Abstract
Emerging evidence regarding the neurobiological basis for many psychiatric disorders has generated increased interest in the significance of neurological signs in psychiatric illness, particularly as the pathophysiology of these illnesses appears more subtle and diffuse than in classical neurological illnesses. Minor neurological abnormalities have been reported in a range of psychiatric conditions and reflect central nervous system (CNS) dysfunction, due either to aberrant neurodevelopment or degeneration of previously healthy CNS structures. Neurological ‘hard’ signs (NHS) reflect clearly localized CNS dysfunction and are suggestive of a focal lesion or regionally specific pathology. Neurological ‘soft’ signs (NSS), which do not signify a localized primary tract or nuclear lesion, suggest a more diffuse pathological process, such as aberrant neurodevelopment or generalized neurodegeneration [1–3]. The boundary between these two categories is less clear than the typology suggests, as ‘hard’ signs may appear when related structures or circuits connected to the expected lesion site are affected [4]. The presence of ‘hard’ signs in psychiatric illness has traditionally been felt to suggest an underlying systemic or neurological disorder, but these signs appear to have an increased prevalence even when no comorbidity is present [5], [6]. ‘Soft’ signs may reflect subtle alterations to neural structures, rendering an individual vulnerable to the development of psychiatric illness, and have been demonstrated in schizophrenia [7], [8], obsessive-compulsive disorder [9], [10] and affective disorders [11].
Cortical release signs (CRS), also known as primitive reflexes, are a group of behavioural or motor signs that, when present in an adult, reflect a loss of cortical inhibition and hence are ‘released’ by pathology [12]. Whilst traditionally viewed due to frontal lesions, they also appear with more diffuse pathology and are best viewed as neither purely hard nor soft signs. Some forms of CRS, such as utilization behaviour, are not simple disinhibited motor reflexes and represent disinhibition of subcortical and probably cortical circuits by lesions to inhibitory afferents. These signs are present during normal foetal and post-natal CNS development when subcortical myelination remains incomplete [13], and the reflex forms of CRS do not require the presence of cortical tissue [14]. They are inhibited by the development of cortical structures and their descending efferents, and can re-appear following damage to cortex, subcortical white matter carrying cortical efferents, or the extra-pyramidal system [15].
The prevalence of CRS in healthy adults is variable, with the grasp reflex being absent [16], although the palmomental reflex is present in some [17]. Prevalence of most CRS increases with age in clinical and nonclinical populations [18–20]. When CRS appear in otherwise asymptomatic individuals, they are commonly associated with small vascular lesions in basal ganglia or subcortical white matter [20]. Healthy controls rarely have more than two CRS, even beyond the seventh decade [21]. Presence and number of CRS correlate with reduced cognitive capacity in the healthy elderly, those with mild cognitive impairment, and those with frank dementia [12], [22], suggesting they are a marker for global cognitive decline. Whilst CRS have been presumed to have localizing value, particularly to frontal areas [23], [24], they may be elicited when pathology is present in other cortical regions [16].
CRS have been grouped into a prehensile group (aimed at drawing an object near) and a nociceptive group (withdrawing from a painful stimulus) [25], shown in 25], in contrast to nociceptive signs which have a higher base rate in the population and appear in early or mild disease [19], [25], [26]. However a number of signs do not fall easily into these categories, including the nuchocephalic & corneomandibular reflexes and utilization behaviour.
Nociceptive and prehensile cortical release signs

Comparing CRS in different populations has been difficult due to the lack of standardized methods of elicitation and rating used in prevalence studies [27]. Similarly, differences in examination for and recognition of CRS renders interobserver reliability low unless standardized assessments are used [27], [28]. The use of the Neurological Examination Scale (NES) to rate CRS and other soft neurological signs in schizophrenia sufferers [29] has allowed for more reliability, and similar scales have been developed for Alzheimer's disease [30], Parkinson's disease [31] and general neurology patients [27].
