Abstract
The Third International Right Heart Summit was organized for the purpose of bringing an interdisciplinary group of expert physician-scientists together to promote dialogue involving emerging concepts in the unique pathophysiology, clinical manifestation, and therapies of pulmonary vascular disease (PVD) and right heart failure (RHF). This review summarizes key ideas addressed in the section of the seminar entitled “Transplantation in End-Stage Pulmonary Hypertension.” The first segment focused on paradigms of recovery for the failing right ventricle (RV) within the context of lung-alone versus dual-organ heart-lung transplantation. The subsequent 2-part section was devoted to emerging concepts in RV salvage therapy. A presentation of evolving cell-based therapy for the reparation of diseased tissue was followed by a contemporary perspective on the role of mechanical circulatory support in the setting of RV failure. The final talk highlighted cutting-edge research models utilizing stem cell biology to repair diseased tissue in end-stage lung disease—a conceptual framework within which new therapies for PVD have potential to evolve. Together, these provocative talks provided a novel outlook on how the treatment of PVD and RHF can be approached.
Keywords
The Third International Right Heart Failure Summit was held in Boston in the fall of 2013 for the purpose of bringing an interdisciplinary group of expert physician-scientists together to promote dialogue involving emerging concepts in the pathophysiology, clinical manifestation, and therapies of pulmonary vascular disease (PVD) and right heart failure (RHF). Specific goals for the symposium were to develop an integrative nomenclature that standardizes and effectively captures right heart disease, to present contemporary theories in the pathobiology and hemodynamics of the right ventricular (RV)–pulmonary vascular axis, and to discuss novel therapeutic approaches that may be used in the treatment of patients with various right heart syndromes. To cover these broad-ranging topics, the symposium was organized into the following 3 sections: (1) Pulmonary Hypertension and the Right Ventricle—Thinking Outside the Box, (2) Emerging Hemodynamic Signatures of the Right Heart, and (3) Transplantation in End-Stage Pulmonary Hypertension. Key concepts addressed in the third section of the summit are the focus of this review.
COMPLEX DECISIONS IN RV RECOVERY: LUNG OR HEART-LUNG TRANSPLANT
Dr. Philip Camp opened the second day's lecture series with a review of the epidemiology, indications, eligibility, and outcomes for patients undergoing heart-lung transplantation, as compared to lung transplantation alone. Considerable advances in medical therapy and the ability to follow patients over time have contributed to a decrease in the acute need for heart-lung en bloc transplantation. According to the International Society for Heart and Lung Transplantation (ISHLT) registry, the number of heart-lung transplants has indeed declined from 1990 to 2010, with less than a third of these being performed for idiopathic pulmonary arterial hypertension (IPAH).1,2 Selection criteria for lung and heart-lung transplant were reviewed, and it was emphasized that appropriate patient identification and transplant discussion must occur early in the treatment process. Eligibility entails the absence of significant multisystem dysfunction, New York Heart Association class III or IV functional status, and failure of medical therapies.
Candidacy for lung transplantation alone in the United States is determined by a scoring system known as the lung allocation score, with groups A–D used to categorize patients according to underlying pathology. Overall median wait time for lung transplantation is now 3.6 months. Patients with PVD (group B) experience the longest wait times, at a median of 9.7 months, compared to the 2.1-month median wait time for patients with restrictive lung disease patients (group D). 3
