Abstract
Most carbonatites were emplaced in continental extensional settings and range in age from Archean to recent. They commonly coexist with alkaline silicate igneous rocks, forming alkaline-carbonatite complexes, but some occur as isolated pipes, sills, dikes, plugs, lava flows, and pyroclastic blankets. Incorporating cone sheets, ring dikes, radial dikes, and fenitisation-type halos into an exploration model and recognising associated alkaline silicate igneous rocks increases the footprint of the target. Undeformed complexes have circular, ring, or crescent-shaped aeromagnetic and radiometric signatures. Carbonatites can be effectively detected by soil, till, and stream-sediment geochemical surveys, as well as biogeochemical and indicator mineral surveys Carbonatites and alkaline-carbonatite complexes are the main sources of rare earth elements (REE) and Nb, and host significant deposits of apatite, vermiculite, Cu, Ti, fluorite, Th, U, natural zirconia, and Fe. Nine per cent of carbonatites and alkaline-carbonatite complexes contain active or historic mines, making them outstanding multi-commodity exploration targets.
Introduction
Mineralised carbonatites and alkaline-carbonatite complexes are highly sought after, multi-commodity, but poorly understood exploration targets (Verwoerd 1986; Mariano 1989a, 1989b; Richardson and Birkett 1996a, 1996b; Pell 1996; Birkett and Simandl 1999). They are the main source of niobium (Nb; Mackay and Simandl 2014a; Simandl et al. 2018) and rare earth elements (REE; Simandl 2014; Verplank et al. 2016), which are considered critical metals for key economic sectors in industrialised countries (European Commission 2017), and have become popular exploration targets for mining companies worldwide. Alkaline-carbonatite complexes are significant sources of Cu, apatite, fluorite, vermiculite, and other commodities. Most modern studies on carbonatites address their origin and aim to improve our understanding of the Earth's mantle (Bell and Tilton 2001, 2002; Bell and Simonetti 2010; Rukhlov et al. 2015).
Carbonatites and alkaline-carbonatite complexes were considered controversial ever since A.G. Högbom began detailed geological work at the Alnö Island alkaline-carbonatite complex in 1889. The history of carbonatite research up to 1966 is well summarised in the 1st and 2nd editions of the widely available benchmark publication entitled ‘The Geology of Carbonatites’ by Heinrich (1980). A number of additional reviews have been performed since that time, including the book edited by K. Bell (1989) entitled ‘Carbonatites: Genesis and Evolution’. However, the most important review from an exploration geologist's point of view is probably the compilation of Wooley and Kjarsgaard (2008a) entitled ‘Carbonatite occurrences of the word; map and database’, available free of charge for download from the Geological Survey of Canada website. The highly descriptive and factual nature of this publication avoids traps and controversies associated with the genesis of carbonatites, and takes into consideration some of the key suggestions of Mitchell (2005) who attempts to improve currently accepted classification of Le Maître (2002) by making it more relevant to geological mappers and exploration geologists. The descriptive approach, in combination with the availability of data as spreadsheets, will make the Wooley and Kjarsgaard (2008a) compilation a valuable source of information for years to come, since spreadsheets can be easily updated and customised by the user.
Herein, we review the definition and classification of carbonatites and summarize information pertinent for carbonatite exploration such as tectonic setting, morphology, carbonatite-related alkali silicate rock associations, accompanying alteration, and temporal distributions. We also discuss carbonatites and alkaline-carbonatite complexes in terms of resources, exploration methods, and prospectivity. The four main objectives of this paper are to (i) alert the exploration community to the multi-commodity exploration potential of carbonatites and alkaline-carbonatite complexes, (ii) highlight the relevance of tectonic setting, carbonatite-related alkali metasomatism (fenitisation-type metasomatism), and alkaline-carbonatite complex morphology to the exploration geologist, (iii) present relevant advances in exploration methods, and (iv) bridge the gap separating carbonatite-related academic researchers and mineral explorers.
Definitions and classifications
Carbonatites are defined by the International Union of Geological Sciences (IUGS) as igneous rocks containing more than 50% modal primary carbonates (Le Maitre 2002). Depending on the predominant carbonate mineral, a carbonatite is referred to as a ‘calcite carbonatite’, ‘dolomite carbonatite’ (Figure 1), or ‘ferrocarbonatite’, where the main carbonate is iron-rich. If more than one carbonate mineral is present, the carbonates are named in order of increasing modal concentrations. For example, a ‘calcite-dolomite carbonatite’ is composed predominately of dolomite. If non-essential minerals (e.g. biotite) are present, this can be reflected in the name as ‘biotite-calcite carbonatite’.
Upper Fir dolomite carbonatite, British Columbia, Canada; slightly weathered surface; dolomite-brown, Na-amphibole – green, apatite – white. Coin for scale is 2 cm in diameter.