Clinical neurobiology of cortical release signs
Palmomental reflex
This reflex, first described in 1920 in a patient with amyotrophic lateral sclerosis [32], occurs when the thenar eminence of the palm is stroked with light touch or a pin proximal to distally [18]. This produces a slow, sustained contraction of the ipsilateral mentalis muscle in most cases, but may also result in contralateral or bilateral contraction [33], [34]. In healthy individuals the trigger area for the reflex is usually the palm, but may include other areas of the arm, the trunk, or the sole of the foot [24], [35]. Elicitation of the reflex outside the thenar eminence is rare in healthy individuals [35–37]. This reflex is present in up to 30% of healthy young adults [17], and greater than 60% of patients aged over 50 [18–20],[33]. It is electromyographically demonstrable in most normal adults [38], [39], and is often the first reflex to appear with age [40]. The habituation seen in healthy individuals after 3–5 stimulations is rare when neurological disease is present, making it an important differentiator between clinically relevant and incidental findings [28], [41], [42]. Contralateral response to stimulation is also suggestive of neurological disease [39]. With clinical improvement in the causative lesion, the reflex may again disappear [34].
Pollicomental reflex
This variation of the palmomental reflex was first elaborated by Bracha in 1958 [24], in patients suffering pathology in the premotor area of the frontal lobe. Stroking the volar surface of the terminal pollical phalanx with a thumbnail or key evokes a brisk contraction of the ipsilateral mentalis muscle. Unlike the palmomental reflex, it has a low base rate in healthy adults [24]. In the Maastricht Ageing Study [43], 5% of adults under 50 and 17% of those over 50 demonstrated a positive response (van Boxtel, pers. comm.). Like the palmomental reflex, lack of habituation is considered abnormal.
Grasp reflex
The grasp reflex was first described in 1914 [44], [45]. It refers to a progressive forced closure of a patient's hand via finger flexion and thumb adduction on an object when the palmar surface of the hand is stimulated by strong pressure moving from the ulnar to radial side [46]. Elbow flexion may also occur [47]. Patients are usually able to release their grip when asked [46], [48]. A similar reflex can be elicited in the foot with stimulation of the sole [48], [49]. A related CRS, the instinctive grasp reaction, is a more complex and less stereotyped hand movement designed to maintain or manipulate an object in the hand [50]. Initially thought to simply reflect frontal lobe damage [50], more recent work suggests that the anterior cingulate gyrus (ACG), supplementary motor area (SMA) or deeper white matter is the lesion site when a grasp reflex or instinctive grasp reaction is present [46], [51]. Grasp reflexes follow contralateral SMA lesions which remove its inhibition of the primary motor cortex, and instinctive grasp reactions follow contralateral ACG lesions which disrupt the modulating input of the ACG on the lateral premotor area [51]. The grasp reflex appears to be very rare in individuals without CNS disease [16], [27], [46], and is almost universally absent in healthy young adults [15], [18], [19], [27].
Snout and pout reflex
The snout reflex (along with the sucking, rooting and pout reflexes) is one of a group of primitive oral reflexes that facilitate feeding in the neonate [52], which were first described more than a century ago [53], [54]. The snout reflex occurs when both lips pucker in response to pressure on the nasal philtrum, producing an elevated protrusion of the lower lip [55], [56]. The pout response is a myotactic stretch reflex elicited by firm tapping on the lips or around the mouth, and electromyographically present in normal individuals [57]. The pout response is reflective of an upper motor neurone lesion when present [55]. Traditionally associated with frontal disease, the snout reflex is prevalent in diffuse CNS disorders [58] and may reflect frontal-subcortical pathology or disconnectivity [19]. Prevalence of the snout reflex has been associated with increasing age in healthy subjects [18], [20], [59]; it rarely appears prior to the fifth decade [18], [20], [37], occurs at a rate of 10–15% between 40 to 60 years and 30% in those over 60 [18], [20].
