Abstract
Abnormal lung microvascular endothelial vascular barrier function may contribute to pulmonary inflammation, such as that occurring during inhalation of cigarette smoke (CS). Cystic fibrosis transmembrane conductance regulator (CFTR), an anion channel expressed in both epithelial and endothelial cells, regulates the organization of tight junctions between epithelial cells and has also been implicated in the transport of sphingosine-1 phosphate (S1P), a vascular barrier–enhancing sphingolipid. Because CS has been shown to affect CFTR function, we hypothesized that CFTR function contributes to lung endothelial cell barrier and that CFTR dysfunction worsens CS-induced injury. CFTR inhibitors GlyH-101 or CFTRinh172 caused a dose-dependent increase in pulmonary or bronchial endothelial monolayer permeability, which peaked after 4 hours. CFTR inhibition was associated with both intercellular gaps and actin stress fiber formation compared with vehicle-treated cells. Increasing endothelial S1P, either by exogenous treatment or by inhibition of its degradation, significantly improved the barrier function in CFTR-inhibited monolayers. Both cultured lung endothelia and the lung microcirculation visualized in vivo with intravital two-photon imaging of transgenic mice deficient in CFTR showed that CFTR dysfunction increased susceptibility to CS-induced permeability. These results suggested that CFTR function might be required for lung endothelial barrier, including adherence junction stability. Loss of CFTR function, especially concomitant to CS exposure, might promote lung inflammation by increasing endothelial cell permeability, which could be ameliorated by S1P.
INTRODUCTION
There is increasing appreciation of pulmonary vascular endothelial activation induced by inhalation of cigarette smoke (CS), associated with subtle loss of endothelial barrier function and lung inflammation which in turn could contribute to the development of chronic lung diseases such as chronic obstructive pulmonary disease (COPD). 1 The maintenance of pulmonary vascular barrier function is dependent in part on a careful balance between sphingolipids sphingosine-1 phosphate (S1P), which maintains a tight barrier, 2 and ceramide, which increases pulmonary endothelial permeability.1,3 Recently, exposure to CS has been directly linked to marked impairment in the function of anion channel cystic fibrosis transmembrane conductance regulator (CFTR). 4 CFTR deficiency has been shown to increase ceramide in bronchial epithelial cells. 5 Because CFTR is a transporter of S1P, plays a role in maintaining sphingolipid balance, 6 and is expressed on lung endothelial cells, 7 we set out to determine whether impairment of CFTR function could, by itself, lead to and/or exacerbate lung endothelial barrier dysfunction induced by the soluble components of CS. A member of the adenosine triphosphate–binding cassette (ABC) transporter superfamily, CFTR is expressed in numerous tissues and is best known as a regulator of anion transport, particularly across epithelia. Its importance has been primarily studied in the context of cystic fibrosis (CF), a disease induced by genetic mutations in the CFTR gene that cause impaired CFTR function. The discovery of an inhibitory effect of CS on CFTR function drew similarities between COPD and CF, both characterized by progressive obstructive pulmonary disease, varying degrees of lung inflammation, and susceptibility to respiratory infections. 8 Because the endothelium is an active participant in inflammation, it is important to determine the impact of CFTR inhibition on its role as a barrier between the circulatory compartment and the lung.
CFTR is not only a chloride channel, but also an ABC transporter that helps shuttle S1P across the plasma membrane. S1P is a metabolite of ceramide in the sphingolipid metabolism cascade, via ceramidase production of sphingosine, which is then phosphorylated to S1P via sphingosine kinases. 9 The importance of sphingolipid homeostasis to the maintenance of multiple cellular functions cannot be overstated. It has been shown that the loss of CFTR function causes sphingolipid imbalances directly linked to lung epithelial cells hypersusceptibility to Pseudomonas aeruginosa infection 5 and to inhibition of lung endothelial cell stress-induced apoptosis. 10 However, the role of CFTR in the maintenance of vascular endothelial barrier, to our knowledge, has not been described. Interestingly, in the respiratory epithelium, the maintenance of barrier function depends on normal CFTR trafficking for the organization and function of tight junctions. 11 In the endothelium, the tethering mechanisms responsible for barrier integrity are largely a function of the adherens junctions. 12 Whether the normal organization and function of adherens junctions similarly requires CFTR is not known.
We used primary lung endothelial cells from multiple species, including human donors, cultured in monolayers, to test the impact of impaired CFTR function on the endothelial barrier function. Complementing these with intravital two-photon microscopic evaluation of microvascular barrier in lungs of mice with Cftr mutations, we demonstrate an important role of CFTR in maintaining the lung endothelial barrier. We further show that endothelial cell barrier dysfunction induced by CFTR inhibition could be restored by S1P supplementation.
METHODS
Reagents
All reagents were purchased from Sigma-Aldrich (St. Louis, MO), unless otherwise specified.
Cells
Primary rat lung microvascular cells (RLMECs) were kindly provided by Dr. Troy Stevens (University of South Alabama, Mobile, AL) and maintained in Dulbecco's modified Eagle medium (DMEM) high-glucose supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin. Human primary lung microvascular endothelial cells and their culture media EBM2-MV and bullet kit were from Lonza (Walkersville, MD). Sheep bronchial artery endothelial cells were obtained as described 13 and maintained in DMEM with 20% fetal calf serum, 150 μg/mL endothelial cell growth supplement, 100 μg/mL penicillin-streptomycin, 0.25 μg/mL amphotericin B, and 0.1 mM minimum essential medium with nonessential amino acids. All primary cell cultures were maintained at 37°C in 5% CO2 and 95% air. Experiments were performed up to passage 10, with cells confluent in monolayers.