Which transplant is the optimal choice? Single-lung transplantation for pulmonary hypertension (PH) has largely been abandoned because of adverse postoperative outcomes, including pulmonary edema, graft dysfunction, and need for prolonged mechanical circulatory support (MCS) and ventilation.1,2 The gold-standard and most common surgical treatment option is double lung transplantation, which allows for lower rates of postoperative diffuse alveolar damage, delayed onset of bronchiolitis obliterans syndrome (which may play a role in chronic rejection), improved pulmonary vascular resistance (PVR), and sustained improvement in RV ejection fraction (EF).4,5 Despite these noted advances, survival for PH patients after transplant remains suboptimal, with a median survival of 5 years. The early postoperative period (3–6 months) remains the time of greatest vulnerability for IPAH patients; however, if survival is maintained beyond years 1–3, long-term outcomes are superior to those for patients with chronic obstructive pulmonary disease (COPD), sarcoidosis, or idiopathic pulmonary fibrosis (IPF) (Fig. 1).1,2
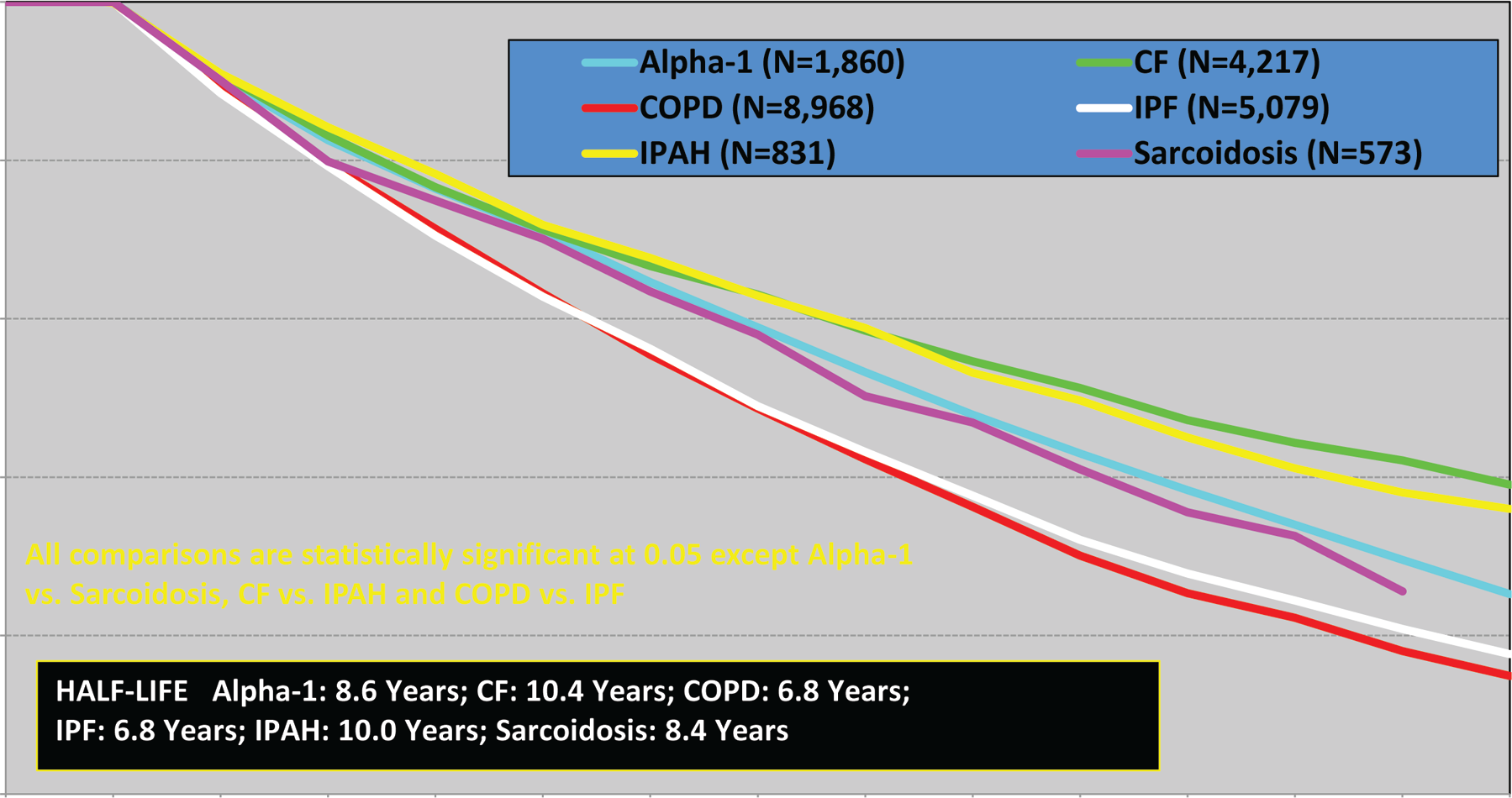
Kaplan-Meier survival by diagnosis for adult lung transplants performed between January 1990 and June 2010, conditioned on surviving to 1 year. Among patients surviving at least 1 year, there were better conditional half-lives after transplant for diagnoses of CF (10.4 years), PAH (10.0 years), sarcoidosis (8.4 years), and Alpha-1 (8.6 years) than for diagnoses of COPD or IPF (6.8 years for both). Alpha-1: α1-antitrypsin deficiency emphysema; CF: cystic fibrosis; COPD: chronic obstructive pulmonary disease; IPF: idiopathic pulmonary fibrosis; PAH: pulmonary arterial hypertension. Reproduced with permission from Figure 18 of Christie et al. 1
Heart-lung transplantation remedies the scenario in which a patient is deemed “too sick” for lung transplantation alone for fear of unrecoverable right ventricle (RV) function. Though historic indications of congenital heart disease and PH with poor RV function remain relevant, contemporary candidates include patients inappropriate for MCS bridging or who have suffered MCS-related complications. The large dual-organ bloc is implanted with a short ischemic time and naturally provides a matched cardiopulmonary relationship. Wait times are increased, and long-term survival is reportedly lower (64% at 1 year),1,2 compared to those for lung transplant alone. These data are not randomized, however, and may be a reflection of pretransplant heart-lung patients being more critically ill, with worse end-organ damage. Survival for PAH patients undergoing heart-lung transplant is similar to patients with congenital heart disease. 1
Despite increases in lung transplantation worldwide, only a minority of these procedures are performed for PH and RHF. Determining the optimal timing of transplantation for these patients remains challenging. Eligibility often follows only when right atrial pressures exceed 15 mmHg and/or when the cardiac index is measured as less than 1.8 L/min/m 2 and patients are in cardiogenic shock, which obviates MCS as a bridge to transplant and may increase chances of adverse events after transplantation. Although posttransplant survival has improved for PH patients, the current allocation scoring system may place them at a disadvantage relative to other groups on the pretransplant wait list. 6 Recent attention to this matter prompted a reevaluation of predictors of mortality and wait times and may lead to the incorporation of other factors that reflect heart failure. 7 Dr. Camp concluded his presentation with a proposed shift in the PH-RV treatment paradigm from “treating the failing organ to rescuing and resuscitating the failing organ,” invoking reports of MCS used to salvage the RV and lungs. 8 With extracorporeal life support, for example, the RV may be allowed a chance to positively remodel, which could lead to improved outcomes after transplant.
