Abstract
Craving is a powerful motivational state associated with the intense desire to consume a drug. For alcoholism as for other forms of addiction, it is a key feature in the diagnosis of substance abuse [1]. Classically, craving for alcohol and relapse have been regarded as being intimately related [2]. In the last decades, various models of drug craving have been proposed [3], the most highly regarded concept being the cue-reactivity model. According to this model, in addition to a specific pattern of autonomic responses, appearance of drug-related stimuli evokes in addicts a cognitive-symbolic response, consisting of subjective craving for the drug, followed by a behavioural response, consisting of drug-seeking behaviour. These responses, conceptually, have developed by classical conditioning [4–6]. This conditioning model implies that a series of extinction trials, consisting of non-reinforced exposure to substance use cues, would be helpful in reducing later craving and relapse when the alcoholic is confronted with alcohol cues. An analogous treatment design had been successfully applied to the treatment of anxiety, obsessive–compulsive and phobic disorders. For cue-exposure treatment in alcohol dependence, two important pilot studies have been conducted by Monti et al. in 1993 [7] and Drummond and Glautier in 1994 [8], who presented patients with their favourite alcoholic beverage for approximately 40 minutes on 10 experimental sessions, giving the instruction to look at it, smell it, think about it, but not drink it. Compared with a standard alcohol treatment, this cue-exposure approach resulted in a higher incidence of abstinence and a higher percentage of abstinent days during the 3–6 months posttreatment monitoring. No difference was found in latency to first drink. In summary, the cue-exposure studies in alcohol dependence conducted hitherto [3, 6, 9] have been promising in several aspects, but much of the research in this area has yet to be carried out, especially considering psychobiological aspects of this matter.
During the last decade, research has increasingly attended to the neurobiological basis of alcohol craving and especially to the brain regions involved. Animal experiments showed that signals for reward, whether cocaine [10], alcohol [11] or food [12], reliably activate dopamine-innervated limbic brain regions. Animal models of addiction consistently implicate key brain structures of the mesocorticolimbic dopamine system, such as the nucleus accumbens, amygdala, septum and also of the ventral striatum [13]. The nucleus accumbens and the amygdala also seem to be involved in opioid peptides and glutamate actions mediating alcohol reinforcement [11].
In recent years, five functional magnetic resonance imaging (fMRI) studies on alcohol craving elicited in alcohol-dependent patients by alcohol-associated cues have been published. In the investigations of George et al. [14, 15], alcoholic patients viewed in the scanner a series of visual pictures, grouped in 24-second blocks of five pictures, each displayed for approximately 5 seconds. Six blocks with alcoholic beverages respectively with neutral content were randomly presented while MRI scans were performed. In response to the alcohol stimulus and compared with the other stimuli, increased activity was found in the nucleus accumbens, cingulate, insula, dorsolateral prefrontal cortex and anterior thalamus. Using a similar visual cue-exposure protocol, Heinz and colleagues found activation in response to the alcohol-associated cues in putamen, anterior cingulate and adjacent medial prefrontal cortex [16, 17]. Apparently, in these studies brain regions were activated during cue-elicited alcohol craving, which are involved in brain reward mechanisms – analogue to the findings in animal experiments – and also in cognitive (memory and attention) processing and emotion regulation. Using an olfactory cue-exposure approach, Schneider et al. observed in alcoholic patients activation in the amygdala/hippocampal region and cerebellum during the alcohol condition and compared with a control condition [18]. According to the authors, the activated regions indicate involvement of emotion and memory processes during cue-induced alcohol craving.
A neuroimaging study on alcohol craving using singlephoton emission computed tomography (SPECT) has been performed by Modell and Mountz [19]. Alcoholic patients were presented during 6 minutes with a drinking glass containing their favourite alcoholic beverage and during another 6minutes with a glass of tap water (control condition). During stimulus presentation, the radiotracer was incorporated into the brain and thereafter patients rated their craving intensity. Compared to the control condition, the alcoholic patients showed significantly increased cerebral blood flow (CBF) in the head of the right caudate nucleus during the alcohol craving condition.
