Abstract
In recent years, there has been an increasing interest in the comorbidity of substance use disorders and mental disorders. Substance use disorders are more common in schizophrenia than in the general population, with a lifetime prevalence of 40–50% in both Australia and the US [1–3]. This comorbidity is not only common but is also associated with poorer symptomatic and functional outcomes in people with schizophrenia [4].
Another powerful predictor of outcomes is the presence of expressed emotion (EE) in the household[5–7]. Expressed emotion is an indicator of attitudes and behaviour that are likely to induce emotional distress in patients (e.g. negative, controlling or highly emotive communication). Surprisingly, there is little research on the relative contribution of EE and substance use, despite the predictive importance both factors appear to have. No papers examine the predictive influence of EE in a comorbid sample. In one study [8] predictors of relapse in 97 patients with recent-onset schizophrenia were examined, 25% of whom used cannabis daily or almost daily. Expressed emotion was the only prognostic factor to enter a multivariate survival function, but frequent cannabis use significantly predicted relapse within the high-EE subgroup.
In this paper we compare EE and substance use as predictors of relapse in patients with coexisting schizophrenia-related disorders and substance use disorder. We also examine a range of other prognostic factors to check that predictions from EE or substance use are not because of these potential confounds.
Method
Participants
Sixty patients aged between 16 and 65 years with a DSM-IV schizophrenia-related diagnosis were screened. Participants had to fulfill criteria for a DSM-IV substance use disorder and lived with or had frequent contact with at least one key relative in 3 months before admission(≥2 face-to-face contacts and ≥5 telephone calls per week). Patients were excluded if they were dependent on intravenous narcotics or were diagnosed with an organic brain disorder or developmental disorder, or if a parent had a history of psychotic illness. Interviewed relatives were aged over 18 years and lived with the patients or had frequent contact with them in the 3 months before admission.
Procedure
Consecutive patients and relatives who met the criteria were assessed within 1 week of admission to the acute psychiatric ward of Hornsby Hospital in northern Sydney. The Structured Clinical Interview for DSM-IV (SCID) [9] was conducted to confirm the diagnosis of psychotic and substance use disorders. The SCID was supplemented by the review ofhospital records and information from service providers and family members.
The first investigator administered the diagnostic scale and a quan-tity/frequency measure of all substance use and rated medication adherence. They were independent of clinical care. An independent clinician blind to the EE status administered the Brief Psychiatric Rating Scale (BPRS) [10] at discharge. This rater was trained to a high level of interrater reliability (κ = 0.82), in relation to the ratings of the first and third author on standard videotaped interviews [11] and reliability was confirmed on a random sample of interviews used in the current study. The same clinician readministered the BPRS interview and assessed substance use at all assessments.
Key relatives were interviewed with the Camberwell Family Interview (CFI) [13], by the first investigator, who is an accredited EE rater. An interrater reliability check with another accredited rater on 18 randomly selected interviews obtained a minimum percentage agreement of 82% on individual scales. Relatives were interviewed within 1 week of admission and interviews were tape-recorded for analysis.
The BPRS was administered at 3-month intervals during a 9-month follow-up and during any readmission. In 57 of the 58 participants, the assessment of cannabis use from self-reports and file data was supplemented at baseline by information from urinalysis within the first week of admission. Because of the difficulty in obtaining accurate data on consumption, we coded substance use at baseline and during followup according to broad frequency categories, where regular substance use was consumption on at least 5 days per week over the previous 3 months and non-regular use was usage at lower frequency.
A Type I psychotic relapse was defined as a change from normal or non-psychotic state to a state of psychosis. A Type II psychotic relapse involved a marked exacerbation of persistent florid psychotic symptoms. In either case, the relapse assessment was based on BPRS core psychotic symptoms (i.e. ≥5 for unusual thought content, ≥6for conceptual disorganization or hallucinatory behaviour, or ≥7 for suspiciousness) [10], [12], whether or not the episode required hospitalization. In the case of participants with ongoing symptoms (Type II relapse), a rise of at least two rating points across the sum of psychosis items was also required [12].