APPROACHES TO RV SALVAGE THERAPY, PART 1: CELLS
The next segment was devoted to current evolving therapies used to salvage RV function in the setting of end-stage disease. In the first talk, Dr. Aaron Waxman reviewed the mechanisms of altered cellular metabolism in PVD and RHF, providing the conceptual framework within which cell-based therapies are developing to restore RV function. He reminded the audience that the RV is the key determinant of clinical presentation and survival in patients with PH, rather than mean pulmonary arterial pressure, which correlates poorly with patient symptoms. 9
Alterations in cellular metabolism
As PVR approaches systemic vascular resistance, flow to the RV may be impeded. Regional hypoxia develops in the myocardium, which in turn results in alterations in several downstream regulatory pathways, causing capillary rarefaction and worsening hypoxia. As the hypoxia progresses, decreased mitochondrial density is observed, along with a shift in mitochondrial function from the highly efficient process of oxidative phosphorylation to the less efficient process of glycolysis. Specifically, the combination of (1) decreased vascular endothelial growth factor (VEGF) expression, (2) increased transcription of glucose transporter 1, hexokinase, and lactate dehydrogenase kinase, which inhibit glucose oxidation, and (3) reduced coronary perfusion and capillary density results in ischemia and drives the shift to glycolysis.10–13 Work done by Xu et al., 14 as well as by Dr. Waxman's group, has demonstrated decreased mitochondrial dehydrogenase, number of mitochondria per cell, and mitochondrial DNA content in diseased PA endothelial cells, all of which improved after exposure to nitric oxide donors. Similarly, PA smooth muscle cells exhibited disordered mitochondrial metabolism and redox signaling, resulting in decreased oxygen extraction and production of adenosine triphosphate (ATP). Patients with PAH and RHF have increased oxygen requirements, lower oxygen extraction reserve, and increased dependence on coronary blood flow.15–18 Positron emission tomography imaging has been used to demonstrate the shift to the energetically less favorable glycolysis, where increased 18-fluorodeoxyglucose uptake is seen in untreated PAH-induced RV hypertrophy. 19 The vicious cycle of RV failure ensues (Fig. 2), 17 with various potential targets for ameliorating RV function.
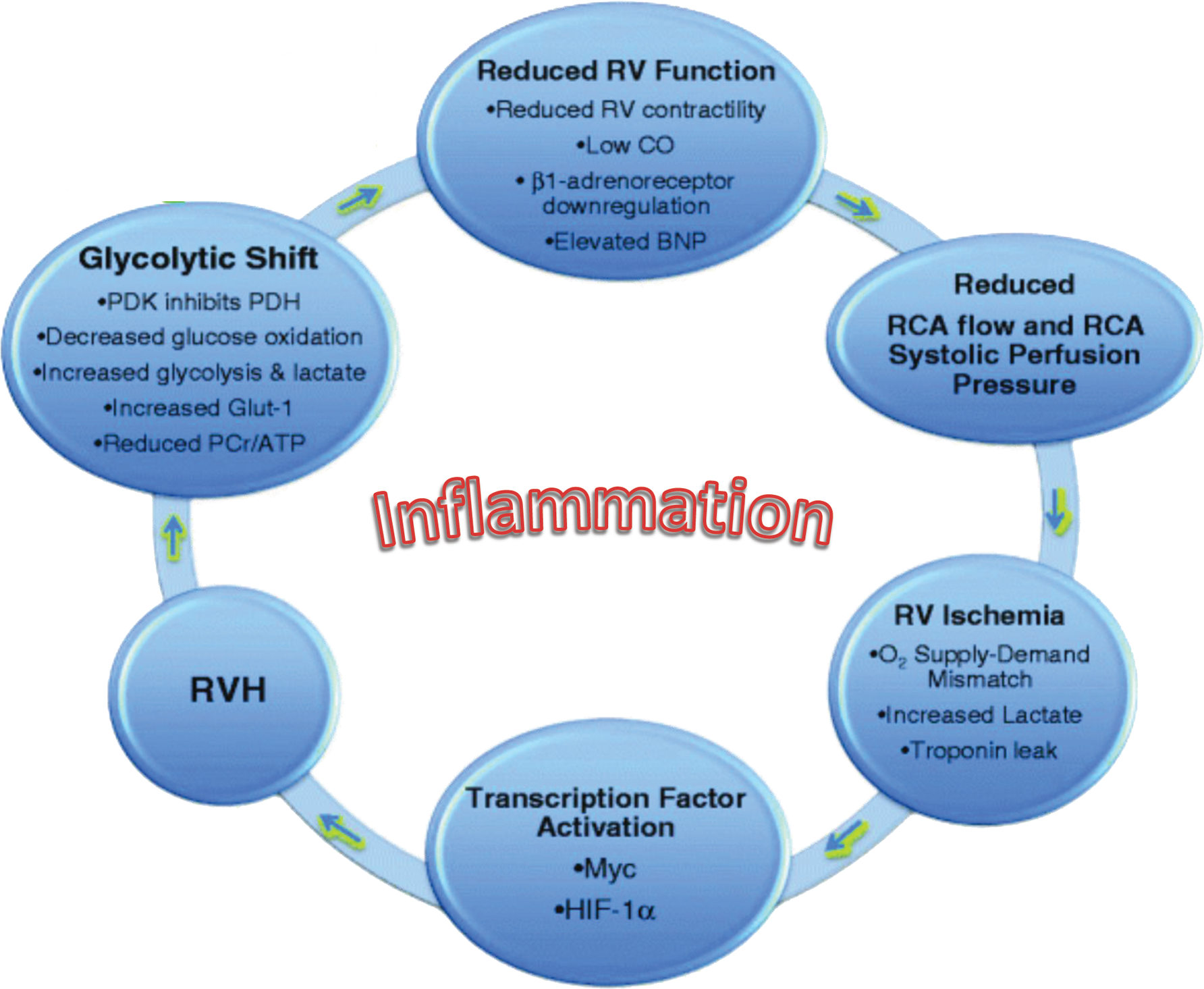
Cycle of right ventricular (RV) failure. Reduced RCA flow and RCA systolic perfusion pressure in RVH are ultimately unable to support the increased oxygen demand of increased RV mass. This leads to RV hypoxia and ischemia. Transcriptional factors (HIF-1α and Myc) are activated by RV ischemia, which contributes to a switch from mitochondrial metabolism (glucose oxidation) to glycolysis. RV function is then further reduced by the depressed ATP production and myocardial acidosis, leading to RV failure as a part of a continuous cycle. BNP: brain natriuretic peptide; CO: cardiac output; Glut-1: glucose transporter 1; HIF-1α: hypoxia-induced factor-1α; PCr: phosphocreatine; PDH: pyruvate dehydrogenase; PDK: pyruvate dehydrogenase kinase; RCA: right coronary artery; RVH: RV hypertrophy. Adapted, with kind permission of Springer Science+Business Media, from Figure 1 of Piao et al. 17
Stem cell therapy
Having outlined a comprehensive rationale, Dr. Waxman turned to the focus of his talk, cell-based therapy in regenerative medicine. Conventional stem cell therapy is thought to develop from totipotent stem cells, which have the ability to differentiate into many different adult cell types and form specialized tissue needed for embryonic development. More pertinent approaches today involve the use of pluripotent (plural) or multipotent (mature) cells, which have potential to form differentiated adult cell lines. There are essentially 2 types of stem cells: embryonic stem cells, which are received from aborted embryos created via in