Rooting reflex
The rooting reflex can be elicited with a visual stimulus (by approaching the mouth with an orally unfamiliar object from within visual fields) or a tactile stimulus (by scratching the cheek or lateral lips), resulting in opening of the mouth and turning of the lips towards the stimulus [55]. It is, like the snout reflex, traditionally associated with frontal syndromes but occurs in a range of diffuse CNS diseases.
Suck reflex
A related reflex, the suck reflex occurs when a sucking or licking movement of the mouth and buccal region occurs in response to stimulation of the oral region, particularly insertion of an object in the mouth [56]. It is rarely found in healthy adults [6], [29], [37].
Glabellar tap
Also known as Myerson's sign [60], this is elicited with a subject's gaze fixed on a distal object whilst the glabellar region is rapidly tapped from above (outside the visual field); normal subjects will habituate and show reflex closure of the eyelids to only the first two or three taps [56], [61]. It was initially considered specific to Parkinson's disease, but has been demonstrated in a variety of CNS disturbance, most particularly Alzheimer's and other dementias, and vascular or neoplastic lesions [12], [16], [42], [59], [62], [63]. The disinhibited glabellar blink may be seen in healthy individuals at rates of up to 30% [16], [59], [63] with prevalence rising sharply beyond the 7th decade [20].
Corneomandibular reflex
The corneomandibular reflex, or ‘winking jaw phenomenon’ was reported by vön Solder in 1902 [64]. It is elicited by light pressure on the cornea, and produces an anteroinferolateral deviation of the contralateral mandible due to pterygoid contraction [65]. This reflex, like other reflexes that unite disparate muscle groups such as the palmomental reflex, may represent a ‘de-differentiation’ of muscles once developmentally associated in fetal life [52]. It has been reported in a wide variety of focal and diffuse illnesses, including vascular and neoplastic disease and neurodegenerative disorders [35],[66–68]. It occurs rarely in healthy adults [69].
Nuchocephalic reflex
Described by Jenkyn in 1975 [70], the ‘doll's eye’ reflex is elicited by a rapid turning of a standing subject's shoulders to one side. A reflex is positive if forceful neck muscle contraction maintains the original head position. It occurs with a frequency of less then 10% until the eighth decade in healthy controls [19].
Utilization behaviour
Lhermitte first described utilization behaviour (UB) as an extension of the grasp reflex, whereby objects are not only grasped but utilized in a compulsive fashion – without regard to need to do so or social context – in lesions in left, right and bilateral frontal cortex [71]. These activities included ‘pouring’ from an empty jug into a glass and using a knife and fork on a plate without food. Like the grasp reflex, UB was felt to result from a lack of frontal lobe inhibition of parietal structures [71–73], but at a ‘higher level’ than in the grasp reflex such that disinhibition of more complex and purposeful movements result in a patient being bound to respond to visual and tactile stimulation [73]. It frequently occurs with imitation behaviour (IB) [73], [74], but appears less commonly and has suggested to be a more severe form of IB [75]. UB has also been demonstrated with vascular, neoplastic, traumatic and infective lesions to the thalamus [76], [77], caudate nucleus [72], [78], frontal white matter [79], supplementary motor area (SMA) [80] and medial frontal regions including the anterior cingulate [72], [74], [81], [82]. It has more recently been suggested that UB results from interruption to a distributed network involving medial frontal structures and subcortical structures [83], [84], and can thus be viewed as a ‘disconnection syndrome’ [79]. This model of understanding a cortical release phenomenon as a result of an interrupted distributed network may be applicable to all release signs, although networks involved in other CRS have yet to be clearly delineated.
