Abstract

INTRODUCTION
Right from the start of this essay I must dispense with the royal “we” to make it clear for the readers: what comes next is the product of a personal gedankenexperiment (thought experiment). Pulmonary vascular disorders, which frequently lead to pulmonary hypertension, are complex and multifactorial. One glance at the pulmonary hypertension classification list, which is revised every decade, makes this clear. According to Daniel Kahneman, 1 complexity reduces the validity of a forecast, claim, or hypothesis—hence our need for algorithms and large databases, which help us in making decisions. Both diagnostic or treatment algorithms and databases are hypothesis-free. These tools, which are based on statistical information gathered from large but sometimes not homogenous cohorts, are used for forecasting and prognostication. As the clinical pulmonary hypertension community is surviving the 6-minute walk crisis in search of new end points, it has occurred to many in this community that hypothesis testing first requires hypothesis generation. According to Kahneman, this gets us into the territory of uncertainty and unpredictability—in his words, into “low-validity environments.” Depending on your degree of risk aversion, you may not be comfortable in such an environment. However, comfort comes from the testing of the hypotheses, which is the step that follows the hypothesis-generating first step. There are many ways to skin a cat and many ways to generate a hypothesis. For this particular exercise, my approach is to identify the spokes—and I will need to explain why I call a spoke a spoke, and I will need to declare to what degree the identification or recognition of a spoke may be biased. Similar to the level or validity of evidence rating, I will rate my bias the following way: little bias (A), moderate bias (B), and probable high bias (C). My hope is that the reader will enjoy this exercise as much as I did.
THE SPOKES
The process of spoke identification and recognition is a difficult one, as mentioned, because of the enormous complexity of clinical pictures, etiologies, and histological presentations of pulmonary hypertensive disorders. This is immediately intuitive when considering that a pulmonary vascular disease due to a congenital cardiac abnormality characterized by high blood flow through the lung circulation and a mean pulmonary artery pressure elevation of 60 mmHg in a patient with hereditary pulmonary arterial hypertension (PAH) have the same outcome: death from right heart failure. A patient with sickle cell anemia–associated pulmonary hypertension instead may die from a stroke or the ill-defined “acute chest syndrome.” Consequently, any further discussion needs a better focus. My biased opinion is that it is legitimate to distinguish severe and progressive PAH (A). Severe and progressive PAH may not be the same: New York Health Association functional class I patients have severe PAH, but their disease may not be progressive. Next, we know that the majority of severe PAH patients have remodeled lung vessels (A); among them are patients with lung vascular lesions, which obstruct the lumen and increase resistance to blood flow (A).
Many investigators feel an explanatory burden when it comes to describing these lesions and to understanding their underlying pathobiology. We can tentatively categorize these patients and say that they have “severe angio-obliterative PAB” (B). Feeling this explanatory burden myself, I need to begin to identify the spokes.
A healthy lung circulation has vessels where the intactness depends on the endothelium being a monolayer. I rate this a B, because I recognize that I have strong feelings about this being a spoke, and because I see biology through the lens of homeostasis. In these forms of angioobliterative PAH, the lung endothelium is not healthy (A), whether the obliteration is due to blood cell adhesion (followed by thrombus organization) or to growing and proliferating cells.2,3 This fact emerges even more clearly as we remember that PAH based on the trigger of chronic or intermittent hypoxia usually remodels the lung vessels without lumen obliteration; the newborn calf may be the exception to this rule.4,5 Many of the lesions in angioobliterative forms of PAH show a halo of inflammatory cells (A), and this suggests the activation and participation in the pathobiology of the innate and adaptive immune systems (B).2,6
We have learned from the hereditary forms of IPAH that the disease penetrance among mutation carriers is low (A);7,8 this calls for a second (and perhaps third) hit. Hence the postulate: without damage to lung vessels, there is no severe angioobliterative PAH. So I add to the two spokes of the unhealthy endothelium and inflammation/immune response the spoke of lung vascular damage or injury. Based on this is the next postulate: angioobliterative PAH can be explained as a process of lung vessel wound healing gone awry. 3 Under most circumstances, the homeostatic program restores the injured endothelial monolayer, and apoptosis or necrosis remain without long-term consequences. Endothelial cell damage and repair may be frequent and are expertly handled by a vascular maintenance program (A). Perhaps there are injuries that are so severe that return to a normal vessel structure is impossible, but we have no evidence of endothelial cell–denuded lung vessels while we appreciate lung vessel loss, rarefaction, and pruning of the pulmonary vascular tree. Patients with severe emphysema lose lung vessels (A), and endothelial cells undergo apoptosis. 9 We do not know whether in human angioobliterative PAH the initiating event is indeed endothelial cell apoptosis, but this is my bias (C). Clearly, lung vessel endothelial cell apoptosis per se is insufficient to trigger angioobliteration and severe PAH; otherwise, emphysema patients would develop severe PAH, which the majority does not (A). The Sugen 5416/chronic hypoxia rat model, 10 as far as we can use it to translate pathobiological insights from it to the human condition (C), suggests that there may be no angioproliferative PAH without initial apoptosis (A).10,11
THE HUB
It is (for me) difficult to see how deducing information from clinical or genetic data alone will let us connect the spokes with the hub. On the other hand, there is the predicament that Kahneman has identified: complexity reduces the validity of the forecast and “inconsistency is destructive of any predictive environment,” or in the context of hypothesis generation, “intuition cannot be trusted in the absence of stable regularities in the environment.” 1 My biased opinion (C) is that the angioobliterative lesions in severe PAH are not rare events but rather are regular occurrences that can be predicted, not with 100% certainty but better than 10% (B). Is a disease model that is built on the hub of lung endothelial cell apoptosis (Fig. 1) too complex? I believe it is not (C), but there is plenty of complexity in the process of inserting the spokes correctly, yet the wheel may roll with a few spokes missing or inserted the wrong way. The schematic (Fig. 1) in fact shows only one horizontal and one vertical axis, thus ordering sequential events that likely occur in parallel or simultaneously in the vascular tissue. The three processes—programmed cell death (apoptosis), self-eating (autophagy), and phagocytic removal of the dead cells (efferocytosis)—are arranged horizontally on the basis of a wealth of published information that these mechanisms are linked.12–16 All three serve a homeostatic purpose, as will be discussed now.