Where the modal classification cannot be applied, the IUGS chemical classification may be used (Figure 2(a)). This classification, based on wt.% ratios, subdivides carbonatites into calciocarbonatites, magnesiocarbonatites, and ferrocarbonatites. For calciocarbonatites, the ratio of CaO/(CaO + MgO + FeO + Fe2O3 + MnO) is greater than 0.8. The remaining carbonatites are subdivided into magnesiocarbonatite [MgO > (FeO + Fe2O3 + MnO)] and ferrocarbonatite [MgO < (FeO + Fe2O3 + MnO)] (Woolley and Kempe 1989; Le Maitre 2002). If the SiO2 content of the rock exceeds 20%, it is referred to as silicocarbonatite. When the IUGS chemical classification is used, iron content in sulphides and oxides should be excluded from consideration, otherwise, magnetite and hematite-rich calciocarbonates or magnesiocarbonatites may be erroneously classified as ferrocarbonatites (Gittins and Harmer 1997). A natrocarbonatite is a special variety of carbonatite consisting mainly of Na–K–Ca carbonates, such as nyerereite [(Na, K)2Ca(CO3)2] and gregoryite [(Na, K,Ca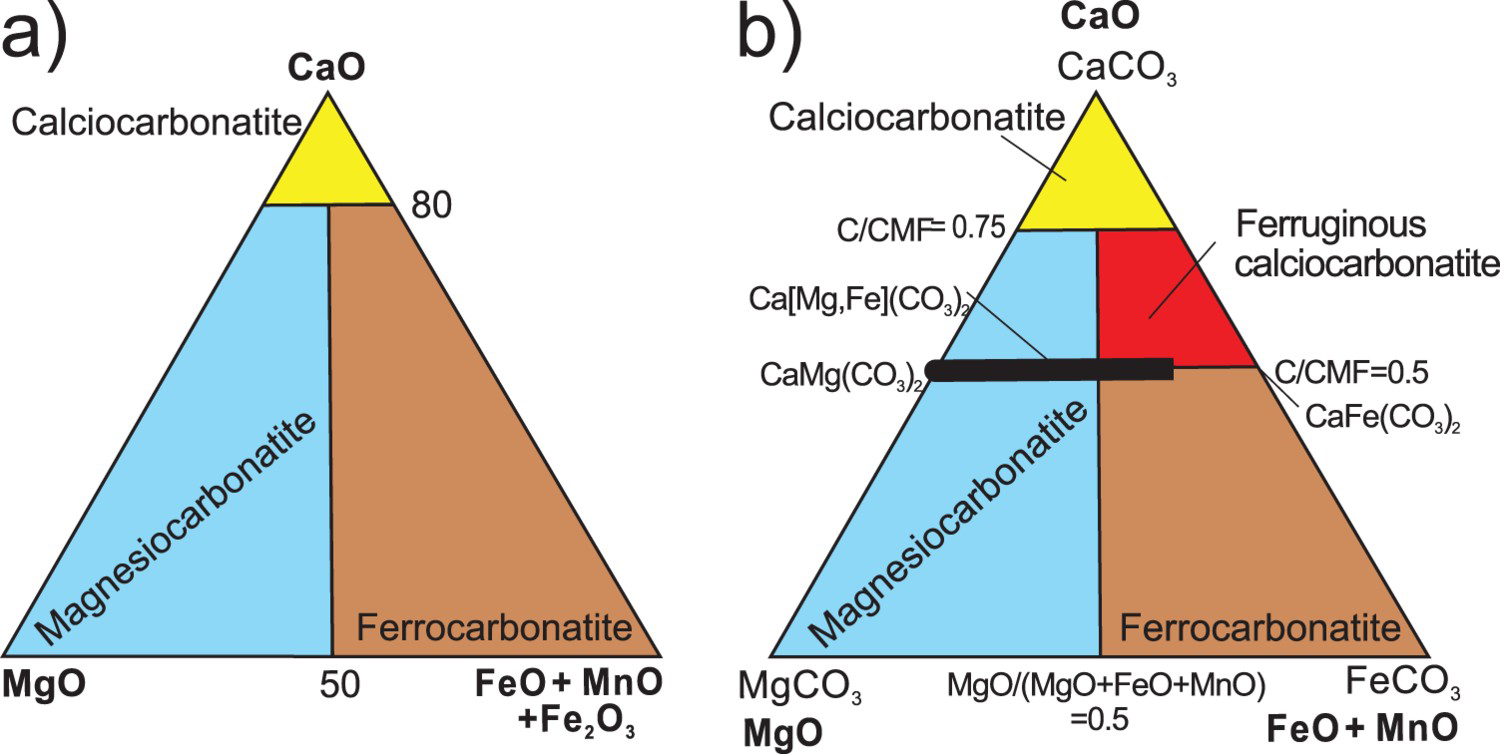
A refinement to the IUGS chemical classification based on molar proportions, proposed by Gittins and Harmer (1997), introduces the term ‘ferruginous calciocarbonatites’ (Figure 2(b)). The boundary separating calciocarbonatites from magnesiocarbonatites and ‘ferruginous calciocarbonatites’ is set at 0.75, above which carbonatites contain more than 50% calcite on a molar basis. Although not universally accepted, Gittins and Harmer's classification is commonly used in studies of carbonatite-hosted ore deposits (e.g. Trofanenko et al. 2016).
A mineralogical-genetic classification of carbonatites was proposed by Mitchell (2005). His benchmark paper points to pitfalls of the IUGS classification, and subdivides carbonatites into ‘primary carbonatites’ and ‘carbothermal residua’. The introduction of the term ‘carbothermal residua’ is significant as it alerts mantle specialists to the fact that not all rocks currently called ‘carbonatites’ by academicians satisfy IUGS definition. In their well-known and widely referenced compilation, Woolley and Kjarsgaard (2008a) have taken into consideration suggestions proposed by Mitchell (2005), but instead of using ‘primary’ and ‘carbothermal residua’ they adopted the terms ‘magmatic’ and ‘carbohydrothermal’. The recognition of ‘carbohydrothermal carbonatites’ by academia is beneficial to the exploration community as it eliminates a deep historic divide between researchers with an interest in the Earth's mantle studying igneous carbonatites sensu stricto (as defined by IUGS), and those that study or explore for carbonatite-related ore deposits. The term ‘carbohydrothermal carbonatite’ is defined by Woolley and Kjarsgaard (2008b) as carbonatite which precipitated at subsolidus temperatures from a mixed CO2–H2O fluid that can be either CO2-rich (i.e. carbothermal), or H2O-rich (i.e. hydrothermal). The carbohydrothermal aspect of some carbonatite phases is in line with number of recent studies highlighting the role of fluids in formation of carbonatite-related REE deposits, such as at Bayan Obo Fe-REE-Nb deposit, Inner Mongolia (Smith et al. 2015; Lai et al. 2016) and Wicheeda Lake carbonatite-related REE mineralisation, Canada (Trofanenko et al. 2016), and with studies that provide constraints on transportation of REE by hydrothermal fluids and their deposition (e.g. Migdisov et al. 2016). Similarly, it may play an important role in the genesis of other carbonatite-related deposit types (e.g. apatite ores at Seligdar apatite deposit, Russia; Prokopyev et al. 2017).
