Abstract
For many patients with severe pulmonary arterial hypertension, heart failure—and, in particular, right heart failure—is the final chapter of their chronic illness. Targeted therapy for pulmonary hypertension is effective only if the right ventricular ejection fraction is maintained or improved. Because improvement of right heart function and reversal of right heart failure are treatment goals, it is important to investigate the cellular and molecular mechanisms that cause right heart failure. Here, we propose that right ventricular capillary rarefaction is an important hallmark of right heart failure and consider that the “sick lung circulation” and the pressure-overloaded right ventricle constitute a functional unit.
Historically, the right ventricle (RV) was—and occasionally continues to be—the forgotten chamber of the heart. However, as the study of lung vascular diseases advanced in recent years, the importance of the RV became clear: patients suffering from severe forms of pulmonary arterial hypertension (PAH) frequently die of RV failure (RVF). The relative uncoupling between increased RV afterload and RVF in subgroups of patients has raised the question of whether any load-independent factors contribute to the development of RVF, and, if so, which ones may be potentially reversible.
In this perspective, we summarize the factual and hypothetical molecular mechanisms of RVF and attempt to formally emphasize the importance of the “sick lung circulation” and its contribution to the development of RVF. We also address the importance of recognizing the lung circulation as part of a cardiopulmonary system, particularly when testing new pharmacotherapies.
THE LUNG CIRCULATION–RV AXIS: A BRIEF HISTORICAL PERSPECTIVE
The founding fathers of lung vascular research at the University of Colorado—most prominently, Dr. Robert Grover, after whom this American Thoracic Society Grover Conference has been named—distilled the essence of the concept of heart-lung interaction in the name of their laboratory: the Cardiovascular Pulmonary (CVP) Research Laboratory. Trainees at the CVP lab already had a systems approach to pathobiology in mind when utilizing the clinical method of Fulton to assess RV hypertrophy in chronic hypoxia exposure studies together with hematocrit to apportion the impact of pulmonary arterial pressure elevation and polycythemia on RVH.1,2 Back then, little could be done to measure RV function with the exception of obtaining cardiac output. 3 Yet both the importance of RVF and the variable survival time of patients with severe idiopathic PAH (formerly known as primary pulmonary hypertension, or PPH) had already been recognized. 4 In 1998, Bristow et al. 5 provided an overview of the pathobiology of RVF, and a quote from his state-of-the-art talk is particularly worth remembering: “One of the fundamentally important questions surrounding primary pulmonary hypertension is that subjects differ substantially in their tendency to develop RVF. This leads to a variable natural history with some subjects living a surprisingly long time and others dying more rapidly. Therefore, mechanistic hypotheses for the development of RVF from chronic pressure overload need to accommodate this biological variability. One such hypothesis that our group is testing is that chronically elevated wall strain leads to changes in gene expression that produce contractile dysfunction and perhaps other deleterious biological phenomena such as programmed cell death (apoptosis)” (p. 101S).
In the following years, a workshop sponsored by the Heart, Lung, and Blood Institute, National Institutes of Health, focusing on the forgotten RV 6 finally challenged the research community with the question, why does the RV fail? Today, the knowledge base of RVF mechanisms is far broader, certain molecular and histological hallmarks are now considered part of a RVF signature or program, and many new hypotheses continue to be investigated.
