Abstract
Hematopoiesis and vascular homeostasis are closely linked to each other via subsets of circulating bone marrow–derived cells with potent activity to repair endothelial injury and promote angiogenesis. As a consequence, abnormalities in hematopoiesis will eventually affect vascular health. Pulmonary arterial hypertension (PAH) is a vascular disease characterized by severe remodeling of the pulmonary artery wall. Over the past decade, circulating hematopoietic cells have been assigned an increasing role in the remodeling, such that these cells have been used in new therapeutic strategies. More recently, research has been extended to the bone marrow where these cells originate to identify abnormalities in hematopoiesis that may underlie PAH. Here, we review the current literature and identify gaps in knowledge of the myeloid effects on PAH.
INTRODUCTION
Pulmonary arterial hypertension (PAH) is a devastating disease characterized by severe remodeling of the pulmonary artery and, as a consequence, increased pulmonary artery pressure and ultimately right ventricular failure. Bone marrow (BM)–derived cells are essential to vascular homeostasis, and circulating hematopoietic cells and the marrow-resident mesenchymal stromal cells equally affect vascular health. Among these cells, the fibrocytes and mesenchymal stem cells (MSCs) are well characterized using a combination of cell surface markers and/or cell culture techniques, with fibrocytes being a specific subset of collagen-I-producing myeloid-derived cells1,2 and MSCs as a subset of BM stromal cells with the capacity to differentiate into adipocytes, chondrocytes, and osteoblasts. 3 The population of BM-derived “endothelial progenitor cells,” a subset of BM cells with potent angiogenesis activity, is less well defined. For the past several years, it was believed that a subset of these cells exhibited the capacity to differentiate into endothelial cells. Endothelial progenitor cells define a heterogeneous group of cells that are classified into 3 groups in humans on the basis of the method of isolation, including circulating angiogenic cells (CACs), colony-forming unit Hill (CFU-Hill; sometimes also referred to as CFU-EC), and endothelial colony-forming cells (ECFCs). 4 The latter are true endothelial cells, as evidenced by their in vivo capacity to form blood vessels, and are defined as endothelial cell colonies appearing after 7–21 days of culture of blood mononuclear cells (MNCs) in endothelial conditions. 5 CAC do not form colonies but appear earlier during cell culture (starting on day 4) as spindle-shaped or cobblestone-morphology cells and typically bind Ulex europeas lectin and uptake acetylated low-density lipoprotein (acLDL).6–8 Peripheral blood CAC originate from proangiogenic myeloid cells. 5 Murine BM-derived endothelial progenitor cells are isolated using a similar method. CFU-Hill colonies contain a mixture of proangiogenic myeloid progenitors and angiogenic T cells.9–12 In addition, circulating or BM cells expressing hematopoietic stem cell markers such as CD34, CD133, or c-Kit in humans or SCA-1 and c-Kit in murine in combination with VEFGR-2 have also been referred to as endothelial progenitor cells. 4 Thus, it is evident now that, except for ECFC, the methods used to identify endothelial progenitor cells in the peripheral blood circulation and BM in fact detect or enrich for proangiogenic myeloid hematopoietic cells.13,14 Plating of mononuclear cell on fibronectin or gelatin substrates, used to isolate endothelial progenitor cells, selects monocytic cells. 15 Two frequently used fluorescent labels, Dilconjugated acLDL and fluorescently labeled plant lectin U. europaeus agglutin 1 (UEA-1), are not specific for endothelial cells and will also stain myeloid cells.10,13,16,17 In vitro tube formation assays used to access angiogenic cord formation by endothelial progenitors are not useful to identify these cells, because many other types are able to form these structures. 13 Moreover, although several in vivo studies initially reported that endothelial progenitor cells contribute to endothelial repair and regeneration by differentiating into endothelial cells and integrating into the endothelium,18–25 subsequent in-depth studies using genetically tagged BM or endothelial-cell-specific reporter genes26–29 showed that the endothelial progenitor cells localize adjacent to blood vessels or only temporarily incorporate into the endothelium. Current paradigm in vascular biology is that these BM-derived myeloid cells acquire endothelial-like cell mimicry. Nevertheless, these cells are essential paracrine actors as proangiogenic hematopoietic cells critical for new blood vessel formation and endothelial repair30–35 that are composed of a heterogeneous group of myeloid hematopoietic progenitor and mature cells, including mast cells. Their diverse phenotype and angiogenic activities and the controversy surrounding endothelial progenitor cells have been reviewed in detail elsewhere.13,36 Smooth muscle cells of hematopoietic origin,37–39 derived from BM stromal cells, 40 or of unidentified BM origin41,42 have also been reported. Here, we provide a review of the roles of the BM-derived cells in PAH, including proangiogenic hematopoietic cells, mast cells, fibrocytes, and MSCs. We provide review of studies involving animals and patients and discuss the clinical implications of the findings and future perspectives.