Discussing the discrepant findings between the SPECT and the fMRI studies on cue-elicited alcohol craving, one has to consider that Modell and Mountz analysed the SPECT scans only in three regions of interest (orbitofrontal cortex, head of the caudate nucleus and thalamus), according to the research hypothesis of the authors. However, the fMRI scans were analysed using statistical parametric mapping, avoiding restriction on particular brain regions. Furthermore, because of the technique of image acquisition, the protocol of cue exposure differed substantially. Presentation of stimuli by multiple blocks, each of approximately 20 seconds, in the fMRI studies and by two single epochs of 6minutes each in the SPECT study might possibly result in varied psychodynamics of elicited craving experience. In the SPECT study, a traditional cue-exposure design was used which had been established in numerous studies, investigating selfreports of craving and various vegetative psychophysiological parameters in response to alcohol-related cues [5, 20]. From these studies useful aspects concerning development and maintenance of alcohol dependence, of relapse mechanisms and possible therapeutic strategies have evolved which merit further neurobiological investigations including functional neuroimaging [4, 21].
Using the traditional cue-exposure design, we studied cue-elicited alcohol craving administering positron emission tomography (PET). In 21 detoxified alcoholic patients while looking at and smelling their favourite alcoholic beverage, regional CBF was measured using radioactively labelled H2 15O as tracer. Whole brain PET scans were analysed by statistical parametric mapping, avoiding restriction on particular brain regions.
Method
Patients
Twenty-one male patients, who met the diagnostic criteria for alcohol dependence according to ICD-10 and DSM-IV, participated in the study. Exclusion criteria included a history of neurological disease or trauma, major psychiatric disorders, substantial abuse of drugs other than alcohol, or current use of psychoactive drugs. The patients were recruited from the Psychiatric University Clinic in Freiburg and the Psychiatric State Hospital in Emmendingen, where they had been detoxified on a ward. Thereafter they underwent an inpatient rehabilitation program in these clinics. On the day of the PET scan, the patients were abstinent for 15.5±8.0 days (range: 7–35 days). They were free from benzodiazepine or chlormethiazole medication for at least 5 days. Immediately before the scanning session the patients were assessed for alcoholwithdrawal symptoms, using the Clinical Institute Withdrawal Scale for Alcohol (CIWA-A [22]; translated and modified by Stuppaeck et al. [23]), and completed the State–Trait Anxiety Inventory (STAI; scale X1 [24]). The study was approved by the institutional review board of the University of Freiburg School of Medicine, and written informed consent was obtained from all patients. Exposing patients with dependence to situations that may result in relapse was, from the view of ethics, an particularly critical point of our experimental design. The ethics committee agreed, because all patients underwent an inpatient treatment program and were to be controlled for possible relapse risks after participating in the experimental session.
Apparatus and measurements
Image registration and analysis were performed according to previously defined standards [25, 26]. In brief, the patients were transferred to the scanner (Siemens CTI ECAT EXACT tomograph, 10.8 cm FOV, 6.8mmfull width at half maximum, FWHM), where the patients' heads were positioned according to the orbitomeatal line. Exposure of alcoholrelated and control cues is shown in Fig. 1. An experimental design was used as established in numerous studies on alcohol craving assessing psychophysiological measures [5, 27]. The first 20minutes were provided to the patient as an adaptation phase, during which the patient could become familiar with the lab setting and was asked to relax. The patient was then twice presented mineral water and thereafter twice his favourite alcoholic beverage. For the alcohol condition, for example, a tray with a full manufacturer's bottle of the patient's favourite alcoholic beverage together with a matching drinking glass was put in front of the patient's face, the glass was filled, a sponge was dipped into the beverage and was placed immediately before the nose of the patient. Then a bolus of 27 mCi (1000 MBq) H2
15O was applied i.v. and a dynamic PET scan was acquired in 2D mode. Each PET acquisition was started when the rate of true counts exceeded that of random counts and was continued for 240 seconds (12 frames per 60 s). The patient had been instructed to look at the items on the tray and to smell the sponge while imagining drinking the beverage for the whole duration of the scan. At the end of each recording, the patient had to rate his desire to drink the beverage presented. The interscan intervals were 16 minutes. To obtain the craving score, a visual analogue scale was used ranging from 0 (‘not at all’) to 10 (‘extremely’). At the end of the last recording, the Profile of Mood States (POMS) was administered additionally. A transmission scan with a 68Ge source was performed to correct for attenuation. Images were reconstructed using filtered back-projection by Shepp–Logan filter (cut-off 0.35 cycles/pixel). The dynamic frames were then corrected for motion artefacts and summed up to generate a single dataset of 31 transaxial planes.