THE LUNG CIRCULATION–RV AXIS: FACTS AND HYPOTHESES
Fact: the right ventricular failure molecular signature is characterized by abnormal metabolism, mitochondrial dysfunction, impaired angiogenesis, increased oxidative stress, adrenergic desensitization, and fibrosis
A clinical definition of right heart failure remains a work in progress. As a mechanistic definition of RVF remains incomplete, perhaps we could define RVF as a state that is morphologically and functionally distinct from compensated RVH. To begin characterizing the molecular signature of RVF, studies utilizing microarray technology have been conducted. Urashima et al. 7 studied the transcriptional differences between the RV and the left ventricle (LV) subjected to pressure overload in rodents and demonstrated a differential expression of genes between the pressure-overloaded RVs compared with the LV counterparts, suggesting that the RV and the LV do not respond to pressure overload identically. If so, this could be very important when translating heart failure treatment strategies to PAH-associated RVF. Next, on the basis of the premise that in rat models of pulmonary arterial banding (PAB) and chronic hypoxia exposure functional RV hypertrophy (RVH) but not RVF develops, Drake et al. 8 evaluated gene expression changes between these two models of PAH with adaptive hypertrophy (chronic hypoxia) and compared them to a model of RVF (SU5416/hypoxia [SuHx]). This study delineated a set of cell signaling pathways that were specific for RVF tissue, particularly highlighting pathways involving mitochondrial dysfunction and metabolism. These large-scale gene expression studies have been complemented with enzymatic rate analysis, electron microscopy, mitochondrial respiration analysis, and protein expression measurements, which altogether suggest that RVF, but not adaptive RVH, tissue is characterized by decreased fatty acid and glucose oxidation, increased glucose uptake and glycolysis, abnormal mitochondrial oxidative capacity, and abnormal mitochondrial ultrastructure.9–14
Two independent laboratories have confirmed another salient feature of the RVF molecular signature: impaired angiogenesis. Bogaard et al. 10 first described that increased RV pressure overload was not sufficient to explain all of the histological and molecular changes observed in RVF tissue, including a reduction in vascular endothelial growth factor (VEGF) expression and loss of capillaries in the RV. Sutendra et al. 11 confirmed this observation in the model of PAH and RV dysfunction caused by monocrotaline in rats, postulating that the reduction in angiogenesis could drive the changes in RV substrate utilization. While a mechanistic role for impaired angiogenesis in the development of RVF has not been formally tested, the role of VEGF in cardiac homeostasis has been studied. May et al. 15 elegantly demonstrated that cardiac-restricted inhibition of VEGF signaling was sufficient to induce not only capillary loss but also hypoxic microdomains and functional changes characteristic of cellular hibernation in, most interestingly, a reversible fashion. However, whether decreased expression of VEGF and the accompanying reduction in capillaries is cause or consequence of RVF remains to be tested. Perhaps a direct consequence of capillary rarefaction in RVF tissue would be cell death and replacement interstitial fibrosis, which have been documented in RVF tissue.13,16 The implications of cardiac fibrosis in heart failure have long been discussed; however, its mechanistic role in RVF-PAH is largely unclear. Recently, Rain et al. 13 demonstrated that patients with severe PAH develop not only systolic but also diastolic dysfunction, which was partially explained by intrinsic stiffening of the RV cardiomyocyte sarcomeres and cardiac fibrosis. Reactive oxygen species (ROS)–induced damage has been characterized in RVF tissue. Gomez-Arroyo et al. 9 demonstrated that RVF tissue exhibits increased staining for 8-oxo-2′-deoxyguanosine, a major byproduct of ROS-mediated DNA oxidation, and the role of ROS in RVF has been mechanistically tested in heme oxygenase 1 knockout mice, which on exposure to chronic hypoxia develop severe RVF. 17
Adrenergic desensitization has long been considered part of the pathobiology of cardiac disease; however, the role of adrenergic desensitization or the potential use for adrenergic receptor blockade in PAH-induced RVF had not been investigated until recently. Bogaard et al. 18 demonstrated that treatment with the nonselective adrenergic blocker carvedilol improved maladaptive RVH functionally and histologically without affecting the lung circulation, and similar effects have been demonstrated utilizing other adrenergic receptor blockers. 19 More recently, Piao et al. 12 evaluated the role of adrenergic desensitization in RVF and demonstrated that G protein–coupled receptor kinase 2 mediated an uncoupling of β-adrenergic receptor signaling, therefore impairing inotropic reserve. A particularly interesting finding from this study is that, at least in rats, downregulation of the β1, α1, and dopamine 1 receptors was partially independent of pressure overload. Indeed, compared with the PAB model of mechanically induced chronic RV pressure overload, the SuHx model of RVF demonstrated a larger impairment of the RV contractile reserve and adrenergic desensitization in spite of similar levels of RV afterload. These studies raise many questions, and here we highlight only three: (1) Can β-blockade treatment have cardiac effects that could improve RV function independently from heart rate modulation? (2) To what extent is adrenergic desensitization dependent on pressure overload? (3) Could adrenergic desensitization be explained by circulating factors perhaps released from a sick lung circulation?
