Abstract
Atypical antipsychotic medications have replaced conventional medications as the first-line pharmacological treatment for psychotic illness in Australia. Olanzapine, an atypical antipsychotic, is now the most commonly prescribed of these medications. Between July and December 2001, prescriptions for olanzapine accounted for 65% of total prescriptions for atypical antipsychotic medications [1].
The popularity of olanzapine has emerged due to its tolerability and equal or greater efficacycompared to typical antipsychotics [2], [3]. Although many of the adverse side-effects commonly associated with typical antipsychotics are reduced, weight gain can be pronounced while on atypical antipsychotics. Previous studies have shown that weight gain in patients taking olanzapine is greater than with most other antipsychotics, second only to clozapine [4].
Up to 94% of people taking olanzapine gain weight [5], with approximately 40% of individuals gaining more than 7% of their baseline weight [6]. Based on a meta-analysis of 78 studies, the degree of weight gain appears to be an average of 4.15 kg during 10 weeks of olanzapine treatment [4]. More recently, a mean weight gain at 12 weeks of 7.3 kg has been reported [7]. The majority of weight gain occursin the first 3 months following commencement of olanzapine [8], highlighting the importance of early intervention for weight gain mitigation. Weight gain tends to plateau between 36 and 39 weeks [8], [9].
People with a mental illness have a 2.9 times greater mortality rate from natural causes compared to the general population [10]. The greatest contributor to this rate is cardiovascular disease. People with a mental illness have a higher prevalence of risk factors for cardiovascular disease and other comorbidities such as smoking, overweight and obesity, high alcohol consumption, poor diet, lack of exercise, diabetes, dyslipidaemia and hypertension [11], [12]. Despite this greater risk, this population is less likely to have their physical illnesses diagnosed and managed effectively [11], [12]. Some atypical antipsychotic medications can further exacerbate these factors. Olanzapine is not only associated with increased weight but also with increased rates of diabetes and dyslipidaemia [13–16]. Although the increased risk of diabetes and dyslipidaemia is not fully dependent on weight gain, clearly preventing weight gain in patients with schizophrenia will significantly reduce their cardiovascular risk factors in the domains of blood pressure, susceptibility to diabetes and activity levels.
Psychological consequences of weight gain are also important factors and include a sense of inability to control one's own behaviour, a sense of demoralization, physical discomfort and being the target of social stigma – factors that may already be pronounced in mental illness[17]. In addition, it is well recognized that weight gain secondary to antipsychotic medication reduces medication compliance [18].
In acknowledging that olanzapine is associated with weight gain and that being overweight is associated with chronic disease, recent research has investigated both pharmacologic and behavioural interventions to prevent or treat weight gain associated with antipsychotic therapy.
Reflecting an increasing understanding of the neuroendocrine mechanisms underlying the weight gain associated with atypical antipsychotic medications (described elsewhere), a variety of pharmacological interventions have been trialled in an attempt to reverse or minimize the weight gain [19]. In a double-blind placebo-controlled trial, nizatidine (an H2 antagonist) was found to have an early transient effect in limiting weight gain in patients commencing olanzapine, but this effect appeared to be eliminated by 16 weeks [20]. Following administration of Amantadine, weight losses of 3.5 kg over 6 months and of 2.2 kg over 4 months have been demonstrated in patients who gained over 7.3 and 15 kg, respectively on olanzapine [21], [22]. There is some evidence, however, that amantadine can worsen psychotic symptoms [23]. Similarly, fluoxetine (a selective serotonin re-uptake in-hibitor[SSRI]) was shown to reduce clinical effectiveness of Olanzapine [24], while the use of Sibutramine (an anorectic agent) in the mentally ill population is not recommended because of its interactions with SS-RIs [25]. The weight control potential of sibutramine for people with a mental illness not on SSRIs has not been examined. Only a small number of case reports have discussed the effect of orlistat, a lipase inhibitor, for weight loss in patients on antipsychotic medications [26–28]; however, there are no well-controlled clinical trials examining this approach. At present, there is limited evidence on the effectiveness and safety of current pharmacological interventions for weight control in patients taking antipsychotic medications [29], [30].
Psychosocial interventions that focus on empowering the patient to take control of their diet and activity levels and their health and wellbeing, intuitively offer much more to this group. Several authors have trialled various interventions in an attempt to reverse or prevent the weight gain associated with atypical antipsychotics. Table 1 summarizes some examples of such research.