vitro fertilization, and adult stem cells, which are received from limited tissues, namely, bone marrow, muscle, and brain, but also from the placental cord and baby teeth. Stem cells exist in micro-environments called “niches,” where they are in direct contact with and exposed to soluble factors that provide support and signals regulating self-renewal and differentiation. Depending on what kind of environment they are nurtured in, there is a tendency to generate cell types of the tissue in which they reside. Recent experiments have raised the possibility of stem cells from one tissue giving rise to other cell types, forming the basis of a concept known as plasticity.
Cell-based therapy can be used to repair, regenerate, or replace damaged organs. For the purposes of treating patients with RHF and PVD, the hope is to stimulate tissue at the cellular and molecular level to repair itself. In work by Dr. Duncan Stewart's group, 20 bone marrow–derived endothelial progenitor cells (EPCs), which function to repair and regenerate blood vessels, as well as endothelial nitric oxide synthatase (eNOS)–engineered EPCs, were used to prevent injury and restore microvasculature structure and function in monocrotaline-induced PAH. Cumulative survival was improved most with eNOS-engineered EPCs, followed by regular EPCs, compared to controls, in the setting of monocrotaline-induced lung injury. 20 Dr. Waxman expressed the increased facility of using mesenchymal stem cells (MSCs), which are easier to isolate and can be obtained from cord, fat, and bone marrow.21,22 They have paracrine and angiogenic properties, which include the secretion of growth factors (VEGF, fibroblast growth factor, and hepatocyte growth factor) and stimulation of endothelial cell proliferation. These cells also have anti-inflammatory properties, conferring the inhibition of T cell proliferation, downregulation of proinflammatory factor secretion, and reduction of oxidative stress and endothelial damage, as seen in murine models of hind-limb ischemia. 23 MSCs can repair a variety of tissues after injury and are currently being used in clinical trials to treat patients with cardiovascular disease. The precise mechanism of action and the role of myocyte regeneration versus angiogenesis, however, are controversial.24–27 In a canine model of anterior infarction and chronic ischemia, large numbers of MSCs were infused without causing damage. In fact, MSCs differentiated into smooth muscle cells and endothelial cells, resulting in significant left ventricular EF improvement at 60 days and a trend toward reduced fibrosis and greater vascular density. 28
Understanding possible mechanisms by which stem cells restore function compels returning to the essential roles of mitochondrial function, which involve not only the regulation of metabolism and energy production but also control of stress responses, apoptosis, necrosis, and organ hibernation.29–31 When healthy MSCs are mixed with vascular stem cells, they bridge to one another with nanotubes wherein the transfer of functional mitochondria can be observed. 32 Spees et al. 33 demonstrated this process by using a lung cancer cell line called A549 cells; they rendered mitochondria is this line dysfunctional, so that they no longer were able to generate substantial ATP. After these cells were mixed with MSCs, the formation of nanotubes was observed, enabling transfer of healthy mitochondria to diseased cells and restoration of A549 cells' ability to generate increased ATP. 33 Similarly, a protective murine model showed mitochondrial transfer from healthy bone marrow–derived stromal cells (BMSCs) to pulmonary alveoli in lipopolysaccharide-induced lung injury. Lung injury, as indicated by protein leak and leukocytosis, was retracted, and increased ATP generation was observed with transfer of healthy mitochondria from BMSCs. 34 Although mitochondrial transfer has been demonstrated as such in vitro, it is unclear whether this occurs in vivo.
