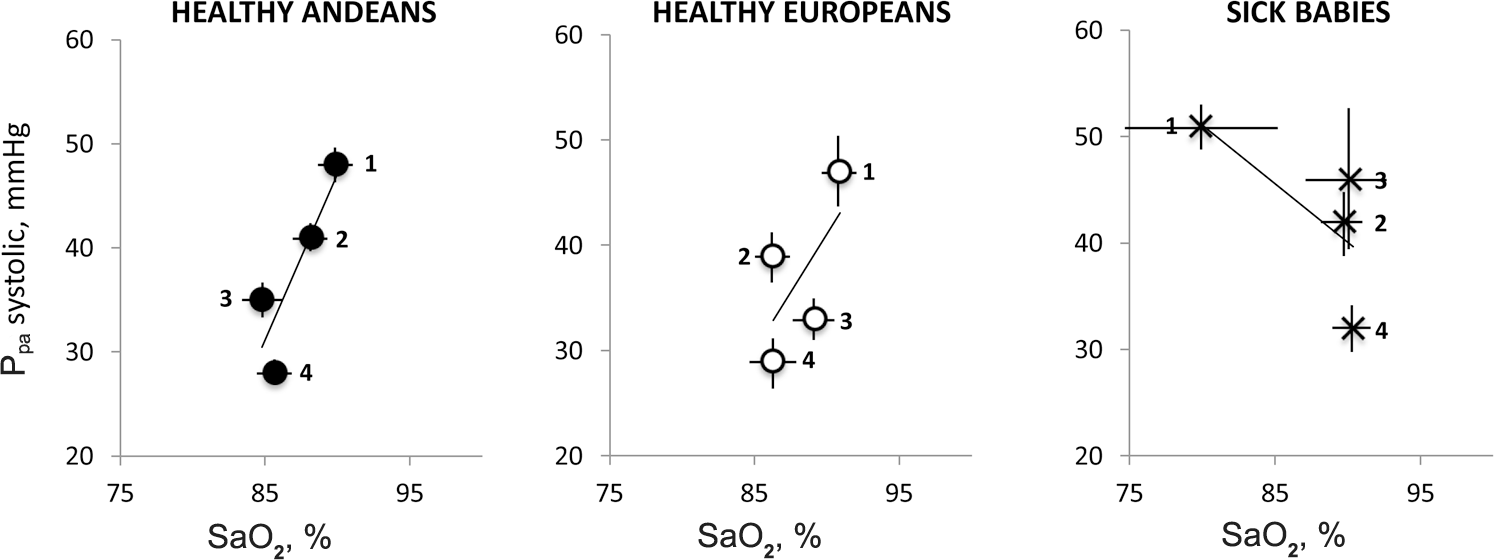
Mean ± SEM values for estimated pulmonary artery systolic pressure (
When healthy Tibetans, Han, and Andeans were compared across all study times, SaO2 was higher in the Tibetan than the Han or Andean babies during wakefulness, active sleep, and quiet sleep (Fig. 2). The SaO2 while feeding was higher in the Tibetans or Andeans than in the Han. Lower SaO2 values in the Han babies were chiefly the result of greater age-related declines, whereas the Tibetan-Andean differences were due to small but consistent differences at each study time.
There were no group differences in SaO2 between the healthy Andean, healthy European, and sick babies from 1 week to 4–6 months of age (Fig. 3). There was, however, significant interaction between the effects of group and time, which stemmed largely from the lower and more variable values seen in the sick babies. The European and sick babies had a greater age-related rise in SaO2 during active sleep and more variable values during quiet sleep than the Andean babies.
Echocardiography studies
All babies had structurally normal hearts on initial exam. Several minor anatomic variants without physiologic significance were identified, such as supernumerary chordae tendinae, Chiari network, persistent left superior vena cava draining to coronary sinus, and mild branch pulmonary stenosis. One small apical ventricular septal defect became apparent on the final study, after estimated right-sided pressures had declined from 48 to 29 mmHg, presumably allowing visualization of a left-to-right shunt across the defect. None of the healthy infants had a measurable tricuspid regurgitation jet. Thus, right heart pressures were estimated with systolic time intervals of the pulmonary valve.