PROANGIOGENIC HEMATOPOIETIC CELLS IN PAH
Proangiogenic hematopoietic cells are subsets of myeloid hematopoietic progenitors and monocytic cells. The majority of the studies involving BM in PAH have investigated these cells in animal models as well as in patients as endothelial progenitor cells. Tables 1 and 2 provide a detailed overview of the characterization and terminology used for these cells in the individual publications.
Phenotype of bone marrow (BM)–derived proangiogenic cells in animal models of pulmonary arterial hypertension (PAH)
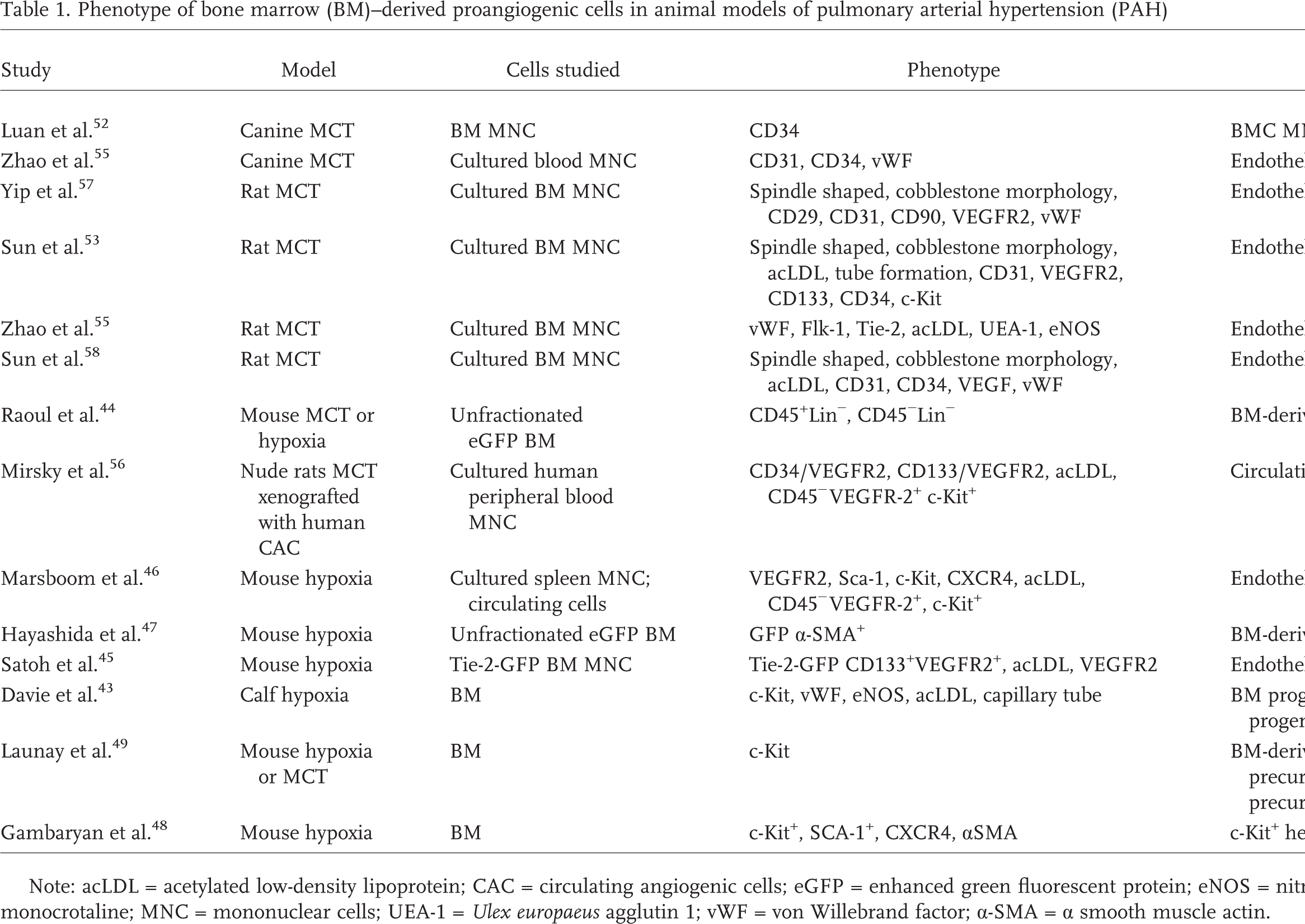
Note: acLDL = acetylated low-density lipoprotein; CAC = circulating angiogenic cells; eGFP = enhanced green fluorescent protein; eNOS = nitric oxide synthase; MCT = monocrotaline; MNC = mononuclear cells; UEA-1 = Ulex europaeus agglutin 1; vWF = von Willebrand factor; α-SMA = α smooth muscle actin.