Hypothesis: the role of hibernation and autophagy in the failing RV
Myocardial apoptosis is one of the hallmarks of heart failure, and the complicated cross talks between apoptosis and autophagy were recently thoroughly investigated.20–22 Abnormal protein aggregates as well as the accumulation of damaged organelles have been suggested to play a role in the pathogenesis of heart failure (proteotoxicity). 23 Autophagy is a highly regulated homeostatic process involved in the removal of protein aggregates, and both the ubiquitin-proteasome system and autophagy, which serve as a waste management system, can become impaired in heart failure.23,24 Abnormal expression of autophagy-related factors has been described in multiple diseases, including pulmonary vascular diseases; 20 however, a role for autophagy in the adaptation of the RV to chronic pressure overload has not yet been investigated.
Although evidence of autophagosomes in the heart dates back to the 1950s, 25 molecular analysis of autophagy started to develop about a decade ago with the report of the autophagy-related genes (Atg) from genetic screens in Saccharomyces cerevisiae. 25 The autophagy process is divided into mechanistically distinct steps that could be summarized as follows: (1) induction, (2) cargo recognition, (3) vacuole (autophagosome) formation, (4) fusion, and (5) degradation. 26 Each step is a tightly regulated process that requires the interaction of multiple proteins. 22 Although autophagy has been described primarily as a cell survival mechanism, 20 the functional relationship between apoptosis and autophagy is complex. Indeed, whereas autophagy constitutes an adaptive process to prevent cell death, it also constitutes an alternative pathway to cellular demise, named autophaghic cell death (or type 2 cell death). 22 Rotter and Rothermel 27 recently proposed that the outcome of autophagy—whether it is beneficial (homeostatic) or detrimental (leading to apoptosis)—actually depends on the timing and the magnitude of the autophagic activity in relation to the severity of the cellular stress. Although the theory has never mechanistically tested in animal models of RVF, Ma et al. 28 provided evidence for impaired autophagosome clearance in a model of cardiac ischemia/reperfusion injury and demonstrated that impaired clearance contributes to cardiomyocyte death.
As mentioned before, it has been proposed that maladaptive RVH is characterized by capillary rarefaction, 16 myocardial ischemia, 19 fibrosis,2,20 and cardiomyocyte apoptosis, 2 and more recently it has been reported that the dysfunctional RV is characterized by significant metabolic remodeling and mitochondrial dysfunction. 21 These molecular and cellular changes are associated with a decrease in cardiac output, marked RV dilatation, and decreased exercise capacity. 2 Gomez-Arroyo et al. 9 previously demonstrated that maladaptive RVH in the SuHx rat model also exhibits multiple features of abnormal mitochondrial biology. Furthermore, the authors showed that RV dysfunction is characterized by decreased expression of genes encoding proteins required for mitochondrial biogenesis, such as TFAM, Top1mt, POGL2, and POLRMT. Importantly, it was reported that maladaptive RVH exhibits a significantly low mitochondrial yield, decreased mitochondrial oxidative capacity, and severely abnormal mitochondrial ultrastructure by electron microscopy in comparison to control RVs. A further analysis of electron microscopy images provided evidence for an increased number of autophagosomes (Fig. 1A, 1B) as well as increased mitophagy (selective autophagy of mitochondria). Because autophagy can cause a decrease in mitochondrial mass (as much as 50%) as part of an adaptive cell response 23 and because both PAB and SuHx are characterized by a significant decrease in net mitochondrial protein, 9 we propose that autophagy is highly induced in the RV during pressure overload. A large number of proteins interact and cooperate to initiate autophagy and are useful in order to assess effective autophagic flux, such as the conversion of LC3-I to LC3-II, degradation of p62, and parkin, a ubiquitin E3 ligase required for selective autophagic degradation of mitochondria (mitophagy).22,24 We propose that autophagy is induced in the pressure overloaded RV, and Figure 1C illustrates that conversion of LC3-I to LC3-II is increased, as determined by Western blot analysis of proteins obtained from SuHx and PAB RV tissues. Many transcriptional regulatory factors link upstream signaling pathways to autophagy. 5 The AMP-activated protein kinase (AMPK) and the mammalian target of rapamycin (mTOR) signaling pathways tightly regulate autophagy. Active AMPK leads to phosphorylation and activation of the tuberous sclerosis proteins 1/2 complex, which inhibits mTOR activity, increasing ATP levels via recycling of nutrients. 26 Tissue from failing SuHx RVs has increased levels of phosphorylated AMPK (Fig. 1D), which supports our finding of increased autophagy as evidenced by increased LC3 conversion. In mammalian cells, accumulation of unfolded proteins induces endoplasmic reticulum stress (ERS), which induces autophagy in a highly conserved manner. 26 Indeed, the RV from SuHx rats exhibits increased markers of ERS, such as GRP94 protein and CHOP (growth arrest and DNA damage-inducible, Gadd153) and Gadd34 transcript levels (Fig. 2A–2C). Thus, increased autophagy could be an adequate response to chronic pressure overload. ROS can favor the proteolytic activity of ATG4 in the RV, thereby inducing autophagy 27 and cellular stress markers, which, like ceramides, are increased in SuHx RV tissues. These sphingolipids are prominent inducers of apoptosis but also stimulate autophagy and are associated with lipotoxicity. 29 Utilizing chromatography electrospray ionization tandem mass spectrometry, we have performed lipid profiling and found increased levels of ceramide 16, 14, and 24 species (Fig. 2D–2F), which (together with enhanced AMPK activity and increased ROS and ERS) support our finding of increased autophagy induction in the pressure-overloaded RV.