Cortical release signs in neuropsychiatric illness
Schizophrenia
A large body of evidence now implicates both hard and soft signs in schizophrenia, where abnormalities of sensory integration, motor co-ordination, motor sequencing and CRS are over-represented [85–88]. The publication of the Neurological Evaluation Scale (NES) in 1989 [29], which includes the snout, suck and grasp reflexes and glabellar tap, has allowed for the comparison of different populations of schizophrenia patients with controls and other mental illnesses. Over half of all schizophrenia sufferers demonstrate at least one CRS [89–91], and elevated rates of the palmomental [6], [89],[91–93], grasp [89],[91–93], snout [89], [91], [93], [94], corneomandibular [91] and glabellar tap [91], [94], [95] have been reported. Other studies have shown global elevations in CRS [96], with few studies finding non-significant or no elevation of CRS rates in schizophrenia [8], [97]. A meta-analysis suggests that the palmomental, snout and grasp reflexes all occur at significantly higher rates than in controls [98]. Patients with childhood onset schizophrenia demonstrate even higher rates than in adult-onset illness, suggesting that paediatric schizophrenia is a more ‘biologically driven’ form of the disease and that degree of neurodevelopmental disturbance is related to time of illness onset [99]. Grasp reflexes have been reported as state markers for psychosis [100], [101], although other authors suggest that CRS represent trait markers with NSS acting as state markers [102]. The presence of CRS has been associated with tardive dyskinesia by some [91] but not all [90], [103], [104] authors. They have been demonstrated at onset of the first psychotic episode [105], including in neuroleptic-naïve patients [106], and prevalence increases with duration of illness [102], [105]. The rate of CRS in unaffected first-degree relatives is higher than controls but less than patients [94], [96], [107], suggesting a contribution to CRS by genetic factors. The most commonly reported CRS in schizophrenia is the palmomental reflex, although most studies do not comment on bilaterality, strength of response or habituation making its significance in the illness unclear. The glabellar tap reflex may result from the Parkinsonian effect of antipsychotics [8], [108]. Although they may be associated with indicators of poor outcome, the clinical utility of CRS is limited as they are not universally present and are seen in other psychiatric illnesses [98]. NSS that reflect high-demand cognitive processing (such as verbal memory and audio-visual integration) have proven to have greater specificity and predictive value than CRS [88], [98].
Bipolar disorder
CRS have been reported in bipolar disorder by a number of authors [11], [91],[109–111]. Higher rates are found in poor-prognosis patients [112], and signs such as the grasp and palmomental reflex may diminish with clinical recovery [113], [114], suggesting CRS may be a state if not trait marker. Up to half of bipolar patients may demonstrate CRS, most commonly the palmomental reflex, with the snout reflex and disinhibited glabellar tap occurring in up to one quarter of patients [91]. In childhood and adolescent mania patients, CRS have been shown to occur no more commonly than in controls whereas signs reflecting motor co-ordination, sensory integration and inhibition occur at significantly higher rates [114]. The paucity of studies limits firm conclusions, although in light of the strong shared genetic diathesis between schizophrenia and bipolar disorder [115], CRS in these illnesses may reflect a shared biological heritage.
Major depression
Very few studies have examined CRS in depression. A controlled study of patients with mania and depression showed increased prevalence of the nuchocephalic reflex and glabellar tap, but predominantly in patients with associated cognitive impairment [11]. Utilization behaviour was present in one cohort in all patients with major depression and one fifth of patients with dysthymia, suggesting it reflected the degree of hypofrontality in mood disturbance [116]. CRS have been associated with depression in cerebrovascular disease but not in Parkinson's disease [117]. The relationship between CRS and psychomotor retardation, which may result from frontostriao-limbic dysfunction [118], has not been examined.
Obsessive-compulsive disorder
The two studies that examined CRS in OCD using the CNI as the rating instrument have shown discrepant findings, with the first study suggesting OCD sufferers had higher rates of release signs than controls, on par with schizophrenia sufferers [10]. A later study found equally high rates of other neurological signs but no elevation of CRS [9]. Motor tics have been associated with OCD [119], but their association with CRS has not been examined.