Phenotype of bone marrow (BM)–derived proangiogenic cells in patients
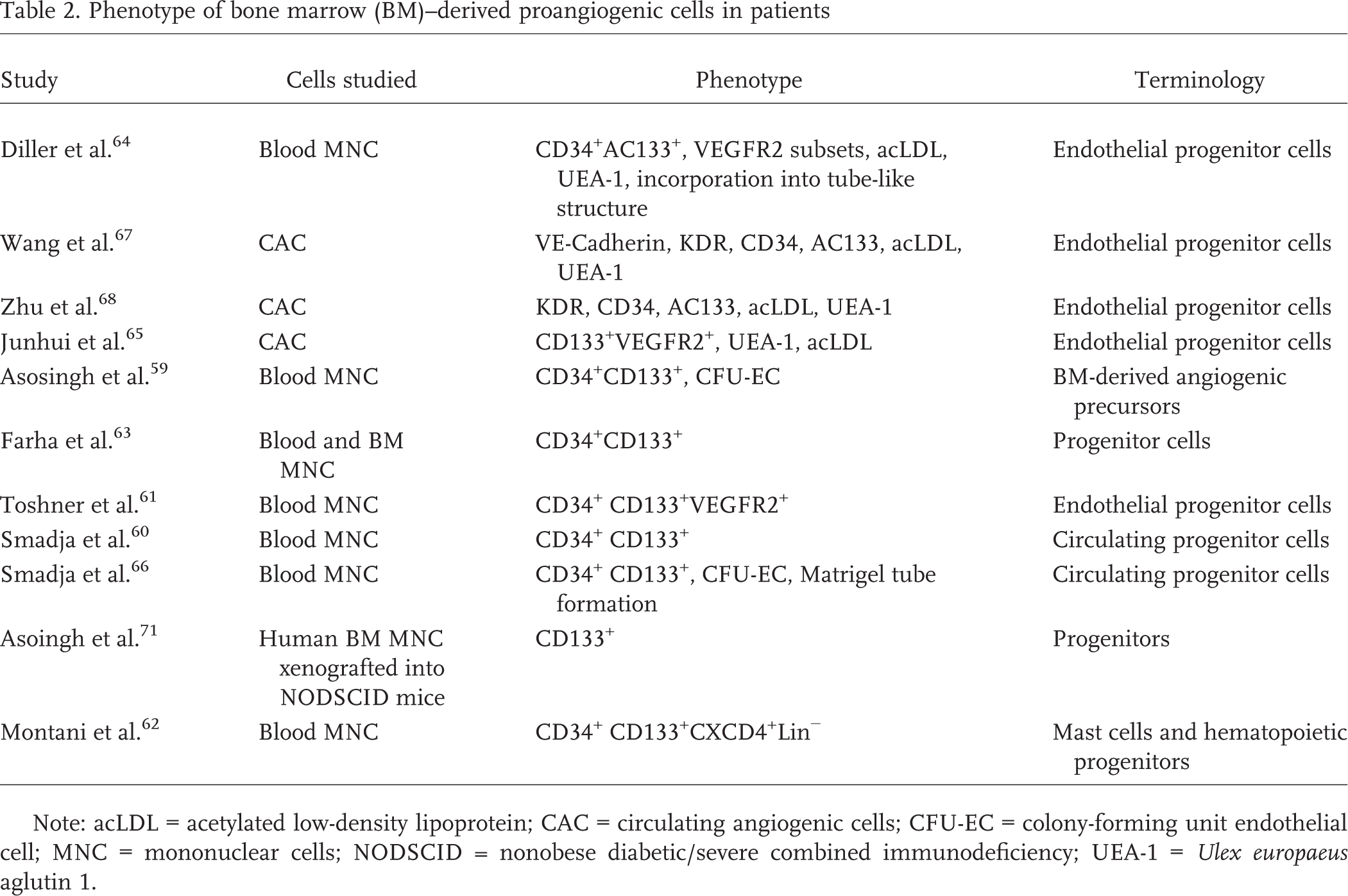
Note: acLDL = acetylated low-density lipoprotein; CAC = circulating angiogenic cells; CFU-EC = colony-forming unit endothelial cell; MNC = mononuclear cells; NODSCID = nonobese diabetic/severe combined immunodeficiency; UEA-1 = Ulex europaeus aglutin 1.
