Abstract
The role of frontal–subcortical circuits (FSCs) and human behaviour has been of interest since Alexander et al. [1], following in the evolutionary, hierarchical, systemic and sensori-motor tradition of Hughlings Jackson [2], described five functionally segregated basal-gangliathalamo- cortical circuits, consisting of motor, oculomotor, dorsolateral prefrontal, lateral orbito-frontal (OF) and anterior cingulate (AC) networks. While the authors limited their functional speculations, they pointed out that anatomic and physiological evidence accumulated over the previous two decades had revealed a level of organization and functional specificity of the basal ganglia, in parallel to that of the cerebral cortex itself. Each circuit appeared to receive input from several separate but functionally related cortical areas, to traverse specific portions of the basal ganglia and thalamus, and project back onto one of the cortical areas providing input to the circuit, thus forming a partially ‘closed loop’.
The neuro-anatomical work of Heimer [3] provided a further understanding of the relationship between brain and behaviour in discussing the importance of the ‘new anatomy’ of the basal forebrain. A classical dichotomy, which reinforced a ‘limbic versus basal ganglia’ or ‘limbic versus extrapyramidal’ view of emotional expression, was the concept that limbic forebrain structures projected to the hypothalamus, rather than to basal ganglia, whereas non-limbic cortical areas including most of the isocortex (neocortex) projected to the basal ganglia. The use of selective silver degeneration procedures combined with electron microscopic methods allowed for the demonstration of a ventral-striatal-pallidal system, leading to the notion of parallel cortical–subcortical re-entrant circuits. Heimer described cortical projections from the olfactory cortex and hippocampus (allocortex) and nonisocortical (mesocortical) areas such as the entorhinal, insular and cingulate cortex as well as the posterior orbital-medial cortex and the temporal pole. The ventral striatum (nucleus accumbens, olfactory tubercle, ventral caudate-putamen) receives projections from the entire cortical mantle. According to Heimer, the realization, that the whole cortical mantle is related to the basal ganglia, has provided a new blueprint of forebrain organization, the ventral striatum and ventral pallidum being integral parts of a new theoretical framework for adaptive responding and neuropsychiatric disorders.
Lichter and Cummings [4] have pointed out that the understanding of FSCs provides a unifying framework for understanding the similarity of behavioural changes that accompany cortical and subcortical lesions. They describe at least five circuits, namely ‘a motor circuit originating in the supplementary motor area, an oculomotor circuit originating in the frontal eye field and three circuits with origins in the dorsolateral prefrontal cortex (DLPF) mediating “executive” function, an AC circuit involved in motivational mechanisms, and an OF circuit involved with the medial portion allowing integration of visceral-amygdal functions with the internal state of the organism, while the lateral portion is involved with the integration of limbic and emotional information into contextually appropriate behavioural responses’. The authors point out that the five circuits share common structures, namely projection sequentially to the striatum, to the globus pallidus (GP) and substantia nigra (SNr), and then to specific thalamic nuclei, with a final link back to the frontal lobe. Each circuit has two pathways (i) a direct pathway, featuring a monosynaptic link between the striatum and GP interna–substantia nigra (Gpi–SNr) complex; and (ii) an indirect pathway that projects from striatum to GP externa (Gpe) linking to the Gpi–SNr, via the subthalamic nucleus.
Weingarten and Cummings [5] confirmed that while the striatum is the input segment of the basal ganglia, there are two routes, direct and indirect from the striatum to GP, which then utilize gamma-aminobutyric acid (GABA) to connect to the thalamus and complete each fronto–subcortical circuit by connecting to the circuit specific area in frontal cortex using glutamate as its neurotransmitter. While the separation of neurobehavioural, neuropsychiatric and movement syndromes in terms of FSC involvement is most specific at the cortical area, the organization is recapitulated at the striatal, pallidal and thalamic levels of the circuits.