Results
Sample characteristics
Sixty-two patients met the criteria for inclusion. All but two eligible patients (97%) agreed to participate. The remaining patients were primarily male (92%), never married (85%), unemployed (73%) and living with relatives (92%). Only three participants (5%) had less than 35 hours of contact with their relatives per week and the average across the sample was 43.9 h/week (SD = 10.1). Since there were only three participants with less than 35 h of contact per week, contact could not be examined as a moderator of the predictive effect of EE in the current study. Ages ranged from 17 to 42 years (M = 24.6, SD = 5.5) and they had 11.7 years of education on average (SD = 1.9). Mean duration of treated psychosis was 2.7 years (SD = 3.6) and 62% were within 2 years of initial treatment. Twenty-nine patients (48%) had a SCID diagnosis of schizophrenia, 20 (33%) schizoaffective disorder, eight (13%) had mood disorders with psychotic features and three patients (5%) had a diagnosis of drug-induced psychosis. A history of childhood disorder was reported by 27 (45%), of whom 17 had attention deficit hyperactivity disorder (ADHD). At baseline, cannabis was used by 88%, followed by 7% amphetamine and 5% alcohol. Forty-nine (82%) were multiple substances users. Thirty-six participants (60%) used one or more substances on a regular basis. Average duration of drug use before the first hospital admission was 5.8 years (SD = 1.8). The most commonly reported motives for using substance were coping with problems (58%), recreation (35%) and socializing (7%). Participants reporting regular use of cannabis at baseline had a mean of 368.4 ng mg−1 of Delta(9)-tetrahydro-cannabinoid (THC) in their urine (SD = 356.8), compared with 277.0 ng mg−1 (SD = 253.3) for those reporting non-regular use, but this difference was not statistically significant (M = 55, t(1.07), p = NS).
All but two patients (97%) were available for follow-up assessments and 70% of the available sample appeared to be fully adherent to medication during follow-up (based on all available data). However, 43 participants (74%) continued to use one or more substances, 23 of whom (40%) reported being regular users. Fifteen participants (26%) reported that they had stopped using any substance and a further 12 (21%) changed from regular to irregular use. Only five participants (9%) reported that they had increased from irregular to regular use.
Eighty-two (87%) of the eligible relatives from 60 families agreed to participate, comprising 55 mothers (67%), 25 fathers (31%), one sibling and one spouse. Mothers were the primary carers of 54 participants (90%).
Expressed emotion and its correlates
Standard cut-offs were used to classify relatives as high or low in EE [13]. A high-EE environment was observed in 37 (62%) households, but in only six of these did both key relatives have high-EE interviews. Half the relatives had interviews rated high in EE. High-EE interviews were from 58% of mothers, 32% of fathers and from the only interviewed partner. Overall, 40% of mothers and 24% of fathers showed emotional overinvolvement. No interview was categorized as high EE by only critical comments. Fifteen interviews displayed only emotional overinvolvement, five showed hostility, nine exhibited overinvolvement and hostility and nine were high in criticism and hostility. A further three showed high emotional over-involvement (EOI) and criticism, and two showed all three EE components.
Patients from high EE environments were more likely to have a schizoaffective or mood disorder (54%) than schizophrenia (45% ). Their BPRS psychotic index at baseline was (3) also slightly higher (M = 10.0) than in low-EE environments (M = 9.4, t(58) = 2.2, p = 0.032).
The 23 patients with more than 2years oftreated psychosis were more likely to be in high EE environments (78%) than the 37 patients with 2 more recent-onset psychosis (51%; ). High EE environments were also associated with childhood disorder (ADHD: 83%; other disorder: 80%; no disorder: 45%; χ2 = 8.1, df = 2, p = 0.017). Expressed emotion did not predict medication compliance during follow 2 up (). Seventy per cent of patients from high-EE households were regular users of substances at baseline, compared with 2 only 44% of those in the low EE group (). How 2 ever, EE at baseline did not predict use over the follow-up ().