An overview of previous psychosocial interventions to reverse or prevent weight gain associated with atypical antipsychotics
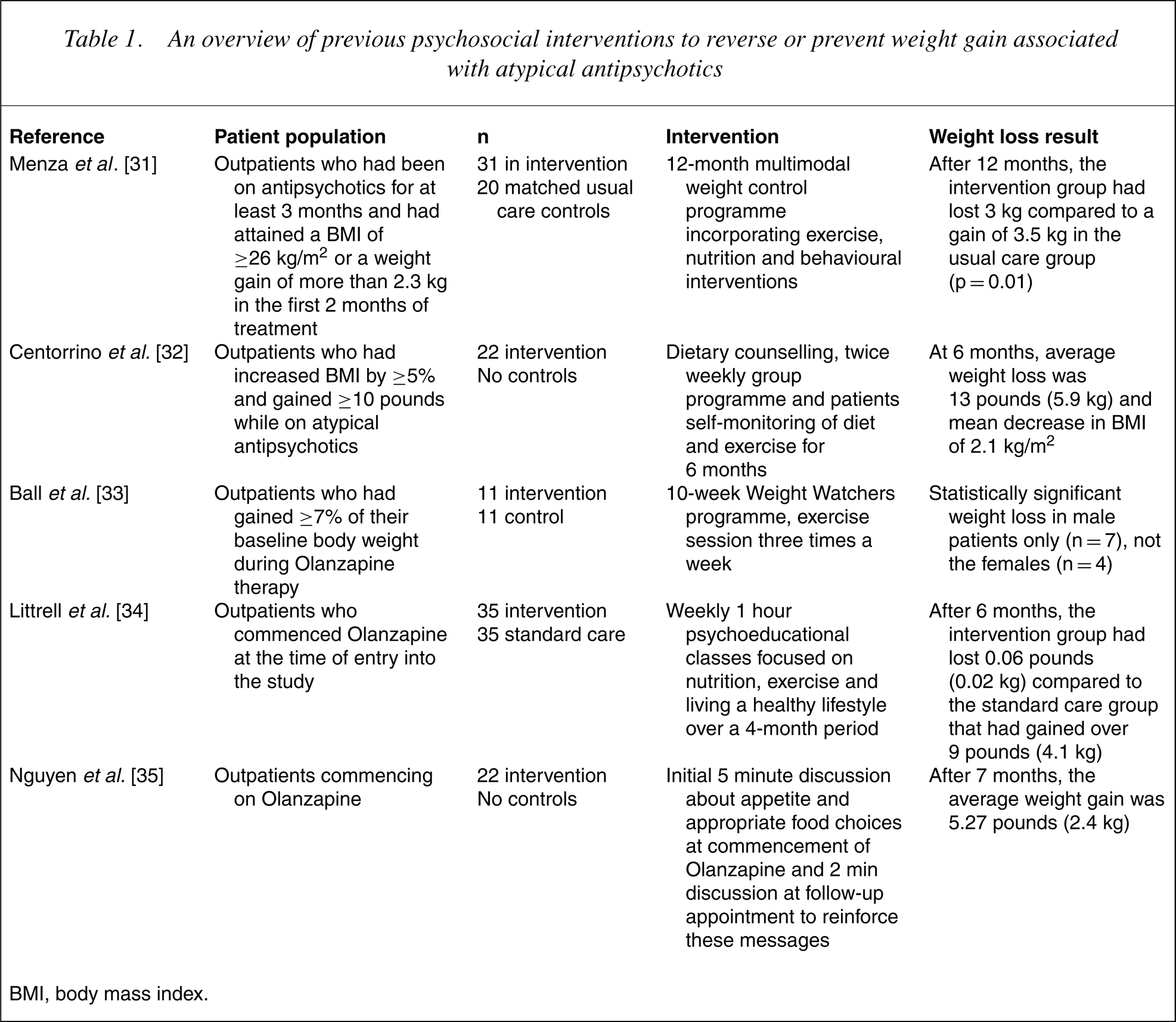
BMI, body mass index.
Although some of these studies have demonstrated success in reversing the weight gain associated with atypical antipsychoticsvia group education, early intervention to prevent the weight gain from ever occurring is preferable. Several studies have been successful in preventing or minimizing the weight gain from the commencement of the medications; however, these have either been via group education or were not randomized controlled trials.
In summary, while there is some evidence that nutrition education can be effective in reversing and preventing weight gain in individuals on olanzapine, there are no randomized controlled trials investigating whether weight gain associated with olanzapine can be prevented via individual one-on-one nutrition education.
The purpose of this randomized controlled study was to determine if the weight gain associated with olanzapine could be minimized or prevented in the 3 and 6 months after its commencement via individual nutrition education provided by a dietitian. It was hypothesized that individuals participating in a structured individual nutrition education program would gain less weight than those receiving standard psychiatric care. The study also aimed to determine the effect of individual nutrition education on quality of life, body image, overall health and activity levels.
Method
Subjects
Subjects were recruited via community and inpatient settings associated with the Peninsula Health Psychiatric Services Frankston Hospital, Victoria, Australia. All participants were outpatients when commencing the study. The study was approved by the Peninsula Health Research and Ethics Committee. Written informed consent was obtained from all participants after they received a complete explanation of the study.
All individuals had commenced olanzapine within the 12 weeks before entering the study. Both first-episode and previously diagnosed subjects were recruited. Those who had conditions likely to significantly affect metabolism(e.g. diabetes, hypo-or hyperthyroidism), a body mass index (BMI) of more than 35, currently or previously diagnosed with an eating disorder or clinically significant alcohol/substance abuse, were excluded.