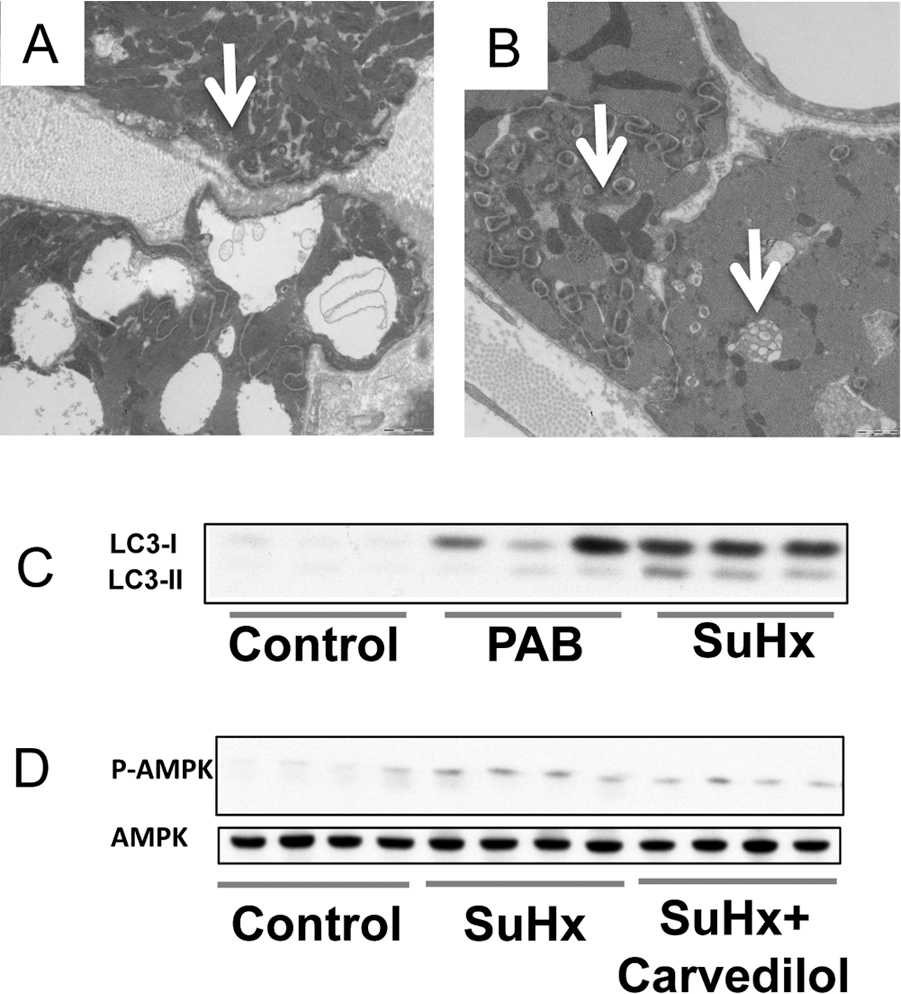
A, B, Electron microscopy snapshots demonstrating the presence of what appear to be autophagosomes (arrows). C, Western blots showing increased levels of LC3-I to LC3-II conversion and increased 5′ AMP-activated protein kinase (AMPK). PAB: pulmonary arterial banding; SuHx: SU5416/hypoxia; P-AMPK: phospho-AMPK.