Univariate predictions of relapse
Over the 9-month follow-up, 19 patients (33%) had a psychotic relapse, 13 of whom were admitted for inpatient care. Five relapses were Type I and 14 Type II. Table 1 shows the predictive effect of individual variables. Expressed emotion was the strongest predictor of relapse. Patients from high EE environments were 5.6 times more likely to relapse than were those from low EE environments (49% vs 9%). A survival function predicting time to relapse is shown in Fig. 1. Expressed emotion produced a beta of −1.95 (SE = 0.738; Wald = 6.80, df = 1, p <0.01; Exp(beta) = 7.04). The increased risk from high EE was distributed relatively evenly throughout follow-up, rather than clustering in particular time periods. We examined whether EE predicted a 9-month relapse in a subsample of 37 patients who were within 2 years of their initial inpatient treatment. None of the 18 patients from low EE environments relapsed, compared with nine of 19 (47%) from high EE contexts ().
In the full sample, there were only three other significant predictors of relapse. Fewer patients with ADHD in childhood relapsed during follow-up than did patients with no disorder or another childhood disorder (Table 1). Women were more likely to relapse than men, but this result was based on only five women. Four of the five women were from high EE environments characterized by hostility, all were polysubstance users and four had affective or schizoaffective disorders and were on mood stabilizers.
Prediction of time to relapse from expressed emotion.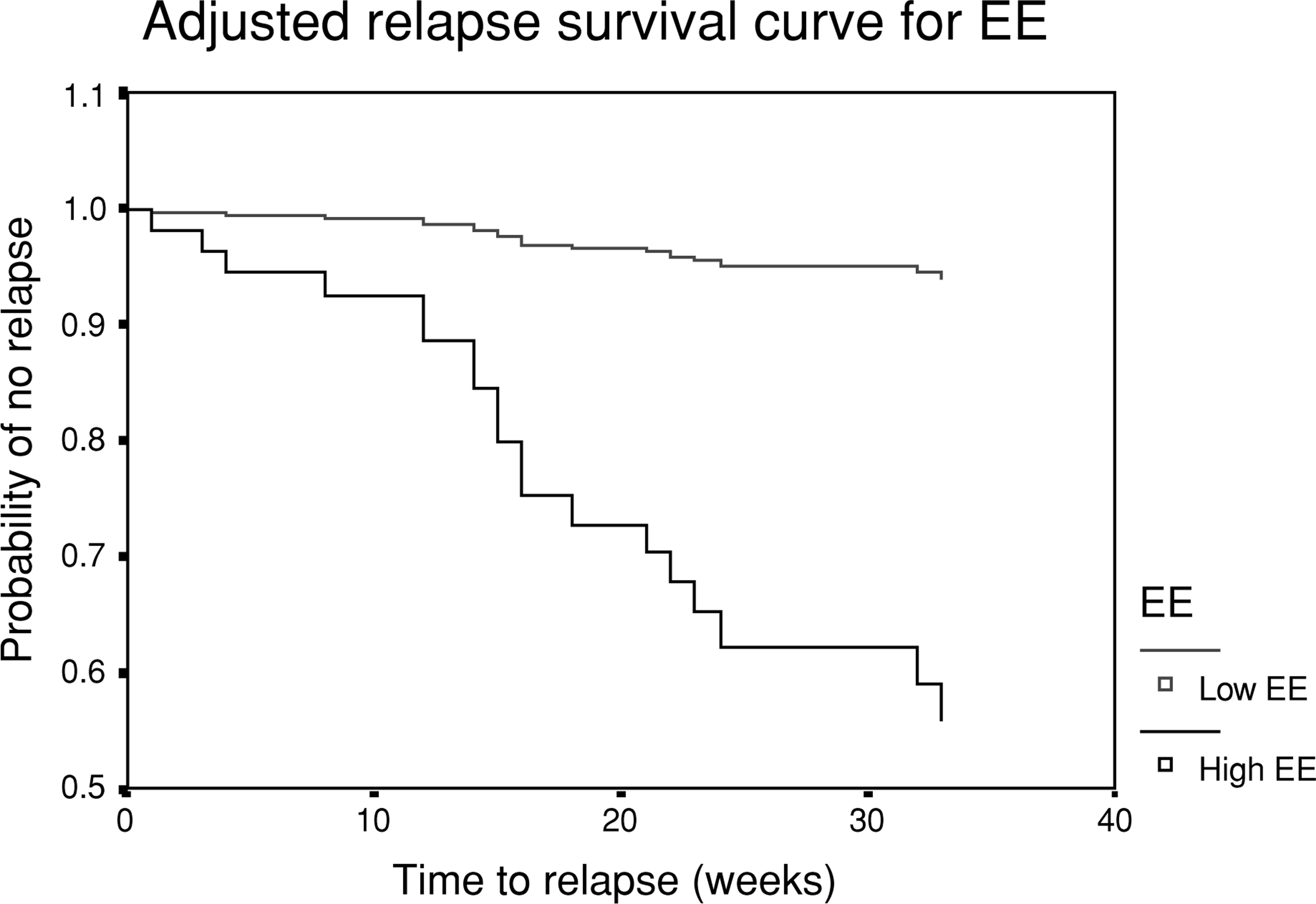
Prediction of relapse
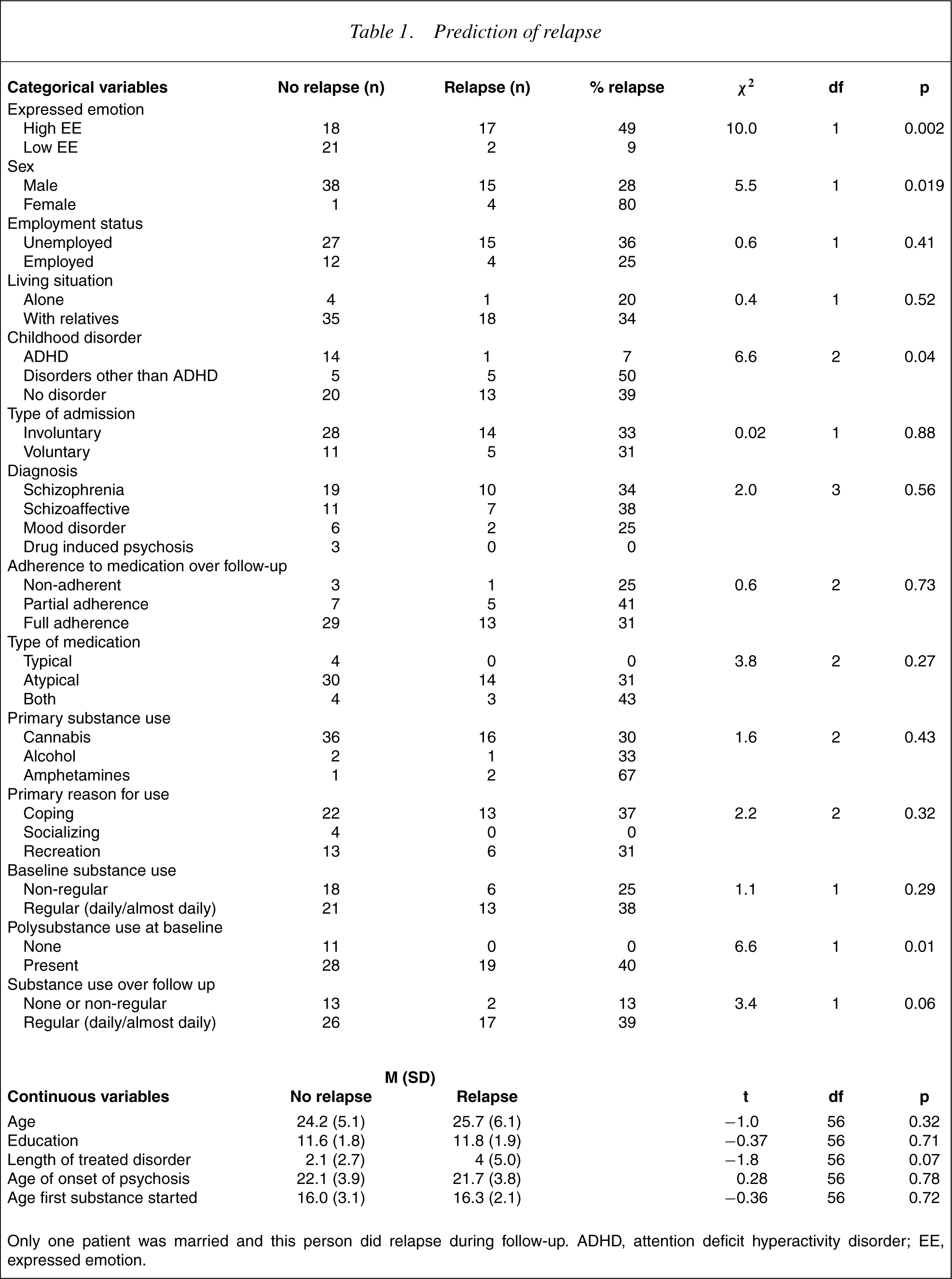
Only one patient was married and this person did relapse during follow-up. ADHD, attention deficit hyperactivity disorder; EE, expressed emotion.