CS extract treatment of cells
Aqueous CS extract was prepared from filtered research-grade cigarettes (1R3F) from the Kentucky Tobacco Research and Development Center at the University of Kentucky. A 100% CS extract was prepared by bubbling smoke from 2 cigarettes into 20 mL of basal culture medium (DMEM) as described elsewhere 1 and was used in cell culture experiments within 20 minutes. A similar procedure was used to prepare the control extract, replacing the CS with bubbling ambient air. Treatments were performed with CS extract concentrations ranging from 1% to 10% (vol : vol), as indicated.
CFTR studies
The following were added to cultured cells for the indicated time before the addition of CS extract: CFTR inhibitors GlyH-101 (Calbiochem, Billerica, MA; 1–10 μM, 1 hour) and CFTR (inh)-172 (5–20 μM, 1 hour).
S1P studies
To increase exogenous S1P, cultured cells were treated with S1P (Cayman Chemical, Ann Arbor, MI) simultaneous with the addition of CS extract and CFTR inhibitors. Pyridoxine analog 4-deoxypyridoxine (4DP), an inhibitor of pyridoxal phosphate-dependent enzyme sphingosine-1-phosphate lyase (S1PL), was used to increase endogenous S1P and was administered to cultured cells concomitant to addition of CS extract and CFTR inhibitors.
Transcellular electrical resistance (TER) measurements
Electric cell substrate impedance sensing (ECIS; Applied Biophysics, Troy, NY) was employed to measure the electrical resistance across the endothelial cell monolayer over time. Cells were cultured to confluence on specialized 8W10E ECIS cell plates (Applied Biosystems), until TER achieved a steady state. Nutrient-rich media was removed and replaced with serum-free media 2 hours before experimental treatments, and TER was recorded for up to 20 hours. TER values (expressed in ohms) for each time point were normalized to the initial TER value (at the beginning of the recording) and plotted as normalized TER. Only wells with an initial TER greater than 1,200 ohm were used.
Immunocytochemistry
Endothelial cells were grown on gelatin-coated cover slips overnight and treated as indicated. After fixation in paraformaldehyde (4%), cells were stained for actin using Texas red phalloidin (Molecular Probes/Invitrogen; 1 : 200), as previously described. 1 Adherens junctional proteins were visualized using immunostaining for VE-cadherin (fluorescein isothiocyanate [FITC]–conjugated V-CAD, Molecular Probes/Invitrogen; 5 μg/mL) and β-catenin (EMD chemicals, Darmstadt, Germany). Nuclei were visualized with 4′,6-diamidino-2-phenylindole (Molecular Probes/Invitrogen). Confocal images were collected using either an Olympus FV1000-MPE confocal microscope mounted on an Olympus IX81 inverted microscope stand with an Olympus Plan Apo 60 × water immersion objective, numerical aperture (NA) 1.4, or a Nikon Eclipse (TE200S) inverted fluorescence microscope with a 40 × objective, NA 1.4.
Intact mouse preparation and intravital microscopy
All animal studies were conducted in compliance with the Institutional Animal Care and Use Committee guidelines of Indiana University. B6.129P2(CF/3)-CftrTgH(neoim)Hgu (abbreviated CftrMHH) congenic mice were used. The inbred CF strain CF/3-CftrTgH(neoim)Hgu was established by strict brother-sister mating from the original CftrTgH(neoim)Hgu mutant mouse that produces low levels of CFTR and was generated using insertional mutagenesis in the Cftr exon 10. 5 Then, the congenic CftrMHH strain was generated by backcrossing the targeted mutation into the Bl6 inbred background. Homozygous mice were used for experiments, and wild-type littermates were used as controls.
Mice were anesthetized by inhaled isoflurane (5% in oxygen), orotracheally intubated with a 20-gauge catheter, and ventilated at a rate of 130 breaths/minute. For the remainder of the experiment, general anesthesia was maintained with isoflurane (2% in oxygen). The right internal jugular vein and the carotid artery were cannulated via cutdown with a 26-gauge catheter for administration of fluid and fluorescent probes and for monitoring of systemic blood pressure, respectively. A thoracotomy was performed in the fifth left intercostal space and the sixth rib was excised. The window, measuring 0.5 cm in diameter, was interfaced with the lung via this thoracotomy. A dextran (150 kDa amino dextran [TdB Consultancy, Uppsala, Sweden] conjugated to FITC [Invitrogen, Carlsbad, CA] 20–22 mg/kg) was administered intravenously (i.v.) to label the circulating plasma. Nuclei were stained with Hoechst 33258 (Invitrogen; 10–12 mg/kg i.v.), and leukocytes were labeled with Rho-G6 (0.2 mmoL in saline; 0.3 mL/kg i.v.). Two-photon excitation microscopy was conducted on a Zeiss LSM-510 Meta Confocal/Multiphoton Microscope system mounted on an upright Axioplan 2 stand (Carl Zeiss Microimaging, Thornwood, NY) and a vibration-isolation table. The illumination source was a tunable titanium-sapphire laser (using a 10-W Millennia diode solid-state pump laser; Spectra Physics Lasers, Mountain View, CA). The band-pass filters used for the red and green channels were 605/690 and 525/550, respectively. The excitation wavelength was set at 820 nm for all experiments, and a nondescanned mode was used. A Zeiss C-Apochromat 40×/1.2-W Korr UV-VIS-IR objective was used for imaging and was warmed with an objective heater (Warner Instruments, Hamden, CT). All image series were collected at a constant pixel dwell time, which yielded a 1.1-second frame time for the frame size of 512 × 512 pixels (169 × 169 μm).