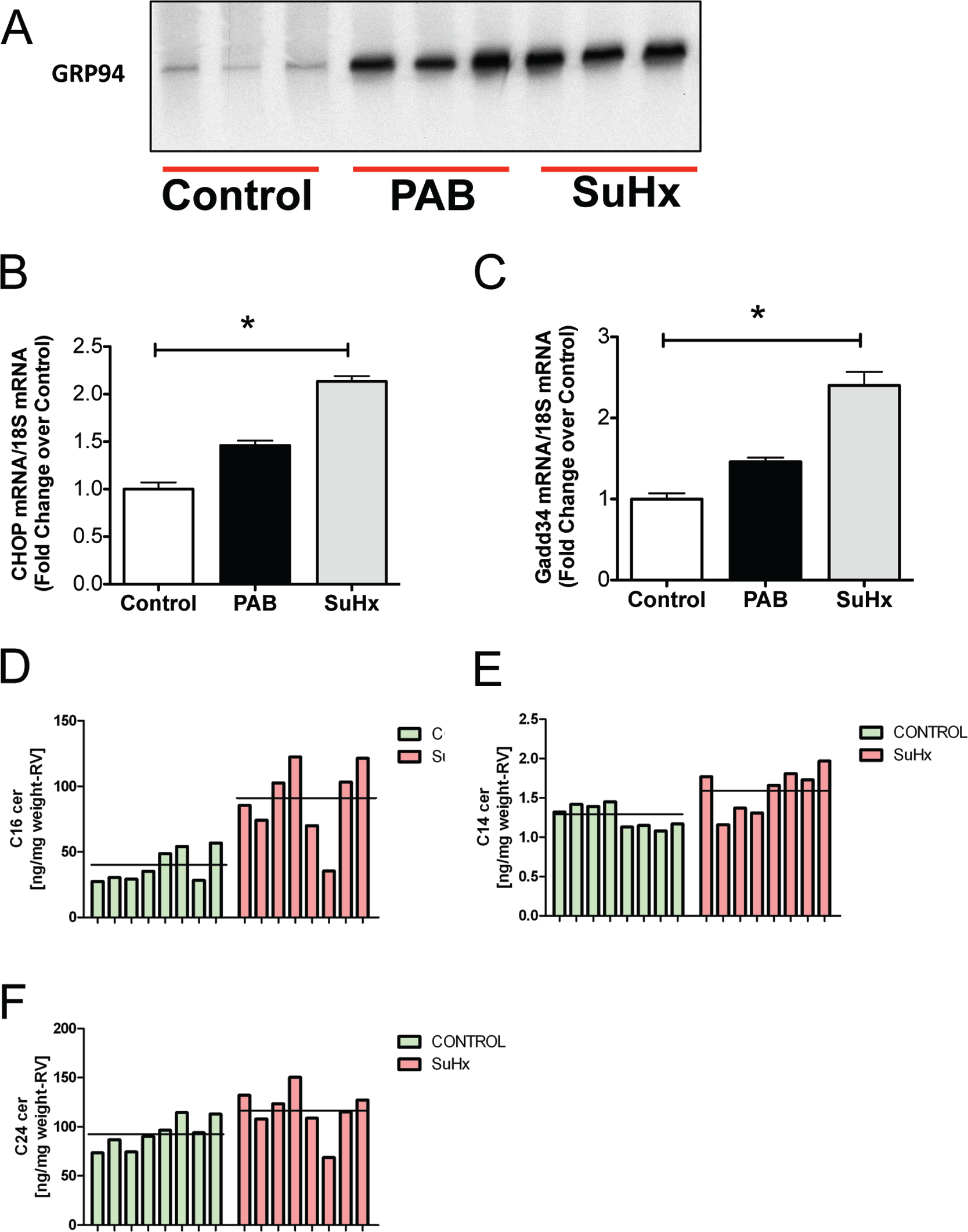
A, Western blots showing increased levels of GRP94. B, C, Quantitative polymerase chain reaction demonstrating increased levels of CHOP (growth arrest and DNA damage-inducible, Gadd153) and Gadd34. GRP94, CHOP, and Gadd34 are all markers of endoplasmic reticulum stress. D–F, Chromatography electrospray ionization tandem mass spectrometry demonstrating increased levels of ceramide 16 (C16; D), 14 (C14; E), and 24 (C24; F) species. PAB: pulmonary arterial banding; SuHx: SU5416/hypoxia; mRNA: messenger RNA; RV: right ventricle.