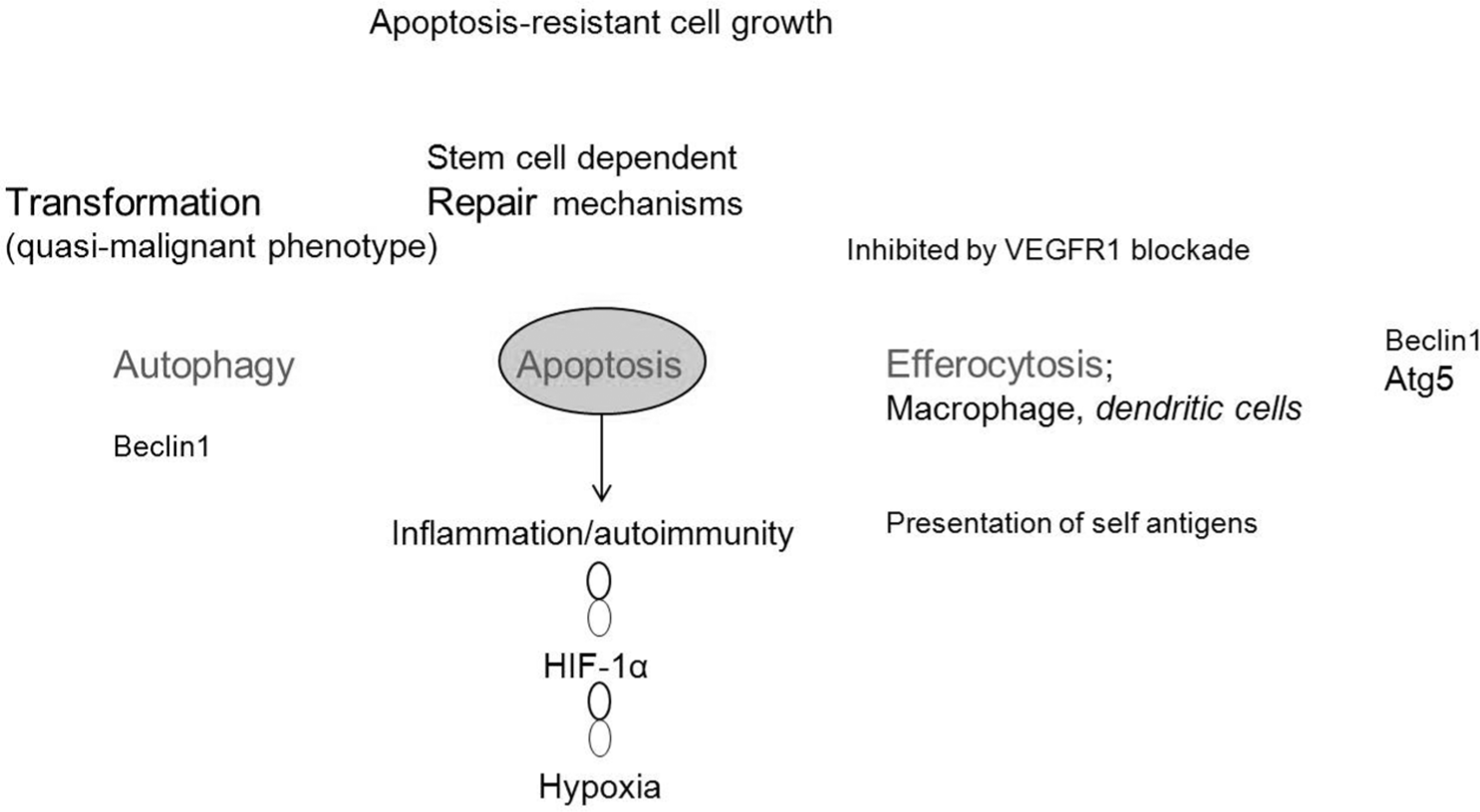
Disease model built on the hub of lung endothelial cell apoptosis. VEGFR: vascular endothelial growth factor receptor; HIF: hypoxia-inducible factor.
