Abstract
Orogenic gold deposits are the main source of gold for humanity. These deposits formed from the Archaean to Phanerozoic at crustal depths of more than 4 km. The key parameters accounting for their formation remain under discussion. I review these parameters in light of recent scientific advances using a systemic approach, where the relative pertinence of each component of a complex system in time and space is addressed. From the base to the top of the mineralising system, the following components and processes are reviewed: (1) the fluid sources; (2) the appropriate ligands and their sources; (3) the solubility of gold and its potential sources; (4) the function of faults as conduits; and (5) the mechanisms for precipitating gold. This review provides further support for the role of carbon-rich and pyritic sedimentary rocks in the formation of orogenic gold deposits throughout geological time.
Keywords
Introduction
Orogenic gold deposits account for an estimated 75% of the gold extracted by humans (Phillips 2013), including the tonnage of Witwatersrand, the world's largest district (∼30% of gold extracted: Phillips and Powell (2011) – either as the erosion of primary gold deposits or from hydrothermal accumulation. Such deposits are thus the largest, single genetic type of deposit for providing gold. Nevertheless, as a genetic class, their formation remains a matter of debate (Tomkins 2013a). The main reason for this debate lies in their depth of formation in the crust, which is interpreted to be >4 km (Goldfarb et al. 2005 – Figure 1). As a consequence, we do not have direct access to active mineralising systems, as is the case for some other types of gold deposits (Hannington et al. 2016).
Anatomy of an orogenic gold hydrothermal system with the key parameters. Note: The conceptual representation is based on a systemic analysis approach, where the formation of an orogenic gold deposit, from bottom to top and in time, requires a succession of processes for providing key parameters; otherwise, the mineralising system aborts. The timeframe is adapted from Cox (2016).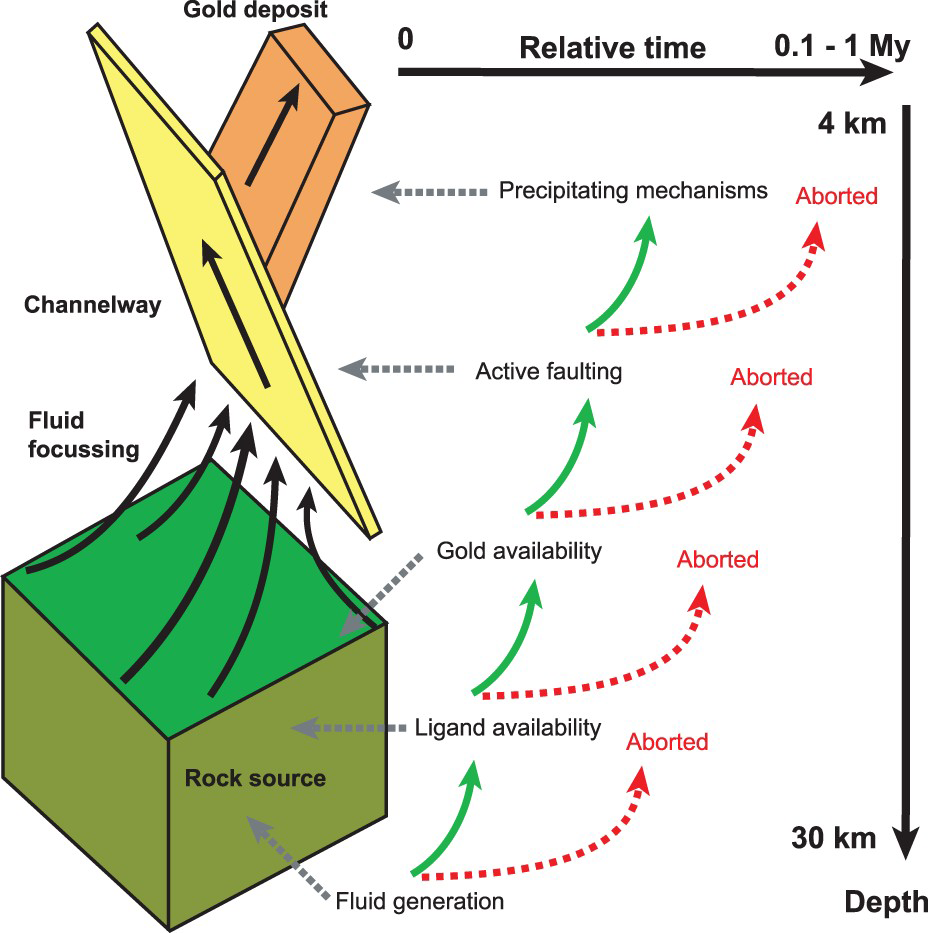
The term ‘orogenic’ was first introduced by Bohlke (1982), but the term ‘orogenic gold deposit’ was mostly used by Groves (1993). Groves and his team dominated the academic publishing system for almost 20 years. Their major contributions suggested that orogenic gold deposits could have been formed vertically along crustal faults at pressures and temperatures covering a wide range of depths, from the sub-greenschist to granulite metamorphic facies (Groves 1993; Groves et al. 1998, 2003). This concept is known as the ‘crustal continuum model’. However, the crustal continuum was an empirical, not a genetic model. As a consequence, for more than 20 years, key parameters for the formation of orogenic gold deposits have been difficult to address because deposits could have been formed in a range of extreme conditions. In short, given such a large range of physicochemical conditions, recognising the key parameters for addressing the genesis of orogenic gold deposits was very difficult. At the end of the 2000s, some papers were published (Phillips and Powell 2009, 2010; Tomkins and Grundy 2009; Tomkins 2010), demonstrating that the ‘crustal continuum model’ was not valid over the large spectrum of temperatures and pressures as initially proposed but only for restricted ranges of depth and temperature mostly corresponding to the greenschist metamorphic facies. By fixing more restricted conditions, it became possible to address the most important genetic aspects: (1) the fluid sources; (2) the appropriate ligands and their sources; (3) the solubility of gold and its potential sources; (4) the role of faults as conduits; and (5) the mechanisms for precipitating gold. The scientific advances related to these parameters are addressed below.