Dr. Waxman concluded the presentation by sharing a swine model developed in his laboratory to study this paradigm. Injection of amide-coated glass beads was used to provide for an inflammatory response and obstructive vasculopathy–induced RHF, defined as a decrease in cardiac output by 35%, a decrease in Po2 to less than 70 mmHg, and RV hypertrophy. This model allows for stem cell therapy delivery directly via the right coronary artery and assessment of its effect on right-heart response over time. Questions that remain in this emerging field include, but are not limited to, the following: (1) What is the mode of action by which mitochondria improve function? (2) What is the fate of these stem cells? (3) Can these cells migrate to nontarget sites? (4) Is there potential for uncontrolled cell proliferation or tumorigenicity? (5) Are there potential graft-versus-host effects that can ensue? The goal of this science will be the translation of therapy to clinical practice, to potentially allow for alternative means of rescue therapy and bridging to transplantation for patients with RHF in the setting of end-stage PVD.
APPROACHES TO RV SALVAGE THERAPY, PART 2: MACHINES
Dr. Francis Pagani delivered the second half of this 2-part segment by providing an experienced surgeon's insight into the perhaps more commonly employed salvage therapy, MCS. Optimal timing for initiation of MCS is vital to ensure favorable outcomes and is based on a number of variables: the level of and response to medical support, hemodynamics, end-organ perfusion, past history, transplant status, and institutional experience with various MCS devices, among others. In addition to timing, several important questions warrant careful consideration to determine appropriate MCS selection: (1) Does the patient require partial, versus full, hemodynamic support? (2) Is there univentricular or biventricular involvement? (3) Is significant hypoxia present? (4) Is the inciting event acute or an exacerbation of chronic disease? (5) What devices are available, and what is the level of local expertise? Germane to the focus of this symposium, Dr. Pagani reiterated that the most common shortcoming in providing support to the left heart is the failure to recognize significant right heart compromise and the need for biventricular support. Improved patient selection, patient management, and device technology have reduced post–left ventricular assist device (LVAD)–implant need for RV assist device (RVAD) support. Still, 15%–20% of the MCS patient population suffers from consequences of RV failure, exhibiting failure to thrive, poor functional improvement, and dramatically accelerated short-term mortality. 35
Perhaps what remains most elusive is defining an end point for MCS therapy. If the end point is recovery, then clinicians have to be prepared to withdraw mechanical support when recovery is not feasible. If the end point is transplantation, then the goal of MCS is to optimize nutrition and other organ function while awaiting an appropriate donor. The utilization of short-term percutaneous devices as bridges to transplant provides for suboptimal outcomes, however, and thus these devices are often transitioned to more durable devices—a “bridge-to-bridge” tactic. 36 Dr. Pagani presented an algorithmic approach to right-sided circulatory failure in which, after an etiology is determined, the extent of pulmonary failure, biventricular involvement, and the need for short-term versus long-term support are considered to determine the optimal MCS device. Options include percutaneous and durable devices as well as venoarterial, venovenous, and right atrium–left atrium extracorporeal membrane oxygenation (ECMO) configurations (Fig. 3).
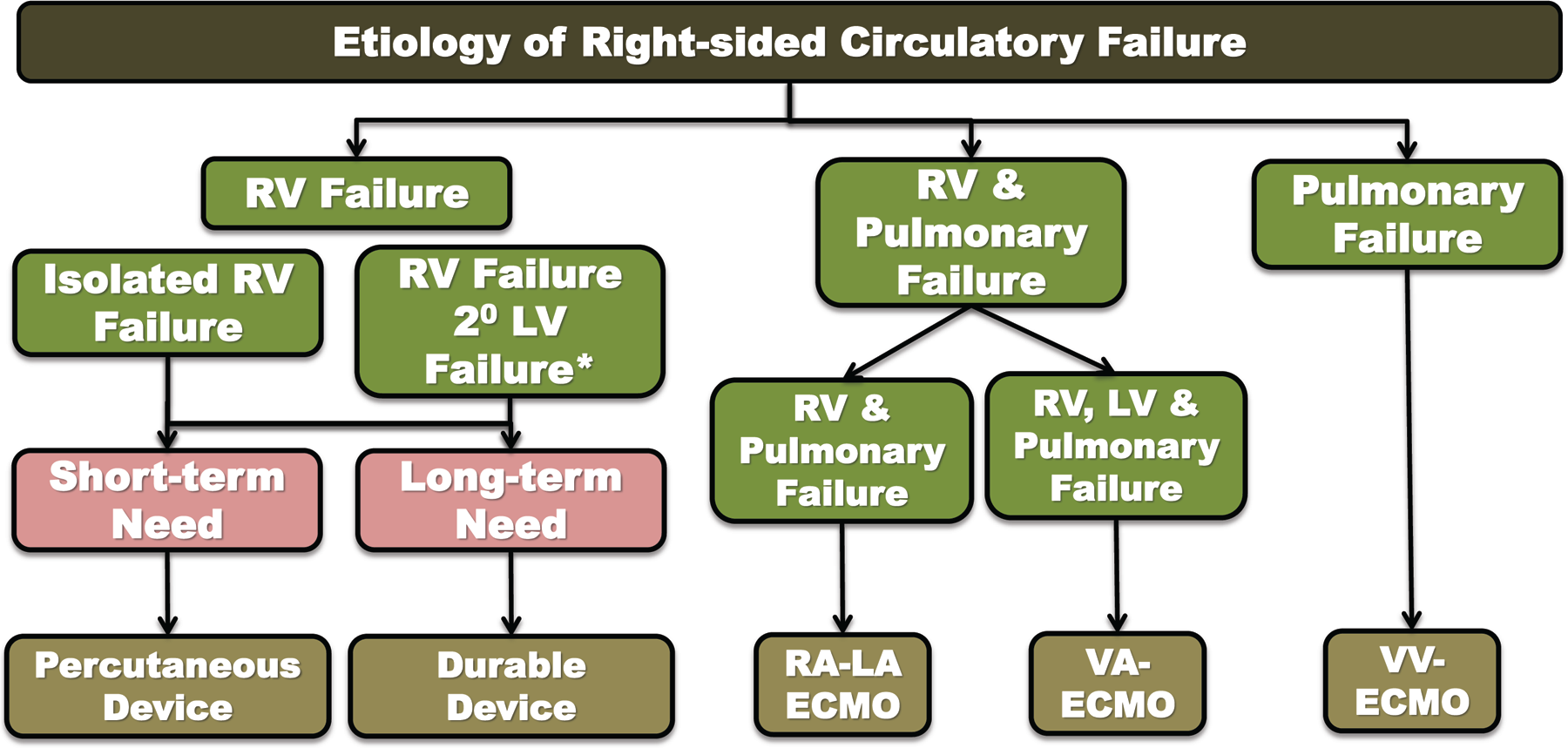
Algorithmic approach to device consideration for right-sided circulatory support in the setting of right-sided circulatory failure. ECMO: extracorporeal membrane oxygenation; LA: left atrial; LV: left ventricular; RA: right atrial; RV: right ventricular; VA: venoarterial; VV: venovenous; 2°: second-degree. Figure provided by Dr. Francis Pagani and reproduced with his permission.