Statistical analysis was performed using SigmaStat 3.5
Comparisons among groups were made using analysis of variance. For experiments in which two conditions were being compared, a two-tailed Student t test was used. All data are expressed as mean ± SEM, and statistically significant differences were considered if P < .05.
RESULTS
Treatment with CFTR inhibitors GlyH-101 or CFTR(inh)-172 caused a dose-dependent increase in human and rat pulmonary endothelial monolayer permeability (Fig. 1A, 1B) with TER decreases by up to 40% and 50%, respectively (P < .05). The maximal rate of decrease was reached at 4 hours after CFTR inhibition. Similarly, a dose-dependent increase in sheep bronchial artery endothelial monolayer permeability was observed in response to GlyH-101 (Fig. 1C).
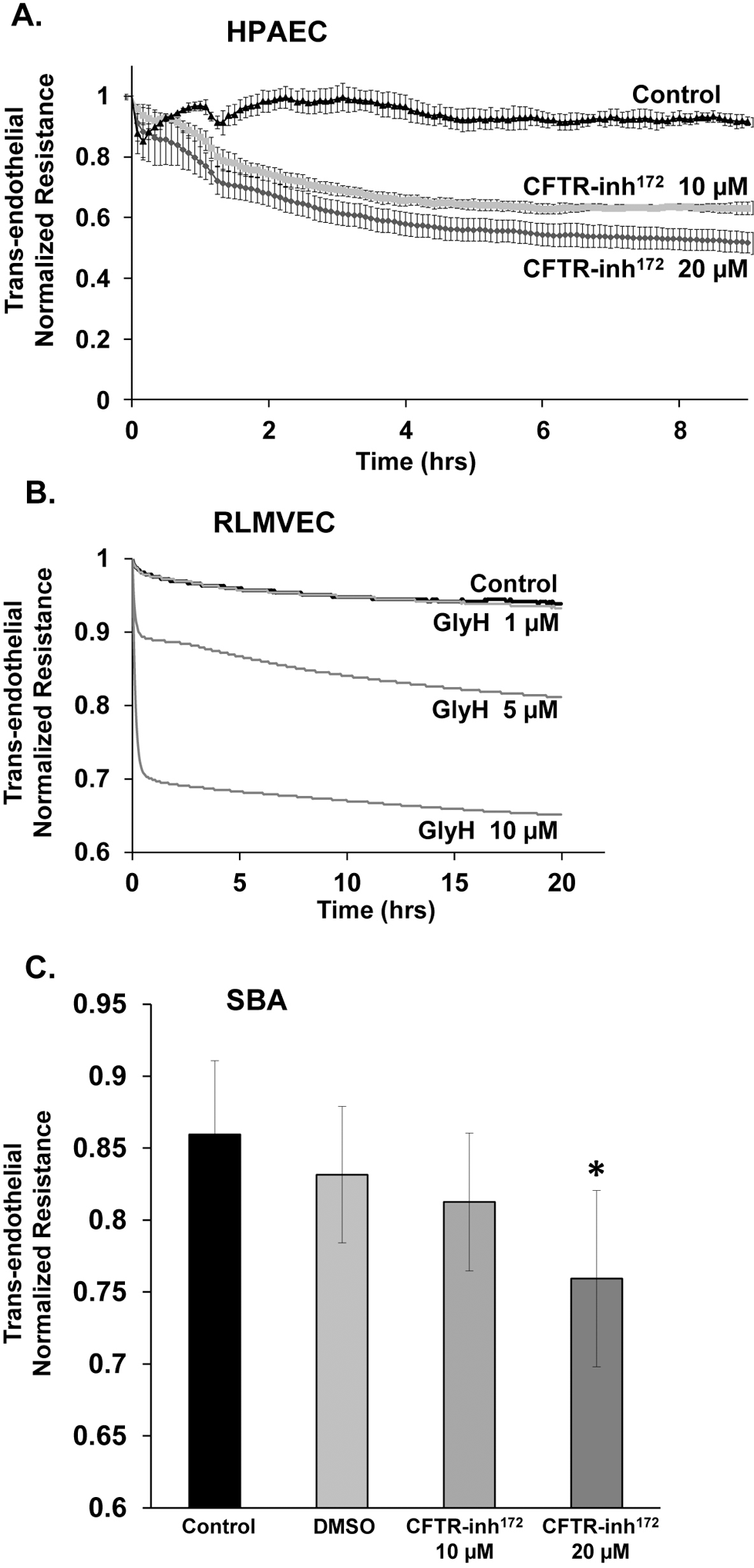
Dose-dependent increase in lung endothelial monolayer permeability (decreased transendothelial normalized resistance) in response to treatment with cystic fibrosis transmembrane conductance regulator (CFTR) inhibitor CFTR(inh)-172 in human pulmonary artery endothelial cells (HPAECs; A; n = 3–5; P < .05 vs. control) and CFTR inhibitor GlyH-101 in rat lung microvascular endothelial cells (RLMVECs; B; n = 1) and in endothelial cells isolated and purified from sheep bronchial artery (SBA) with CFTR(inh)-172 (C; n = 5). Asterisk indicates P < .05 vs. control, shown at 3.5 hours. DMSO, dimethyl sulfoxide.