Proangiogenic hematopoietic cells in animal models of PAH
Proangiogenic progenitors in experimental PAH have been studied in small and large animal models, as contributors to the remodeling and as cellular vehicles for gene therapy. Animals exposed to hypoxia or monocrotaline (MCT) are used as models of PAH. In the hypoxia-induced PAH model, animals were placed in a hypobaric chamber, using 10% oxygen (O2) for an extended period of time.43–49 The levels of circulating progenitors expressing hematopoietic cell surface markers were consistently increased in these models, which suggests a role in pathogenesis.43–46 In green fluorescent protein BM chimeric mice, mobilization and recruitment of BM cells into pulmonary arteries and adventitia were observed. A fraction of he BM-derived cells detected in the angiogenic remodeled areas expressed α-smooth muscle actin (α-SMA), suggesting that these cells differentiated into myofibroblasts or smooth muscle cells.43,47 It is possible that these α-SMA cells are derived from BM stromal cells or fibrocytes. Stromal-derived factor 1α (SDF-1α) is a primary factor in the recruitment of BM progenitors into tissues. Animals exposed to hypoxia showed increased expression of SDF-1α in the lung tissue as well as increased presence of SDF-1α-receptor-positive (CXCR4 or CXCR7) c-Kit+ progenitors in the remodeled areas. 48 Pharmacological blockade of SDF-1α receptors before the exposure to hypoxia, but not in animals with established PAH, showed parallel reduction in c-Kit+ cell recruitment and decreased right ventricular systolic pressure. 48 In 2 other studies, the development of PAH was related to proangiogenic hematopoietic cells with impaired angiogenesis capacity.45,46 Serotonin binding its receptor 2B receptor (5-HT2BR) induces vasoconstriction of the pulmonary arteries by stimulating smooth muscle cell contraction. 50 5-HT2BR is overexpressed in patients with PAH, and 5-HT2BR-deficient (5-HT2B−/−) mice fail to develop PAH under hypoxia. 49 Early evidence that the BM contributes to the origin of PAH came from a landmark BM transplantation study demonstrating that wild-type BM led to development of PAH in 5-HT2B−/− mice. 49 In contrast, engraftment of 5-HT2B−/− BM into wild-type mice prevented PAH in animals under hypoxia or treated with MCT. 49 Similar mechanisms may operate in large animals. For example, increased numbers of c-Kit+ cells were found in the expanded vaso vasorum in remodeled areas of the pulmonary artery wall in calves exposed to chronic hypoxia. 43 Overall, these reports suggest that proangiogenic hematopoietic cells contribute to PAH in hypoxic animal models.
MCT is a potent pyrrolizine alkaloid endothelial toxin that causes pulmonary vascular disease in rodents.51,52 Therapeutic effects of ex vivo expanded mature hematopoietic proangiogenic cells have mostly been studied in this model.5,13–18,52–55 In all but one of these studies, 56 administration of the proangiogenic cells before pulmonary hypertension onset decreased right ventricular systolic pressure and/or reduced remodeling.44,52,54,55,57 One mechanism of benefit seems to be via increased expression of endothelial nitric oxide synthase (eNOS).44,57 Additional improvement was observed when the hematopoietic cells were given in conjunction with vasodilator therapy.53,58 Animals with established pulmonary hypertension showed further reduction of right ventricular systolic pressure and survival when proangiogenic cells were transduced with eNOS. 55 Thus, ex vivo expanded differentiated proangiogenic hematopoietic cells have beneficial effects in MCT models of pulmonary hypertension.
Proangiogenic hematopoietic cells in human PAH
Although animal studies of pulmonary hypertension have high levels of circulating progenitors, findings are controversial in human PAH. Some groups reported elevated numbers in the peripheral circulation,59–63 whereas others found a reduction64,65 or no difference, 66 compared with healthy control subjects. There are several possible explanations for these discrepancies. First, all groups analyzed different CD34+, CD133+, or c-Kit+ progenitor cell subsets with or without a combination of lineage markers. Second, studies vary in the stage of patients evaluated. Patients with end stage of PAH who are on a waiting list for lung transplantation have significantly reduced numbers of circulating progenitors (KA, personal observation). Another confounder is insufficient number of events acquired and/or nonstandard gating strategy. The latter is critical for detection of circulating progenitors, which are rare events. Although there is no single correct way to analyze for these cells, a uniform set of specific guidelines should be followed (Fig. 1).