A systemic approach was developed by the author for addressing orogenic gold deposit formation. Similar approaches have been used for reviewing some concepts regarding the formation of orogenic gold deposits (Goldfarb and Groves 2015; Wyman et al. 2016). Here, the systemic approach is used to evaluate the relative pertinence of each component of a complex system in time and space (Figure 1). The internal combustion engine provides a good example to illustrate the systemic analysis approach: combustion, air and fire should be present but at specific concentrations and timing. If any of these constituents is lacking, out of proportion or poorly timed, neither combustion nor kinetic energy can be produced. The orogenic systems are reviewed from the base to the top with an emphasis on timing, sources and processes for addressing the key parameters for their formation.
As a cautionary notice, any review attempting to integrate natural and complex systems such as orogenic gold deposits into a coherent model or framework is inevitably biased, especially regarding the integration of all deposits ranging in age from Archaean to Phanerozoic time. This review is based on various papers, including deposit- to regional-scale studies, thermodynamic modelling, new and emerging analytical techniques and experimental studies. One particular aspect of this review is the use of papers that have demonstrated or proved the processes. In this sense, most of the reviewed processes have been mentioned in the literature as long as 50 years ago but as possibilities and are not cited here. Finally, this review aims to drive research on specific aspects of these complex mineralising systems.
Sources of fluids
The origin of the fluids involved in the formation of orogenic gold deposits has been debated for a long time, mostly because deuterium and oxygen isotope data plot in the superposed fields of magmatic and metamorphic fluids (Ridley and Diamond 2000). Recent developments in thermodynamic modelling have demonstrated that metamorphic dehydration of seafloor rocks is a viable mechanism for producing abundant aqueous-carbonic and low-salinity fluids, as documented in orogenic gold deposits from fluid inclusions (Ridley and Diamond 2000). Elmer et al. (2006) and Phillips and Powell (2010) demonstrated that hydrated seafloor rocks release fluids at the metamorphic transition of greenschist to amphibolite, mostly when chlorite is converted to amphibole. Hydration of seafloor rocks is induced by hydrothermal convection at oceanic ridges, where the rate of spreading controls in part the seafloor topography, the exposed rocks and consequently the intensity of hydration reactions (Gretchen and Früh-Green 2010). Sedimentary rocks, which are commonly richer in hydrous silicates, provide even more abundant fluids when rocks are crossing the greenschist to amphibolite facies boundary (Connolly 2010; Tomkins 2010; Zhong et al. 2015).
One important aspect, however, lies in the possibility of releasing large amounts of fluids in a relatively short period of time, thus limiting the direct consumption of fluid by local fluid-rock reactions along the pathway. In other words, a dynamic fluid-flowing system must be established to transfer a large volume of fluids up to the deposit site without losing much fluid on the way. Thermodynamic modelling by Connolly (2010) demonstrated that a large volume of fluid is released by metamorphic reactions at the transition of greenschist to amphibolite when hydrated sedimentary rocks (pelites) are buried following a P-T-t (pressure–temperature–time) typical of mountain belt evolution (Spear 1993). In such a case, hydrated rocks first experience a pressure increase related to the physical mountain building process and second, a thermal re-equilibration (Figure 2). The burial of rocks is rapid (∼1 My), but the subsequent thermal prograde metamorphism is slower, in the range of 10–100 My (Connolly 2010). This evolving path is coherent with observed field relationships, where orogenic gold deposits are formed at the end of convergence when belts have acquired most of their structural architecture (Kerrich and Cassidy 1994). Furthermore, metamorphic reactions at the transition of greenschist to amphibolite are essentially controlled by temperature (∼550°C: Figure 2) over a wide range of pressures (∼2–10 Kbar). Since temperature is the more important control for fluid generation at the transition of greenschist to amphibolite, Connolly (2010) demonstrated that a higher geothermal gradient, which induces metamorphic fluid release at lower pressure, is more favourable for establishing a dynamic flowing system (by solitary wave propagation). Conversely, hydrated rocks that undergo progressive increases in pressure and temperature would progressively release their fluids (Connolly 2010; Yardley and Cleverley 2015), hence limiting the development of a dynamic fluid-flowing system.