Devices designed for RV support. Temporary support options (extracorporeal systems) include the Levitronix CentriMag, Abiomed AB5000, and Berlin Pediatric Excor VADs, which all require surgical implantation. These systems are most frequently used in postcardiotomy shock scenarios and are less commonly employed for RV failure after LVAD implantation. The most commonly utilized device in the United States is the Levitronix CentriMag, which can be configured to provide full or partial support for the RV, the LV, or both. This device is a continuous-flow centrifugal pump with an internal rotor that is magnetically levitated with no internal bearings, allowing for low levels of hemolysis. It provides 6–9 L/min of support, is durable, can be used for ECMO circuits, and is approved by the US Food and Drug Administration (FDA) for RVAD use for up to 30 days. 37 In ideal cases, patients can ambulate, allowing for optimization and recovery. Unfortunately, however, rates of stroke, bleeding, and infection remain very high. A new percutaneous option is offered by the Abiomed Impella RP RVAD, which has yet to be approved by the FDA. It is a centrifugal pump with an axial design and a mechanical pivot that is inserted percutaneously through the femoral vein and positioned in the pulmonary artery. Blood from the inferior vena cava is aspirated and expelled into the pulmonary artery at a maximum rate of 4.4 L/min. It is currently under investigation for post–LVAD implantation and postcardiotomy RHF in the Recover Right trial (http://clinicaltrials.gov, NCT01777607). Dr. Pagani pointed out, however, that despite consistent improvement in hemodynamics, percutaneous support devices have failed to demonstrate a survival benefit in several clinical trials.38–42 This may be due to the fact that RV or LV dysfunction did not have sufficient time to resolve, and thus temporary support did not suffice or translate to a survival benefit.
Durable devices for RV support include the Thoratec IVAD, the Thoratec pVAD, and the SynCardia Cardio-West Total Artificial Heart (TAH). The 2 Thoratec VADs represent flexible, pneumatically actuated systems that use older technology and are FDA approved as bridges to recovery or transplantation. Although the pneumatic systems are large and cumbersome and require tethering to a portable drive console, these devices allow for patients to be discharged to home. The SynCardia TAH is FDA approved for bridge to transplantation for severe biventricular failure, intractable arrhythmias, unusual anatomy, or complicated reoperation. 43 Data from the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) show TAHs confer a higher 3- and 6-month survival compared to biventricular assist devices (85% vs 70% and 68% vs 62%, respectively). 44 New opportunities to utilize continuous-flow pump technology for a TAH system are on the horizon. This technology results in pulseless circulation, can be used for adult congenital heart disease, and allows for dynamic responses to physiologic demand. Dr. Frazier 45 was the first to implant 2 modified Thoratec HeartMate II devices to provide for a continuous-flow total heart replacement in the United States.
Exciting prospects exist for the implementation of durable devices to provide right-sided support. The HeartWare HVAD is a continuous-flow rotary pump with centrifugal flow designed for LV assist that has also been used for RV and biventricular circulatory support,46,47 although it is not approved for these indications. The challenge of using LVAD technology for RV support, however, lies in the required modification of current devices to cater to RV morphology and physiology. The inlet cannula, for example, is designed for the thickness of the LV and is too long for the thin RV free wall. Differences in afterload may also require the pump to run outside of its normal operating range. Dr. Krabatsch and his group 47 in Berlin were the first to report a modified approach to biventricular support using the HVAD technology, in 17 patients for whom 30-day survival was 82% (Fig. 4). As systems shrink in size, applications for biventricular support become more promising. The HeartWare Miniaturized Ventricular Assist Device (MVAD) centrifugal pump will provide up to 6 L of flow and will enable minimally invasive surgical approaches. In fact, the multicenter trial to study this technology (http://clinicaltrials.gov, NCT01831544) may include an arm to evaluate efficacy in providing biventricular support.

