Abstract
Kraepelin and Freud both regarded endocrinology as potentially important in the causation and treatment of major psychiatric disorders [1, 2], and the role of endocrine systems in the pathogenesis of depressive illness has been a focus of research for many decades. However, limited understanding of complex endocrine systems and their interaction with neural networks and primitive scientific methodology frustrated early attempts to establish links between endocrine dysfunction and mood disorders [3]. More recent developments in the field of psyconeuroendocrinology have highlighted the significance of endocrine systems in the pathophysiology of mood disorders and suggested scope for future developments in treatment. This article selectively reviews these recent developments, with a particular emphasis on the potential for the development of new treatments.
The hypothalamic-pituitary-adrenal (HPA) axis
Cortisol, a glucocorticoid released from the adrenal cortex, is the end-product of the hypothalamic-pituitaryadrenal (HPA) axis. The HPA axis comprises the tissues of the hypothalamus, pituitary and adrenal cortices, regulatory neuronal inputs, and a variety of releasing factors and hormones (Fig. 1). Neurosecretory cells within the paraventricular nucleus of the hypothalamus secrete corticotropin-releasing hormone (CRH) and arginine vasopressin (AVP) into the microportal circulatory system of the pituitary stalk. CRH and AVP cause the release of adrenocorticotropic hormone (ACTH) from the anterior lobe of the pituitary. Cortisol is released from the adrenal cortex in response to ACTH. Cortisol has a panoply of central and peripheral effects which are mediated via in part by glucocorticoid receptors (GRs).
The hypothalamic-pituitary-adrenal axis. CRH, corticotropin-releasing hormone; ACTH, adrenocorticotropin hormone; AVP, arginine vasopressin; GR, glucocorticoid receptor.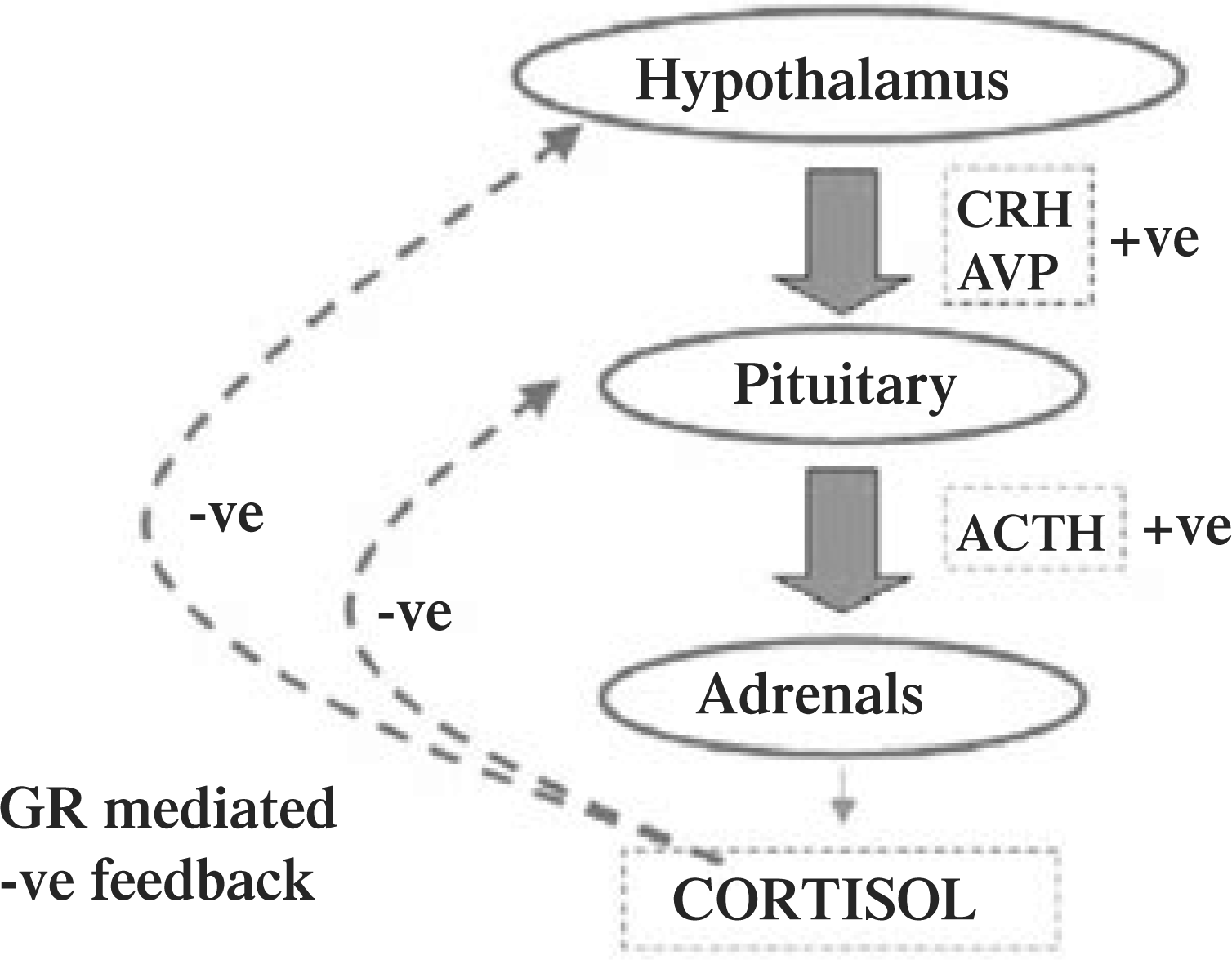
The activity of the HPA axis is highly regulated. Secretory cells within the paraventricular nucleus receive neuronal inputs from a number of brain regions including the amygdala, hippocampus and nuclei within the midbrain. The HPA axis also has an autoregulatory mechanism mediated by cortisol. Endogenous cortisol binds to GRs in the HPA axis and acts as a potent negative regulator of HPA activity. These regulatory mechanisms are important in determining basal levels and circadian fluctuations in cortisol levels. Changes in GR number or function may be important in altering the homeostatic function of the HPA axis observed in healthy individuals.
Cortisol and GR abnormalities in depression
The first observations of abnormalities of cortisol levels in patients with depression were made in the late