Pressure – temperature – time metamorphic diagram (P-T-t). Note: This diagram illustrates the idealised path of hydrated seafloor rocks that experienced a rapid burial and subsequent thermal rebound during exhumation, according to the modelling of Connolly (2010) for optimal metamorphic fluid generation. The schematic blocks illustrate for various points (1–3) along the path the thickening and exhumation of the seafloor rocks and the related position of the 400°C isotherm for reference. During exhumation (3), the tectonic stress field can be inverted as the mountain load (gravity) becomes the principal constraint (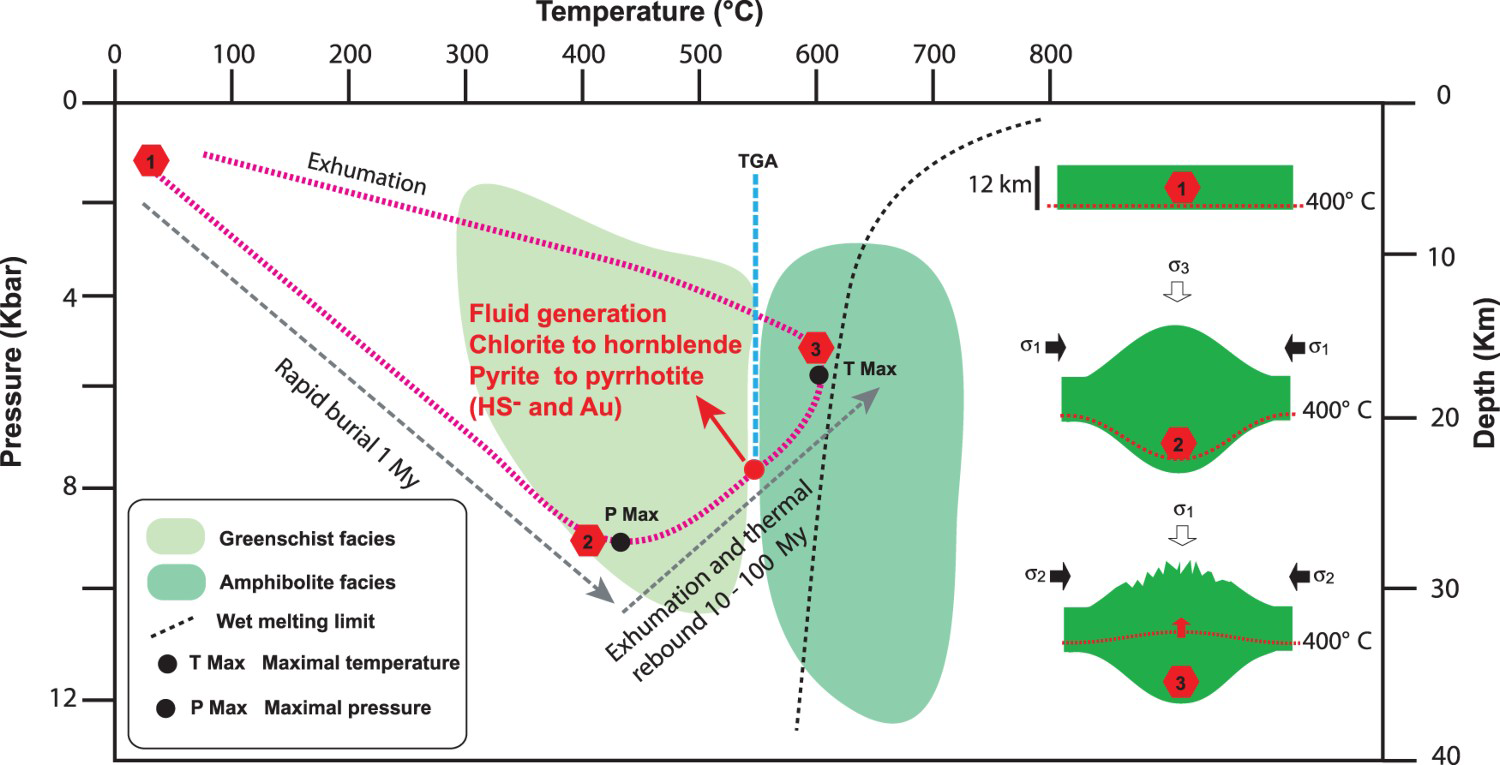
Nevertheless, if thermodynamic modelling of fluid generation accounts well for the metamorphic sources, in some cases, magmatic fluids were also involved (Yardley and Cleverley 2015). These deposits commonly contain a granitophilic suite of elements including Bi, Te, As, Mo, Sb, Sn and W (Thompson et al. 1999; Helt et al. 2014), and accordingly, some fall within the intrusion-related type. This debate was addressed two decades ago (Sillitoe and Thompson 1998; Groves et al. 2003) and remains today. For example, Xue et al. (2013), based on S isotopes (32S, 33S, 34S) of pyrite in four Archaean orogenic gold deposits, suggested a granitic source for the fluids. With the accumulation of laser ablation inductively coupled plasma mass spectrometry (LA-ICP-MS) analyses from pyrite in orogenic gold deposits, it is now obvious that some deposits with typical characteristics of orogenic gold deposits recorded sporadic magmatic fluid and gold contributions. As example, LA-ICP-MS pyrite mapping shown that gold is associated with silver (ratio of Au/Ag < 5) and with higher contents of granitophilic elements surrounding gold grains, especially Bi, Te, Mo, Sn and W (Augustin and Gaboury 2019).