Study design
The participants were randomly assigned to either the nutrition intervention or control group. All the participants in the intervention and control groups received passive nutritional education from the booklet Food for the Mind, a publication produced by the manufacturers of Zyprexa. At the time of recruitment, the manufacturers were recommending the distribution ofthis booklet to all patients commenced on Olanzapine.
The intervention group received six one-on-one individual nutrition education sessions over a 3-month period. The sessions were conducted by an accredited practicing dietitian on a fortnightly basis and were 1 hour in duration. The content of the sessions included discussions between the participant and the dietitian on healthy eating, exercise, label reading, energy density, high fibre diets, non-hungry eating and maintenance of healthy eating and changes in activity levels. One of the primary focuses of each week was the setting of healthy eating and lifestyle goals, the progression of which was reviewed and revised on a fortnightly basis. The sessions were carried out by a pool of dietitians working in the Peninsula Health Network.
Weight, waist circumferenceand BMI were assessed at baseline and at 3 months (end of the nutritional intervention) and 6 months. At 3 months, the subjects were also asked to complete self-report scales based on the Clinical Global Impressions (CGI) to assess their subjective view of their own quality of life, health, body image and assessment of their activity levels [36]. Neither group received any intervention between the 3-and 6-month data collection.
Statistical analysis
All subjects randomized were included in the initial analysis, but only data from people remaining in the study were included in the 3-and 6-month analyses. All withdrawals from the control group were assessed only at baseline and therefore to carry forward the baseline data from this group would have made it impossible to identify treatment effects. Independent sample t-tests were used to assess differences between intervention and control groups. All data were analysed using SPSS 11.0 statistical software with the advice of a statistician where appropriate.
Results
Ninety patients were referred for involvement in the study. Thirtynine were not included due to being non-contactable (n = 18), declining to participate (n = 13), unable to travel to sessions (n = 4) or being too unwell as deemed by the psychiatrist (n = 4). Fifty-one patients were enrolled in the study (22 males, 29 females). The nutritional intervention group consisted of 29 participants (11 males, 18 females) and the control group 22 participants (11 males, 11 females).
Table 2 describes the baseline characteristics of the participants. As seen in Table 2, before intervention there were no significant differences between the control and intervention group in clinical characteristics, anthropometric values and psychometric rating scales scores.
Baseline characteristics of 51 participants randomly assigned to either intervention or control group
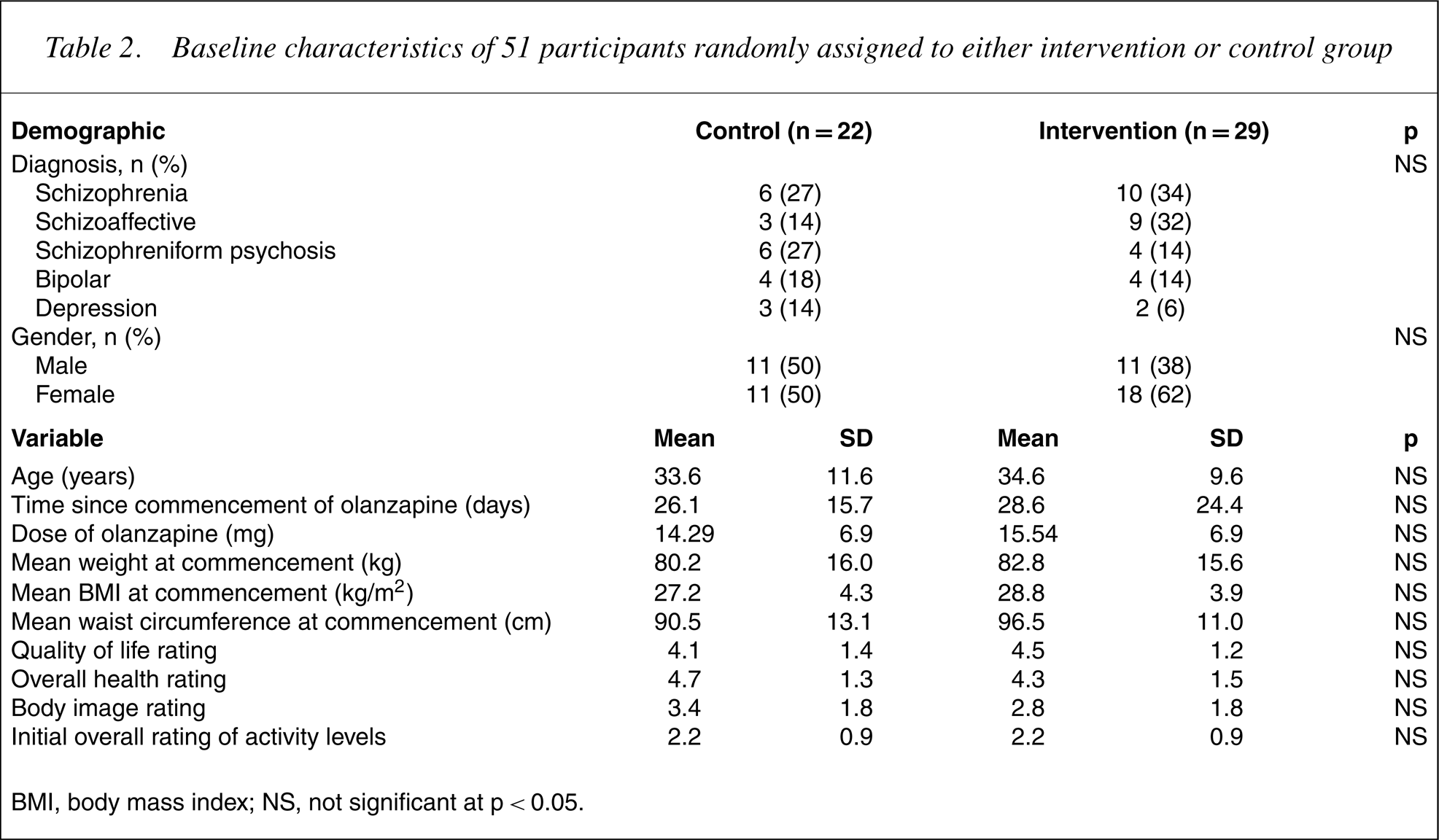
BMI, body mass index; NS, not significant at p<0.05.
Seventeen participants were withdrawn from the study before the 3-month follow-up. Eleven of these withdrawals (65%) were from the control group and were withdrawn due to ceasing or non-compliance with olanzapine (n = 6), or due to being non-contactable (n = 5). The six withdrawals from the intervention group were due to ceasing olanzapine (n = 1), being non-contactable (n = 1), pregnancy (n = 1), moving from the area (n = 1) and not wanting to participate (n = 2). There were no differences in demographic, clinical characteristics or anthropometric values between the patients who withdrew from the