1950s by Board and colleagues, and these observations have been repeatedly replicated. Subsequent studies have shown that HPA hyperactivity, as manifested by hypersecretion of CRH, increased cortisol levels in plasma, urine and cerebrospinal fluid, exaggerated cortisol responses to ACTH, and enlarged pituitary and adrenal glands, occurs in individuals suffering from severe mood disorders. Hypersecretion of CRH causing hypercortisolaemia may be a result of impaired feedback mechanisms resulting from GR abnormalities, such as decreased receptor number or altered function. This view is supported by the demonstration of GR abnormalities in postmortem studies of patients with severe mood disorders [4]. The dexamethasone suppression test is a measure of the functional integrity of the GR-mediated negative feedback mechanism: the cortisol-suppressing activity of the synthetic glucocorticoid, dexamethasone, is an approximate indicator of GR status. Reports of cortisol non-suppression in response to dexamethasone in both unipolar and bipolar disorders do indeed suggest a primary GR abnormality in these disorders [5]. Recent work has shown that abnormal HPA axis function is also found using the combined Dex/CRH test in bipolar disorder [6]. Responses to the Dex/CRH test have been found to resolve with treatment in depression, although doubts remain about chronic depression [7, 8] and the abnormality may be more temporally stable in bipolar disorder [9]. Post-dexamethsaone arginine vasopressin levels are also abnormal in severe mood disorders, both bipolar and unipolar [10].
Consequences of hypercortisolaemia
It is now established that in conditions in which there are raised endogenous or exogenous corticosteroids (including Cushing's disease and severe mood disorders) there is also a significant degree of cognitive impairment [11]. Studies in experimental animals have shown defi-cits in learning and memory following chronic administration of glucocorticoids [12], as well as marked atrophy of neurones in the hippocampal formation. It has been postulated that a similar neurodegenerative effect of cortisol may underlie some of the cognitive deficits observed in humans suffering from severe mood disorders [13, 14].
Recent clinical data suggest that cortisol treatment induces cognitive deficits in healthy humans, and these deficits appear to be mediated in part via the frontal lobe, suggesting that this brain area may also be sensitive to the neurodegenerative effects of cortisol [15]. The defi-cits in healthy volunteer subjects are reversible, but this may not be the case with the cognitive deficits induced by hypercortisolaemia associated with mood disorders [15, 16]. An early re-establishment of normal HPA activity in mood disorders before permanent deficits in cognitive function occur may therefore be an important therapeutic goal.
Therapeutic targets
There is increasing evidence to suggest that the consequences HPA dysfunction described above are central to the pathogenesis of affective disorders and cognitive de-ficits. Modulation of the effects of hypercortisolaemia may provide potential treatments for mood disorders, and such strategies are the focus of considerable research interest.