Source of ligands and gold solubility
Gold is a noble metal that is non-reactive under atmospheric conditions. It requires a ligand to enhance its solubility for hydrothermal transport. The availability of ligands in solution is a fundamental parameter; thus, establishing which ligand is involved in forming orogenic gold deposits is essential in order to address the source of the gold. Chloride (Cl−) and hydrogen sulphide (HS−) are possible complexing agents, and gold has 3 oxidation states: Au0, Au1+ and Au3+ (Williams-Jones et al. 2009). Phillips and Powell (2010) proposed that hydrogen sulphide should be the most suitable ligand because Au1+ has an elevated electronegativity value for sulphur (a value of 2.58 in Pauling unit), hence favouring covalent bonding. The validation lies in the metallic assemblages found in orogenic gold deposits, where As, Sb, B, Se, Te, Hg, Bi, Mo and W are common trace metals and have Pauling values >2. Conversely, Cu and Zn are commonly lacking in orogenic gold deposits and have lower Pauling values (<2), which are more compatible with Cl− for forming stronger ionic complexes (Yardley and Cleverley 2015; Zhong et al. 2015).
Experiments performed under hydrothermal conditions have confirmed that gold is soluble under HS− complexes (Stefansson and Seward 2004; Williams-Jones et al. 2009). These data have been used to develop binary diagrams of gold solubility as a function of the oxygen fugacity (ƒO2) and pH of the aqueous solution. Most of these diagrams (Figure 3), calculated for different temperature, pressure and moles of sulphur in solution, reveal two fundamental conditions controlling the field of highest gold solubility: (1) a very restricted range of pH close to neutral value (pH∼7); and (2) ƒO2 just below the sulphate–sulphide (oxide–sulphide) mineral stability boundary (Figure 3). These specific conditions are discussed below.
Schematic logƒO2 – pH diagram for gold solubility. Note: This diagram shows contours (0.1, 1.0 and 10.0 ppm) for the solubility of gold–bisulfide complexes, modified from Hodkiewicz et al. (2009) and Phillips and Powell (2011). From the maximal solubility field, an increase of the oxygen fugacity (white arrow) as well as a decrease (grey arrow) will induce rapid gold precipitation, although the decrease in solubility is less abrupt. For the pH, any changes from approximately neutral values (black arrows) induce gold precipitation.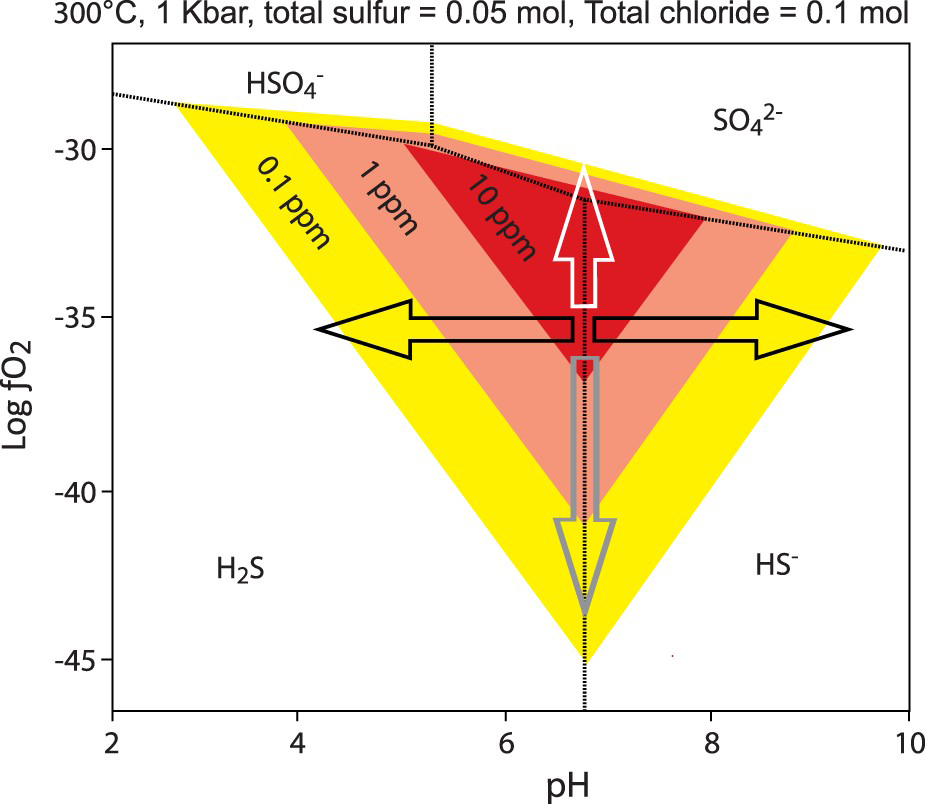
Sulphur occurs naturally as gas (H2S, SO2), minerals (sulphides and sulphates) and as a native element. In volcano-sedimentary rocks, pyrite is the dominant source of sulphur. Pyrite is well known to convert to pyrrhotite by desulphidation reactions under metamorphic conditions (Toulmin and Barton 1964). Since pyrite contains more S than pyrrhotite, S is expected to be released from this reaction. However, not all the S liberated from this conversion remains in solution to act as a ligand for gold. The temperature–pressure conditions, as well as the content of Fe-silicates consuming S for forming pyrrhotite, mostly control the excess S available for gold complexing (Tomkins 2010; Zhong et al. 2015). Using thermodynamic modelling, Tomkins (2010) demonstrated that pelitic rocks rich in organic matter can generate up to 10 times more free aqueous sulphur (HS−, S2-) than basaltic rock, according to the reaction:
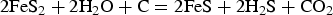
The optimal window for releasing S from the pyrite–pyrrhotite conversion is at temperatures of 500 to 550°C, corresponding to the transition of greenschist to amphibolite, with the more efficient release of S at lower pressure, i.e. under a higher geothermal gradient. Recently, Finch and Tomkins (2017) also concluded that pyrite–pyrrhotite conversion occurs at chlorite dehydration conditions. Thus, both fluid and sulphur can be released from metamorphic reactions occurring at the transition of greenschist to amphibolite facies mostly from sedimentary rocks.
Sources of gold
Numerous studies have attempted to link orogenic gold deposits with a specific source rock type (Frimmel 2008; Pitcairn 2011), such as basalts (Evans et al. 2010; Phillips and Powell 2010; Fu et al. 2012; Pitcairn et al. 2015), iron-rich formations (Meyer and Saager 1985; Lambeck et al. 2011), lamprophyre dykes (Rock and Groves 1988a, 1988b), plume-related basaltic rocks (Bierlein and Pisarevsky 2008; Webber et al. 2013; Augustin and Gaboury 2017) and felsic magmas (Kerrich 1988; Connors et al. 1993; Xue et al. 2013). Such a lack of consensus clearly demonstrates that the source remains under discussion, as reviewed by Tomkins (2013a). From an exploration perspective, clearly, no conceptual models would be correct if the gold source is not constrained.
Recently, primary pyrite formed during seafloor bacterial reduction of sulphate was proposed as the main source for gold for orogenic gold deposits. This concept was pioneered by Pitcairn et al. (2006) in the Otago Schist, where they demonstrated that higher-grade metamorphic rocks were leached of their metals (As, Mo, Au, Ag, Sn, W, Cd and Hg). The Otago Schist was ideal for study because it is devoid of known evidence of magmatic activity. Furthermore, the host sedimentary rocks thought to be of the same composition, exhibit various metamorphic facies up to amphibolite grade. Later, Pitcairn et al. (2010) demonstrated that the leaching of metals from the higher-grade metamorphic rocks was related to the expulsion of trace metals present in nodular pyrite before prograde metamorphism.
In parallel, Large and his team published numerous studies using LA-ICP-MS mapping of pyrite (Large et al. 2007, 2009, 2011, 2012; Thomas et al. 2011) to demonstrate that gold in orogenic gold deposits was sourced from primary pyrite. These researchers argued that gold in arsenic-bearing syngenetic to diagenetic pyrite (with Au values at the ppm level), hosted in black shale is a critical requirement for the later development of orogenic gold deposits during metamorphism and conversion of pyrite to pyrrhotite. Furthermore, Tomkins (2013b) reinforced the sedimentary model by demonstrating that oxidising deep oceans were more favourable for gold uptake into sedimentary pyrite. Zhong et al. (2015) demonstrated that metamorphic devolatilisation of pelites, essentially by chlorite dehydration, can liberate ∼2 ppb of gold in autogenous fluids upon crossing the greenschist-amphibolite facies boundary.