study and those who did not. A significantly greater attrition rate was evident in the control group, with 50% withdrawing compared to only 20% of the intervention group (p = 0.08). After withdrawals had been removed from the dataset, there were still no significant differences at baseline between the control and intervention group in clinical characteristics, 70 anthropometric values and psychometric rating scales scores.
Body weight, BMI and waist circumference changes from base-60 line to 3 months are presented in Table 3. The nutritional intervention group gained significantly less weight (intervention = 2.0 kg, controls = 6.0 kg; p = 0.002), had a significantly smaller change in BMI (intervention = 0.7 kg/m2, controls = 2.0 kg/m2;p = 0.03) and had a significantly smaller change in waist circumference (intervention = 0.4 cm, controls = 5.9 cm; p = 0.001) than the control group.
Three-month data for control and intervention groups
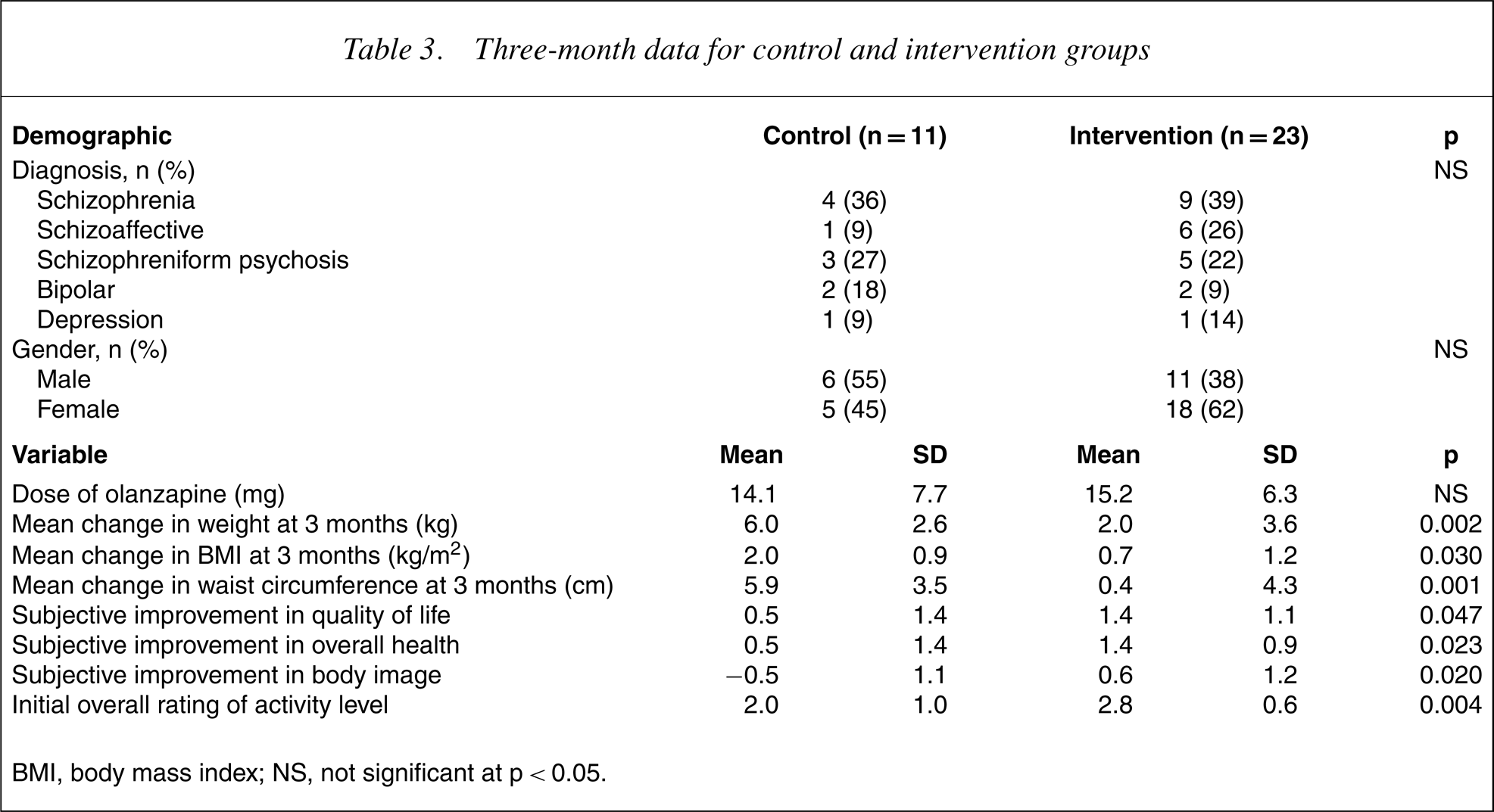
BMI, body mass index; NS, not significant at p<0.05.
Only 13% of the intervention group, compared to 64% of the control group, gained more than 7% of their initial body weight. In addition, 22% of the intervention group actually lost weight, whereas none of the control group lost weight. Figure 1 compares the amount of significant weight gain at 3 months between the intervention and control groups.
Comparison of significant weight gain at 3 months between the intervention and control groups.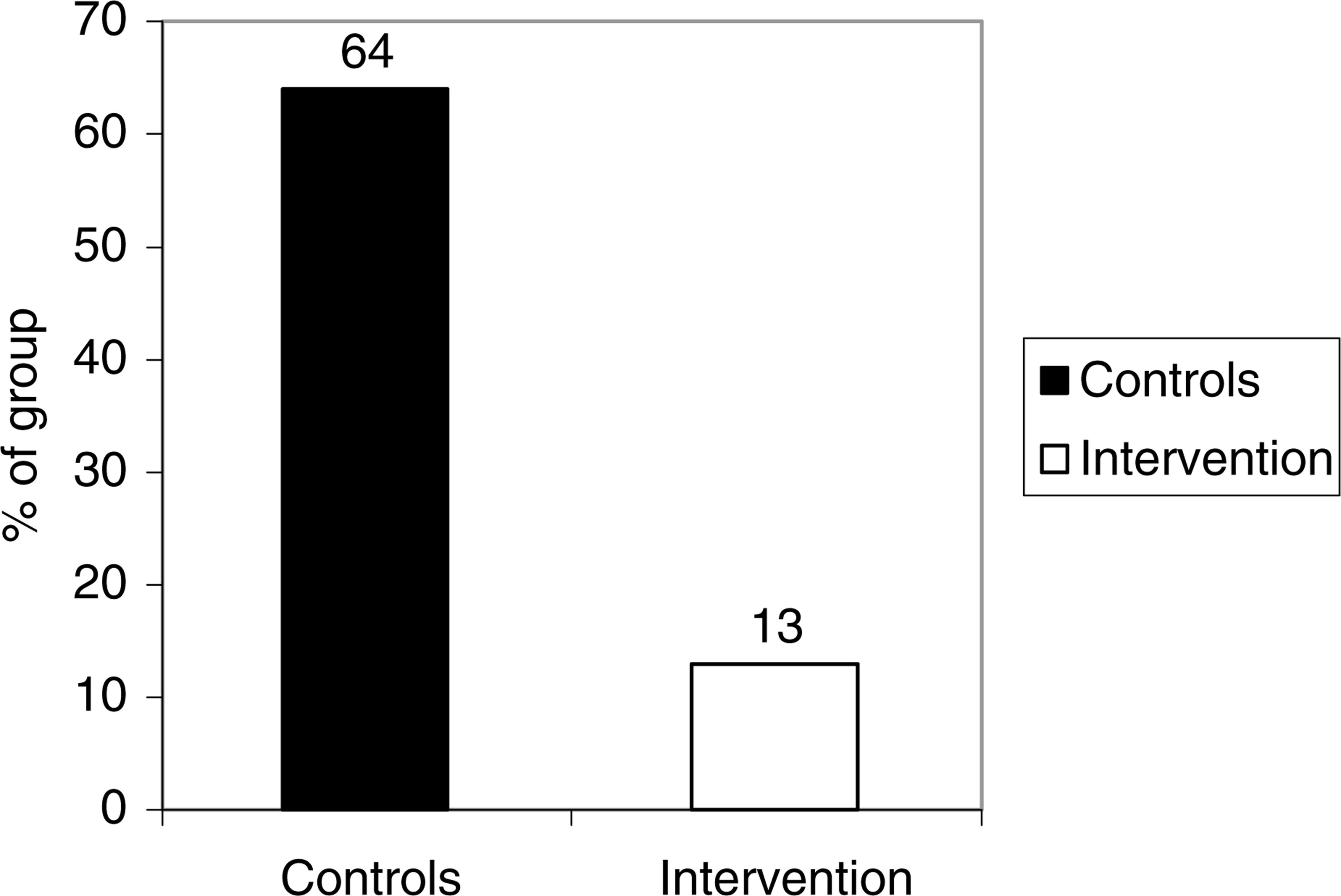
At 3 months, significant differences were seen between the groups in their descriptions of their current activity levels, with the intervention group describing themselves as more active than the controls. In addition, the intervention group reported significantly greater improvements in quality of life, health and body image compared to the controls (see Table 3).