For the benefit of exploration geologists and field mappers, in this paper, we use the term carbonatite to broadly include both magmatic carbonatites and related carbohydrothermal phases, as it may be extremely difficult to distinguish between them on the outcrop or hand specimen scale without follow-up laboratory studies. We use the term alkaline-carbonatite complex to denote occurrences where magmatic and or carbohydrothermal carbonatites are spatially associated with alkaline silicate rocks of igneous or metasomatic origin.
In this review paper, the term ‘carbonatite-related ore deposit’ is used in the broadest sense. It refers to deposits that are genetically or spatially associated with carbonatites or alkaline-carbonatite complexes. A carbonatite-related deposit could consist of unaltered magmatic minerals; minerals that crystalised from carbohydrothermal, hydrothermal, or metamorphic fluids transporting metals; or minerals concentrated by weathering or supergene enrichment, which commonly overly carbonatite protore. Consequently, a ‘carbonatite-related ore deposit’ could be hosted by a carbonatite or alkaline-carbonatite complex, be situated at the contact between the intrusion and host rock, or be distal to the intrusion.
Origin of carbonatites
There are currently three main hypotheses explaining the origin of carbonatite melts: (1) immiscible separation of parental carbonated silicate magmas at crustal or mantle pressures (Kjarsgaard and Hamilton 1989; Lee and Wyllie 1998; Wyllie and Lee 1998); (2) crystal fractionation of parental carbonated silicate magmas such as olivine melilitites or kamafugites (Veksler and Lentz 2006); and (3) low-degree partial melting of carbonated mantle peridotite below 70 km depth (Wyllie and Huang 1976a, 1976b; Eggler 1978; Bell 1989; Dalton and Presnall 1998; Harmer and Gittins 1998; Bell and Rukhlov 2004; Rukhlov et al. 2015). Hypotheses invoking or supporting a possible derivation of carbonatites from the Earth's crust (Lentz 1999; Ferrero et al. 2016), or from the Earth's mantle with some crustal contribution (Cheng et al. 2017; Song et al. 2017), have also been proposed. Furthermore, a recent study based on boron isotopes of carbonatites worldwide suggests that, although most carbonatites may originate in the upper mantle, younger carbonatites (<300 My) probably involve at least partial subducted crustal component (Hulett et al. 2016). It is likely that not all carbonatite-forming melts are of the same origin. However, regardless of their mode of formation, most researchers agree that alkalis (Na and K) play an important role in the genesis of calcite and dolomite carbonatites, and ferrocarbonatite intrusions.
The importance of alkalis in the genesis of carbonatites is consistent with studies of low-temperature (<600°C) natrocarbonatitic lavas from Ol Doinyo Lengai, Tanzania that contain 38–40 wt.% combined Na2O and K2O, 4.5 wt.% F, 5.7 wt.% Cl, approximately 15 wt.% Ca, and less than 1 wt.% combined Mg and Fe (Keller and Krafft 1990; Keller and Zaitsev 2012). Petrographic and geochemical evidence from extrusive carbonatites, such as Homa Mountain, Kenya (Clarke and Roberts 1986); Tinderet, Kenya (Deans and Roberts 1984; Zaitsev et al. 2013); Kerimasi, Tanzania (Hay 1983; Zaitsev 2010; Guzmics et al. 2011); and Kaluwe, Zambia (Ngwenya and Bailey 1990), as well as evidence from intrusive carbonatites such as Bol'shaya Tagna, Russia (Andreeva et al. 2006); Oka, Canada (Chen et al. 2013); Guli, Russia (Kogarko et al. 1991); and Palabora, South Africa (Sharygin et al. 2011), suggests that calcite- and dolomite-rich carbonatites are residues or cumulates derived from alkali-bearing (moderately alkaline) melts. Recently, ‘halite intergrown with, and included in, dolomite, calcite, apatite, REE- fluorocarbonates, pyrochlore, fluorite, and phlogopite of a dolomitic carbonatite’ was reported at the St.-Honoré alkaline-carbonatite complex (Canada) by Kamenetsky et al. (2015), further supporting the argument that parental magmas of primary carbonatites are enriched in Na and Cl. A comprehensive review of fluid and melt inclusion studies of intrusive carbonatites by Veksler and Lentz (2006) further documents the alkaline nature of melts to which intrusive carbonatites are related or from which they originated. A hypothesis suggesting that calcite carbonatites evolve from natrocarbonatite parental magmas by loss of alkali due to fenitisation was advanced by Le Bas (1981) and Woolley (1982); however, this hypothesis was challenged, if not discredited, by Twyman and Gittins (1987), Mitchell and Belton (2004), and Mitchell (2005).
A thermal barrier, as established by Cooper et al. (1975), was a major obstacle to most hypotheses proposing that natrocarbonatites evolved from moderately alkaline calcite carbonatites. However, recent experimental work suggests that natrocarbonatite lavas may evolve from a moderately alkaline calcite carbonatite precursor (which is unmixed from nephelinites) by crystal fractionation of calcite and apatite (Weidendorfer et al. 2017). This appears possible because of the ability of halogens to suppress the calcite liquidus, thereby eliminating the thermal barrier (Weidendorfer et al. 2017). There is no consensus regarding the origin of magnesium- and Fe-rich carbonatites. They are believed to either evolve from early calcite carbonatites by fractional crystallisation or be of hydrothermal origin (Thompson et al. 2002), essentially ‘carbohydrothermal’.