Using a solid-probe mass spectrometer (Gaboury et al. 2008), Gaboury (2013) demonstrated that ethane (C2H6) is present in fluid inclusions from orogenic gold deposits covering a wide time spectrum from 100 to 2800 Ma. He also demonstrated that ethane is sourced from thermally degraded organic matter, hence providing a reliable tracer for the involvement of carbonaceous shale at depth. Gaboury (2013) provided the first independent validation of the pyrite-sourced sedimentary gold model. More recently, evidence for gold sourced from carbon-rich sedimentary rocks in orogenic gold deposits was provided by numerous other independent techniques, such as (1) noble gases in fluid inclusions at the Phanerozoic world-class Macraes mine (Goodwin et al. 2017); (2) stable carbon isotope compositions of CO2-rich fluid inclusions hosted in mineralised quartz from Palaeoproterozoic gold deposits (Lüders et al. 2015); (3) MIF-S (mass independent fractionation) isotope anomalies of Δ33S in pyrite from Archaean orogenic gold deposits (Agangi et al. 2016; Selvaraja et al. 2017; Godefroy-Rodríguez et al. 2018; Kresse et al. 2018); and (4) boron stable isotopes from tourmaline in Archaean (Molnár et al. 2016) and Palaeoproterozoic orogenic gold deposits (Lambert-Smith et al. 2016a).
Extracting some gold from other rocks listed above is certainly possible, especially from basaltic rocks, as reviewed by Pitcairn et al. (2015). However, the sedimentary model is more robust because it provides both a source and mechanism for extracting gold from gold-rich primary pyrite (Zhong et al. 2015). The sedimentary rocks are also more favourable for providing the HS− ligand (Tomkins 2010). One missing link is the demonstration that pelites can generate abundant CO2 in the autogenous metamorphic fluids by decarbonation reactions (Zhong et al. 2015; Yakymchuk 2017), as previously demonstrated for seafloor hydrated basaltic rocks using thermodynamic modelling (Elmer et al. 2006). In conclusion, the fluid, ligand and gold all appear to be sourced from the same carbon- and pyrite-rich sedimentary rocks and generated or liberated under the same metamorphic reaction conditions at the greenschist – amphibolite facies boundary.
Role of faults
The formation of orogenic gold deposits is related to structural discontinuities (faults, shear zones, fold noses, competency contrasts) as reviewed by Vearncombe and Zelic (2015). These discontinuities act as conduits for fluid migration from the metamorphic dehydration zone, below the transition of greenschist to amphibolite, to the precipitation site in the upper crust (Figure 1), which is commonly under greenschist facies conditions. During the 1990s, orogenic gold deposits were understood to form in accretionary tectonic regimes (Kerrich and Wyman 1990), in accord with the necessity of bringing hydrated rocks at amphibolite depth (Connolly 2010; Figure 2). In the 1990s, researchers were hoping to define a particular type of fault with a specific kinematic setting more favourable for forming an orogenic gold deposit. Nevertheless, hundreds of structural studies worldwide proved that mineralisation is hosted in various types of faults and dilatational features: (1) high-angle reverse faults (Abitibi belt, Canada: Robert and Brown 1986; Gaboury et al. 2001); (2) low-angle thrust faults (Macraes mine, New Zealand: Teagle et al. 1990); (3) normal faults during exhumation (Otago Schist, New Zealand: Upton and Craw 2014; Yukon, Canada: Mackenzie et al. 2008); (4) saddle-reef dilatation zone (Cox et al. 1991); and (5) transcurrent faults (Archaean Yilgarn belt; Australia: Weinberg et al. 2005; Palaeoproterozoic Birimian belts of western Africa: Augustin et al. 2017). More examples are also given by Vearncombe and Zelic (2015).
Regardless of the kinematic setting, faults should provide permeability for channelling fluid flow during earthquakes (Sibson 1987; Sibson et al. 1988; Cox et al. 1991; Cox 2005, 2016), and in this regard, their geometries are important. Typically, irregular fault systems, such as curviplanar or helicoidal, appear to be more favourable for transferring large volumes of fluids (Gaboury et al. 2001).
One aspect that has been neglected over time is the necessity of having tectonic activity after the main accretionary event. As described, favourable source rocks are first buried during the shortening event (Figure 2), referred to here as the mountain building phase. This phase is typically the main event yielding the high topography and all the tectonic features (Figure 2). However, thermal re-equilibration, which is responsible for the key reactions at the transition of greenschist to amphibolite boundary, is a diachronic event (Figure 2), arriving later (Simard et al. 2013). How long after the mountain building phase remains to be established, but numerous mineralisation ages suggest much later, in the range of 100 My after the end of this phase (Jemielita et al. 1990; Kerrich and Cassidy 1994; Olivo et al. 2007; Augustin et al. 2017). If faulting is no longer active at the time when the fluids are available at their generation site (greenschist-amphibolite boundary), then no favourable draining system would be established and no orogenic deposit would form in the upper crust. If some faults are available, they would be used regardless of their or inherited kinematics, hence accounting for the variety of mineralisation settings documented worldwide.