Next, we investigated whether the loss of endothelial barrier function following CFTR inhibition was associated with microscopic changes in intercellular contacts or in the actin cytoskeleton. Endothelial cells treated with vehicle maintained their tight intercellular contacts and showed a fine actin cytoskeletal network, visualized by immunocytochemistry using specific antibodies for β-catenin, a main component of adherens junctions, and fluorescently labeled phalloidin, respectively. In contrast, inhibition of CFTR function following treatment with either GlyH-101 or CFTR(inh)-172 was associated primarily with redistribution of the junctional protein β-catenin and scattered actin stress fiber formation (Fig. 2).
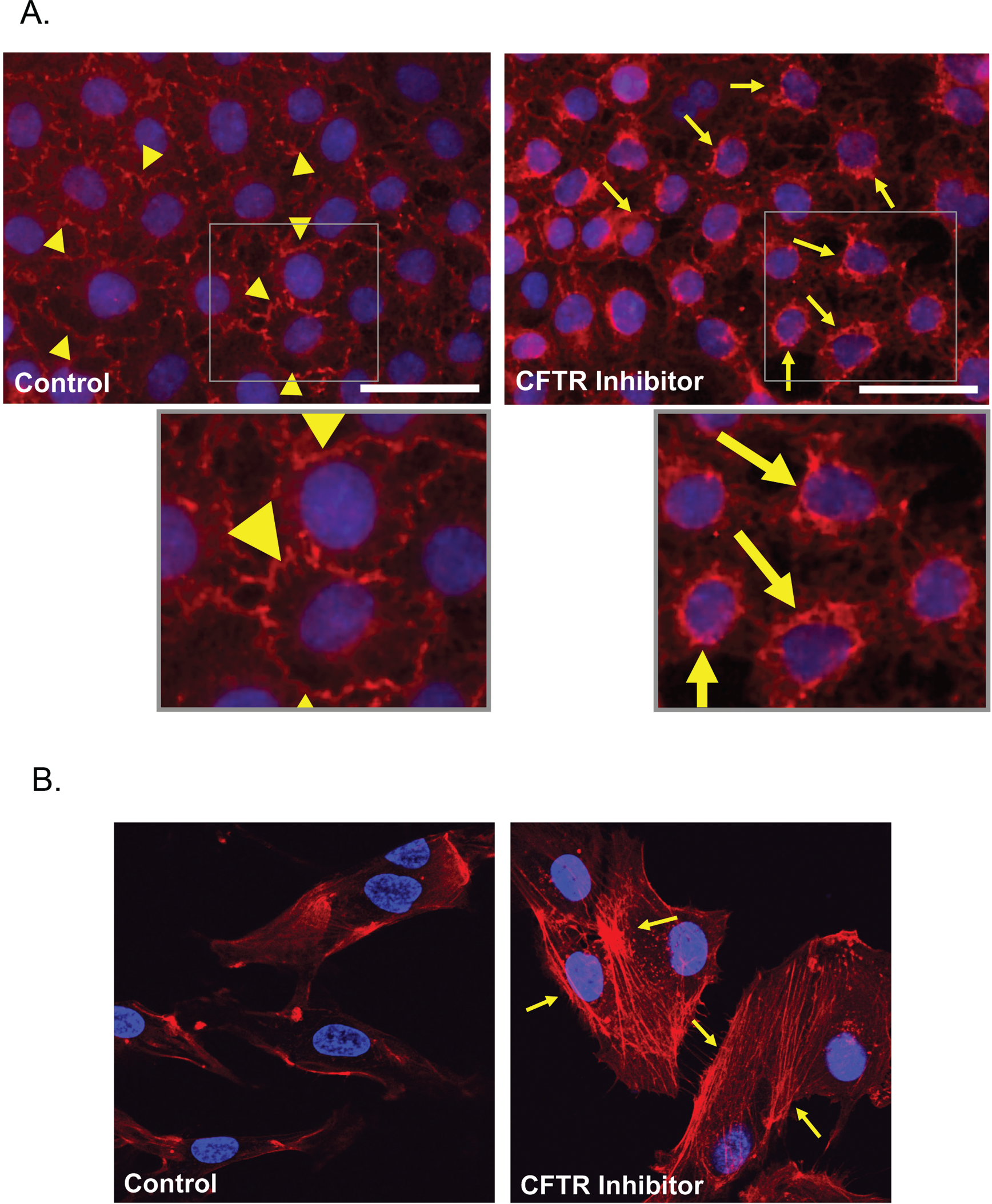
Fluorescence microscopy of rat lung microvascular endothelial cells (RLMVECs) treated with specific cystic fibrosis transmembrane conductance regulator (CFTR) inhibitors CFTR(inh)-172 or GlyH-101 (not pictured) demonstrated internalization of β-catenin from the cell periphery (yellow arrows vs. arrowheads; A); and increased actin stress fibers (yellow arrows; B) compared with vehicle-treated cells. Images were acquired by epifluorescence (A) and confocal (B) microscopy of CFTR-inhibited versus vehicle-treated endothelial cells after staining for nuclei (4′,6-diamidino-2-phenylindole, blue; A and B) and β-catenin (red; A) or actin (TR-phalloidin, red; B). Scale bar in A = 70 μm, and insets are magnified by 100%.