Alzheimer's disease
The grasp and snout reflexes are seen more commonly in sufferers of Alzheimer's disease (AD) than controls [30], [120] and their presence correlates with the degree of cognitive impairment and illness severity in most [12], [22], [30],[120–126] but not all studies [59], [127]. CRS are also associated with functional limitation and behavioural disturbances in AD [127] and the presence of ventricular enlargement [128] and extrapyramidal motor signs [22], [125], [127], [129]. Nociceptive signs appear early (GDS stage 3) [30], whereas prehensile signs appear later (GDS stage 6) [22], [30]. The presence of CRS in AD may reflect frontal atrophy [126] and a loss of large pyramidal cells in cortical layers III and IV in the frontal lobes [121]. Presence of the snout reflex in Down's syndrome may herald the onset of AD in this group [130]. There appears to be no single CRS or combination thereof pathognomonic for AD, although the prehensile reflexes may be more specific [131].
Cerebrovascular disease & vascular dementia
Like AD patients, patients with vascular dementia (VaD) have an increased rate of CRS [12], [22], [132], particularly when pathology predominates in frontal regions [21], [133]. CRS rates have been correlated with degree of cortical atrophy [21] and subcortical white matter disease [21], [134], [135] in VaD patients. No pattern of CRS appears pathognomonic for VaD [131], [136], although unilateral rather than bilateral signs are seen more frequently in VaD compared to AD [22]. In patients with cerebrovascular disease, patients with less than three CRS are rarely demented [21]. Rates of CRS are higher in patients with both transient ischemic attacks (TIAs) and completed strokes [137] than controls, suggesting CRS appear as the load of vascular pathology increases. CRS have been associated with carotid stenosis in VaD patients [138] as has frontal lobe dysfunction [139], suggesting that anterior lacunar infarction and resultant frontal ‘deafferentation’ may be responsible for loss of cortical inhibition. CRS also occur in almost half of patients in the postoperative period of coronary artery bypass grafting [140], where embolic stroke is a recognized complication [141]. Elevated rates of CRS are seen in patients with peripheral vascular disease, itself a marker for cerebrovascular disease [142].
Frontotemporal dementia
Early proposed criteria for frontotemporal dementia (FTD) included CRS [143]. The grasp and pout reflexes have been shown to be more prevalent in FTD than AD [144]. Grasp, pout, snout, glabellar tap and palmomental reflexes are present in more than half of FTD patients [144–146]. The presence of CRS, presumably reflecting a greater load of frontal cortical pathology, reliably differentiated FTD from AD when combined with verbal fluency and judgement scores [144], and CRS are significantly more common in FTD than VaD with frontal signs [145], suggesting that frontal-type dementias may show an earlier presence and higher rate of CRS.
Attention-deficit hyperactivity disorder
Children with attention-deficit hyperactivity disorder (ADHD) have been found to have a greater rate of utilization behaviour compared to controls [147]. ADHD has been associated with hypofrontality [148], and some ADHD-associated behaviours have been proposed as a form of UB [149]. No other CRS have been described in this illness.
Other psychiatric illness
A single study comparing rates of CRS in chronic PTSD sufferers to controls found significantly higher rates of the palmomental reflex and impaired motor sequencing [150]. Human immunodeficiency virus carriers demonstrate significantly higher rates of nociceptive reflexes than controls [151–153], and CRS are associated with illness stage [151–153] and dementia [153]. CRS have been well-described in Parkinson's disease [31], [62], [66], [117], where number of reflexes correlate with disease severity and cognitive impairment [31], [117].
The reported presence and prevalence rates of CRS in psychiatric disorders is shown in Table 2.