The circuits of most interest in the present context are the OF circuit and the AC and DLPF circuits. In obsessivecompulsive disorder (OCD), positron emission tomography studies have demonstrated aberrant OF and AC circuits [6, 7], while the DLPF may be important in attention deficit hyperactivity disorder (ADHD). The DLPF has been demonstrated by Goldman-Rakic [8] to be important for working memory (WM) in primates and humans. According to Goldman-Rakic [8, 9] the most significant experimental discovery for understanding the functions of the prefrontal cortex (PFC) was the demonstration by Jacobsen [10] that PFC damage produced a profound and selective deficit on tests of spatial delayed responses, with a syndrome characterized by distractibility and impulsivity. Delayed response tests are believed to measure the ability of animals and humans to respond to situations on the basis of stored information rather than on the basis of immediate stimulation. Goldman-Rakic [8] has shown that the principal sulcus neurones demonstrate activity that is time-locked to the delay period of a delayed response and thus participate in an essential mechanism for holding spatial and representational information ‘online’ for the time necessary to guide a response.
In addition, Goldman-Rakic [11] has described a larger system of connections between circuitry linking prefrontal, parietal, limbic and temporal cortices, allowing for ‘multiple functional nuances. She notes that delayenhanced discharge during delayed response tasks has been reported in several key structures with which posterior parietal areas and principal sulcus and frontal eye fields are connected including hippocampus, head of caudate nucleus and mediodorsal nucleus of the thalamus’. More recently Sherman and Guillery [12] have argued that the thalamus functions as a relay in cortical–cortical pathways in addition to thalamocortical pathways. Thus the role of cortical–cortical re-entrant circuits in higher cortical representational processes is of considerable interest, but representation and inhibition may also require subcortical input.
While Goldman-Rakic emphasized the importance of cortical–cortical circuits in representational processes, it has been postulated that subcortical circuits are involved in inhibitory processes. According to Houk [13] signal processing operations that occur in the subcortical loops are quite powerful computationally, and by a recursive process use the results of pattern classification to update the cortical pattern that provides its own input. Thus recursion or re-entry is fundamental to higher cortical executive functions, including WM, while ‘yes’ or ‘no’ decisions appear to be made at subcortical levels. Houk and Wise [14] suggest that pyramidal neurones of the cerebral cortex have combinations of ion channels that promote graded frequencies of firing in response to graded strengths of input, which are advantageous when results of neural computations need to be expressed over a range of magnitudes rather than ‘yes’ or ‘no’ decisions. Neurones with appropriate sensitivity to serial order are distributed throughout the cortical-basal ganglionic loop.
According to Houk and Wise [14] a long history of connectionist literature [15] has shown that highly interconnected recurrent networks are an excellent architecture for associative memories, perceptual operations and the solution of optimization problems. The ability to respond to graded inputs allows many individual factors to be appropriately weighted, and the availability of many loops of interconnection facilitates recursive readjustments of multiple factors. Houk and Wise hypothesized that cortical-basal ganglia modules might function as detectors of specific contexts, providing information that could be useful in planning and gating actions, while cortical-cerebellar modules might function in the programming, execution and termination of actions. Moreover, cortical–cortical modules might implement collective computations, while also serving as a repository for distributed representations.
Neural network models
The Parallel Distributed Processing (PDP) paradigm, which was introduced in the 1980s mainly by Rumelhart and McClelland [15], offers what we believe to be a suitable framework to model the processes in the architecture as suggested in Fig. 1. Parallel Distributed Processing models rely to a large extent on computational models that are based on artificial neural networks comprising large numbers of interconnected units that represent either individual neurones or functional groups of neurones. Harnad [16] describes connectionism as ‘dynamic patterns of activity in a multilayered network of nodes or units with weighted positive and negative interconnections’. The strengths of the interunit connections are individually modifiable in a way that is analogous to the changes to the synaptic strength in response to activation following Hebb [17]. Several principle network architectures with distinct properties have been used successfully not only as models of cognitive functions but also as models for brain structures [18, 19].