Diagram of the cue-elicited alcohol craving paradigm administering positron emission tomography (PET) imaging. Sequence of stimuli presentation, PET scans and craving ratings. Exposure to the control beverage and alcoholic beverage for 4 minutes each. At exposure begin i.v. application of a H2
15O bolus followed by PET acquisition, with rating of the craving score carried out upon exposure end.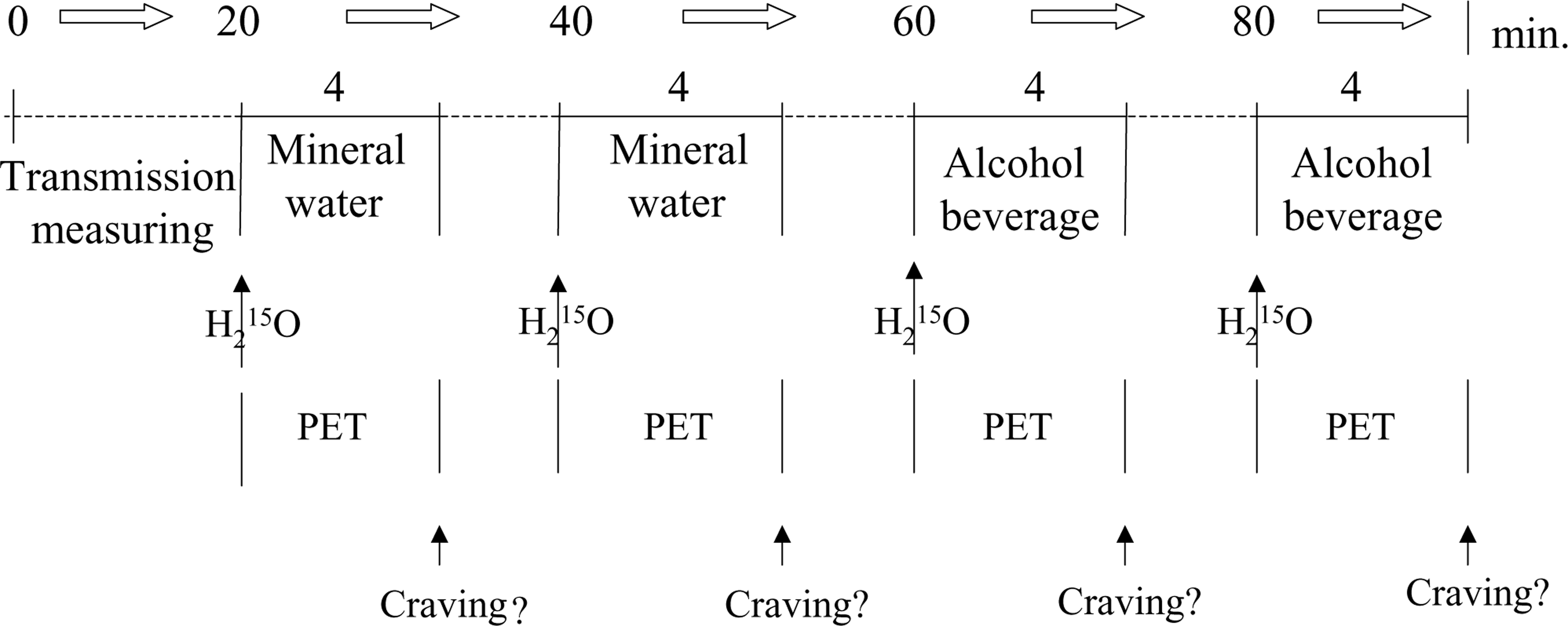
Data analysis
For analysis of the PET images, statistical parametric mapping was used (SPM99, www.fil.ion.ucl.ac.uk/spm). Individual PET scans were realigned using the SPM99 realignment algorithm, with the first scan as a reference in order to correct for head movements between scans. Images were spatially normalized into a standard space using the Montreal Neurological Institute (MNI) template. Before voxel-based statistical analysis, images were smoothed using an isotropic Gaussian kernel of 10mm FWHM. The condition and subject (block) effects were estimated according to the general linear model at each voxel [28]. An analysis of covariance (ancova) was performed. Global flow (calculated as global mean counts) was entered as a confounding covariate before global normalization to 50mL/100 mL/min. To test the hypotheses regarding regionally specific condition effects, the estimates were compared using linear compounds or contrasts. The resulting sets of voxels for each contrast constitute a statistical parametric map of the t-statistic SPM {T}. The SPM {T} were thresholded in terms of spatial extent using distributional approximations from the theory of Gaussian fields [28] and were transformed to the unit normal distribution (SPM {Z}). Additionally, an ancova using the craving score as a covariate of interest was performed for conjunction analyses to determine which brain areas showed changes that covaried with craving. As we hypothesized the dopaminergic brain reward system located in the accumbens nucleus to be activated, a small volume correction was performed for the voxel cluster centred at x=±14, y=6, z=−12 (10mm spherical radius). Only those voxel clusters were kept which exceeded t-values corresponding to p<0.05 (corrected, using the small volume correction algorithm). For additional visualization, the z-score voxel clusters exceeding t-values corresponding to p<0.001 (uncorrected) were projected onto the standard MRI template provided by SPM99, using the SPM projection routine. For display of the Talairach coordinates allowing anatomic identification, a scaling factor was calculated for a nonlinear transformation of the MNI space to Talairach coordinate space [29], using the appropriate algorithm (http://www.mrccbu.cam.ac.uk/Imaging/mnispace.html).