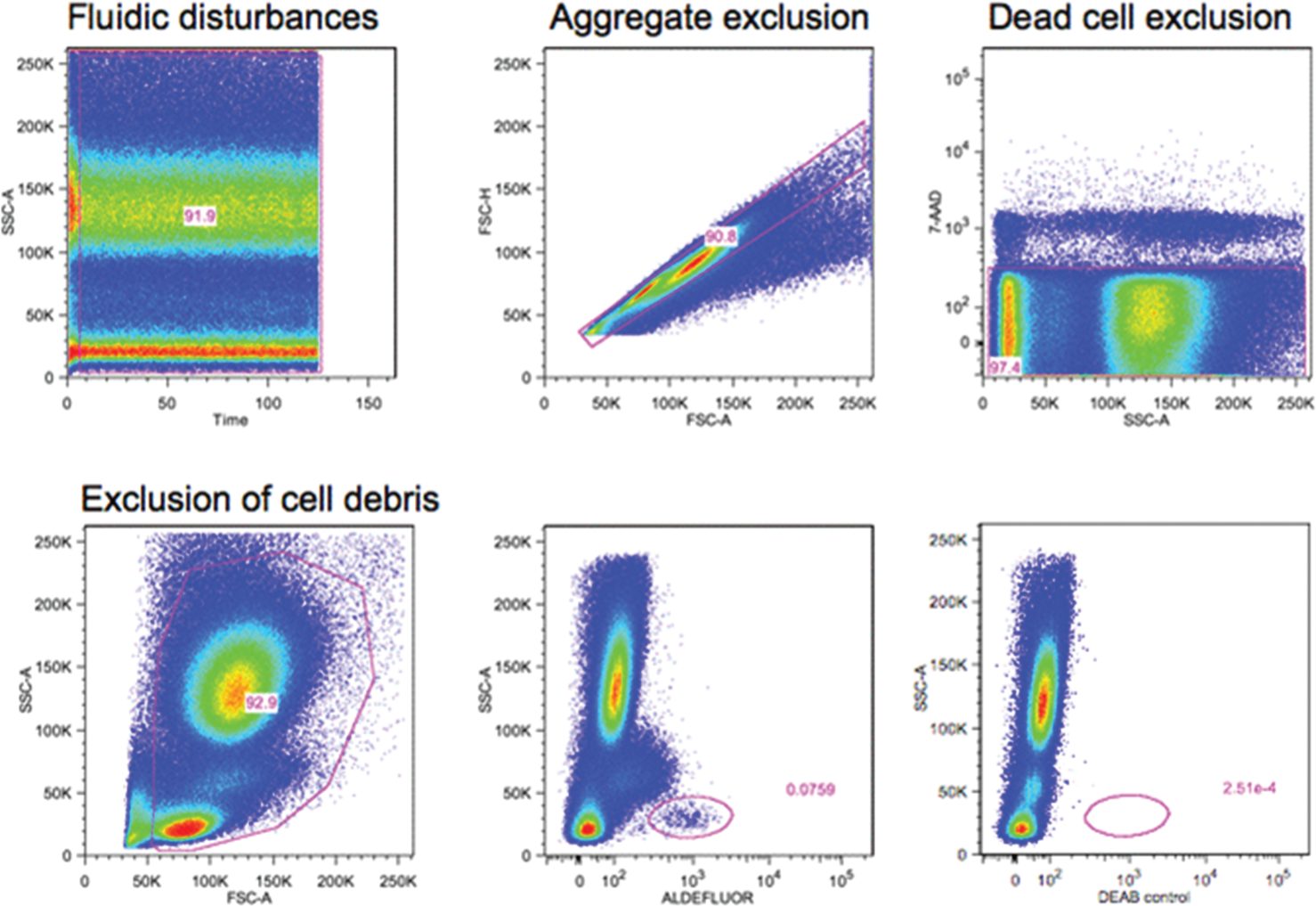
Guidelines for flow cytometric quantification of circulating proangiogenic progenitors. Proangiogenic progenitors are infrequent in the peripheral blood, and therefore all guidelines for rare event analysis apply. 91 The number of events acquired is critical and should be based on the desired coefficient of variance (CV). Poisson distribution best describes the random and independent occurrence of rare events. In this statistic, CV = 100/(positive events acquired)1/2; for example, acquisition of 100 positive events will give a CV of 10%. If the frequency of positive events is 0.02%, 500,000 events need to be acquired. After acquisition, gating-out sources of artifacts should be part of the gating strategy and applied to each sample. Fluidic disturbances in the flow cell will increase variability in the measured parameter. Time-gating should be performed to control for this; any irregularities and burst of events should be excluded. Aggregate correction should be applied to exclude cell aggregates. Dead and dying cells can lead to nonspecific binding and mask true frequency; therefore, a dead cell exclusion dye should be used. Some dead cells will fragment into cell debris during the staining process and should be excluded by gating out events with light scatters too low to be intact cells. Knowing the spatial distribution of the positive events is helpful to gate them, even when the number of events acquired is low. Backgating of the positive events is always recommended to check which events have been excluded during the logical gating process. In this illustration, red blood cells in whole blood were lysed using fixative free ammonium chloride followed by staining for aldehyde dehydrogenase activity (aldefluor), a recently described enzymatic probe for circulating proangiogenic progenitor cells.2–9 Blocking of the aldehyde dehydrogenase activity using diethylamino benzaldehyde (DEAB) in parallel staining was used as control. 7-AAD was used as a dead cell exclusion dye. The progenitors are identified as aldefluor positive cells with low side scatter (SSC) properties. Identical principles apply for other markers of circulating progenitors.