Prevalence rates of cortical release signs in psychiatric disorders
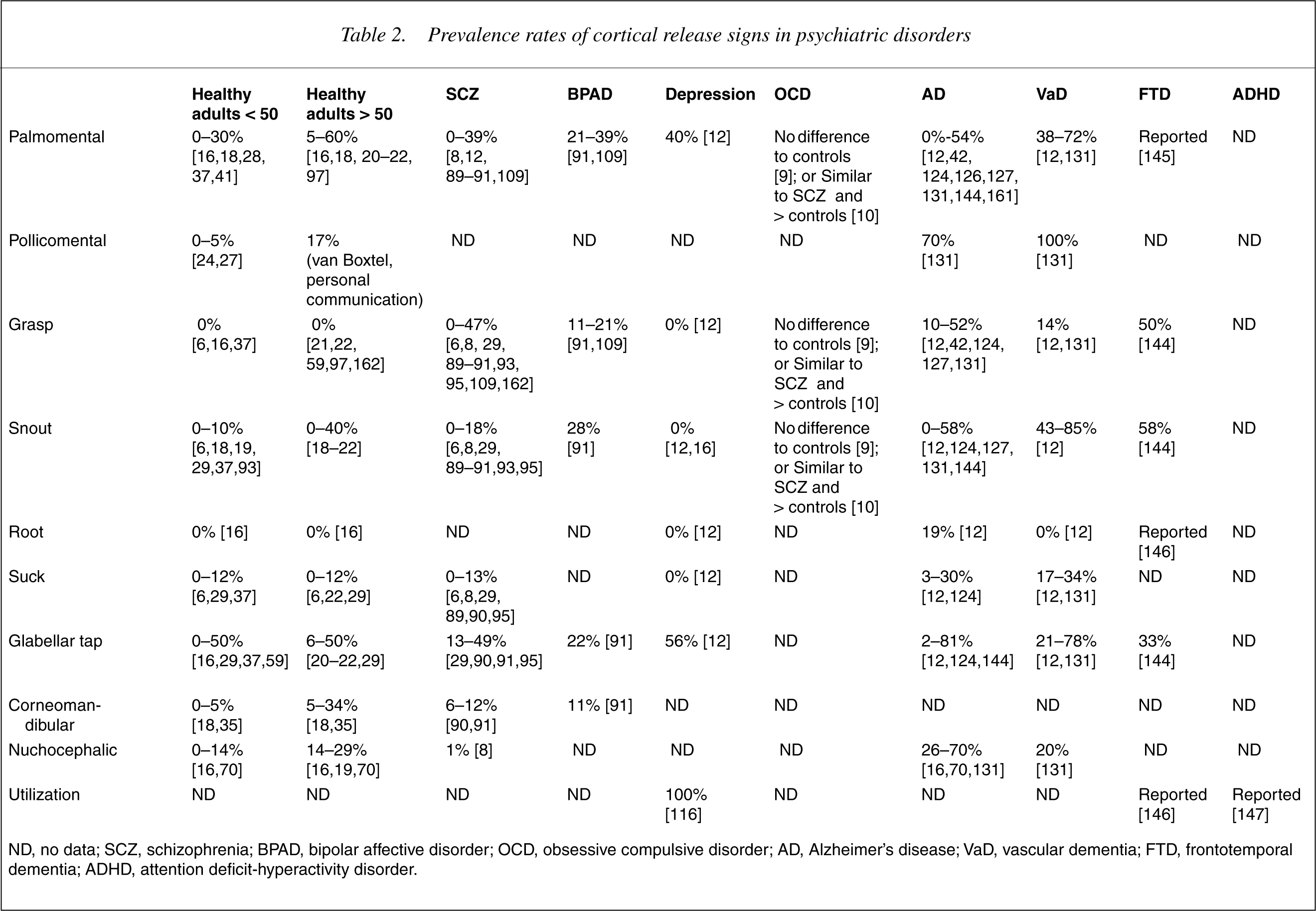
ND, no data; SCZ, schizophrenia; BPAD, bipolar affective disorder; OCD, obsessive compulsive disorder; AD, Alzheimer's disease; VaD, vascular dementia; FTD, frontotemporal dementia; ADHD, attention deficit-hyperactivity disorder.
Discussion
The use of CRS is established in the neurological examination of the newborn [154] but their role in adult medicine and psychiatry is less clear [27], [155]. In a metaanalysis of 258 studies to determine the rate of neurological signs including CRS in schizophrenia and mood disorders, Boks et al. only found 17 studies that provided analysable data [98]. The use of standardized scales goes some way to alleviate the problem of correct application and interpretation [30], [31], [131], but the integration of these findings into clinical care remains problematic.