Polysubstance use at baseline also predicted relapse significantly, with no one relapsing in the group that was not using multiple substances. However, despite being linked to three times the risk of relapse, the frequency of substance use during follow-up fell just short of the 0.05 level of significance as a predictor of relapse (p = 0.06). Relapsing patients had almost double the length of treated disorder than those who did not relapse, but this also failed to reach significance (p < 0.08).
Multivariate predictors of time to relapse
The prediction of time to relapse from EE together with other predictors with a significance level of p< 0.10 was examined in a survival function using Proportional Hazard Model (Cox 1972) simultaneous 2 entry. The regression was highly significant (). As shown in Table 2, high EE remained a significant predictor of shorter time to relapse when the effect of other potential predictors was controlled. Absence of ADHD in childhood was the only other significant predictor. If only EE and childhood disorder history were entered into 2 a survival function, the regression was highly significant () and the equation was 2.388 EE (SE = 0.759) −2.474 ADHD (SE = 1.098) + 0.144 other disorder (SE = 0.533).
Cox regression with simultaneous entry, predicting time to relapse
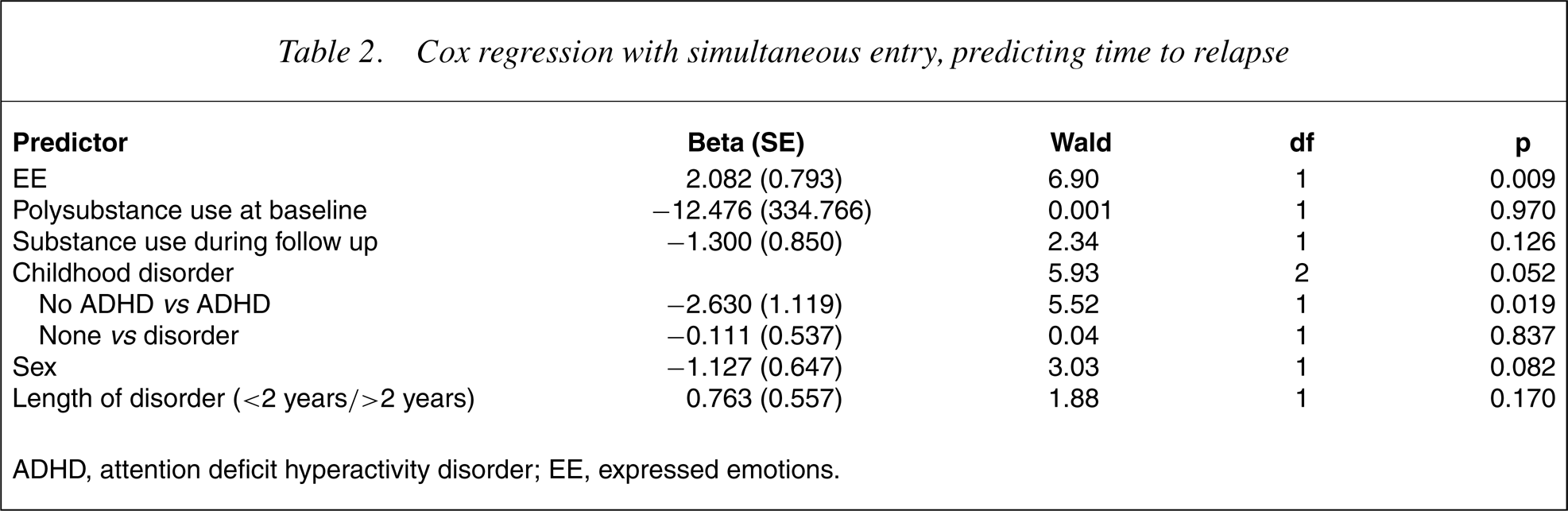
ADHD, attention deficit hyperactivity disorder; EE, expressed emotions.