Exploring for carbonatites: Where to look and what to look for
Tectonic setting
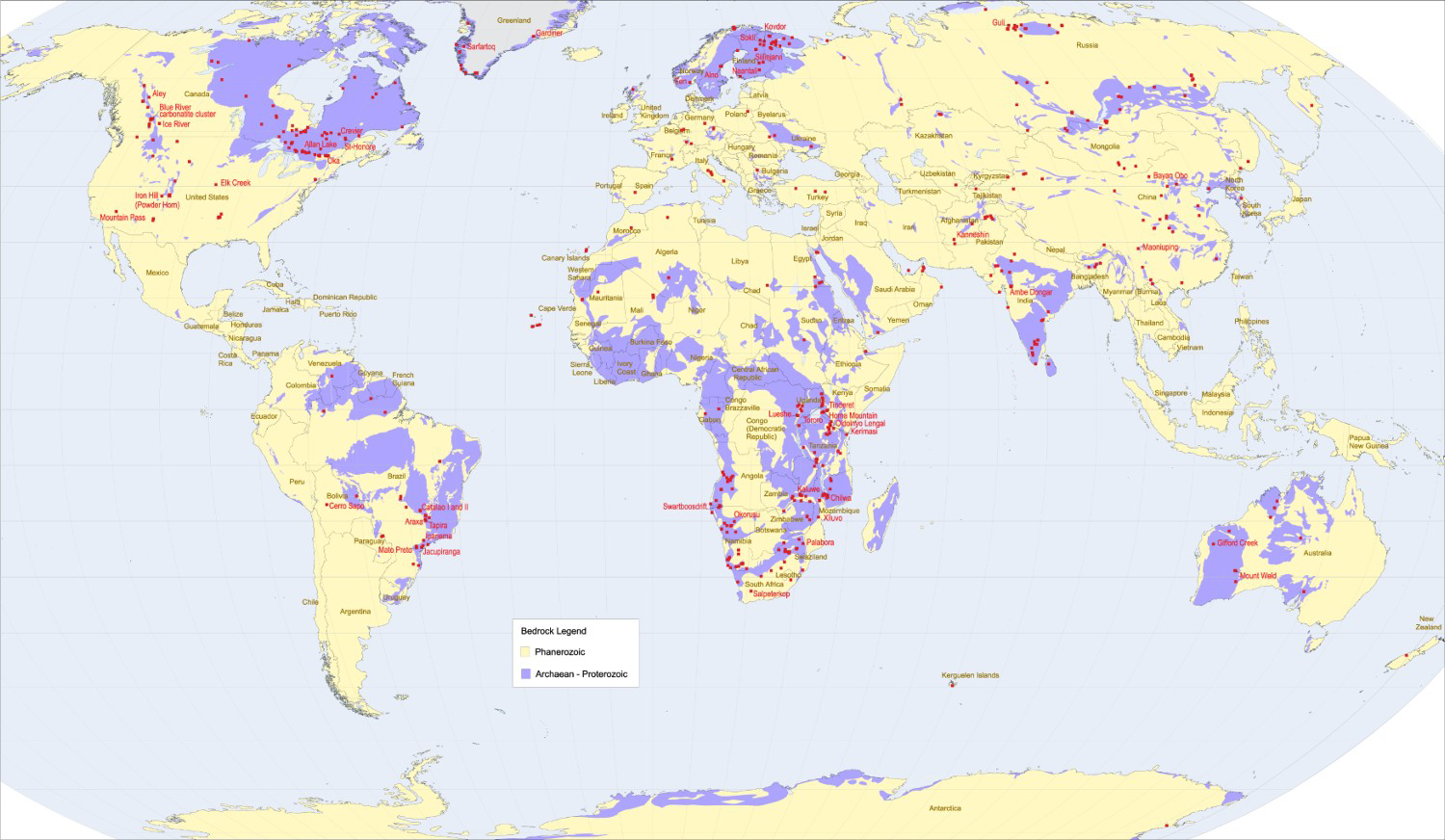
Most carbonatites and alkaline-carbonatite complexes are emplaced in continental (88% cratonic, 10.5% non-cratonic) settings (Figures 3–4) in Archean and Proterozoic rocks, or in Phanerozoic rocks underlain by a Precambrian basement (Sage and Watkinson 1991; Woolley and Kjarsgaard 2008a; Pirajno 2015). They form in extensional tectonic settings (Bailey 1974, 1977, 1992), along major linear trends related to large-scale intra-plate fracture zones, in association with doming features (crustal arching), or in relation to slab windows in subducting plates (Duke 2009; Duke et al. 2014). The link between these tectonic features and intense magmatic activity means that many carbonatites are also temporally and spatially related to ‘large igneous provinces’ (Pirajno 2000; Ernst and Bell 2010). Some researchers (e.g. Nelson et al. 1988; Bell 2001, 2005; Pirajno 2015) consider mantle plumes essential to carbonatite genesis.
Tectonic setting of carbonatites and carbonatite complexes. Based on data from 527 carbonatites from Woolley and Bailey (2012). World map of carbonatite occurrences and their spatial relationship to Proterozoic and Archean rocks. Areas underlain by unsubdivided Proterozoic and Phanerozoic rock are not shown. Modified from Woolley and Kjarsgaard (2008a).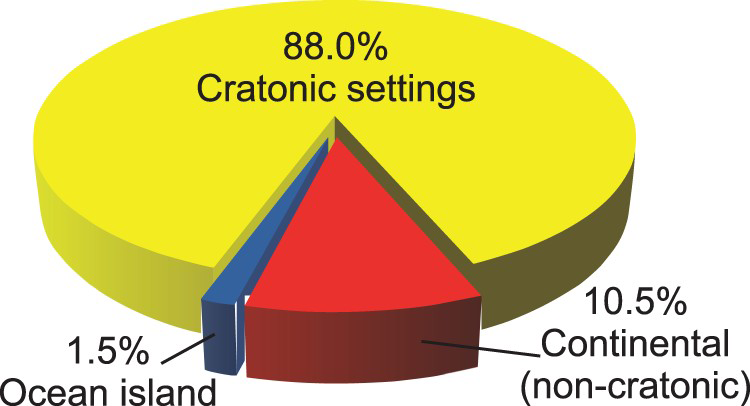
Carbonatites in orogenic settings are sometimes referred to as ‘post-collisional’ (Chakhmouradian et al. 2008; Woodard and Hetherington 2014). This is an unfortunate term because carbonatites that are found in orogenic settings may have been emplaced before a transition from extensional to compressional tectonic regimes, or during post-orogenic extensional relaxation and collapse prior to dynamothermal metamorphic climax. Examples of specific orogenic settings containing carbonatites are: (1) British Columbia Alkaline Province, Canada (Pell 1994, Millonig et al. 2012); (2) The Himalayan collision zone in western Sichuan, China (Hou et al. 2006, 2015); (3) Northwest Pakistan (Tilton et al. 1998); and (4) The Great Indian Proterozoic Belt, India (Leelanandam et al. 2006). The Naantali carbonatite in southwest Finland is also located in orogenic setting (Woodard and Hetherington 2014).