Before 6-month data collection, 12 patients from the intervention group were withdrawn due to being non-contactable (n = 7), not taking olanzapine anymore (n = 4) and unable to come in for assessment (n = 1). Three controls were withdrawn due to being non-contactable (n = 2) and having ceased olanzapine (n = 1). This left 11 in the intervention group and eight in the control group. With these withdrawals removed from the dataset, there were still no significant differences between the control and treatment group in clinical characteristics, baseline weight and BMI values. Psychometric rating scales and waist circumferences were not completed at 6 months.
Body weight and BMI changes from baseline to 6 months are presented in Table 4. As seen in this table, since 3-month data collection the control group had gained on average an additional 3.9 kg, whereas the intervention group had not gained any further weight. After the 6-month period, the intervention group had gained significantly less weight (intervention = 2.0 kg, controls = 9.9 kg; p = 0.013) and had a much smaller change in BMI than the control group (intervention = 0.8 kg/m2, controls = 3.2 kg/m2;p = 0.03). Figure 2 compares the amount of weight gained by the intervention and control groups at 3 and 6 months.
Weight gain comparison in kilograms between intervention and control groups at 3 and 6 months.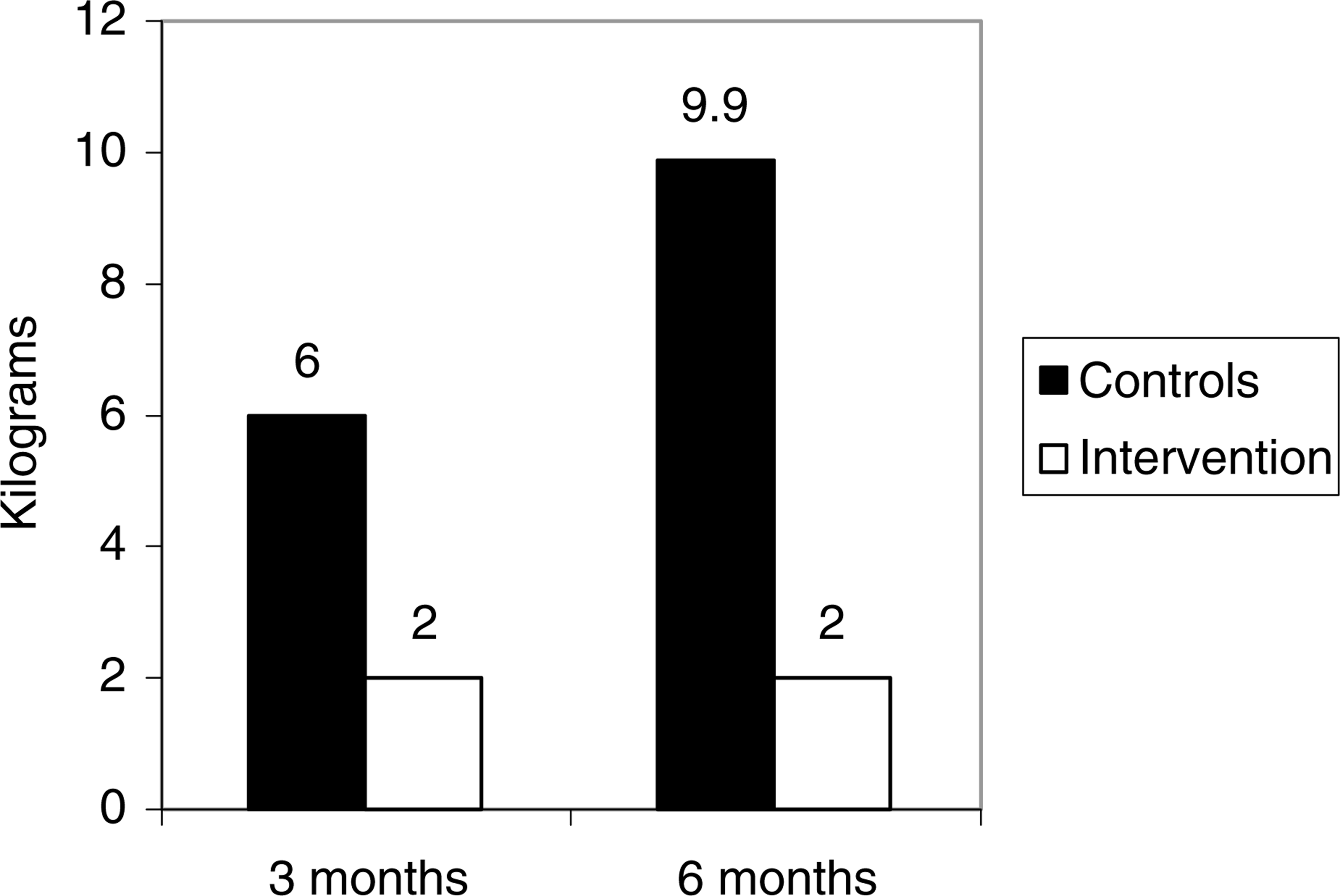
Six-month data for control and intervention group
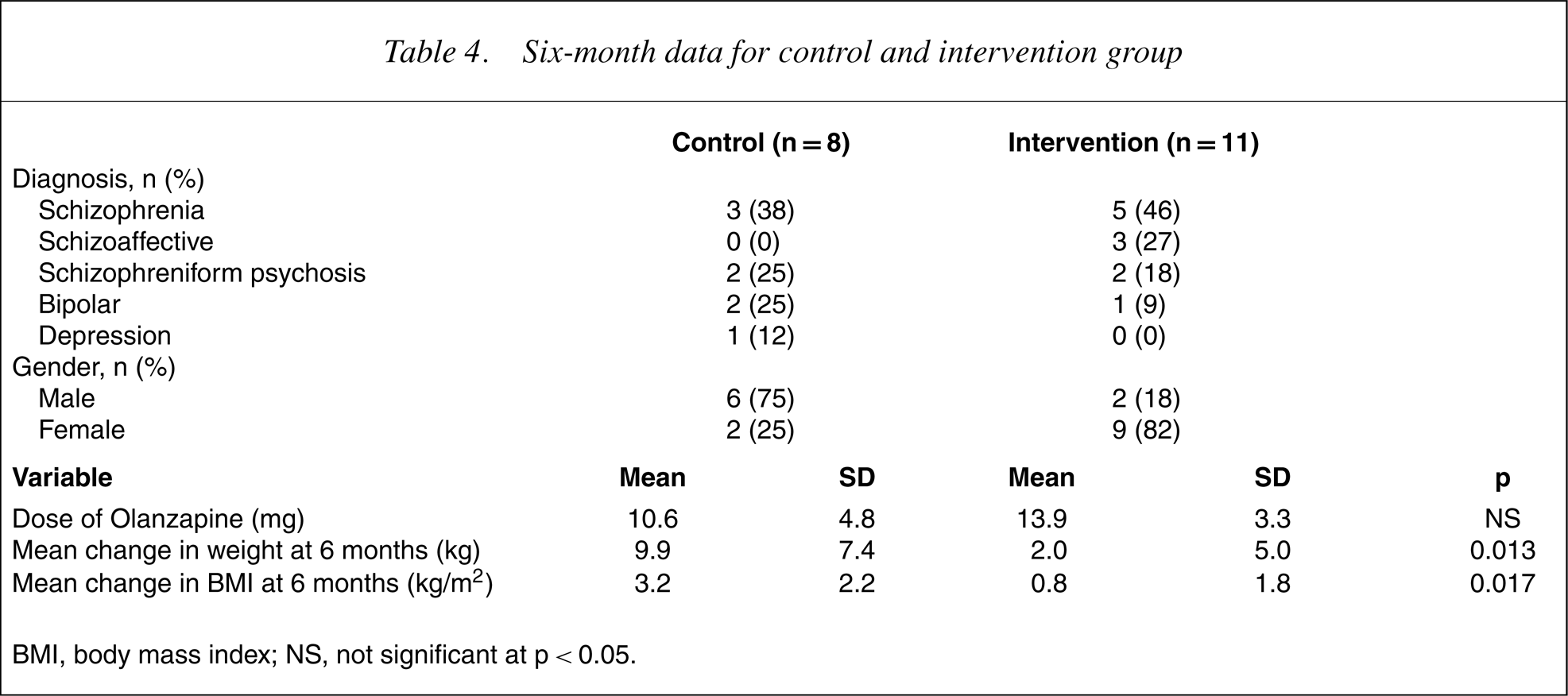
BMI, body mass index; NS, not significant at p<0.05.