Recurrent architecture.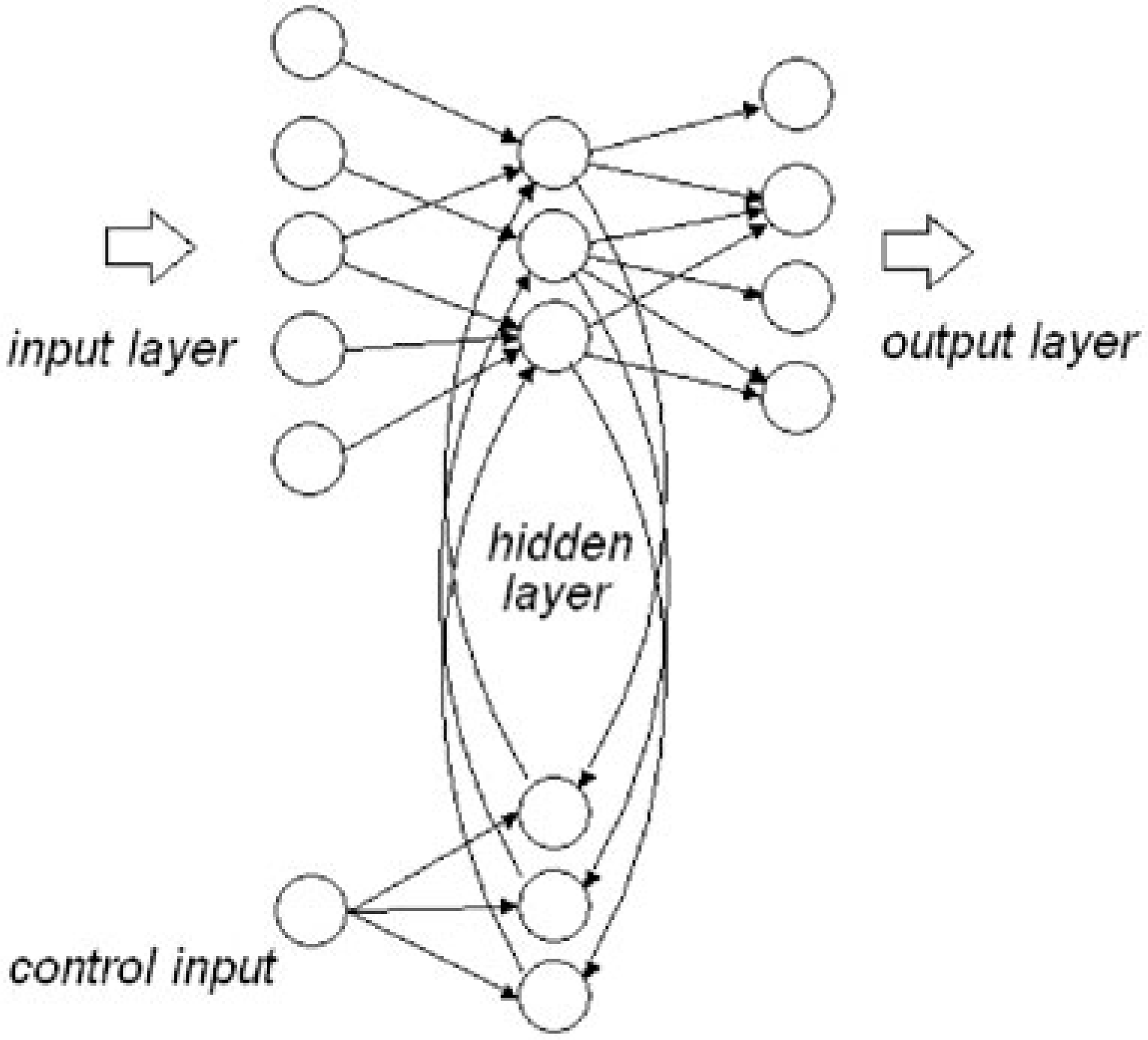
Churchland [20] likens the brain's information processing to that of artificial neural networks with both feed-forward and recurrent capacity. If recurrent or descending pathways are added to the basic feed-forward architecture ‘we lift ourselves into a new universe of functional and dynamical possibilities’. Networks with recurrent connections are able to process temporal relationships in data, because during training the previous internal states influence the network's next state. Recurrent networks contain circuits that allow for part of the information at the output of the hidden layers to be fed back (Fig. 2). The feedback mechanism provides a rudimentary short-term memory that allows for the modelling of temporal events (see, e.g. Elman [21]). According to Churchland and Sejnowski [22] feedback endows a network with several important capacities, including the incorporation of multiple time scales into processing units and the ability to process temporarily extended sequences of inputs. However, simple recurrent networks cannot be trained on more difficult tasks because the temporal memory provided by the recurrent net decays in time.