Carbonatites are identified in three oceanic island regions: (1) The Canary Islands; (2) The Cape Verde Islands, and (3) The Kerguelen Islands; all of which are located off the African continent (Figure 4; Woolley and Kjarsgaard 2008a). However, it is possible that these islands are underlain by remnants of continental lithosphere stranded during drifting of the African plate (Bonadiman et al. 2005; Woolley and Bailey 2012).
Temporal patterns of carbonatite emplacement: Can age be used to screen exploration targets?
Ages of carbonatite emplacement on a global scale
Radiometric ages of cabonatites vary from Archean (e.g. Short Lake, Canada, 2684±170 Ma; Rukhlov and Bell 2010) to very recent (e.g. Ol Doinyo Lengai, Tanzania, which is considered an active volcano). Frequency plots of carbonatites with well-established ages indicate that younger carbonatites are more abundant than older ones (Figure 5). This trend may be due to changes in tectonic activity during the Neoarchean Era that permitted emplacement of carbonatites. As favourable conditions became more widespread, carbonatites became more common (Woolley and Kjarsgaard 2008a). The difficulty to distinguish carbonatites from carbonate-rich metasediments in areas affected by a post-carbonatite dynamothermal metamorphism may be another reason (Woolley and Kjarsgaard 2008a). Veizer (1992) proposed that the overall exponential decrease in the probability of carbonatite preservation with increasing age of crustal segments is the main reason for this trend. Variations in boron (B) concentrations in young (<300–400 Ma) carbonatites and their isotopic composition (δ11B) suggest that greater quantity of subducted material may have reached plumes, and indirectly contributed increase in carbonatite activity during that time period (Hulett et al. 2016).
The frequency of all known carbonatite occurrences with isotopic ages decreases with time. With the exception of carbonatites younger than 150 Ma, the frequency of mineralised carbonatite occurrences shows a similar decrease. Figure based on data from Woolley and Kjarsgaard (2008a), with minor updates.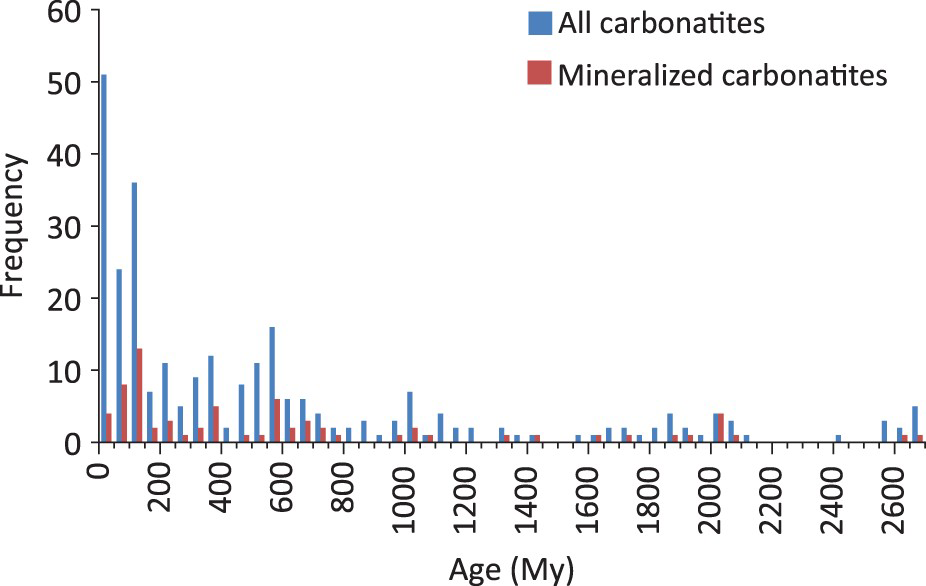
Carbonatites with mineralogy similar to Ol Doinyo Lengai natrocarbonatite may have been relatively common in Earth's history; however, because they convert rapidly into calcite carbonatites in the near surface environment (Zaitsev and Keller 2006), they are not preserved in the geological record. Destruction of nyerereite (the main natrocarbonatite-forming mineral) in surface environments takes place over a few months or less according to the following alteration sequence: nyerereite → pirssonite → calcite → shortite (Zaitsev et al. 2008).