Translating the concept of modified use of LVAD technology to the interventional laboratory, Dr. Navin Kapur and colleagues 48 reported a multicenter experience of a percutaneous centrifugal-flow RVAD (TandemHeart, Cardiac-Assist, Pittsburgh) as a part of a registry in 46 patients. Their findings suggest that this technology is feasible in a variety of acute clinical care settings and warrants prospective evaluation.
In closing, Dr. Pagani touched on the provocative consideration of MCS for RHF in the setting of PVD. A primary concern for RVAD support in this setting is that the generation of high flows across a diseased circulation with high resistance may increase PA pressures and elicit pulmonary hemorrhage. 49 Axial and centrifugal pumps, in particular, are exquisitely sensitive to high afterload, which may proscribe their application for this patient population. Partial-assist devices, such as the CircuLite device or the Impella RP RVAD, may provide adequate hemodynamic support without overwhelming the pulmonary circulation; however, their use in patients with end-stage PVD remains speculative. Where RVAD technology may play a pivotal role is for the select patient with controlled PH and residual RV failure, as a bridge to recovery or transplantation. Because of the high economic burden and unclear outcomes that these potential treatments pose, employment of RVAD technology in patients with RHF and PVD should be considered in the context of a clinical trial.
GROWING NEW LUNGS: DREAM OR REALITY?
Current active clinical trials examining the role of stem cell therapeutics in lung disease are devoted to the study of IPF and COPD but not PH. In the fourth and final talk, delivered by Dr. Edward Ingenito, the focus returned to evolving cell-based therapy as it may apply to regeneration of lung tissue and parenchyma in advanced disease. He shared an ongoing investigation within the context of emphysema and hypothesized how this developing therapy may be applied to the field of PVD.
Although considerable progress has been made in medical therapy for PH, when compared to other advanced lung diseases, including asthma, emphysema, and IPF, limited treatments exist outside of transplantation for any end-stage pulmonary pathology. The umbrella of restorative therapy to reinstate the structure and function of damaged tissue encompasses adaptive remodeling (lung volume reduction surgery for emphysema or myectomy for cardiomyopathy), orthotopic tissue transplant, and regenerative therapy. The latter involves gene therapy, pharmacologic approaches, and cell therapeutics to improve function, with an increasing emphasis placed on stem cell therapy over the past decade. Emphysema involves the non-fibrotic progressive destruction of lung parenchyma distal to the terminal bronchi, resulting in the loss of tissue and lung hyperexpansion. A design for regenerative stem cell therapy in this model requires a system for targeting the alveolar compartment and an adaptive cell that retains its regenerative capacity in vivo. Lung structure is comprised of a rich capillary network for gas exchange, a strong, resilient extracellular matrix, and epithelial cells to ensure an intact surface. Therefore, a lineage-committed cell is needed to influence and orchestrate residual cells to rebuild structure and function.
Dr. Ingenito's group 50 developed progenitor cells from explanted intact lung tissue, allowing them to retain their cellular replicative and regenerative potency. Lung-derived mesenchymal cells (LMSCs) play a critical role in activating resident cells to heal tissue. Unlike BMSCs or fibroblasts, LMSCs are highly clonogenic and do not require input from adjacent cells to survive. These cells are also capable of secreting key components of lung basement membrane, which are important not only for developing the extracellular matrix in emphysema but also for expanding the vasculature in disease states such as PH. When LMSCs are added to a basement membrane–like environment, they spontaneously organize into alveolar structures, secreting potent growth factors and mature elastogenic fibers. Although multiple factors block the sustained activation and survival of these cells on both the endovascular and the endobronchial sides, the creation of a scaffold via cross-linking surface integrins was discovered to mediate this process. The scaffold stimulates activation of focal adhesion kinase and integrin link kinase, which permit survival and retained reparative capacity. Ovine models were used in Dr. Ingenito's laboratory to demonstrate successful in vivo delivery of functional reparative cells to target sites in the lung by way of this engineered scaffold system. 50 Elastase was used to induce unilateral emphysema in 4 sheep lungs. Healthy tissue was biopsied and used to generate autologous LMSCs, which were then injected into both diseased emphysematous lung tissue and healthy lung tissue to ensure that no damage ensued upon engraftment. The treatment with autologous cells using a fibrinogen-fibronectin-vitronectin hydrogel, or scaffold, was well tolerated clinically. Computed tomography and nuclear scintigraphy were used to assess tissue mass and lung perfusion, respectively, indicating emphysema test sites. At 4 weeks, cell proliferation and improvement in structure were observed at diseased locations, with no pathology at healthy treated sites (Fig. 5). This model was next tested in a randomized controlled trial, where 10 diseased sheep underwent implantation of scaffold alone (5) or scaffold containing LMSCs (5). At 4-week follow up, LMSC transplantation was well tolerated and resulted in increased tissue mass and perfusion, compared to the control treatment. Increased cell proliferation and extracellular matrix were confirmed on histology. The authors 50 concluded from these studies that autologous LMSC transplantation is feasible and safe and that it may promote regeneration via secretion of growth factors, elastogenesis, and alveologenesis. The FDA has granted permission to initiate a phase 1 clinical trial using this therapy for advanced emphysematous disease for lung transplant candidates on an active waiting list. This experimental model has not been tested in PVD. However, in closing, Dr. Ingenito proposed a rationale for why such a construct may indeed be restorative in the setting of PVD, namely, that activated LMSCs secrete angiogenic growth factors that could potentially contribute to the regrowth of the compromised vasculature. The field of cellular therapeutics is rapidly evolving, with some studies in humans, and may have broader clinical applications with time, including the disease state of PH.