Results
Demographics and subjective ratings
Demographics and subjective ratings of the patients are shown in Table 1. Rated immediately before the scanning session, their mean score on the CIWA-A was 15.9±3.9 points (range: 10–28 points). The possible total scores of this scale range from 10 to 60 and a significant alcohol withdrawal is usually rated greater than 25 points. Only one of the patients exceeded this value. On the STAI, the patients showed for the subscale State Anxiety, on average, a score of 44.1±4.6, not substantially deviating from the basic score of healthy male patients.
Demographics and subjective ratings of alcohol patients studied (n=21)
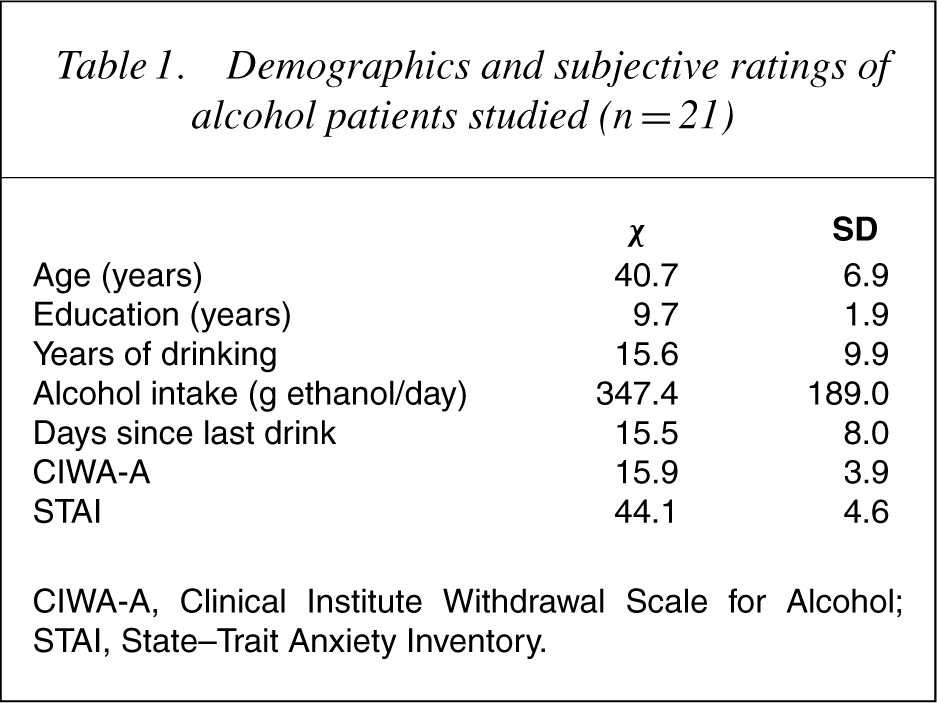
CIWA-A, Clinical Institute Withdrawal Scale for Alcohol; STAI, State–Trait Anxiety Inventory.
Craving indices
Figure 2 shows the craving scores of the individual patients obtained following exposure to mineral water and to an alcoholic beverage. Each score is the average across the both trials presenting neutral respectively alcohol cues. On average, the score for the alcohol condition (8.06) was significantly greater than the score for the neutral condition (3.86). On testing using the Wilcoxon signed rank test, the difference proved to be significant (z=−3.923, p<0.001).