Woolley and Bailey (2012) covered the importance of tectonic controls on carbonatite emplacement. Their study focused on six well documented carbonatite provinces: (a) East Africa (Kenya, Uganda, Tanzania, and one occurrence in Zambia); (b) Namibia and Angola; (c) Eastern Russia (East Tuva, Enisei, East Sayan, Baikal, and Aldan); (d) Greenland; (e) Ontario and southwest Quebec; and (f) Northern Europe, including the Kola Peninsula, northern Norway, Sweden, and Finland. They recognised up to five episodes of carbonatite emplacement separated by hundreds of millions of years in some of these provinces. They pointed out that within at least three of these carbonatite provinces, including one comprising Ontario and southwest Quebec (Figure 6), plumes can be eliminated as a mode of carbonatite emplacement because of the lack of relationship between the direction of the plate movement and the age of intrusions. Instead, carbonatite magmatism appears to coincide with periods of reactivation of faults and fissures associated with changes in tectonic regimes (extensional period). Consider as an example the Ontario and Southwest Quebec carbonatite province covered by Woolley and Bailey (2012). Although age dates from K-Ar on biotite and Rb-Sr on whole rock may have been reset by medium grade metamorphic events, the U-Pb age dates on zircon and baddeleyite corroborate the original data and confirm that the timing of each carbonatite emplacement episode coincides closely with a period of relaxation linked to a major orogeny (Figure 6).
Temporal relationship between intensive carbonatite magmatism and main orogenic events in Ontario, Canada and southwest Quebec, Canada. Potassium–Ar dates on biotite and Sr–Rb dates on whole rock from Woolley and Bailey (2012) and U–Pb dates on zircon and baddeleyite from Rukhlov and Bell (2010). Age estimates for Grenville, Trans Hudson, and Kenoran orogenies from Tohver et al. (2006), Corrigan et al. (2009), and Moser et al. (2008), respectively.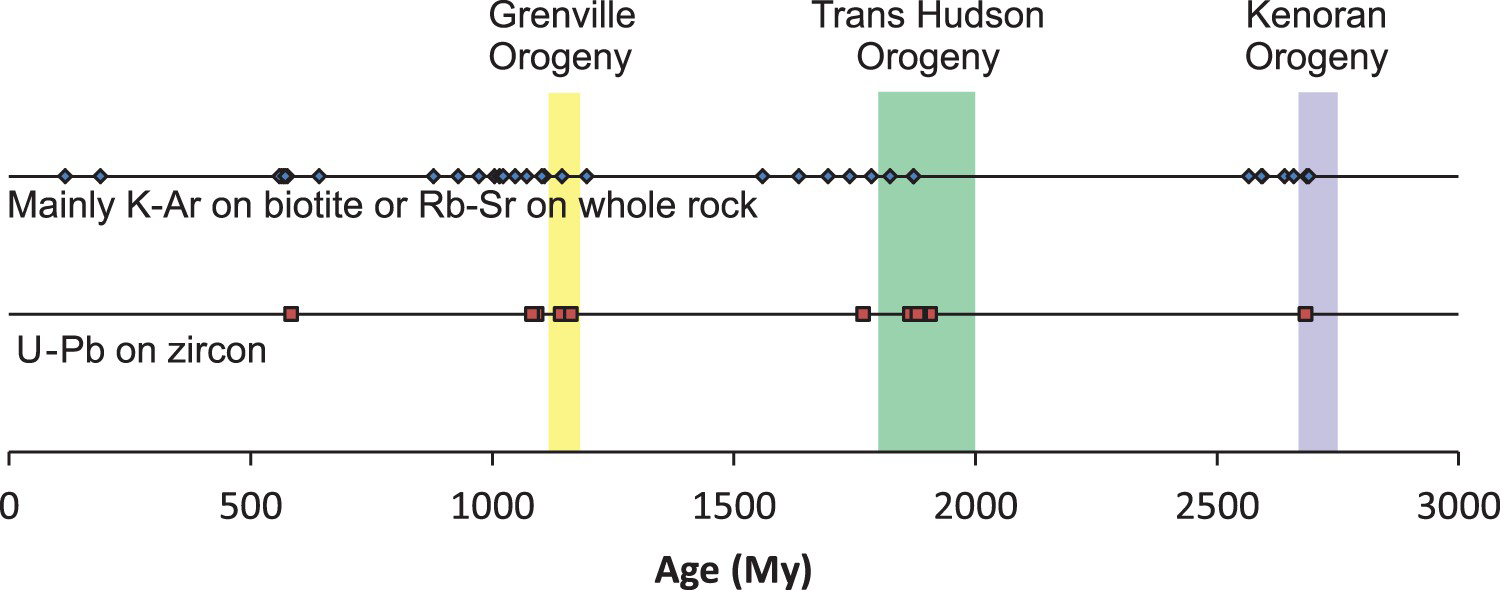
The frequency of mineralised carbonatite occurrences follows a similar pattern to the frequency of all carbonatites, except for the interval between 150 and 0 Ma (Figure 5). Relatively shallow erosional levels may explain this divergence, with few exceptions, as most carbonatite-related mineralisation is spatially related to intrusive carbonatite phases at depth, whereas their volcanic (near surface) equivalents are commonly low grade or barren (unless subjected to supergene enrichment). In view of the broad temporal range of carbonatites on the world scale and the divergence of trends between non-mineralised and mineralised carbonatites within the 150 to 0 Ma time interval, the age alone is of limited use for screening carbonatite exploration targets.
Age relationships between individual barren and mineralised carbonatite pulses on the complex-scale
Within the same alkaline-carbonatite complex, calcite carbonatites are generally the oldest (Richardson and Birkett 1996a). Radiometric ages are seldom used systematically to age date discrete intrusive phases of the same alkaline-carbonatite complex; however, where field relationships are observable, calcite carbonatites are cut by magnesian (dolomite carbonatite) pulses, which in turn predate more Fe±Mn-rich (ferrocarbonatite) pulses as illustrated by the Chilva Island Complex (Garson and Smith 1958; Woolley 1982). However, in many field studies of cabonatite complexes, the cross-cutting relationships are either not discernable or only dolomite carbonatite or ferrocarbonatite pulses were observed. Magmatic mineralisation (e.g. Nb, apatite, magnetite, and baddeleyite) is commonly hosted by early to intermediate magmatic pulses (Richardson and Birkett 1996a). Such mineralisation may be remobilised at later stages, possibly as a result of post-magmatic overprint (e.g. Aley carbonatite, British Columbia; Chakhmouradian et al. 2015). The REE mineralisation commonly post-dates Nb mineralisation (Richardson and Birkett 1996a), as exemplified by the dolomite carbonatite-hosted REE mineralisation at Wicheeda Lake deposit, British Columbia (Trofanenko et al. 2016), where the third generation dolomite and REE mineralisation is interpreted as hydrothermal. Another example of where Nb mineralisation is post-dated by REE mineralisation is the St. Honoré alkaline-carbonatite complex, Quebec. Here, the principal host to Nb mineralisation is a crescent-shaped, dolomite carbonatite cone sheet, surrounded in part by barren calcite carbonatite; the late core of this complex is strongly enriched in REE (Lafleur and Ayad 2012).