Because CFTR is important for S1P transport, and S1P treatment of endothelial cells is known to enhance endothelial barrier function primarily by enforcing cortical actin formation, we supplemented CFTR-inhibited lung endothelial cells with S1P. First, pulmonary microvascular endothelial cell monolayers were treated with exogenous S1P (1 μM). Endothelial cells treated with S1P exhibited significantly less decrease in TER in response to CFTR inhibition (Fig. 3A). We next investigated whether a similar response could be achieved by increase in endogenous S1P, via administration of 4DP, an inhibitor of S1P lyase (the enzyme responsible for S1P degradation). 4DP treatment attenuated the decrease in TER in response to CFTR inhibition (Fig. 3B).
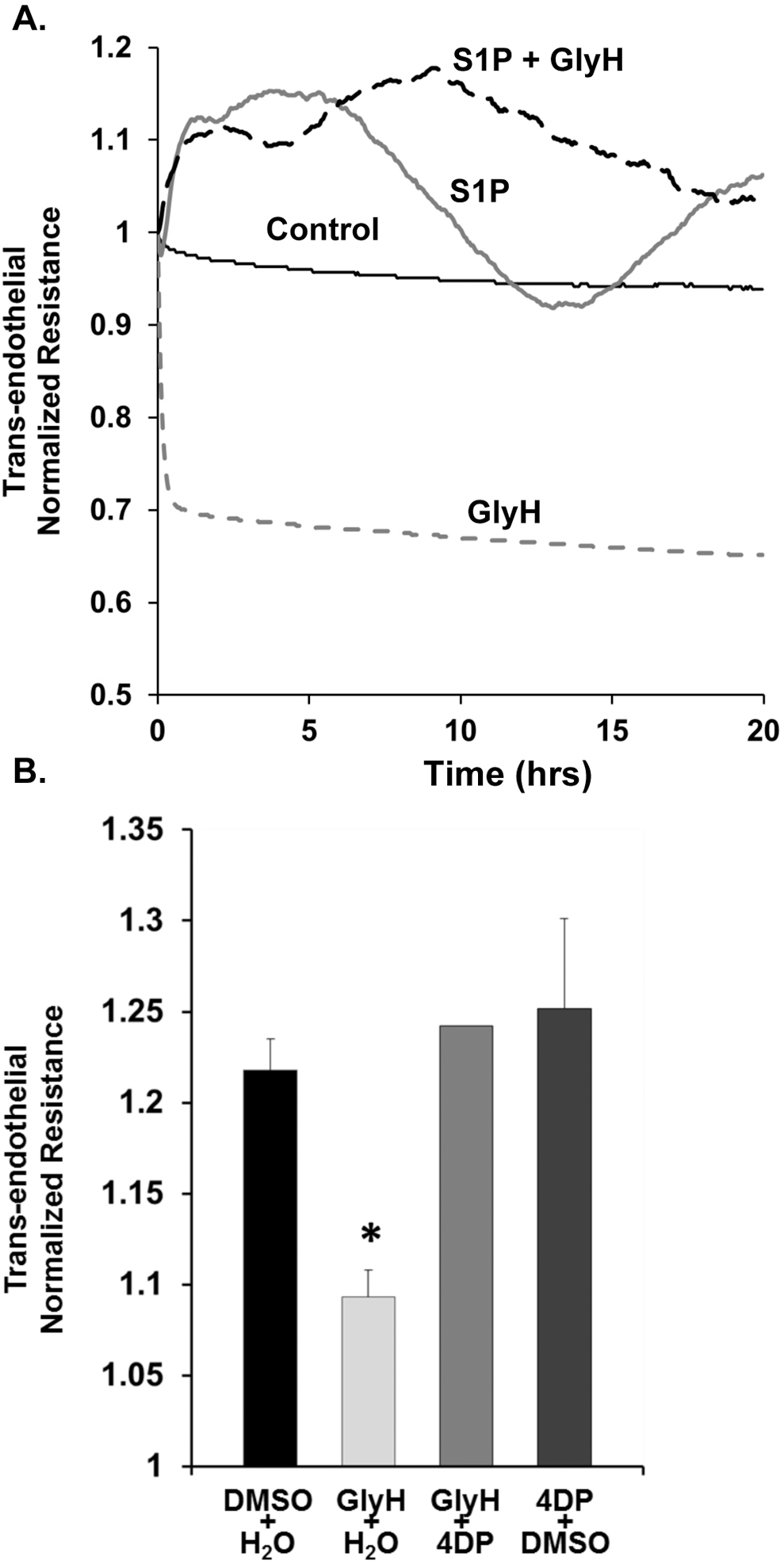
A, Exogenous sphingosine-1 phosphate (S1P; 1 μM) attenuated the decrease in transendothelial resistance (TER) in response to cystic fibrosis transmembrane conductance regulator (CFTR) inhibitor GlyH-101 (10 μM) in rat lung microvascular endothelial cells (RLMVECs). B, Increase in endogenous S1P, stimulated by administration of 4-deoxypyridoxine (4DP; 5 mM), also attenuated the decrease in TER in response to CFTR inhibitor GlyH-101 (10 μM) in RLMVECs, shown at 2-hour time-point, P < .01 GlyH + H2O versus every other condition, n = 1–4. DMSO, dimethyl sulfoxide.