Self-reports of craving induced by alcohol-related and control cues. Self-reported urges of the individual 21 alcoholic patients to drink the exposed beverage, assessed on a 10-point visual analogue scale. Note: Increased mean craving score (horizontal bar) for alcohol condition compared with control condition.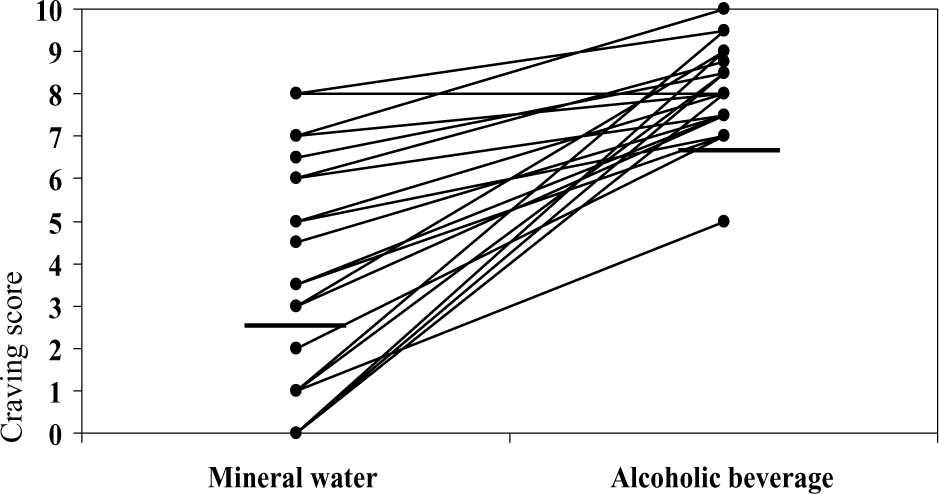
Brain activity analysis
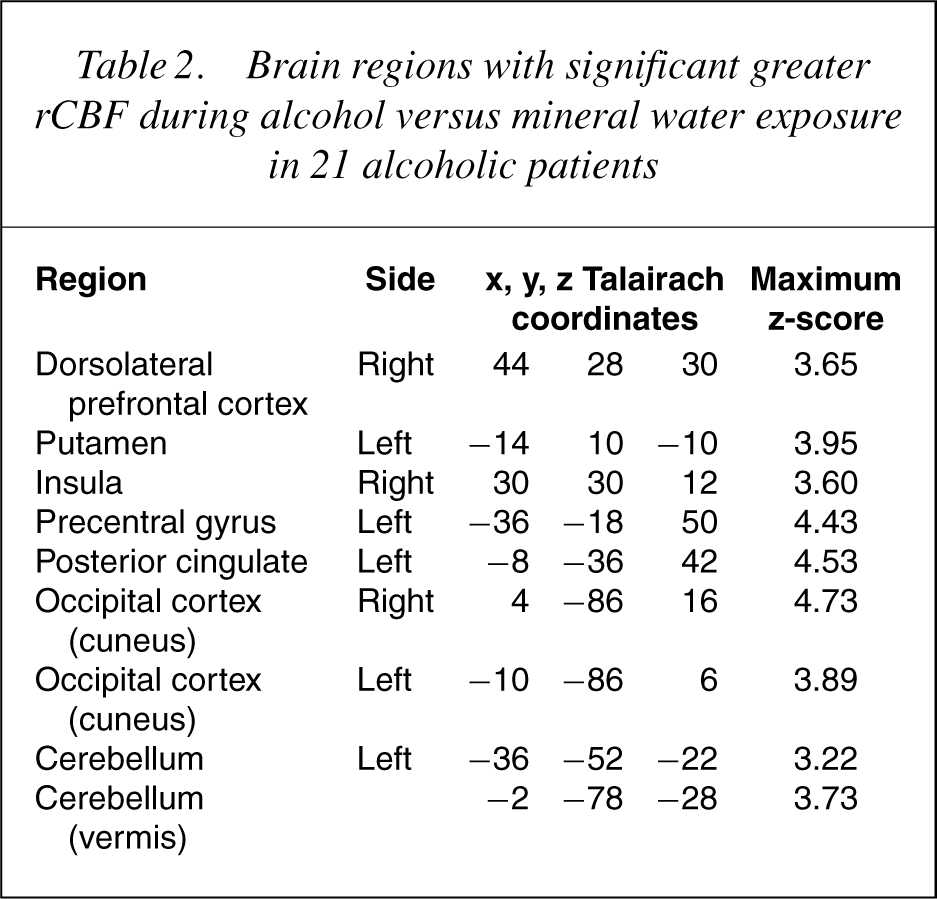
Comparing alcohol exposure with mineral water exposure, brain areas were identified with statistically significant (p<0.001, uncorrected) differences in rCBF. Seven brain regions had significantly greater rCBF during alcohol than during mineral water condition: right insula, left precentral and postcentral gyrus, left posterior cingulate gyrus, right occipital cortex (cuneus), left cerebellar hemisphere and cerebellar vermis. Performing conjunction analysis for rCBF difference between the alcohol and mineral water condition with craving score as covariate, a significant CBF increase was no longer found for the postcentral gyrus. But, in addition to the other brain regions mentioned earlier, a significant increase was found in the left occipital cortex (cuneus), right dorsolateral prefrontal cortex and left ventral putamen (Fig. 3). Following the a-priori hypothesis of ventral putamen to be activated, small volume correction for this region was performed; resulting p-values for this region, on the left side, were <0.02 (corrected). The Talairach space localizations of the centres of significant voxel clusters and the maximum z-scores are each summarized in Table 2.