Although in most cases REE mineralisation is paragenetically late and appears hosted by carbonatites that could be considered carbohydrothermal, frequently cited research of Mariano (1989a) suggests that, at the Mountain Pass REE deposit (California), carbonatite-hosted REE mineralisation is late magmatic based largely on textures. However, the lack of reported pyrochlore and columbite or their alteration products, which are common in typical magmatic carbonatites, and local enrichment in Ba suggests that by today's terminology, the host to REE (bastnaesite-parisite) mineralisation may be ‘carbohydrothermal’ in origin. A follow-up study may be justified to confirm Mariano's (1989a) original findings and eliminate incertitude.
Carbonatite-associated igneous rocks
Most intrusive carbonatite occurrences are part of alkaline-carbonatite complexes and are spatially tied to one or more intrusive silicate rock groups including melilitolites, ijolites, alkali gabbros, feldspathoid syenites, syenites, kimberlites, and lamprophyres or their volcanic equivalents (Woolley and Kjarsgaard 2008a, 2008b). Worldwide, only 24% of carbonatite rocks are not part of alkaline-carbonatite complexes (Woolley and Kjarsgaard 2008a).
Peridotites and pyroxenites, commonly found near ijolites and melilitolites, are not considered a distinct association because they are interpreted as cumulates (Woolley and Kjarsgaard 2008a). Nevertheless, these rocks are important from the exploration point of view because they are commonly observed in deeply eroded alkaline-carbonatite complexes, providing indirect information about the depth of erosion and, by extension, the mineralisation potential of the complex (Frolov 1971; Epshteyn and Kaban'kov 1984).
Phoscorites are magnetite, olivine, apatite rocks usually associated with carbonatites (Le Maitre 2002) and ultramafic rocks of alkaline-carbonatite complexes (Woolley and Kjarsgaard 2008b). In some cases, there is gradation between ultramafic rocks and phoscorite (e.g. Yonghwa phoscorite-carbonatite pipe; Seo et al. 2016). The definition presented by Le Maitre (2002), is very restrictive because olivine commonly retrogrades into pyroxene, amphibole, and serpentine. A much broader definition and classification of phoscorites are anchored in Russian literature (e.g. Yegorov 1993; Krasnova et al. 2004) and proposes that phoscorite should be redefined as a ‘plutonic ultramafic rock comprising magnetite, apatite, and one of the silicates, forsterite, diopside, or phlogopite’. According to Krasnova et al. (2004), most of the 21 phoscorite (sensu lato) occurrences that they are aware of are mineralised. Should this broader definition be applied worldwide, the number of phoscorite occurrences would increase substantially. For example, in British Columbia, the Aley carbonatite and several carbonatites in the Blue River area would be considered to contain lenses or fragments of phoscorite (Figure 7). This has important implications from an exploration point of view because, within several alkaline-carbonatite complexes, phoscorites are host to Nb, baddeleyite (natural zirconia), apatite and iron, with a potential to recover scandium as a co-product (Rudashevsky et al. 2004; Ivanyuk et al. 2016; Kalashnikov et al. 2016).
Phoscorite lenses and irregular pods (sensu Krasnova et al. 2004) consisting of magnetite with minor apatite, dolomite, and serpentine (possibly replacement of olivine) are enclosed in dolomite carbonatite, Aley carbonatite, British Columbia, Canada. Marker for scale measures 14 cm in length.
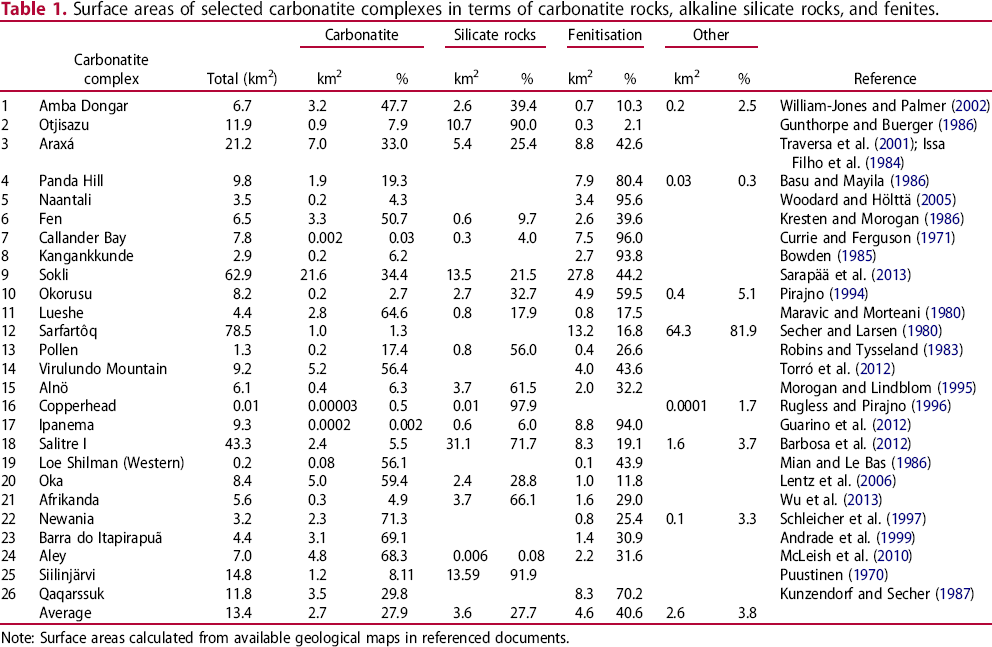
Recognition of carbonatite-related intrusive silicate rocks is also important. In some cases, carbonatite represents only a minor part of an alkaline-carbonatite complex. Consequently, recognising the spatial association of carbonatites to phoscorite and commonly associated intrusive silicate rocks substantially increases the size of an exploration target (Figure 8; Table 1).