Cox (2016) sheds light regarding the key role of fluid pressure and seismicity along faults. Using a modern example of a fluid injection experiment, he reported that high fluid pressure induced swarm seismicity with a magnitude range of 2–4 for individual earthquakes and a frequency of hundred to thousand events per day. The formation of orogenic gold deposits is well known to be associated with fluid pressurisation and rupture of the fault (Sibson 1987; Sibson et al. 1988), but the swarm seismicity has two major implications. First, seismicity is triggered by high fluid pressure, implying that the fault geometry or its kinematics are secondary in importance for forming an orogenic gold deposit as described above. Another major implication is that the period for forming an orogenic gold deposit is reduced to 10, 000–100, 000 years. Such a reduced period is more geologically coherent with the necessity of generating massive amounts of metamorphic fluid as demonstrated by Connolly (2010). Furthermore, this paper emphasises the role of fluid pressure rather than a specific tectonic regime for driving small displacements along gold-bearing structures, as recognised since a long time.
Gold precipitating mechanisms
Gold in orogenic deposits commonly occurs as native gold particles or in sulphides as micro- to nanometric inclusions. Precipitation mechanisms account for such specific gold habits and can be addressed by considering the following reactions and solubility diagram (Figure 3).
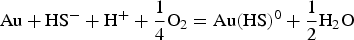
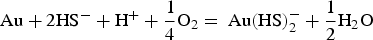
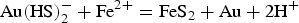
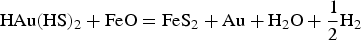
According to Le Chatelier's principle, if the activity of HS− decreases in reactions 2 and 3, gold will precipitate. HS− is consumed when pyrite is formed from reactions 4 and 5, leading to gold precipitation within pyrite. Conversely, native gold is precipitated when HS− is not directly consumed by pyrite formation. According to Figure 3, gold will precipitate if the pH and ƒO2 of the fluid change. CO2 is a ubiquitous constituent of orogenic gold-mineralising fluids. Carbonic acids (H2CO3, HCO−, CO−), which are the aqueous species of CO2 according to pH, act as pH buffers (Phillips and Evans 2004) by keeping fluids in the optimal range of gold solubility at close to neutral pH values. Phase separations, expressed by the coexistence of liquid- and vapour-dominated (CO2) fluid inclusions, imply that CO2 was removed from its initial one-phase fluid (water-carbonic acid). As a consequence, fluids became more acidic, hence precipitating free gold (Figure 3). Fluid acidification is manifested by the occurrence of muscovite as the proximal hydrothermal alteration around gold-bearing veins and the direct association of muscovite with visible gold (Augustin et al. 2017).
Phase separation is a consequence of fluid immiscibility induced by a sudden decrease in pressure (Wilkinson and Johnston 1996). Numerous models have been proposed to link earthquakes along faults in the upper crust and fluid pressure cycles to account for gold precipitation, including as examples the fault valve (Sibson et al. 1988), seismic pumping (Sibson 1987) and the self-equilibrating model (Gaboury and Daigneault 2000). Flashing vaporisation (Weatherley and Henley 2013) has been proposed as a mechanism for precipitating gold and other metals related to earthquakes. This mechanism requires an extreme pressure decrease, well below the hydrostatic fluid pressure, and is thus questionable (Craw 2013), especially considering that even small pressure decreases are sufficient to induce gold precipitation by minor pH changes.
Reactions involving change in ƒO2 have also been proposed. For example, Phillips and Powell (2011) suggested the following reaction to account for gold precipitation in the Witwatersrand basin, where gold is intimately related to carbon seams, veins and nodules.
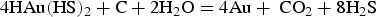
Systemic analysis
From the reviewed key parameters, orogenic gold deposits are evidently the products of successive processes that should occur at specific depths and times with specific rocks, tectonic settings and physicochemical conditions (temperature, pressure, pH and redox conditions). From the lower to the upper crust, the favourable successions required and all the possible ways of aborting the system is presented in Figure 1. First, metamorphic fluids should be generated. Second, ligands (HS−) should be available; otherwise, the system aborts. Third, gold should be present, and fourth, faults should be available and active to transfer the mineralising fluids up to the seismic zone. Finally, efficient mechanisms for gold precipitation are needed; otherwise, fluids will be flushed to the surface (eroded at present exposure) with their gold in solution. This systemic approach also highlights the possibility of forming barren ore zones, which have all the characteristics of a productive system but lack gold. Since most gold exploration geologists have been exposed to such a frustratingly barren system, I review below what appear to be the best criteria for selecting the most promising areas.