Brain areas with increased activation comparing alcohol with control condition. Voxel clusters of areas with significant rCBF increase during alcohol exposure compared with mineral water exposure projected onto surface-rendered standard 3D MRI in 21 alcoholic patients. CE, cerebellum; DLPFC, dorsolateral prefrontal cortex; IN, insula; OC, occipital cortex; PC, posterior cingulate; PU, putamen.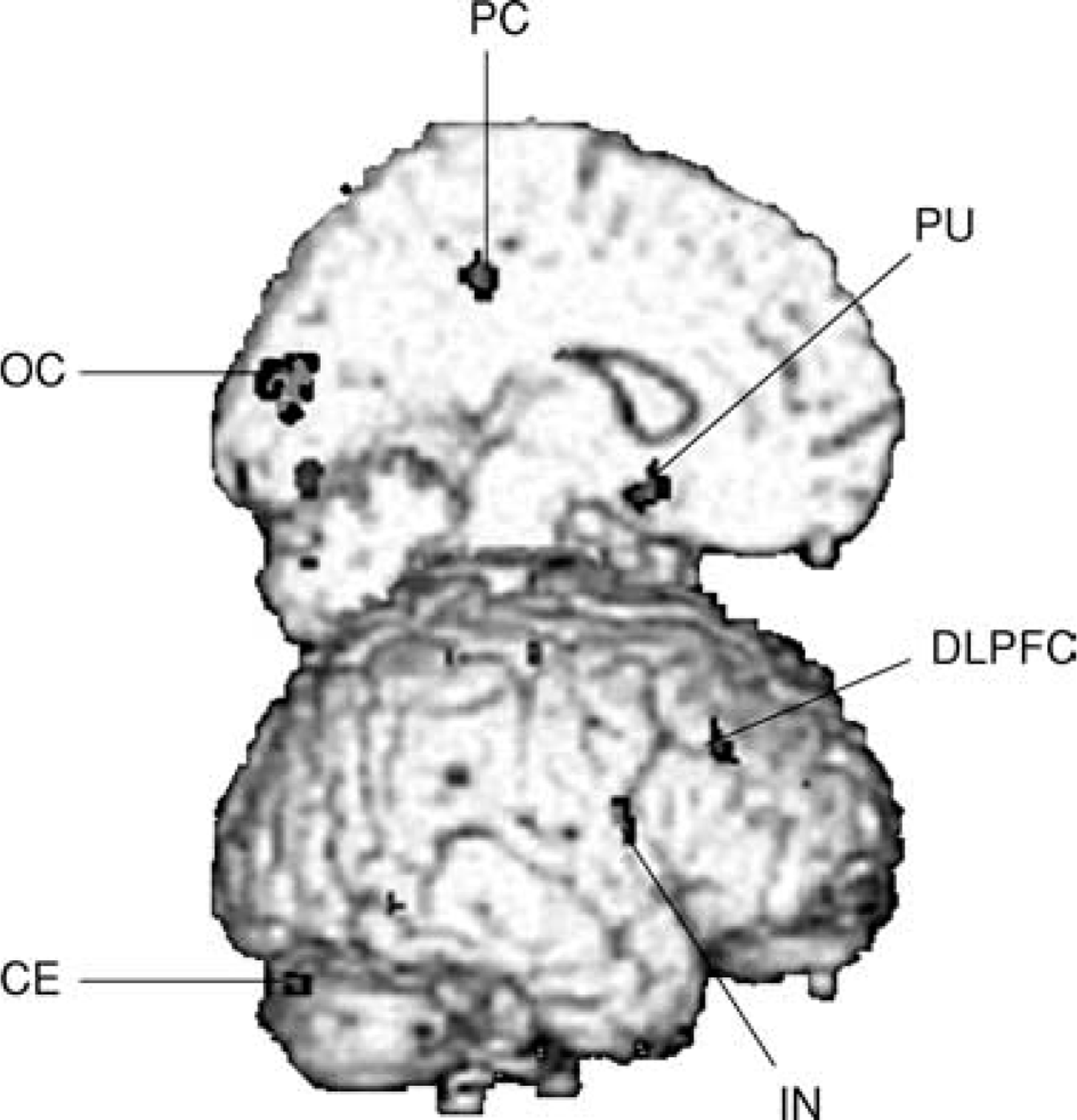
Brain regions with significant greater rCBF during alcohol versus mineral water exposure in 21 alcoholic patients
No brain regions were found with significant rCBF decrements during alcohol exposure compared with mineral water exposure.