Discussion
This randomized controlled study has demonstrated that weight gain following the commencement of olanzapine can be minimized by individual nutrition education provided by a dietitian. It is possible to reduce the percentage of people gaining clinically significant amounts of weight (more than 7% of their initial weight) following the commencement of olanzapine from 64% to 13%. A large body of published work shows a mean weight gain of between approximately 4 and 7.5 kg [4], [7] following 3 months of olanzapine administration. In accordance with these findings, the controls in the present study had gained a mean of 6 kg approximately 4 months after the commencement of olanzapine (i.e. they had been on olanzapine for approximately 1 month before commencing the study) and went on to gain a mean of 10 kg over 7 months. In comparison, the intervention group only gained 2 kg in the 4 months following commencement and then their weight stabilized in the following months. Although not assessed, one would assume that this stabilization would be attributable at least partly to sustaining the lifestyle changes undertaken in the first 3 months. In addition, five of the intervention group lost weight throughout the study period, whereas no controls lost any weight.
The timing of nutrition intervention is extremely important. It is well documented that the most rapid gains occur in the first 3 months of administration [8]. Although several authors have demonstrated some limited ability to reverse weight gain associated with antipsychotic medications mainly via group education [31–33], this is one of the first randomized controlled studies to be published that demonstrates the effectiveness of individual nutrition education to prevent significant weight gain in this critical period. Preventing, rather than treating the weight gain should be the priority and hence weight management strategies should be implemented when initiating olanzapine treatment. Engaging people at this time is likely to be more challenging than when they are more stabilized on their medications and therefore a variety of interventions should be available to ensure a suitable option for each individual.
The clinical importance of preventing weight gain in this population cannot be understated given that the rate of obesity among people with schizophrenia even before starting antipsychotic medication is two to three times greater than the general population [37]. Minimizing weight gain as demonstrated in this study should significantly reduce their cardiovascular risk factors in the domains of blood pressure, susceptibility to diabetes and dyslipidaemia as well as simply reducing their BMI. Although not assessed in this study, the association between weight gain secondary to antipsychotic medication and medication compliance is well documented [18]. Increased compliance is likely to be a significant further benefit of providing appropriate nutritional intervention.
This study has also demonstrated that individual nutrition education led to significantly improved quality of life and general perceptions of health and body image. Demoralization is an important issue for people living with psychoses [38]. One could reasonably assume this would be reduced with this intervention.
The current study does have several limitations. The sample size was relatively small, although the effect size was sufficient to convincingly show a difference between the two groups. The small sample size contributed to some failures in randomization (more females than males in the intervention group), which would be corrected using a larger sample size. The fact that people with diabetes and those who were morbidly obese were excluded from this study limits the generalizability of these findings to this group. Larger studies are therefore needed to explore if anydifferences would emerge among the genders, those who are and are not morbidly obese and those with and without diabetes.
The potential weight gain effect of other medications was not assessed in this study. Given that both firstepisode and established cases of schizophrenia were included, this is particularly pertinent, although one would hope that the randomization procedure distributed such effects equally across both groups. In a replication study it would be important to distinguish between these two groups and to use more objective and well-validated measures of quality of life, health and body image.
Although this study does have some limitations, it also has several strengths. It was a randomized controlled study, with the control treatment reflecting current clinical practice. A pool of dietitians completed the education sessions and therefore it is likely that a real treatment effect was seen rather than the effect of one particular charismatic committed dietitian. The clinical sample used is representative of the range of diagnoses presenting to public mental health services. This relatively unselected sample reflects clinical reality and it is expected that these results would be reflected in clinical effectiveness if applied in everyday clinical settings.
It is vital to be sure that any intervention in the domain of weight or lifestyle change is sustainable in the long-term and is not merely a short-term treatment effect. The current study's 6-month data are suggestive of a robust long-term effect, however, a larger longer-term study, with careful follow-up of all participants, is needed to replicate these findings.
Further studies are also required to investigate the ‘dose’ of intervention required. Although it is difficult to draw comparisons due to a lack of controls, Nguyen et al. [35] demonstrated similar weight gain in their openlabel uncontrolled prevention study that used very brief nutritional interventions [36]. There is certainly scope for a larger study looking at a range of ‘doses’ of nutritional intervention to identify the most cost-effective level of preventative intervention.
The intervention used in this study is easy to replicate in a general practice or adult clinic setting. The highly effective nature of this intervention and the consequences of initiating olanzapine without providing nutritional intervention suggest that all prescriptions of olanzapine should be accompanied by nutritional counselling of a similar nature. It is possible that medicolegal issues may arise in the future if prescribers do not take responsibility for ensuring patients have access to evidence-based preventive weight management programs.
Footnotes
Acknowledgements
This research was funded by an investigator-initiated research grant from Eli Lilly Australia.