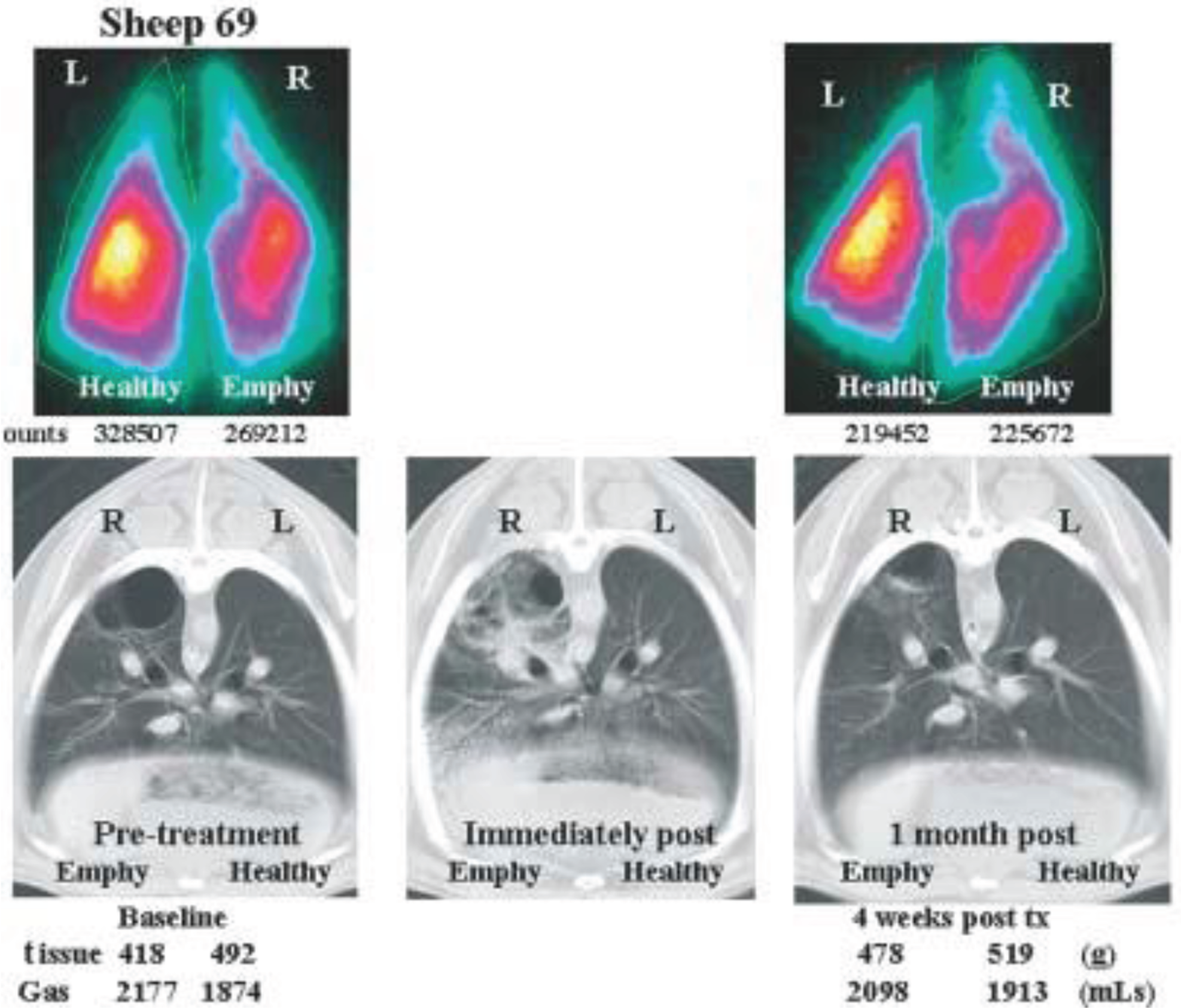
Design and testing of biological scaffolds for delivering reparative cells to target sites in the lung. Quantitative perfusion scans and computed tomography images were used to show elastase-induced emphysematous (emphy) and healthy lung before treatment, immediately after treatment, and 4 weeks after treatment (post tx) with lung-derived mesenchymal stem cells (LMSCs). Copyright 2009 John Wiley & Sons, Ltd.; reproduced with permission from Figure 7 of Ingenito et al. 50
CONCLUSIONS
Although significant progress has been made in the understanding of RV-pulmonary vascular pathophysiology, definitive therapeutic options outside of transplantation are limited for the treatment of end-stage disease. The International Right Heart Summit was a unique forum that enabled cross talk among world-renowned physician-scientists to advance current thinking and consider novel approaches to the treatment of RHF. In this context, currently available and developing technologies for salvaging the RV were reviewed. At one end of the spectrum, models of stem cell proliferation and utilization to repair and regenerate tissue provide for an exciting therapeutic paradigm in the potential treatment of PVD and RV dysfunction. At the other end, advances in the field of MCS may allow for miniaturized temporary and durable assist devices uniquely designed for right heart support as bridges to recovery and/or transplantation. Future work and clinical trials are ongoing to study the clinical application of these wide-ranging novel approaches to the patient with end-stage right heart disease.