Discussion
This is to our knowledge the first PET study on cueinduced
alcohol craving following five fMRI studies on this issue. During exposure to alcohol cues and compared with neutral cues, we found in alcoholic patients a robust activation of the ventral striatum, associated with the brain reward system and, at a lower level of significance, activation in brain regions associated with memory and attention circuits. The CBF increase in these brain regions varied directly with the intensity of alcohol craving. Our findings are consistent with those obtained in the fMRI studies; a result not at all self-evident considering that our cue-exposure protocol, taken from the classical cuereactivity experiments in addiction research, substantially differed from that used in the fMRI investigations.
Involvement of the brain reward system was reflected by craving-associated activation of the ventral striatum (putamen) and, at a lower level of significance, the insula. In primates, the ventral striatal domains of the caudateputamen are the equivalent of the nucleus accumbens in rodents, which has been elaborated as a key structure of the mesolimbic dopamine system implicated in the brain reward circuitry [13, 30, 31]. Signals for rewards such as food, sex and drugs of abuse activate the ventral striatum and this structure reliably contributes to reinforcement of drug-seeking and drug consumption [11, 32]. The insula is considered as a part of the limbic system involved in brain reward processing by connections with the nucleus accumbens [33] and with the orbitofrontal cortex [34], which has recently been discussed as an essential structure of the neural reward circuitry [35]. In the fMRI studies on alcohol craving, activation of main targets of the brain reward circuitry including ventral striatum [16] and insula [15] and furthermore of nucleus accumbens, amygdala/hippocampus and anterior cingulate [15, 17, 18] has been found.
In our alcoholic patients, we infer involvement of memory circuits during craving from activation of the dorsolateral prefrontal cortex and cerebellum. By the cue-exposure approach used in our study to elicit craving, obviously some types of memory processes were involved. Activation of the dorsolateral prefrontal cortex has consistently been shown in animal experiments and in humans during spatial and non-spatial memory tasks and working memory operations [36]. Traditionally, the cerebellum has been viewed solely as a mediator of motor functions, while recent studies have shown implication in memory processes. Cerebellar activation has been observed during performance of cognitive tasks that require explicit, episodic memory while motor activity was controlled [37, 38]. Anatomical pathways connecting cerebellum and dorsolateral prefrontal cortex seem to be involved in such cognitive tasks [39]. Increased activity in both brain regions has also been found in the fMRI studies on alcohol craving [14, 18]. Functions of the cerebellum beyond its classically described activity in motor control have also been reported by Denton and his group working on the neural correlates of thirst. They showed that thirst-associated cerebellar activation was mainly related to vegetative aspects of thirst and to sensory and cognitive aspects, respectively (for references see below).
Activation of dorsolateral prefrontal cortex furthermore indicates involvement in attention processes [40]. We have shown by means of electrophysiological correlates of information processing that in chronic alcoholic patients the types of cognitive processes discussed here are altered in a particular manner [41, 42]. Activation of the posterior cingular region and the visual cortex (cuneus, mainly Brodman area 18) during exposure to alcoholic cues has also been observed in the fMRI studies on alcohol craving described earlier.