We propose that in the setting of chronic pressure overload, the RV remains compensated by inducing adaptive autophagy with an adequate turnover of damaged proteins and damaged organelles. In other words, in compensated RVH (as modeled in rats with PAB) protein aggregates and damaged organelles are effectively removed from the cells. In contrast, we postulate that compensated RVH transitions to RVF when autophagy becomes maladaptive, the damaged proteins and organelles that are not removed accumulate, and the “congested” cells die. In short, we hypothesize that maladaptive autophagy causes RVF (Fig. 3).
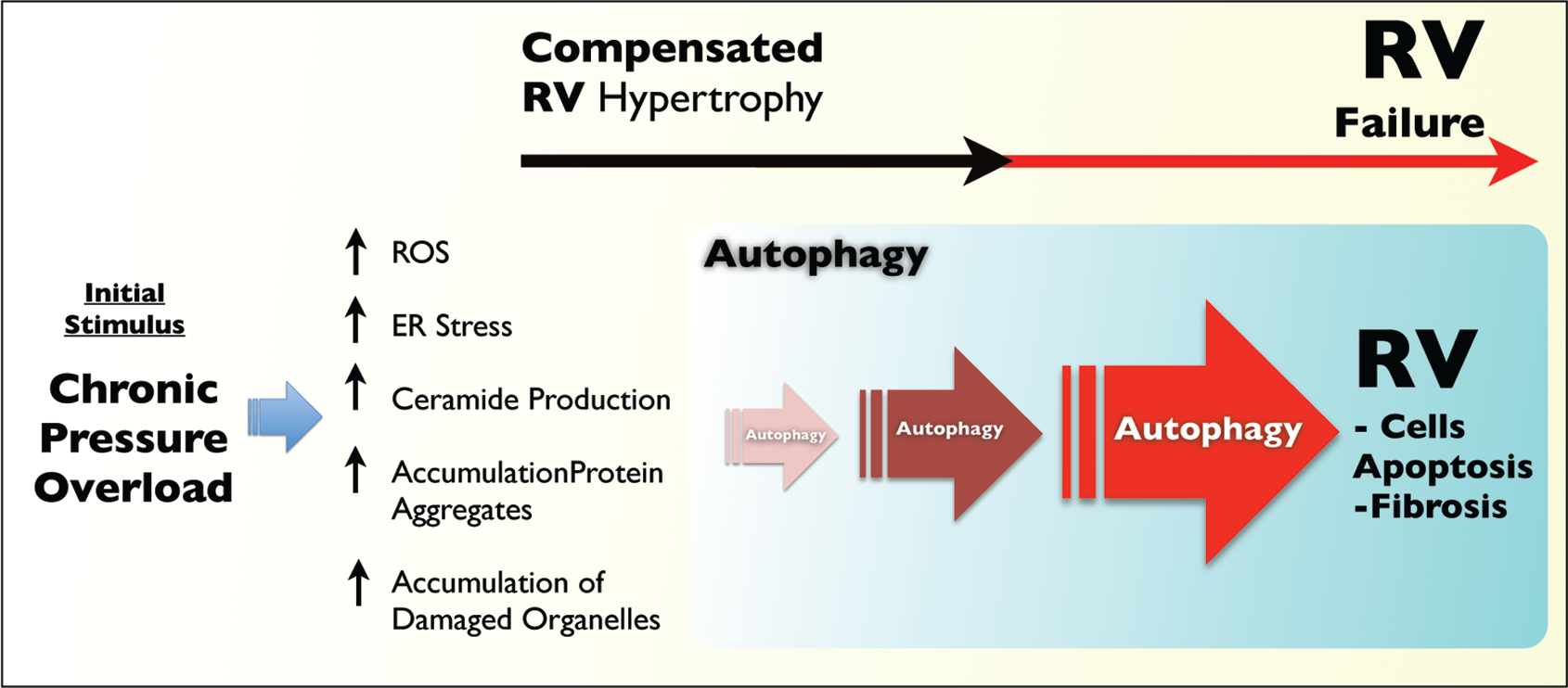
Autophagy/apoptosis concept of a transition of the right ventricle (RV) from compensated hypertrophy to failure. Autophagy is depicted as an initially protective mechanism of cellular waste management. Effective autophagy removes damaged proteins and mitochondria; when overloaded, ineffective autophagy leads to apoptosis. ROS: reactive oxygen species; ER: endoplasmic reticulum.