The healthy European and Andean babies had similar PEP, AT, ET, AT/ET, PEP/ET, HR, and
Echocardiography studies in healthy and sick European and Andean babies at 3,600 m in La Paz, Bolivia
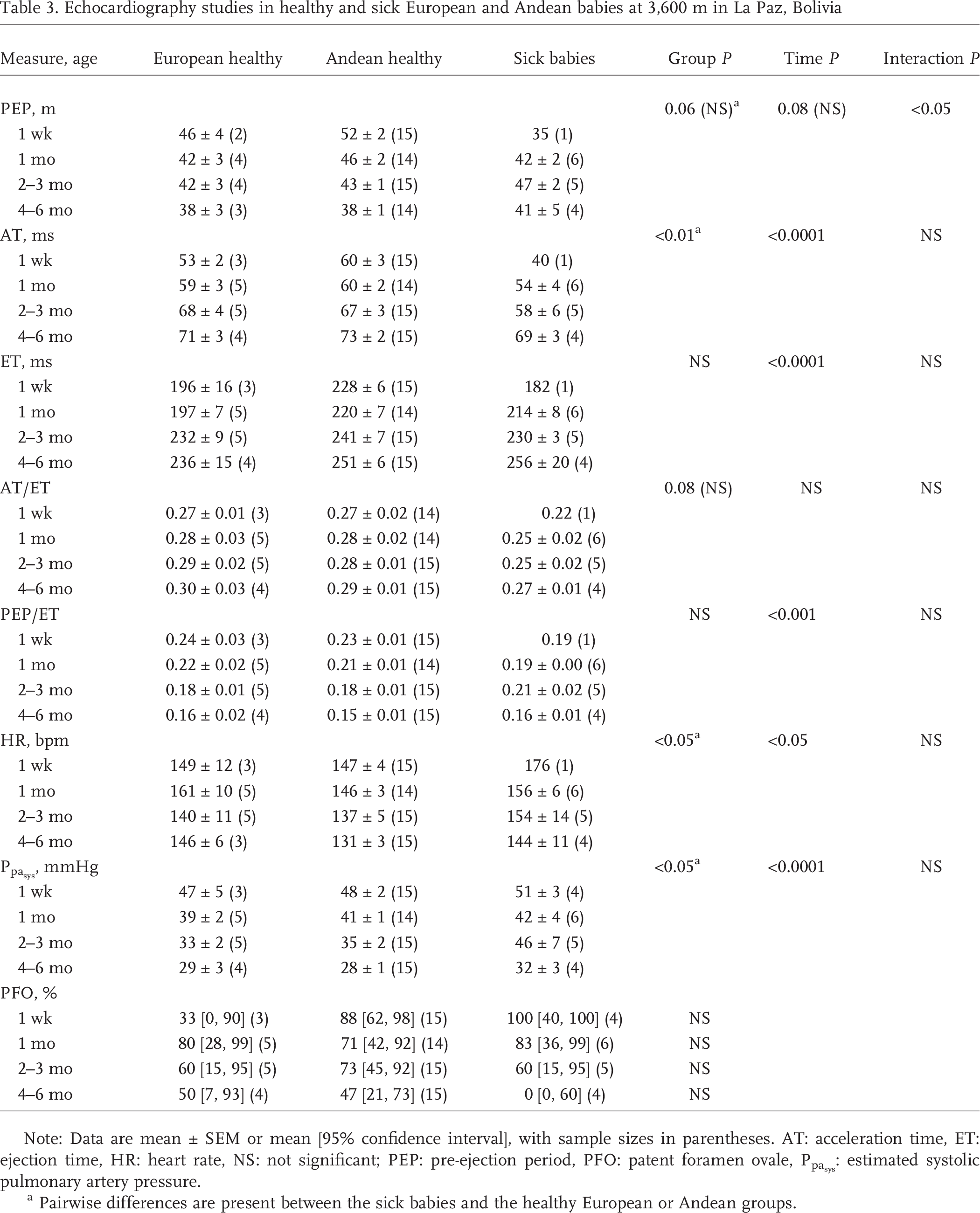
Note: Data are mean ± SEM or mean [95% confidence interval], with sample sizes in parentheses. AT: acceleration time, ET: ejection time, HR: heart rate, NS: not significant; PEP: pre-ejection period, PFO: patent foramen ovale,
Pairwise differences are present between the sick babies and the healthy European or Andean groups.
Compared with healthy babies, the sick babies had lower PEP, lower AT, a trend toward lower AT/ET, higher HR, and higher
Changes in Ppa with respect to SaO2 and clinical course
The
DISCUSSION
At 3,600 m, Tibetans had higher levels of neonatal oxygenation than Andean or Han babies while awake or asleep, and Andean values were higher than Han values during feeding, but neither SaO2 nor
Strengths of our study were the ability to measure SaO2 in each of the 4 recognized activity states and to study it, as well as
Several human and experimental animal studies demonstrate that postnatal normalization of Ppa is affected by residence at high altitude. At 4,540 m, the decline in Ppa is prolonged or even fails to occur in some infants.
63
Lambs gestated at 3,600 or 3,800 m also have elevated Ppa, which persists even after return to sea level.64,65 Greater hypoxia in utero or during the neonatal period also increases the reactivity of the pulmonary vasculature to hypoxia later in life in human and experimental animal studies.44,45,64 At 3,600 m in Bolivia, right ventricular wall thickness, an indicator of the shift from right to left ventricular predominance with declining Ppa, showed essentially no decrease during the 12 months after birth, whereas values at low altitude (400 m) decreased during the first month of life to a dimension that remained unchanged through the rest of infancy.
62
Despite supplemental O2 during the first 6 hours of postnatal life in Leadville, Colorado (3,100 m), echocardiographic Ppa indices were normal to moderately elevated in healthy term infants and did not fully normalize until 2–4 months of age.