To determine whether inhibition of CFTR function potentiates the effect of soluble CS components on endothelial barrier function, lung microvascular endothelial cells were first treated with CFTR inhibitors followed by addition to CS soluble extract. As expected, CS exposure dose-dependently lowered TER in rat pulmonary microvascular endothelial cell monolayers. Inhibiting CFTR function using pharmacological inhibitors before CS exposure markedly potentiated the loss of endothelial barrier function (Fig. 4). Even when cells were treated simultaneously with CFTR inhibitor and CS extract, supplementation of lung endothelial cells with S1P significantly attenuated the reductions in TER (Fig. 4B).
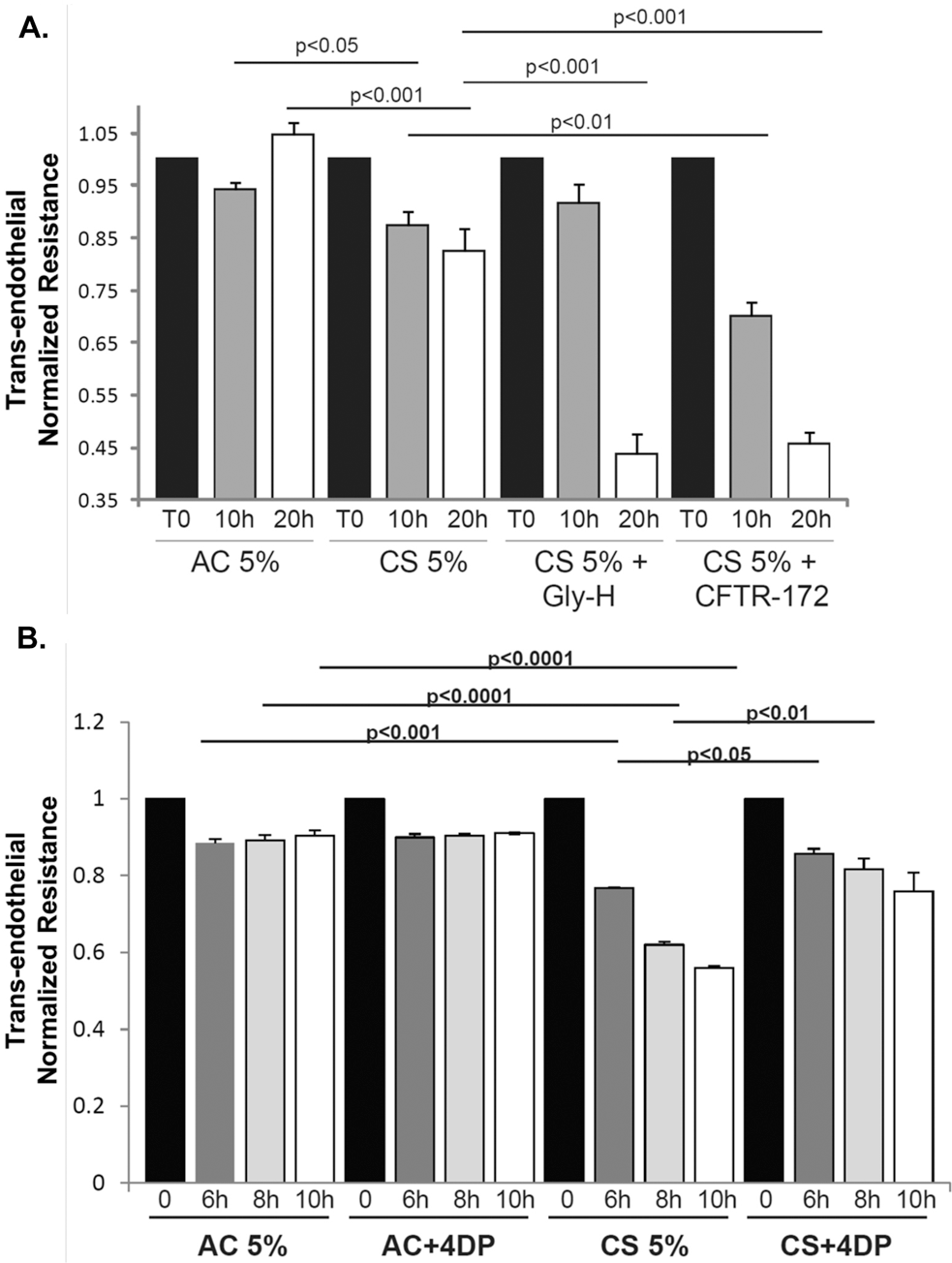
A, Cystic fibrosis transmembrane conductance regulator (CFTR) inhibition with Gly-H (5 μM) or CFTR-inh172 (10 μM) worsened the time-dependent decrease in transendothelial resistance (TER) in response to cigarette smoke (CS) extract (CS, 5%) in rat lung microvascular endothelial cells (RLMVECs). This was compared with ambient air extract used as control (AC). B, Increase in endogenous sphingosine-1 phosphate, stimulated by administration of 4-deoxypyridoxine (4DP), attenuated the decrease in TER in response to CS (5%) in RLMVECs (n = 2–6).