Dehydroepiandrosterone
The adrenal steroid dehydroepiandrosterone (DHEA) has been used with some success in the treatment of depression [17]. The physiological function of DHEA is unclear, but DHEA is known to possess antiglucocorticoid properties which may account for its therapeutic effects. Another explanation is that DHEA is partially metabolized to testosterone and oestrogen, both of which have effects on mood. The ratio between cortisol and DHEA may be a more sensitive measure in depression than basal cortisol alone [18].
Steroid synthesis inhibitors
Inhibiting steroid synthesis is another strategy which has been employed to lower raised cortisol levels. Ketoconazole, administered daily, was shown to reduce both cortisol levels and depressive symptoms within 72 hours in a case of treatment-resistant depression [19]. Subsequent studies have investigated the use of ketoconazole, as well as metyrapone and aminoglutethimide, as antidepressant therapies, with inconsistent results [20]. However, Jahn et al. recently examined whether the addition of metyrapone to standard antidepressants induced a more rapid, more efficacious, and sustained treatment response in patients with major depression [21]. They reported that metyrapone was an effective adjunct in the treatment of major depression, accelerating the onset of antidepressant action. A better treatment outcome compared with standard treatment and an antidepressive effect sustained for the period of observation were both observed.
Corticotropin-releasing hormone antagonists
Over-secretion of CRH, resulting in hypercortisolaemia, may be normalized by blockade of CRH receptors. Preclinical studies have suggested that CRH antagonists will have clinical utility in conditions related to HPA hyperactivity, particularly anxiety disorders. We await the results of ongoing clinical investigations.
Glucocorticoid receptor agonists
Activation of the GR-mediated negative-feedback mechanism that regulates cortisol levels is another strategy for reducing circulating cortisol levels. The synthetic glucocorticoid dexamethasone given at doses of 3–4 mg for 4 days has been shown to have antidepressant effects [22]. At this dose dexamethasone does not enter the central nervous system and consequently central GRs are not activated. Glucocorticoid receptors at the level of the pituitary are activated leading to a lowering of endogenous circulating cortisol. The brief course of administration of dexamethasone in these studies avoids the side-effects associated with longer-term treatment.
Glucocorticoid receptor antagonists
Glucocorticoid receptor antagonists have also been advocated as agents with potential therapeutic properties for mood disorders. This is based on the ability of the GR antagonist to block any detrimental effect of hypercortisolaemia and on the ability of an antagonist to upregulate its receptor. Administration of a GR antagonist results in an acute antiglucocorticoid effect, while presumably causing a compensatory up-regulation of GR numbers, leading to enhanced negative feedback on the HPA axis. Initial clinical studies using the GR antagonist RU-486 (mifepristone) have been encouraging, but some clinical efficacy may have been masked by the prolonged administration of the drug [23]. Animal studies suggest that GR numbers are increased rapidly (within hours) after the administration of RU-486, which may restore normal feedback, thus ‘resetting’ the HPA axis. Such data suggest that a brief period of treatment with the antagonist may be adequate for restoring normal HPA axis function. In a recent pilot study, 20 subjects with bipolar disorder were treated with 600 mg day −1 of RU-486 or placebo for 1 week in a double-blind crossover design [24]. Over the 6-week study, neurocognitive and neuroendocrine function and mood symptoms were measured. Following treatment with RU-486, selective improvement in neurocognitive functioning was observed and significant improvements in mood symptoms were found compared with placebo. The improvement in cognition inversely correlated to basal cortisol levels, adding to the plausibility that this was an antiglucocoticoid effect. These data require replication but provide preliminary evidence that glucocorticoid antagonists may be effective in the treatment of cognitive deficits and mood symptoms associated with bipolar disorder. Interestingly, a similar protocol in patients with schizophrenia suggested no beneficial effect on symptoms or cognitive impairments, possibly because these patients had no evidence of dysfunctional HPA axes [25].
Conclusions
There is robust evidence demonstrating abnormalities of the HPA axis in both bipolar and unipolar depression. Hypercortisolaemia may be central to the pathogenesis of depressive symptoms and cognitive deficits, which in turn may result in part from the neurocytotoxic effects of raised cortisol levels. Manipulation of the HPA axis has been shown to have putative therapeutic effects in both preclinical and clinical studies, and recent data suggest that both glucocorticoid receptor antagonists and steroid synthesis inhibitors may be useful in the treatment of mood disorders.
This paper was presented at the Joint CINP/ASPR meeting, held in Brisbane, December 2005.