In preliminary studies, ex vivo expanded circulating proangiogenic cells have been used as cell therapy in idiopathic PAH. These patients showed clinical improvement as indicated by increased 6-minute walk distance, decreased mean pulmonary artery pressure, improved pulmonary vascular resistance, and increased cardiac output, compared with individuals who received conventional therapy alone.67,68 It is important to note that the angiogenic cells in these studies67,68 were CAC, which are low-proliferative and monocyte-derived cells, as shown by others. 4
Although there is controversy regarding the levels of circulating progenitors, reports consistently show that these cells are present in high numbers in remodeled areas of the pulmonary artery wall.61–63,69 In paraffin-embedded lung tissues obtained from explanted lungs or lungs rejected for transplantation, CD133+ cells were found at consistently higher levels within the lung parenchyma and intima in idiopathic PAH compared with control lungs. 69 In another study, CD133+ cells were found at high levels in concentric lesions and, in higher number, in plexiform lesions, in contrast to healthy lungs. 61 Employing flow cytometry, we found that levels of CD34+CD133+ cells in the pulmonary artery wall were tenfold higher than levels in controls. 63 Using another progenitor cell marker, c-Kit, elevated numbers of c-Kit+ cells were observed in PAH pulmonary arteries with the majority of the cells located in the perivascular area and vasa vasorum. 62 In all these studies,61–63 increased expression of progenitor cell chemoattractant SDF-1α by pulmonary artery endothelial cells in the vascular lesions was found, suggesting upregulated SDF-1α as one of the mechanism involved in the recruitment of progenitors into the pulmonary artery wall.
Key questions remain as to whether these progenitors contribute to the disease or attenuate the pathological remodeling. To address this, studies were extended to the BM where these cells originate. Reticulin, an extracellular matrix fiber, is present in low amounts in the normal BM around the blood vessels as a component of the basement membrane. Increased reticulin deposition, which is an indicator for abnormal hematopoiesis, 70 was observed in the marrow of patients with PAH. 63 Higher numbers of CD34+CD133+ and CD34+CD133− subsets of progenitors were found in the BM aspirates of patients with PAH compared with the BM from healthy control donors. 63 Additional analysis using ex vivo hematopoietic colony-forming assays and hematopoietic transcription factor gene array analysis revealed that the hematopoietic stem cell in PAH is skewed in the myeloid lineage toward erythroid differentiation. 71 Surprisingly, in the familial form of the disease, unaffected family members had similar BM abnormalities and elevated mobilization in the peripheral circulation, suggesting that increased hematopoietic stem cell proliferation may start before the onset of PAH. 63 Engraftment of human BM into nonobese diabetic/severe combined immunodeficiency mice is a standard assay to study hematopoietic stem cells in vivo. Transplantation of PAH BM CD133+ hematopoietic stem cells into these mice confirmed the increased myeloerythroid proliferation and, strikingly, induced pulmonary vascular disease, including endothelial cell injury associated with increased oxidative stress, in situ thrombi, and right ventricular hypertrophy. These findings advance the idea of a causal link between BM and PAH. 71 Intriguingly, independent from these findings, gene expression profiling of peripheral blood mononuclear cells showed increased levels of erythropoiesis-related genes, correlated to disease severity in patients with idiopathic PAH. 72 Collectively, reports point toward increased myeloerthyroid proliferation and mobilization of hematopoietic progenitor cells in human PAH. These cells home to the PAH pulmonary arterial wall, preferentially in the remodeled areas, and contribute to pulmonary vascular disease.
MAST CELLS IN PAH
Mast cells originate from the myeloid lineage. In contrast to other hematopoietic cells, mast cells do not egress the BM fully formed but, rather, as a progenitor, which shares many of the proangiogenic hematopoietic progenitor cell markers and matures in the peripheral tissues. Mast cells release various angiogenic factors, and their angiogenic potential has been well documented, in particular in the field of cancer biology. 73 There is evidence from animal models and samples obtained from explanted PAH lungs that mast cells are involved in the disease. Increased numbers of mast cells in perivascular remodeled areas of the pulmonary artery were detected in MCT-injected rats. Inhibition of mast cell degranulation by inhibiting c-Kit or by mast cell stabilizer cromolyn reduced right ventricular systolic pressure, right ventricular hypertrophy, and vascular muscularization in a preventive (before onset of PAH), but not in a therapeutic approach. 74 In another study, preventive treatment of rats in the MCT model had a similar favorable outcome. 75 Several groups have reported that mast cells are abundantly present in and around vascular lesions in tissue sections of explanted PAH lungs.62,76,77 Mast cell product tryptase was also found to be increased in the serum of patients with PAH and the levels decreased after treatment with mast cell inhibitors cromolyn and fexofenadine. 77 However, no clinical improvement was observed in this small cohort of patients. These studies provide additional support for the myeloid origins of PAH and point to possible cell mechanism (e.g., mast cells and their proangiogenic and vasoconstrictive products).