Recurrent neural network. Gpe, globus pallidus externa; Gpi, globus pallidus interna.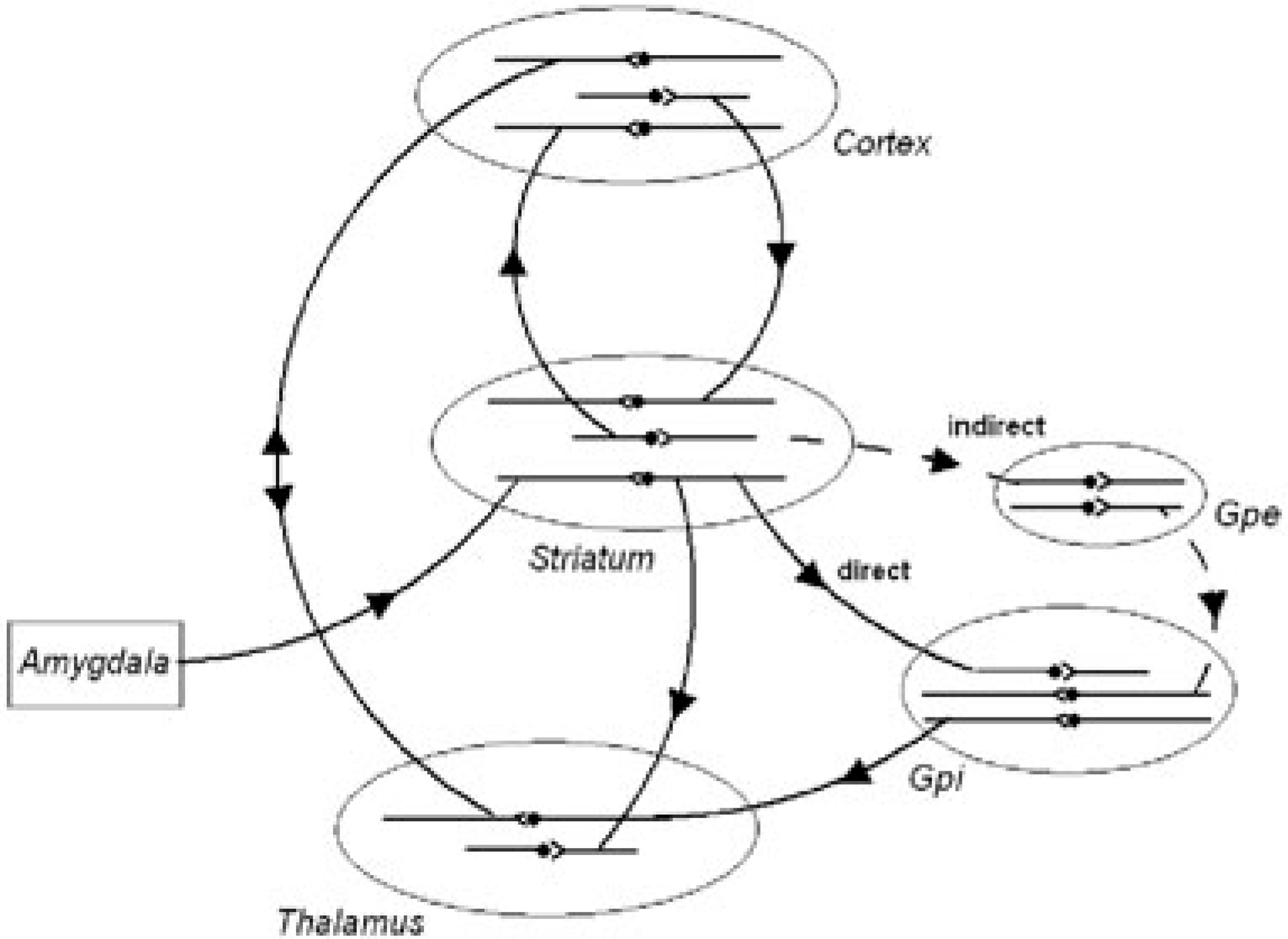
Although the model that is suggested here does not aim to solve the shortcomings of short-term temporal memory, the addition of a relatively simple control circuit for the feedback units will allow for modelling the desired effect. The control input that is used to vary the feedback ratio (the amount of feedback signal in the overall signal to the hidden units) can inhibit the memory decay. Provided a sufficiently high proportion of the output signal from the hidden units is fed back, the network will settle into a stable state that is governed by the feedback loop. However, the signals from the input layer to the hidden layer can effectively be saturated or swamped by the feedback signal, if the ratio between input and feed back at the hidden layer is strongly biased toward feedback. This saturation (arousal/re-enforcement) cannot be overcome by changes to the regular inputs alone because the behaviour is no longer governed by the (regular) inputs, giving rise to a recurrent state.