Hypothesis: the failure-prone RV phenotype
The large variability in the natural history of PAH-induced RVF raises the question of whether long-term survivors or patients who remain in functional class I for many years have a genetic advantage for RV adaptation that patients who progress early to RVF do not. We hypothesize that those individuals who present earlier with RVF symptoms (even when pulmonary hemodynamic changes do not appear to be severe) are genetically prone to express the RVF program (the failure-prone RV phenotype) earlier than others who manifest a more successful RV adaptation. A variation of this hypothesis is that some patients are genetically prone to neurohormonal overactivation, which either triggers or contributes to the transition from compensated RVH to RVF.18,29 In the first scenario, the potential to generate and maintain a strong and resilient RV muscle is genetically controlled; in the second scenario, the ability of the pressure-overloaded RV to meet the metabolic demands produced by a sympathetic-adrenal overdrive is under genetic control. Characterizing the population of patients with an adapted RV could shed light on the mechanisms that mediate cellular adaptation to pressure overload that could serve as potential therapeutic targets.
Hypothesis: the sick lung circulation
As reviewed by others,30,31 there is a strong consensus that in patients with PAH all three layers of the pulmonary arterioles are morphologically and physiologically altered—hence the term “sick lung circulation” (Fig. 4). Because chronic RV pressure overload does not immediately translate into RVF in humans (i.e., patients with Eisenmenger syndrome) and rats (PAB), at least not until later disease stages,10,32 we hypothesize that load-independent factors could also contribute to the molecular RVF profile we have addressed so far. We asked, how does the sick lung circulation affect cardiac function? We hypothesized that phenotypically altered lung vascular cells from the sick lung circulation contribute to the progression from adaptive to maladaptive RVH. In this intricate molecular model of cardiopulmonary interaction, a fraction of the cardiac output and the total lung vessel effluent enters the coronary circulation, and circulating cells and cell fragments, free DNA, and exosomes containing mircoRNAs (miRNAs) from the sick lung circulation come into contact with the myocardial microcirculation and can be taken up and processed by microvascular endothelial cells. For instance, it has been published that circulating free DNA, including mitochondrial DNA, can be cytotoxic, 33 and miRNA can reprogram endothelial cell genes. Microparticles have been shown to activate endothelial cells in an interleukin 1β–dependent manner, 34 and endothelial cell progenitor–derived microvesicles can activate an angiogenic program in endothelial cells via transfer of messenger RNA. 35 Although to date not formally tested, there is a possibility that any of these factors contribute to RVF by affecting the microcirculation of the RV. Indeed, such a “bad-humor” hypothesis of Hippocrates and Galen—which is now undergoing a revival 36 —could be applied to both right and left heart dysfunction in the setting of severe PAH and even pulmonary venous hypertension.
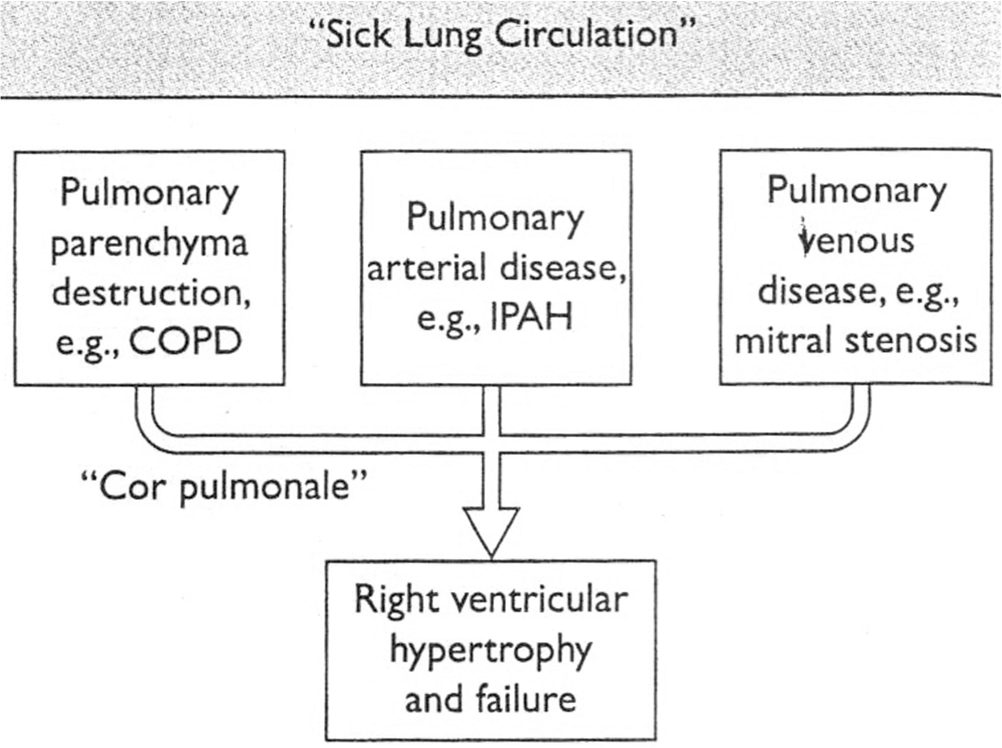
Schematic. In chronic lung diseases that involve the lung microvessels (such as chronic obstructive pulmonary disease [COPD]/emphysema and the group of severe angioproliferative forms of pulmonary arterial hypertension [PAH], including idiopathic PAH [IPAH]), the lung microvessels are histologically and functionally altered—that is, they are sick. In chronic left heart failure, paradigmatically illustrated by mitral valve disease, lung capillaries are damaged, and postcapillary vesicles are remodeled and show signs of inflammation. In these entities, right ventricular hypertrophy and failure are associated with a sick lung circulation.