Although adherence to medication was not a significant univariate predictor of relapse, we did check to see whether it modified the prediction from EE. It did not. A further secondary analysis examined the specific role of regular cannabis use during follow-up. Cannabis use was neither a significant predictor in the multivariate survival function nor was there a significant prediction from an interaction between EE and cannabis use. Finally, no univariate prediction from cannabis use was seen in high EE environments.
Discussion
Patients from high EEenvironments were significantly more likely to relapse and had earlier psychotic relapses than patients from low EE environments. Relapse rates were similar to those in previous studies on EE in schizophrenia [6]. Predictive effects from EE were stronger than from any other measured variable, and prediction of time to relapse remained significant after entry of other predictors that individually approached significance. The predictive effect of EE remained strong in patients with recent disorder onset, and no patients from low-EE environments relapsed during follow-up. These results provide the first empirical evidence that EE is related to relapse in patients with psychosis and substance misuse. They also support the predictive effect of EE in patients with a short history of treated disorder [8], [14].
Polysubstance use at baseline was a significant univariate predictor of relapse, but during follow-up, neither substance use nor cannabis use specifically reached significance as predictors. Poorer symptomatic outcomes from more frequent substance use have typically been obtained in previous research [4]. In one previous study [8], the prediction of relapse from frequent cannabis use in the study on people with recent-onset schizophrenia did not reach significance in the sample as a whole, but (contrary to our results) it did significantly predict relapse in a subgroup from high EE environments. It is possible that the relatively weak impact of cannabis and other substance use on relapse in the present study was because of imprecision of intake measurement, although we took care to create a ‘best estimate’ measure from all available sources of information, including multiple questions to participants and collateral information.
Factors associated with high EE included substance use at baseline and longer duration of treated disorder. This was consistent with high EE being a stress reaction by people in the immediate social environment [4], [6]. This raises the intriguing possibility that in some other studies where a predictive effect of substance use on outcomes has been observed, the pathway of influence may to some extent be via EE and other social reactions to substance use, rather than solely by pharmacological or other direct consequences of substance use on symptoms.
Interestingly, childhood ADHD was associated with a lower risk of psychotic relapse than either other childhood problems or no childhood disorder. This result was unexpected, and the basis of any protective mechanism or lesser risk in these people requires clarification. There is some evidence that attentional problems in childhood are related to more severe neuropsychological deficits in adult schizophrenia [15], and the presence of neurocognitive deficits in childhood would be expected to predict poorer symptomatic outcomes [16]. However, we have not located studies on the specific influence of previous ADHD diagnosis on the risk of psychotic relapse. The issue is complicated by the fact that, although both ADHD diagnoses and ADHD-like features are more common in children at risk of schizophrenia [17], there are differences in the patterns of attentional deficits between ADHD and prodromal schizophrenia [18], and doubt is cast over the validity of the initial ADHD diagnosis. If a protective effect is replicated in prospective research with standardized and reliable diagnostic assessment, it raises intriguing questions concerning the underlying process.
A significantly greater rate of relapse was observed in women, which was counter to expectations [18]. Although the number of women was too small for the result to be given substantial credence, it is possible that women with psychosis who have been substance users are at greater risk of relapse –for example, because of stress induced by social stigma. We note that the rate of hostility-related EE in women was very high (80%) and that a specific predictive effect of sex was not seen in the multivariate survival function.
The strengths of the present study include the high rate of completion, rigorous assessment procedures and the use of multiple information sources on substance use. The key finding of this study is a significant predictive effect from EE in a population with comorbid psychosis and substance use is based on a similar or greater sample to most previous studies [6].
The results indicate that the assessment of household EE is at least as important in this population as it is in patients without comorbidity and that the predictive effects of EE are not because of confounding variables. Further research on the specific impact of family interventions in comorbid populations is eagerly awaited.
Footnotes
Acknowledgements
This research was supported by a grant from the Northern Sydney Area Health Services. The authors thank all the patients and relatives who participated in this study and the staff of the Lindsey Madew Unit at Hornsby Hospital. We also thank Karen Cohen, Yasaman Mottaghipour, Revi Nair, Cherry Wolf, Jenny Timewell and Kathryn Leonards for their assistance.