To demonstrate the in vivo relevance of our in vitro observations, we performed intravital two-photon microscopy of the lung microvascular barrier in mice with Cftr mutations compared with wild-type littermates in response to administration of CSE. As previously reported,1,3 wild-type mice exposed to soluble components of CS in vivo, via intravenous administration of CS extract, exhibited no signs of alveolar fluid extravasation (Fig. 5A). Of note, the final concentration of circulating CS extract (following administration of 100 μL of 20% CSE) was low, corresponding to approximately 1% CSE used in vitro in cell cultures. In contrast, CF-deficient mice exposed to similar CS extract levels showed increased signs of alveolar capillary barrier dysfunction, including extravasation of dextran-labeled plasma and increased adherence of RhoG6-labeled inflammatory cells to the endothelium (Fig. 5B). These results indicated that preexisting CFTR dysfunction resulted in greater CS-induced endothelial dysfunction, suggesting an important role of CFTR in maintaining lung endothelial barrier function.
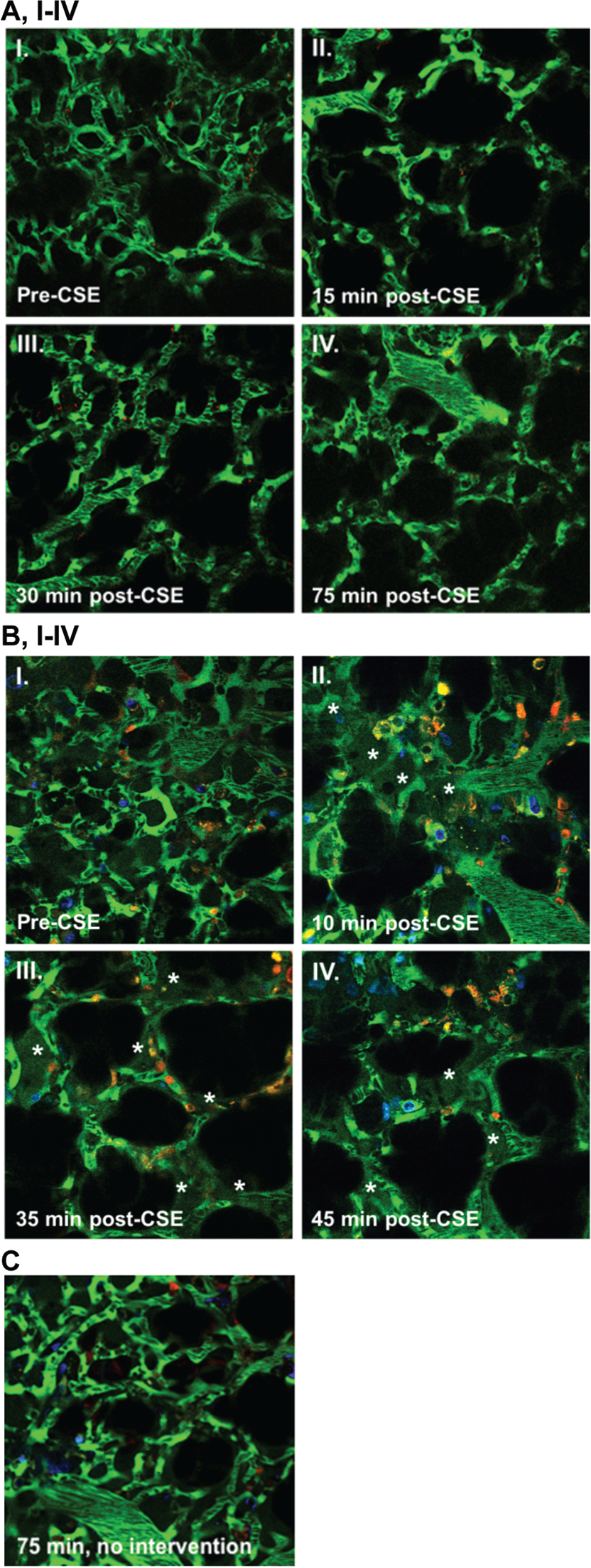
Effect of cigarette smoke extract (CSE) on the lung microcirculation captured in real time in the pulmonary microvasculature of a living wild-type (WT; A) and cystic fibrosis transmembrane conductance regulator (CFTR)–deficient (B) mouse. Three-dimensional reconstruction of fluorescein isothiocyanate–labeled vessels (green) surrounding alveoli (dark regions) and Rho-G6-labeled neutrophils (orange) imaged via intravital 2-photon microscopy before (AI and BI) and after (A and BII–IV) intravenous administration of CSE (100 μL of 20% CSE). Nuclei were stained with intravenous Hoechst (blue). Note increasing neutrophil trafficking and plasma extravasation (asterisks) into airspaces after CSE administration in the CFTR-deficient (B) but not WT (A) mouse and compared to a CFTR-deficient mouse not receiving CSE (C).