Concerning the principal results, our findings are consistent not only with the fMRI studies on cue-elicited alcohol craving but also with functional neuroimaging (PET and fMRI) studies performed in the 1990s on cueinduced cocaine craving. In cocaine addicts, activation during cocaine craving was found in main targets of the brain reward circuitry as ventral striatum, amygdala, anterior cingulate, insula and orbitofrontal cortex. Furthermore, targets of memory and attention circuits as dorsolateral prefrontal cortex, mediotemporal lobe, cerebellum and anterior cingulate were involved [43, 44], for review: [45, 46].
There are remarkable relationships between our work and functional neuroimaging findings on thirst. Denton et al. generated thirst in humans by infusion of i.v. hypertonic saline 0.51M. The patients were neuroimaged by PET and fMRI, when a subjective sense of thirst occurred [47–49]. Remarkably, several brain areas activated in our study during cue-elicited alcohol craving overlap with the brain regions found to be activated during subjective thirst experience, namely the posterior cingulate, the cuneus region, insula, cerebellum and dorsolateral prefrontal cortex. Was thirst a confounding variable not controlled in our study, as was generally the case in the fMRI studies on alcohol craving as yet performed? Concerning our investigation, the alcoholics might have experienced, beside the rated self-report of alcohol craving, a subjective sense of ‘basal’ thirst in response to the neutral and/or alcoholic beverage presented. Yet we identified only those brain areas as possible neural substrates of alcohol craving which showed greater rCBF during alcohol than during control condition, the rCBF changes in addition having to co-vary with the alcohol craving score (ancova testing). Moreover, the brain areas activated during cue-elicited cocaine craving, which were mentioned earlier, overlap, as in alcohol cravingwith several brain regions activated during thirst. Generally, the videos used to induce cocaine craving did not present objects best-suited for triggering thirst as, for example, beverages might do [45]. Thus, the possible influence of ‘basal’ thirst on findings obtained in cue-elicited alcohol craving studies could be assessed definitively if thirst sensation were also rated.
There are potential limitations to the interpretation of our results. Because no control patients were studied,1 it is possible that differences in the physical stimulus properties of the alcohol condition compared with the neutral condition contributed to some portion of the rCBF changes. This especially concerns the odoriferous features of the alcohol cues. In one of the five fMRI studies on cue-elicited alcohol cravingmentioned earlier, alcohol odour, compared with room air, was used as the experimental cue. Activation of the superior temporal cortex was found to be the neural correlate of the olfactory nature of the cues [18]. We did not observe activation of this region during alcohol condition compared with control condition. In contrast to the fMRI study, our alcohol cue varied considerably in its olfactory stimulation parameters. Furthermore, rapid habituation to olfactory stimulation has been shown to occur already after 30–40 seconds [50], whereas our beverage exposition and PET acquisition lasted for 240 seconds. Furthermore, because we intended to avoid a cross-over effect, we did not use a counterbalanced sequential exposure to the cues. Rather, the neutral was always presented before the alcohol condition using an established approach in cue-reactivity studies on alcohol or other substance abuse [4]. In a review of those studies, Carter and Tiffany reported that a counterbalanced cue order was used only in 26% of the alcoholics studies, 11% of the heroin addicts and 25% of the cocaine addicts studies [5]. Admittedly, without randomization of both experimental conditions, an undetected order effect might have influenced our findings. Finally, stress, anxiety and other kinds of emotions could have contributed to the observed rCBF changes with the proviso that they had systematically varied between the two experimental conditions. The only psychological variable we monitored across conditions was craving intensity, and we speculated that subjecting this variable to conjunction analysis might diminish the influence of the other psychological variables mentioned.
In sum, a consistent pattern of brain regions activated during cue-elicited craving was found across our PET and the reported fMRI studies on alcohol craving and the studies on cocaine craving. The functional neuroimaging 1 approach used could offer an adequate strategy for studying the brain substrates of drug desire, a hallmark of drug dependence disorders. Using our PET approach, relevant subsequent investigations could be to study alcoholic patients before and after detoxification or to follow-up detoxified patients neuroimaged by PET with respect to relapse and abstinence, or to compare alcoholic patients with other substance abusers. Research on this field could result in more detailed knowledge concerning important issues of addiction research including development and maintenance of alcoholism and mechanisms of relapse. Useful therapeutic designs and strategies could result from that.
Footnotes
Acknowledgements
This research was supported by grant 01 EB 9804/6 to Olbrich from the Ministry of Education and Research, Federal Republic of Germany.
1We intended to include healthy control patients in the study. Yet, and not unusually in Germany, the University Review Board, following the instructions of local government, does not allow PET studies on healthy patients. Exceptions are made for studies in elderly patients.