Biological networks
In the present context, the importance of neural network (PDP) models is that they demonstrate the capacity of recurrent networks to model feedback effects, which are hypothesized for cortical–subcortical re-entrant circuits. For example, Yang and Seamans [23] described the effects of ventral tegmental area (VTA) activation on prefrontal (PFC) neurones and networks. In control conditions where there is little dopaminergic (DA) tone, VTA neurones fire at a low basal rate, until a better than expected rewarding stimulus is encountered, giving rise to bursting in VTA neurones, and release of glutamate in the PFC. This gives rise to an ‘up’ state in the PFC, which terminates in a few hundred milliseconds, but if large levels of extra-synaptic DA are present, supraoptimal D1 receptor stimulation may push the PFC into a ‘locked state’ with stereotypic thoughts or actions.
Grossberg [24] has described the properties, which allow the brain to self-organize its neural circuits. The inverted-U property enables a ‘gated dipole’ to maintain a ‘Golden Mean’ in response to the circuit's arousal level. The ‘Golden Mean’ implies that circuit sensitivity to input fluctuations is optimal at moderate arousal levels, but degrades in different ways when the circuit is either under-aroused or over-aroused. At low arousal, inputs must be larger than normal to overcome the dipole's increased response threshold, but once the threshold is overcome, the circuit is hyperexcitable above the threshold. The threshold can be brought into normal range by increasing its arousal until it reaches the peak of the inverted-U, but ‘too much of a good thing’ such as amphetamine can create stereotypy or psychosis.
A pharmacological example here is the classical work of Sprague and Sleator [25], who reported that at low psycho-stimulant doses, children with ADHD showed a remarkable improvement on a short-term memory test at all levels of task load, whereas at higher doses, there was a significant decrement in performance with restricted cognition on the more difficult versions of the task. This was referred to as the ‘inverted-U’ effect, was very influential in suggesting that low dose regimens were optimal for attention-disordered children. More recently, Mattay et al. [26] described a similar effect in which the PFC functioned optimally when dopamine activity was neither too low nor too high, corresponding to the top of an upside-down ‘U’. This effect was found to vary according to whether subjects prescribed dexamphetamine were homozygous for Met versus Val alleles of the catechol- O-methyltransferase (COMT) gene. In humans, the COMT gene contains a common variation in its coding sequence, which translates valine to methionine (Val/ Met). At room temperature the Met allele has one-fourth the enzyme activity of the Val allele. It is believed that COMT is important in the metabolism of DA in the PFC, thus accounting for the genetic variation in response to stimulants.
Psychopathological symptoms can thus occur either as a result of either too high or too low neurotransmission (arousal) at cortical synaptic gates. Traditionally the cortex has been conceptualized as the site of executive function [27]. However, the PDP recurrent network analogy places equal or greater emphasis on lower control centres, such as the striatum. When control processes are biased either by anxiety, trauma or medication, with continuing recurrent feedback, a spiral of recurrent behaviour may occur, as in OCD or stereotypy.
Obsessive-compulsive disorder
According to Saxena and Rauch [6] the various FSCs subserve different behavioural functions and mediate the symptomatic expression of different psychiatric syndromes. The circuits originate in nearly every part of the cerebral cortex and project through different subcompartments of the basal ganglia and thalamus, back to the cortex. Saxena and Rauch describe the lateral prefrontal– subcortical circuits as being associated with major depression, whereas orbito-frontal-subcortical circuits are thought to be involved in OCD, while sensori-motor circuits are thought to mediate symptoms of movement disorders.