The proposed systemic analysis involves a simple linear succession of processes (Figure 1), which may appear outdated regarding innovative non-linear current research trends. According to Ord et al. (2012), hydrothermal systems are open-flow chemical reactors where heat and fluids interact under non-linear and non-equilibrium thermodynamic states. This concept involves cyclical changes in the active hydrothermal zone, from exothermic reactions, creating volume loss to endogenic reactions during mineralisation precipitation, as summarised by Vearncombe and Zelic (2015). For flow-controlled reactors, such as orogenic gold deposits, this model accounts for the transition from regional alteration of carbonate/hydrous silicates to more focussed veining – fracturing – breccia hosting the gold mineralisation (Ord et al. 2012). This concept of open reactor accounts well for the characteristics of the gold mineralisation at the deposit-scale. It also implies that fluids were gold-bearing. Alternatively, the proposed systemic analysis is at a larger scale, addressing key parameters for hydrothermal fluid generation and their gold enrichment. Clearly, both approaches are not mutually exclusive but rather complementary.
Implications for gold exploration worldwide
According to Tomkins (2010) and Zhong et al. (2015), pelitic rocks appear to be the best source for gold, ligands and fluid production, although basaltic rocks could also be a significant source. Nevertheless, the largest gold district (Witwatersrand) is notably hosted along the margin of a very large Archaean sedimentary basin (300*100 km), where up to 4.5 km of sedimentary rocks, including shale and argillite constitute the lower sequence (Phillips and Powell 2011). The Muruntau mine, by far the largest single orogenic gold deposit with >5200 T Au (>150 Moz Au), is hosted in iron-rich and carbonaceous Ordovician to Early Silurian marine clastic rocks (Goldfarb et al. 2014). As advocated by Goldfarb et al. (2014), this extraordinary gold concentration may be related to a very large crustal endowment of early Palaeozoic gold, mainly hosted in disseminated syngenetic sulphides from an underlying sedimentary rock source. The Palaeoproterozoic Birimian belts are composed of abundant graphitic shales rich in nodular pyrite, and these belts, in western Africa, have shown probably the highest rate of discoveries of multi-Moz Au deposits in the last decade (Goldfarb et al. 2017). The largest Palaeozoic goldfields are mostly hosted in sedimentary sequences, including Victoria (Australia) and Mother Lode (California, U.S.A.). Archaean goldfields (Superior, Canada; Yilgarn: Australia) are by comparison mostly hosted in volcanic rocks, but graphitic and pyritic shales are present (Gaboury 2013), although less commonly conserved. Ethane has also been documented from the largest gold deposit in Canada (Detour Lake), indicating that sedimentary carbonaceous rocks were involved at depth (Gaboury 2013). For gold exploration, volcano-sedimentary belts containing shale in their pile should thus be selected as a first priority for providing gold, as far as these rocks have their equivalents buried at depths below the transition of greenschist to amphibolite.
Another key parameter lies in the higher geothermal gradient that rocks should have experienced in relatively low-pressure settings, as highlighted by Tomkins (2010) and Connolly (2010). This feature remains difficult to establish because pressure is not well recorded by minerals in the metamorphic facies of interest, as the boundary between greenschist and amphibolite facies ranges from ∼10 to 30 km (Figure 2). The presence of numerous intrusive bodies bordering the volcano-sedimentary belts should be considered as evidence that partial melting occurred below the belts, providing additional heat at lower pressure. More locally, felsic porphyritic dykes, so common in goldfields and earlier than the mineralisation, may also be considered an indication that magmatic underplating occurred after the main event of mountain building and generated high heat flow. Finally, faults showing evidence of long tectonic histories, with abundant reactivation features such as sericite seams, iron-rich carbonate hydrothermal alteration and quartz-carbonate veinlets, are certainly better candidates for having acted as drainage systems when fluids were available at depth.
Conclusions
The scientific advances reviewed above and their implications for our understanding of orogenic gold deposits were possible mostly because of technological advancements. Development of more sensitive analytical apparatus was instrumental for analysing sulphides, fluid inclusions and various isotopic compositions. Thermodynamic modelling provided limits for orogenic gold deposit formation in terms of temperature and pressure and was important for demonstrating that fluids, as well as ligands and gold, could be generated from the dehydration of carbonaceous sediments and basalts and that a higher geothermal gradient is more favourable. Finally, experiments provided conditions for establishing gold solubility with specific ligands and precipitating mechanisms.
Footnotes
Acknowledgements
This paper is based on an advanced course on orogenic gold deposits developed by the author and from field and laboratory work during the last 25 years in various districts around the world. Numerous graduate students have contributed directly by their M.Sc and Ph.D. work. Thanks to all companies that provided access, funding and support for students and in particular to SEMAFO (M.A. Crevier and R. Roy) for their major support since 2007. I am grateful to Professor S.A.S. Dare (UQAC), for proofreading and D. Craw (University of Otago) and R.J. Goldfarb (China University of Geosciences) for their reviews of an earlier version of the manuscript. I thank the anonymous reviewers and the journal editor N. Phillips for their useful comments.
Disclosure statement
No potential conflict of interest was reported by the author.