48
Even healthy controls born at 2,240 m in Mexico City manifested a moderately elevated PEP/AT ratio at 15 and 30 days of age (0.90 ± 0.2 and 0.86 ± 0.24, respectively) and an elevated
While several reports indicate that population ancestry influences resting Ppa and the pulmonary vasoconstrictor response to hypoxia during adulthood (Fig. 1B), little is known concerning population variation in Ppa during infancy or childhood. To our knowledge, the Ppa response to hypoxia has not been measured in Han, but the greater right-axis deviation observed in adult male Han relative to Tibetan residents at 3,600 m73 is consistent with the likelihood of greater pulmonary hypertension. Supporting our catheterization data, recent echocardiographic studies show lower Ppa values in Sherpa highlanders than in acclimatized lowland controls. 74 Higher values in Amhara highlanders than in sea-level controls have also been observed in Ethiopia, 15 suggesting that the Tibetans’ low Ppa at high altitude may be unique. The factors responsible for the Tibetans’ relative protection from pulmonary hypertension at high altitude are unknown. Greater nitric oxide (NO) production and/or NO transfer has been suggested on the basis of higher levels of exhaled NO in Tibetans than in lowlanders in the United States. 75 This may be a body-wide response, since greater lower-limb vasodilator response to ischemia 76 and greater forearm blood flow in association with higher circulating NO products 77 have been observed in Sherpa or Tibetans than in sea-level residents of the United States or acclimatized lowlanders. Population differences in ventilatory sensitivity to hypoxia and hence arterial oxygenation are also likely involved, since Tibetans resemble acclimatized lowlanders by having a brisk ventilatory response to hypoxia, higher levels of alveolar ventilation, 78 and higher SaO2 in most, but not all, studies (see Gilbert-Kawai et al. 79 for a recent review).
We suggest that not only pulmonary hypertension but also CMS has antecedents during perinatal life. Babies born to pregnancies complicated by preeclampsia and/or who weighed less at birth are known to be at increased susceptibility to a range of cardiovascular and pulmonary diseases later in life. 50 It has been shown that Ppa's following ascent to high altitude are greater in young adult men who experienced neonatal hypoxia 45 or were born to mothers with mild to moderate preeclampsia, 80 relative to controls, and that those men were therefore likely subjected to greater hypoxia in utero. We have shown that adult men with CMS had lower birth weights, 51 and Julian et al. 52 recently reported that young men with a preclinical form of CMS had impaired breathing during sleep, compared with age-matched controls. Collectively, these studies are consistent with the concept that reduced fetal growth at high altitude impairs the maturation of pulmonary vascular and respiratory control systems, which in turn could predispose persons to pulmonary hypertension and CMS later in life.
Since Ppa estimates are not available from healthy Tibetan or Han babies at high altitude, we considered that it would be useful to compare their neonatal oxygenation with that of Andean and European infants in whom both SaO2 and
As reported previously, Tibetans have higher SaO2 than Han infants from the time of birth, 49 indicating a better neonatal cardiopulmonary transition. Consistent with our hypothesis, we found that Tibetans had higher SaO2 than Andeans in the awake and active- and quiet-sleep states (Fig. 2). The 2%–5% point differences in SaO2 between Tibetans and Han during wakefulness and the 2% to 8%–9% point differences during the other activity states were greater than the 1%–3% point differences between the Tibetans and Andeans, suggesting a cardiopulmonary transition in Andeans that is intermediate between those of Tibetans and Han high-altitude residents.
However, counter to our hypothesis, neonatal oxygenation did not differ between the healthy Andean and European groups (Fig. 3). Nor did our echocardiographic studies show any differences between healthy Andean and European babies in right heart function or
In summary, the implications of our study findings are threefold. First, population differences in neonatal oxygenation exist such that Tibetans, Andeans, and healthy Europeans did not show the progressive decrease in SaO2 during sleep that characterized Han in our earlier study.
49
Specifically, mean saturations remained at or above 85% in these groups, whereas the Han values dropped into the 70% range and even lower in some cases. In comparisons of high-altitude residents, neonatal oxygenation was highest in Tibetans, intermediate in Andeans and Europeans, and lowest in Han. Values were similar in healthy Andean and European babies, but additional studies in larger numbers of European infants at high altitude are required to confirm (or refute) this observation. Particularly critical will be longitudinal studies of Ppa and SaO2 using well-controlled methodologies to determine whether the postnatal decline in Ppa differs between Andeans and Tibetans or between long- and short-resident groups. Second, while some babies have clinical disease, many show considerable resilience and succeed in maintaining or improving their SaO2 and lowering their
Footnotes
ACKNOWLEDGMENTS
We especially thank all the families who participated for their commitment of time and their interest in the work. We would also like to thank the community of pediatricians, obstetricians, general practitioners, and other health professionals in La Paz who facilitated the participation of their patients in the study, the research staff at the Instituto Boliviano de Biología de Altura and the Clínica del Sur in La Paz for their help with the study conduct, and Nancy West at Denver Health for her statistical consultation. An earlier version of this article was given by LGM as the Estelle Grover Memorial Lecture at the September 2013 Grover Conference on the pulmonary circulation held at Lost Valley Ranch, Deckers, Colorado.
Conflict of Interest: None declared.
