Abstract

Alzheimer's disease (AD) is a neurodegenerative illness characterized histopathologically by deposition of abnormally phosphorylated Tau-protein. The diagnosis of AD is based on consensus criteria and exclusion of secondary causes of dementia [1]. Cerebrospinal fluid (CSF) Tau-protein is the most comprehensively studied diagnostic biomarker that is consistently elevated in AD and is useful in differentiating AD from normal ageing [2]. Major depression is an important differential diagnosis of AD as depression-related cognitive impairment is common in elderly patients.
Case report
A 54-year-old man (AH, see Table 1) with family history (mother) of AD presented to an other psychiatric hospital with severe depression for 3 months. Further exploration revealed loss of interest and episodes of ‘weirdness’ over the past 2 years. He was diagnosed with major depression with psychotic symptoms and treated with mirtazapine, diazepam, tilidine, perazine, risperdone, moclobemide over 3 weeks. Then intravenous maprotiline was added and he developed a confusional state. Intravenous maprotiline was discontinued and neuroleptic medication was tapered off. The symptoms resolved within 4 days; however, cognitive impairment with prominent short-term memory disturbances and mini-mental state test of 23/30 persisted. While magnetic resonance imaging was normal, CSF showed an elevated total protein of 0.92 g L−1 and a high concentration of total Tau-protein (1200 ng L−1, laboratory reference < 250 ng L−1), CSF cell count was normal. A diagnosis of probable AD was made and the patient was referred to rehabilitation. After discontinuation of antidepressive medication, no cognitive impairment was detected at follow-up 6 months later. The patient was then seeking compensation for malpractice. We became involved as legal experts in the case 3 years later and were asked to evaluate the diagnosis of probable AD.
CSF levels of total Tau-protein (pg/mL) in 10 patients who had received high doses of neuroleptic/ antidepressive medication
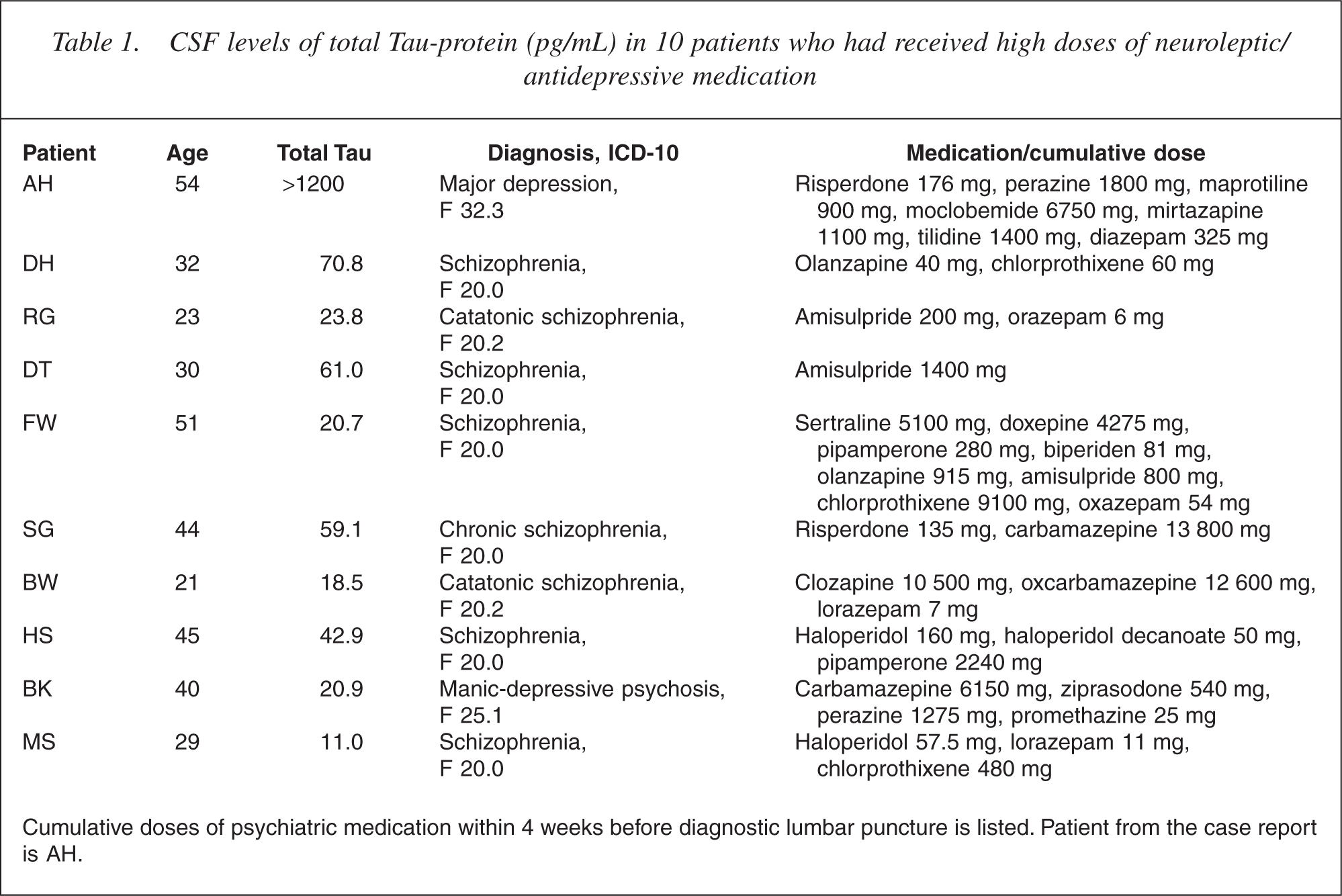
Cumulative doses of psychiatric medication within 4 weeks before diagnostic lumbar puncture is listed. Patient from the case report is AH.
On examination the patient showed mild depressive symptoms without cognitive impairment, neurological examination was otherwise normal. Because there was no evidence of AD or any other neurodegenerative dementia, the question was how to explain the excessively high CSF Tau concentration at the time of acute psychiatric illness.
In retrospect, several explanations seem possible:
Increased levels of CSF Tau-protein have been reported in patients with ‘mild cognitive impairment’ particularly in those who progress towards AD [3]. Therefore, the patient who also had a family history of AD might be at-risk for development of dementia or have ‘incipient AD’. Confirmation of the excessive Tau concentration of 1200 ng L−1 and additional determination of amyloid beta 1–42 would have been desirable, but a follow-up lumbar puncture was refused. In addition to high CSF Tau, there was also a significant elevation of CSF protein indicating blood– brain barrier disruption. However, other neurological diseases – in particular central nervous system infection – were ruled out. Neurotoxicity as a result of the high-dose psychiatric combination therapy might have caused an increase of Tau-protein in the CSF.
Because there are no data on the possible effects of antipsychotic and high-dose antidepressive drugs on CSF Tau in the literature, this case prompted us to investigate whether treatment with combination of different neuroleptic and antidepressant drugs resulted in elevated CSF Tau levels in other cases, too.
The CSF samples were available from nine patients treated at the Department of Psychiatry of the Medical School Hannover between 2002 and 2004 who had received high doses of antipsychotic and/or antidepressants and who had undergone lumbar puncture for diagnostic purposes (Table 1). Cell count, total protein, determination of the albumin ratio and evaluation of oligoclonal bands had been part of the routine CSF analysis. Remaining CSF had been stored at −70°C at the CSF laboratory. Total Tau was measured by ELISA (Innogenetics, Heiden, Germany). Except for one case (SG, also suffered from a herniated disc) with elevated CSF protein, the other CSF parameters including Tau were normal in all nine patients.
CSF Tau is regarded as biomarker for neuronal destruction and has been investigated in patients with dementias and non-demented controls in several studies [4]. High levels of CSF Tau have been closely associated with AD but can also be found in other diseases with extensive or rapid neuronal degeneration such as prion disease, acute stroke or traumatic brain injury. CSF Tau distinguishes between patients with AD and nondemented control subjects with a specificity of 80–90% [2, 5, 6]. In healthy controls or patients with psychiatric diseases, elevated CSF Tau has not been reported so far. It was recently shown that patients with dementia who were treated with neuroleptic drugs had elevated levels of phosphorylated 14-3-3ζ protein in the CSF [7]. In our case, a drug-induced toxic reaction best explains the clinical picture of delirium and elevated CSF Tau and CSF protein. The data from the additional nine patients do not suggest a general association between high-dose psychiatric medication and CSF Tau; however, patient AH had received by far the highest cumulative doses. This report emphasizes the fact that dementia is a clinical diagnosis and that neurochemical marker like CSF Tau should be interpreted with caution particularly in patients under high-dose psychiatric medication. A prospective study of CSF Tau in age-matched cases with drug-induced delirant syndrome would be more conclusive.