THE LUNG CIRCULATION–RV AXIS: IMPLICATIONS FOR FUTURE THERAPIES
Because PAH is a disease of the lung vessels, it was—and continues to be—intuitive to design pharmacotherapies to reverse the vascular remodeling. As the pathobiology of PAH has evolved and new concepts have been introduced, 30 new pharmacotherapies face a critical paradox: whereas PAH is characterized by proliferation of apoptosis-resistant cells, the RV is (or at least appears to be) characterized by the opposite end of the vascular biology spectrum, capillary rarefaction and cardiomyocyte apoptosis. Common targets are yet to be identified, but until discovered any new pharmacotherapy should also address a potential effect on the RV. For example, sildenafil has been shown to increase RV inotropy, 37 and conversely agents like bosentan exert a negative effect on RV function. 38 We propose that when evaluating the effects of a pulmonary hypertension drug, researchers should look for a concomitant improvement in RV function. Alzoubi et al. 39 reported that dehydroepiandrosterone (DHEA) reduced the severity and frequency of lung vascular occlusive lesions in the SuHx rat model of severe PAH, but, most importantly, treatment with DHEA also restored RV function, and this improvement was associated with recovery of the RV capillary density and reduced expression of apoptosis markers. Similarly, Spiekerkoetter et al. 40 treated SuHx rats with FK506 (tacrolimus), which, by means of a high-throughput drug screen, was previously identified to be a potent activator of bone morphogenetic protein receptor type 2 signaling. Tacrolimus reversed the obliteration of the pulmonary vessels and reduced the right ventricular pressure and RVH.
Perhaps one of the important clinical issues in the treatment of patients with severe PAH is prevention of RVF and treatment of RVF. When it comes to pharmacotherapies, the RV remains the forgotten chamber. However, in the experimental setting there have been reports of drug-induced improvement of chronic RVF that did not affect the lung circulation. Bogaard et al. 18 showed that treatment with the nonselective adrenergic receptor blocker carvedilol improved RV function and exercise tolerance. However, the exact mechanism whereby carvedilol treatment improved the function of the failing RV remains to be determined. More recently, Syed et al. 41 showed that treatment with the prostacyclin analogue iloprost improved RVF function and RV fibrosis.
Last, although not yet mechanistically examined, interventions like thrombendarterectomy for treatment of PAH associated with pulmonary embolism, 42 atrioseptostomy, 43 and lung transplantation—all of which reduce the RV afterload and pulmonary vascular resistance to blood flow—reverse RVF and most likely the molecular RVF program.
SUMMARY AND CONCLUSION
Pathobiological concepts come and go—and some celebrate a comeback after decades of hiding in the shadows. We believe that the bad-humors idea of antiquity could now be examined on the basis of our new knowledge of the cardiac secretome, 44 along with the new understanding that microparticles act as carriers of information between organs and cells. The mechanical interaction of the RV–lung circulation unit has been addressed extensively;45–47 in this review, we have proposed an additional or complementary hypothesis that can help explain the transition from adaptive RVH to maladaptive RVH and failure.