FIBROCYTES IN PAH
Fibrocytes are CD45+ collagen-I+ circulating cells in the myeloid lineage. They co-express a specific combination of markers, such as CD45RO and macrophage antigens 25F9 and S100A8/A9, that distinguish them from monocytes, tissue macrophages, and fibroblasts. 2 Two detailed animal studies reported progressive homing of these cells into the pulmonary artery wall, where they obtained an α-SMA-positive myofibroblast phenotype and contributed to thickening of the vessel wall in hypoxia-induced PAH models.78,79 Depletion of the fibrocytes by treatment of the animals with clodronate or gadolinium chloride 78 resulted in reduced myofibroblast accumulation and decreased thickening of the pulmonary artery wall, indicating that the vascular remodeling can be attributed to the recruitment of nonresident blood-borne subsets of mononuclear cells. 78 These findings that a subset of monocytes transdifferentiate into fibrocytes and contribute to the disease may open new therapeutic strategies but need confirmation in human studies.
MSCS IN PAH
MSCs have immunomodulatory benefits in several pathological conditions. In animal models of MCT- or hypoxia-induced PAH, administration of MSCs prevented or attenuated development of disease,80–87 most probably via paracrine mechanisms. 88 Two studies performed autologous MSC transplantation in rats with established MCT-induced PAH.43,44 Both studies found reduced right ventricular hypertrophy and right ventricular systolic pressure and attenuated thickening of the pulmonary artery wall.44,45 In yet another report, MSC therapy did not have beneficial effects in hypoxia-induced PAH. 88 In some studies, MSCs were used as vehicles for gene therapy.85–87 Treatment of animals with MSCs overexpressing eNOS 86 or prostacyclin, 85 a vasodilating and platelet-inhibiting lipid molecule, in the rat MCT model or anti-inflammatory and anti-oxidant enzyme heme oxygenase-1 in the mouse hypoxia model 87 was more effective in reducing right ventricular hypertrophy and pressure and was associated with prolonged survival compared with animals treated with naive MSC. Overall, these findings suggest that use of MSCs as vehicles for gene therapy may be a promising approach, but additional studies are needed.
APPLICATIONS OF BM STEM CELL THERAPIES
In contrast to the available therapies targeting mainly the vasoconstrictive component of PAH, focus has shifted to develop drugs that would halt and reverse the pulmonary vascular remodeling. Therapies modulating the BM progenitors are being looked into on the basis of the established link between the BM abnormalities and the pulmonary vasculopathy. Imatinib, a tyrosine kinase inhibitor that is used for the treatment of chronic myeloid leukemia and has been shown to decrease circulating progenitors in PAH (unpublished data) has been described in case reports to be beneficial for the treatment of PAH. In a phase II study, imatinib therapy was well tolerated by patients with PAH and led to a significant reduction in pulmonary vascular resistance in association with an increase in cardiac output. 89 Results from the phase III study showed clinical benefits in patients treated with add-on therapy with imatinib for 24 weeks compared with placebo, with improved exercise capacity and hemodynamics, but serious adverse events were common. 90 Other drugs targeting the BM progenitors are being considered for the treatment of PAH. Further delineating the interaction between the BM abnormalities and the pulmonary vasculopathy would help develop more selective and targeted therapies.
CONCLUSION AND FUTURE DIRECTIONS
The current paradigm is that abnormalities in hematopoiesis are innate to PAH (Fig. 2). The findings point toward an imbalanced differentiation in the hematopoietic system in which the subsets that promote vascular health are predominated by increased numbers of pathologic progenitors. Additional studies with larger cohorts are necessary to establish a potential therapeutic benefit of modified proangiogenic hematopoietic cells. Because hematopoiesis is a continuous hierarchic process of proliferation and differentiation, the distinction between detrimental and beneficial lineages will be critical to the development of novel therapeutic approaches. Understanding the molecular mechanisms underlying the hematopoietic anomalies and the pathways via which these cells contribute or benefit PAH are still required; likewise, fibrocytes may have a role in pulmonary hypertension, but findings need to be confirmed in patients. MSCs are promising as immunodulatory cells for a wide variety of human diseases, and early findings in pulmonary hypertension models suggest a potential beneficial role in PAH.