The first difficulty arises due to the high base rate of some reflexes in the population, such as the palmomental reflex [17], and interpretation of an isolated finding in an individual should be undertaken cautiously. Secondly, the prevalence of a number of CRS increases with age in healthy individuals, making an isolated finding of a single CRS – other than the grasp reflex – of little diagnostic value in the absence of other neurological signs in individuals over the age of 50 [19], [26]. Furthermore, determining whether a response is ‘abnormal’ requires more than just a positive response to stimulation: the intensity of response, degree of habituation and size of reflexogenic zone are crucial variables [26].
The localizing value of CRS is also questionable, as presence of an ‘abnormal’ CRS may be more representative of diffuse cerebral dysfunction than frontal lobar lesions [16] and should alert the clinician to the necessity to thoroughly investigate all aspects of the central nervous system. No particular CRS or group of signs demonstrates specificity for a particular CNS disease, although the categorization of reflexes into prehensile and nociceptive groups may assist in determining severity of pathology and illness staging [131].
The poorly standardized nature of CRS detection and rating in clinical and research settings [27], [28] is also suggestive of the need of a structured tool for rating CRS in psychiatry. The difficulty in interpretation of CRS in psychiatry due to their relative non-specificity and, for some, high base rate in healthy individuals has resulted in some groups abandoning them entirely when constructing neurological rating scales for psychiatry [156–158]. Of scales containing CRS, the NES [29] was developed for the purposes of schizophrenia research and contains neurological signs felt relevant to this disorder; the CNI, whilst developed for a range of psychiatric settings, only includes a limited number of CRS and takes up to 40 minutes to administer [93]. A pure CRS scale would ideally be brief, portable, have clear standardized instructions and differentiate normal from abnormal reflexes on the basis of stimulus intensity, reflexogenic zone, degree of habituation, and bilaterality of response, none of which are rated in the NES or CNI [29], [93].
For the illnesses seen most often in general adult psychiatry – depressive and anxiety disorders, and the psychoses – the role of CRS is found in the initial thorough diagnostic work-up of patients first presenting with illness, or presenting in an atypical fashion, to aid in the detection of central nervous system disease that may initially present with psychiatric disturbance [159]. Examination for CRS may also be of value as part of diagnostic re-assessment should illness prove unresponsive to treatment or have an atypical course, although no studies have been undertaken to determine if examination for CRS outside the routine neurological examination enhances pick-up rates of neurological abnormalities. On the basis of the available research, a clinician would be well-served to test palmomental, grasp, and oral reflexes at these time points, take careful note of the nature and intensity of the stimulus and response, and integrate these findings with available historical information, mental state and physical/neurological examination, and investigative findings.
In neuropsychiatric practice, the utility of CRS is found in assisting in the initial assessment of undifferentiated cognitive, behavioural or motor disturbance. The presence and nature of CRS in some neuropsychiatric diseases, such as schizophrenia, may be an index of severity of neurodevelopmental disturbance and reflect on illness course and likelihood of development of medication side-effects as well as being state markers [159]. In old age psychiatry, where dementias and other neurodegenerative disorders present with comorbid psychiatric illness, CRS may aid illness staging [30], [160], outcome prediction [126] and diagnostic differentiation [144], [145]. The grasp reflex in particular in this group appears to be a harbinger of significant CNS disease [18–22].
Finally, the understanding of the suppression of CRS with normal neurodevelopment and their re-emergence as the CNS ages or is impinged upon by disease reinforces that psychiatrists place psychiatric diagnosis, treatment and prognosis in the crucial context of the development of the human brain across the lifecycle. Understanding the role of descending cortical inhibition as one of the many developmental trajectories of the CNS, and how damage to these structures results in CRS, may provide the clinician with an additional instrument for diagnostic differentiation, illness staging and monitoring of illness progression.