Saxena and Rauch [6] outline the ‘classical’ route of cortical–subcortical circuits as having two loops (i) a ‘direct’ pathway; and (ii) an ‘indirect’ pathway. In primates the direct pathway is thought to project from cerebral cortex to striatum to the internal segment of the GP, then to thalamus and back to cortex. On the other hand, the indirect pathway has a similar origin from cortex to striatum, but then projects from striatum to the external segment of the GP and then to subthalamic nucleus before returning to GP/SNr, where it rejoins the common pathway to the thalamus and cortex (this is similar to the basic FSC structure described above). Impulses along the direct pathway with two excitatory and two inhibitory projections act as a net ‘positive feedback loop’, whereas activity along the indirect basal ganglia control system, with three inhibitory connections provides negative feedback and decreases thalamo-cortical drive. Obsessivecompulsive disorder symptoms were thought to be due to an imbalance in tone between direct and indirect striato-pallidal pathways. Sachdev and Malhi [7] have described evidence implicating the OF and AC, as well as the striatum and amygdala and it is likely that symptomatology derives from increased feedback, at these sites, preventing appropriate decay of cortical activity.
Attention deficit hyperactivity disorder
Dopaminergic circuits
Strictly speaking ADHD symptomatology may represent the opposite of a recurrent symptom syndrome. That is, distractible behaviour, which is controlled by environmental stimuli, suggests an inability to maintain a sustained, let alone recurrent behaviour pattern. Nonetheless, the PDP architecture may still apply, but in this case there is insufficient neurotransmitter at subcortical levels to maintain a recurrent feedback/reinforcement loop.
Sagvolden et al. [28] have described mesocortical, mesolimbic and nigrostriatal dopaminergic pathways, giving rise to specific symptom classes in ADHD, relating to executive function, emotional regulation and motor symptomatology. The authors postulated that the behaviour and symptoms of ADHD derive from altered dopaminergic function with a consequent failure to modulate non-dopaminergic (predominantly glutamate and GABA) signal transmission. They postulate that a hypo-functioning mesolimbic dopamine branch produces altered reinforcement of behaviour and deficient extinction of previously reinforced behaviour, giving rise to delay aversion, impulsivity and failure to ‘inhibit’ responses or disinhibition. A hypo-functioning mesocortical branch is postulated to cause attentional deficits, while a hypo-functioning nigrostriatal branch gives rise to impaired motor functions. Sagvolden and colleagues have argued that the main component of altered reinforcement processes in ADHD children is a steeper delay-of-reinforcement gradient, or shorter time interval between response and effectivereinforcer, resulting in less effective reinforcement and also less effective extinction of previously established, but no longer reinforced responses.
Sonuga-Barke [29] described a dual-pathway model of ADHD, which integrated executive function and motivational theories of ADHD. Executive function refers to dysregulation of cognitive controls, resulting in failure to modify actions, thoughts, and feelings so that they conform to the social and intellectual requirements of a situation. On the other hand motivational models focus on the inability of ADHD children to delay gratification for later reward (delay-aversion).
Sonuga-Barke has pointed out that previous theories of psychopathology in ADHD had attempted a unitary construct at the level of symptom expression, but given that the ADHD diagnosis was made on the basis of behavioural symptoms, there was no necessary one-toone match between clinical and neuro-bio-psychological characteristics of ADHD. He pointed to the growing body of evidence from neuro-anatomical, psychopharmacological, imaging and clinical studies to support a model of cortical–subcortical interaction, based on parallel but functionally segregated distributed brain circuits connecting specific cortical foci with basal ganglia and thalamic nuclei. The executive circuit, was thought to involve glutamergic excitatory inputs down from the PFC to the dorsal portion of the neo-striatum, with reciprocal excitatory connections back up to cortical regions. These connections were mediated through GABA-based inhibitory inputs to dorso-medial sections of the thalamus via dedicated direct and indirect pathways within subcortical relays (GP, SNr and the subthalamic nucleus). On the other hand, activity within the reward circuit was centred on the ventral striatum (in particular the nucleus accumbens). In this case the excitatory connections from frontal regions (AC and OF) were reciprocated via the ventral pallidum and related structures through the thalamus.