The highly efficient phagocytosis of dead cells (apoptotic bodies) is the main reason why we find very few apoptotic cells in normal tissues, for example, in the normal heart. Professional phagocytes, macrophages, and dendritic cells respond to the “find-me” and “eat-me” signals emitted from dying cells, 13 and nonprofessional cells can phagocytose neighboring cells. If efferocytosis is impaired, the consequences are severe; unengulfed cells perpetuate inflammation, self-antigens are taken up by antigen-presenting dendritic cells, and an autoimmune process can start. 17 In the lungs of patients with severe angioobliterative PAH, one finds it difficult to identify apoptosis-undergoing cells (A), 18 while the opposite is the case in emphysema, where plenty of apoptotic cells are found and the phagocytic removal of dead cells is impaired.9,19,20 It is of interest that there are no published reports of phagocytic clearance of apoptotic cells when it comes to severe angioobliterative PAH, and it is unclear why apoptotic cells are hard to find in and around the lung vascular lesions in severe PAH. There are at least three possible explanations for this observation: (1) there are no apoptotic cells to be seen because the phagocytic removal is highly effective; (2) there are no apoptotic cells because autophagy is highly effective, 21 perhaps preventing apoptosis; and (3) the transition from vascular damage to angioproliferation and cell phenotype switch, 18 resulting in apoptosis-resistant cells, occurs via a dynamic transition of autophagy and efferocytosis from initially being overwhelmed to highly efficient (C). The first studies of the role played by autophagy in PAH were recently published.22,23 It is interesting that the autophagy machinery shares some mechanistic components with those involved when macrophages after corpse engulfment digest the apoptotic bodies. Beclin 1 and Atg5 are proteins that are involved in both processes,24,25 and autophagy proteins regulate the innate immune response. 26 The antiapoptotic Bcl-2 and survivin proteins are expressed in plexiform lesions in the lungs of patients with severe angioobliterative PAH, 18 while the proapoptotic Bim is not expressed in the lungs of pulmonary hypertensive Su/Hx rats. 27
It is highly probable that precursor and stem cells, either mobilized from the bone marrow 28 or resident in the lung vessels, participate in the process of lung vessel remodeling,29–32 including the exuberant endothelial cell growth and phenotype switch of lumen-obliterating cells.
SYNOPSIS
For some of the readers, this essay may appear to be nothing but an example of repetitive reiteration of priors;2,3,10,11,18 however, Figure 1 orders many of the key words and concepts of the present view of PAH pathobiology. Experimental data derived from the Su/Hx rat model of severe angioobliterative PAH were invaluable for the generation of such an apoptosis-centered hypothesis. This hypothesis can now be put to the test and challenged, or it can be remodeled, using most of the same keywords and building blocks, as illustrated in Figure 2. The major limitation of this hypothesis-generating exercise is clearly the bias of this writer: my opinion as a conjecture rather than one based on rigorous analysis of all possible experiments. In so many words—and now seriously self-critical—anyone in the field can connect any number of spokes to a hub; this is of course the real insight of this exercise.

Hypothesis modeling. The difference between the left and the right image is a different geometry, shape, and space enclosed by the building elements (struts rather than spokes). The shape change is achieved by applying a deforming force (squeezing into a different shape). There are more than two possible configurations that one can generate by applying the deforming force.
However, in my opinion the risk/benefit balance of such a hypothesis-generating exercise is tilted toward the benefits. If indeed lung vessel apoptosis is the initiating event, perhaps in some patients triggered by circulating factors,33–36 we may begin to realize that the apoptosis trigger is likely different in different patients: in AIDS patients different from that in scleroderma patients, and different again in patients with portopulmonary hypertension. We may start to search for evidence that indeed during some phase of lung vascular lesion formation (and perhaps only in a subset of patients) efferocytosis of apoptotic vascular cell bodies is inefficient and opens the door to an antiself immune response. We also may appreciate that the reason that established PAH is so difficult to treat lies in the nature of an abnormal stem/precursor cell–driven repair mechanism31,32,37—difficult under these circumstances because effective treatment (i.e., the reopening of obliterated vessels) will require that apoptosis-resistant cells will have to be removed and precursor/stem cells will have to be reprogrammed in order to differentiate and behave as a competent lung arteriolar lumen-blood border, obeying the law of the endothelial cell monolayer.