DISCUSSION
This study suggests a role for CFTR function in the maintenance of the pulmonary vascular barrier function and provides a putative mechanism for the recent observations that relate CS-induced CFTR dysfunction to the type or severity of COPD phenotype. In particular, CS induced endothelial activation and breaches in its barrier function and may facilitate leukocyte and minor plasma extravasation in the lung parenchyma, which could contribute to chronic inflammation. The relevance of our results in pulmonary microvascular cells to the pathogenesis of CF is not immediately apparent, because the alveolar parenchyma damage is not a prominent feature of CF. Nevertheless, the barrier-disruptive action of CFTR inhibitors on bronchial artery endothelial cells may indicate that a leaky bronchial endothelium could contribute to large airway inflammation and impaired mucosal immunity characteristic of CF. 14 Not least, our work may also be interpreted in the context of a role of CFTR on alveolar fluid clearance, noted several years ago in vivo. 15 Mechanistically, our results showing protective effects of S1P on the barrier dysfunction that resulted from a deficient CFTR or from CS exposure suggest that CFTR may regulate vascular permeability, including during CS exposure, via accumulation of ceramide and/or sphingolipid transport regulation and availability. In other words, CFTR-deficient endothelium may be more susceptible to CS-induced inflammation.
Our results suggest that CFTR inhibition leads to disrupted β-catenin distribution at the endothelial intercellular junction and to actin rearrangement. The involvement of CFTR in the endothelial barrier has not been previously described. Interestingly, however, a link between CFTR and β-catenin has been previously recognized in the context of stretch-induced signaling and lung development. Cohen et al. 16 have shown that stretch-associated CFTR expression during lung development was closely related with patterns and levels of Wnt/β-catenin activity in the lung parenchyma. However, the pathways by which CFTR affects β-catenin function remain to be elucidated. The involvement of CFTR, a membrane ion channel and transporter, in endothelial cell intercellular junction integrity was expected nevertheless, given the reports of CFTR involvement in tight junction organization in epithelial cell monolayers 11 , in close interactions with ezrin and actin. 17
Outside-in S1P signaling is essential for endothelial barrier function, via S1P1 receptors which help maintain a robust cortical actin distribution. 2 Intracellular-released S1P is also required to retain β-catenin plasma membrane distribution. 18 In contrast, both exogenous and endogenous ceramides disrupt the barrier, acting on intercellular junction and increasing stress fiber formation via Rho kinase activation. 1 In the context of CS exposure, ceramide was a partial mediator of CS-induced endothelial permeability, downstream of oxidative stress generation, but independent of p38MAPK-induced barrier dysfunction. Here, we show that S1P augmentation was sufficient to prevent CS-induced permeability. This result highlights the importance of a balance of ceramide to S1P signaling in the lung endothelium to maintain homeostasis. It also suggests a targetable pathway for alleviation of CS or CFTR-related endothelial activation in the setting of unwanted inflammation.
We have previously demonstrated that, although CFTR inhibition alone did not affect lung endothelial cell viability, it was sufficient to attenuate oxidative stress-induced apoptosis, linked to altered intracellular pH and inability to increase ceramides in response to oxidative stress. 10 Here, we report that CFTR inhibition markedly augmented CS-induced permeability, possibly due to a synergistic effect of potent additional blockade of CFTR by CS and combined effect on loss of tethering forces. Extrapolating from our previous results using oxidative stress (hydrogen peroxide), 5 it is possible that the combination of CFTR inhibition and CS exposure synergistically increases ceramide and decreases S1P levels/and or signaling required for cell-cell tethering and cortical actin preservation. Apoptosis is less likely to be involved in CFTR-inhibition and CS-induced endothelial permeability for several reasons: in our previous work, CFTR inhibition alone was not sufficient to cause apoptosis, 7 CS-induced permeability was only marginally attenuated by apoptosis inhibitors, 1 and CFTR inhibition has been shown to inhibit oxidative stress–induced apoptosis. 7 Nonetheless, future studies will have to directly dissect the effect of CS on lung endothelial cells CFTR and the putative signaling pathways involved in the synergy of CFTR inhibition and CS exposure on vascular barrier function. This relationship was noted first in the airway, in recent studies linking CS-induced inflammation to decreased airway CFTR expression and activity, decreased mucociliary transport, and COPD symptom severity.19–23 Smokers with and without COPD exhibit reduced CFTR-dependent upper airway 20 and lower airway 19 potential difference. Although the magnitude of the response to CFTR inhibitor was similar across all species of endothelial cells studied, it is possible that distinct vascular beds will have specific mechanistic responses to CFTR inhibition and CS exposure in this context, as previously shown for other edemagenic agents. 24
The requirement of CFTR for endothelial barrier function and the synergy of CFTR loss of function and CS exposure on the barrier function loss will have to be further explored in vivo, because they may have important clinical implications in conditions affecting both patients with CF and smokers who have a functional loss of CFTR. Our results fit with the report that CFTR is required for proper alveolar fluid clearance in mouse models. 15 The clinical implications of these findings include the potential for heightened risk for both ongoing inflammation or injury due to a leaky pulmonary or systemic vascular barrier, especially following a second, pro-edemagenic hit. Indeed, edema is a rare presentation of CF and may be attributable to hypoproteinemia.25,26 Similarly, edema is not clinically evident following habitual CS exposure. However, CF-deficient mice are more susceptible to developing edema in the setting of experimental acute pancreatitis, 27 and CS exposure is a risk factor for the development of pulmonary edema in adult respiratory distress 28 or that occurring as a complication of blood transfusions 29 or severe trauma. 30 These results suggest that an optimal CFTR function may be necessary for barrier maintenance of both epithelial and endothelial structures and that CFTR inhibition may worsen CS-induced inflammation via an effect on vascular endothelial cells, which could be ameliorated by harnessing the barrier protective effects of S1P.
Footnotes
ACKNOWLEDGMENTS
We thank Amanda Fisher for her assistance with animal preparation for intravital imaging experiments.