In both cases, dopamine was thought to be a key neuromodulator of both the executive and reward circuits. Activity within the executive circuit was modulated by the meso-cortical and nigro-striatal dopamine branches, while on the other hand, meso-limbic branches of the dopamine system from the VTA, modulated motivational circuits. According to Floresco et al. [30] the mesolimbic dopamine system plays a central role in reward and goaldirected behaviour, and has been implicated in multiple psychiatric disorders. Understanding the mechanism by which dopamine participates in these activities requires comprehension of the dynamics of dopamine release. Floresco et al. [31] describe the DA system as compartmentalized consisting of a synaptic (and potentially peri-synaptic) compartment and a tonically maintained extrasynaptic compartment, each of which is differentially affected by uptake processes.
The description of separate synaptic and peri-synaptic compartments would imply separate cortical and subcortical genetic influences, and separate pharmacological influences at these levels. For example, DA metabolism is predominantly via the dopamine transporter (DAT) at subcortical levels, but via COMT at the PFC [32]. Bilder et al. described a tonic/phasic theory of dopaminergic actions where subcortical phasic DA is subject to fast reuptake by DAT, whereas most DA in the PFC is removed via re-uptake into noradrenergic terminals, requiring DA to diffuse long distances before inactivation via this route. While the tonic/phasic model differs from the present feedback models, which are based on the neural network analogy, it is similar in emphasizing the importance of cortical–subcortical relationships.
Continuous Performance Task
The Continuous Performance Task (CPT) has been used to measure sustained attention in ADHD [33, 34]. The CPT was originally described by Rosvold et al. [35] and requires subjects to press a button or lever in response to a visual or auditory stimulus, randomly and displayed among non-stimuli over a sustained period of time. It requires retention of a discriminative stimulus and inhibition of response to non-stimuli. Levy [33] utilized the CPT to demonstrate a clear development in capacity for sustained attention in normal children at age 4–6 years, as well as clear differences in inhibitory capacity in children with ADHD related to dopaminergic function [36, 37].
Háger et al. [38] have utilized a combination of the Continuous Performance Test (CPT) and functional magnetic resonance imaging (fMRI) to investigate an attentional network mediated by the dorsolateral prefrontal and mesial frontal cortex, thalamus, basal ganglia, posterior parietal and superior temporal lobe. They demonstrated a right lateralized pattern of activation, not only in cortical, but also in subcortical structures, including the right caudate nucleus. Good performers showed activation in the superior frontal gyrus, posterior insula and thalamus, while areas of deactivation were localized in both left and right posterior insular region, superior colliculi and left fusiform gyrus/hippocampus. In contrast the low-performance group showed no significant activation in the mesial and lateral superior frontal gyrus and extended activation in the right dorsolateral PFC.
In terms of the present models, the CPT is ideally suited to model and measure WM, as well as inhibitory capacity, by measuring both the consistency of reaction times to the index stimulus (WM) and errors of commission (lack of inhibition). Because of the continuous nature of the task, it models ongoing cerebral processing and is thus suited to functional imaging of cortical– subcortical processing, and pharmacological effect on the task parameters such as interstimulous intervals.
Conclusion
The work of Goldman-Rakic describes reciprocal prefrontal- parietal processes involved in WM and representational processes, while subcortical re-entrant circuits provide temporal and inhibitory modulation. The present review postulates that recurrent symptomatology originates in dysfuctions in recurrent cortical–subcortical circuits, controlled by subcortical ‘hidden units’ that allow for ongoing information at the output of the ‘hidden layers’ to be fed back to cortical synaptic gates. The hypothesis allows for conscious modulation of recurrent symptomatology, but places inhibitory control at the interaction of feedback from subcortical levels (namely basal ganglia, cerebellum and amygdala), and cortical receptors in parallel re-entrant circuits. The model implies that there can be separate genetic influences on recurrent behaviours at cortical and subcortical levels. The particular resulting symptomatic syndrome will depend on the particular circuit (motor, oculomotor, dorsolateral prefrontal, lateral OF or AC), neurotransmitters and receptors involved, but greater understandings of recurrent symptomatology should allow more targeted pharmacological interventions.