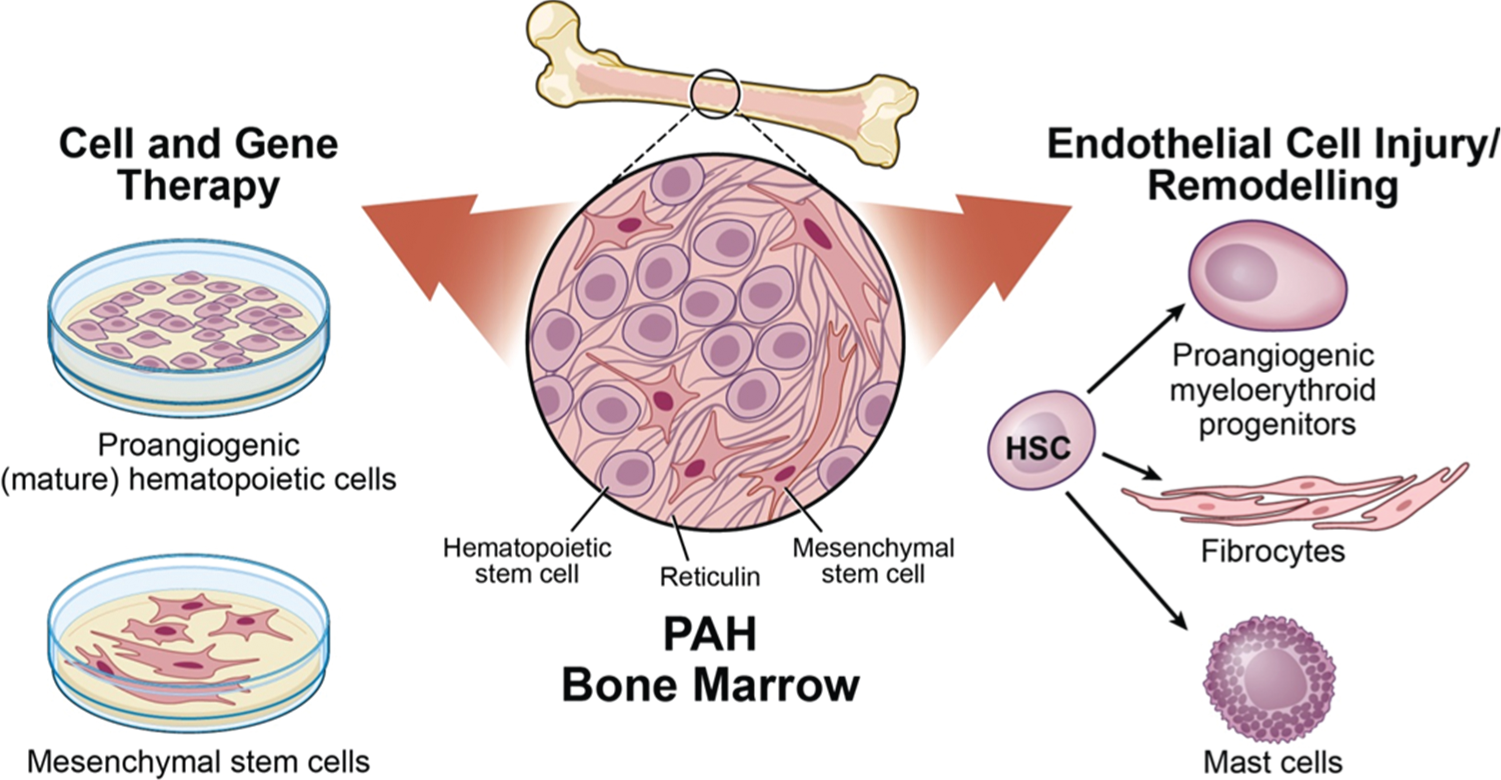
Bone marrow in pulmonary arterial hypertension (PAH). Both the hematopoietic and mesenchymal compartments have been implicated in PAH. Ex vivo expanded monocytic proangiogenic cells, isolated from the peripheral circulation, and mesenchymal stem cells, directly derived from the bone marrow, are used as cell therapy to attenuate the vascular remodeling and/or dampen down the inflammation. These cells have also been used as vehicle for gene delivery. Conversely, increased mobilization of myeloid-erythroid hematopoietic progenitors contributes to the disease by inducing endothelial cell injury and coagulopathy. A subset of the circulating myeloid cells differentiate into fibrocytes that home into the media and adventitia and add to the remodeling by collagen I deposition and by differentiation into myofibroblastic cells. Mast cells also accumulate in the remodeled areas and have been associated with disease progression. HSC: hematopoietic stem cell. Illustration by David Schumick, BS, CMI. Reprinted with the permission of the Cleveland Clinic Center for Medical Art and Photography.