The average surface area of carbonatites is approximately 3 km2. In most cases, the carbonatites are surrounded by alkali silicate rocks, which are surrounded by a zone of fenitisation. If an explorationist recognizes the carbonatite, related silicate rocks (∼2 km2), and associated fenitised zone (∼5 km2), the target area increases to 10 km2. This figure is based on data from 26 carbonatite complexes, with surface areas varying from 0.01 to 78.5 km2, listed in Table 1. Surface areas of selected carbonatite complexes in terms of carbonatite rocks, alkaline silicate rocks, and fenites. Note: Surface areas calculated from available geological maps in referenced documents.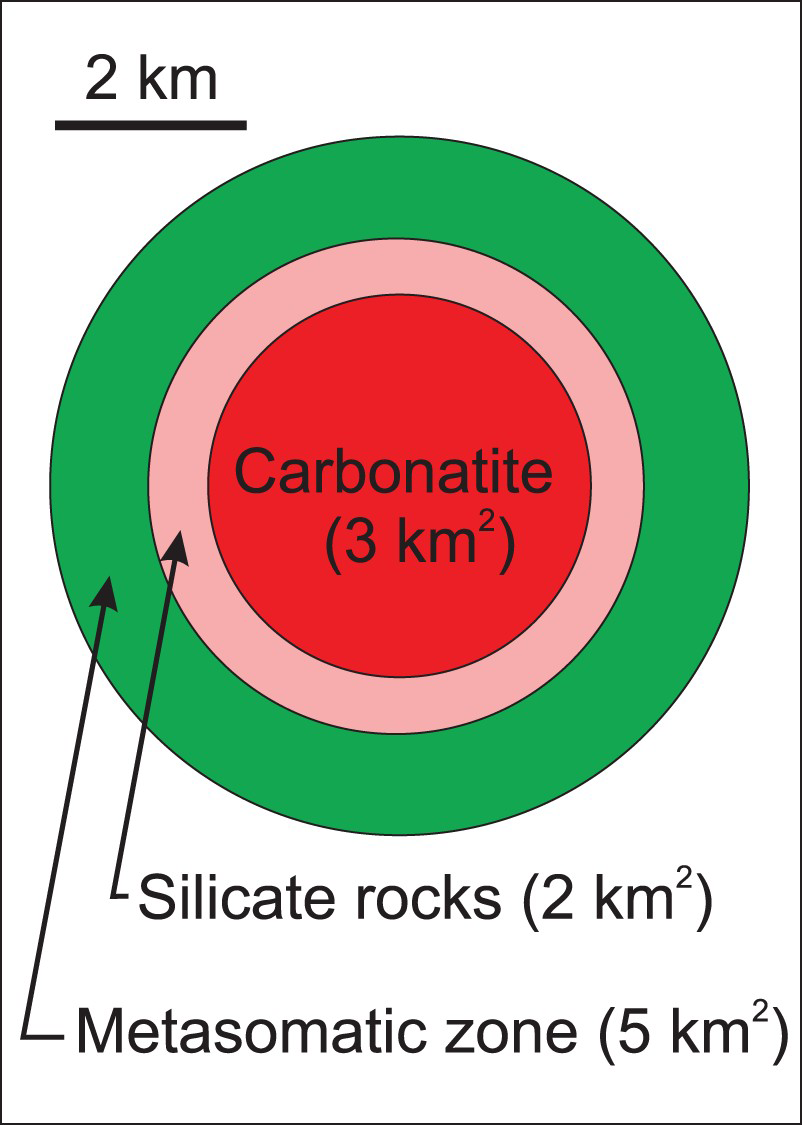
Alkali metasomatism
Most intrusive carbonatites, alkaline-carbonatite complexes, and many agpaitic and miaskitic alkaline intrusions are surrounded by country rock affected by intrusion-related metasomatism. Metasomatism is defined as: ‘a solid state process by which the chemical composition of a rock is altered in a pervasive manner and which involves the introduction and/or removal of chemical components as a result of the interaction of the rock with fluids’ (Zharikov et al. 2007). Fenitisation is defined as: ‘a metasomatic process leading to the formation of fenites’ Schematic representation of bi-metasomatic fenitisation-type interaction between carbonatite melt and related fluids with country rock. Direction of migration of elements is indicated by arrows. Minerals commonly observed in country rock affected by fenitisation-type metasomatism are listed. Nearly monomineralic, massive, K-feldspar-rich fenite locally observed at the contact of Lonnie carbonatite with metapelite country rock, British Columbia, Canada. Green-blue rock consisting of Na-amphibole, Na-clinopyroxene, and feldspar produced by fenitisation-type metasomatism observed in contact with carbonatite (pale to medium brown, left upper corner of picture). The late centimeter-thick white vein, cutting green-blue rock, consists of calcite; Aley Nb deposit, British Columbia, Canada. Variation in intensity of fenitisation-type metasomatism perpendicular to the strike of a 30 m thick carbonatite lens. Intensity of metasomatism decreases with increasing distance from calcite carbonatite contact, as recorded by disappearance of aegirine and Na-amphibole over the distance of approximately 25 m. Lonnie carbonatite, British Columbia, Canada.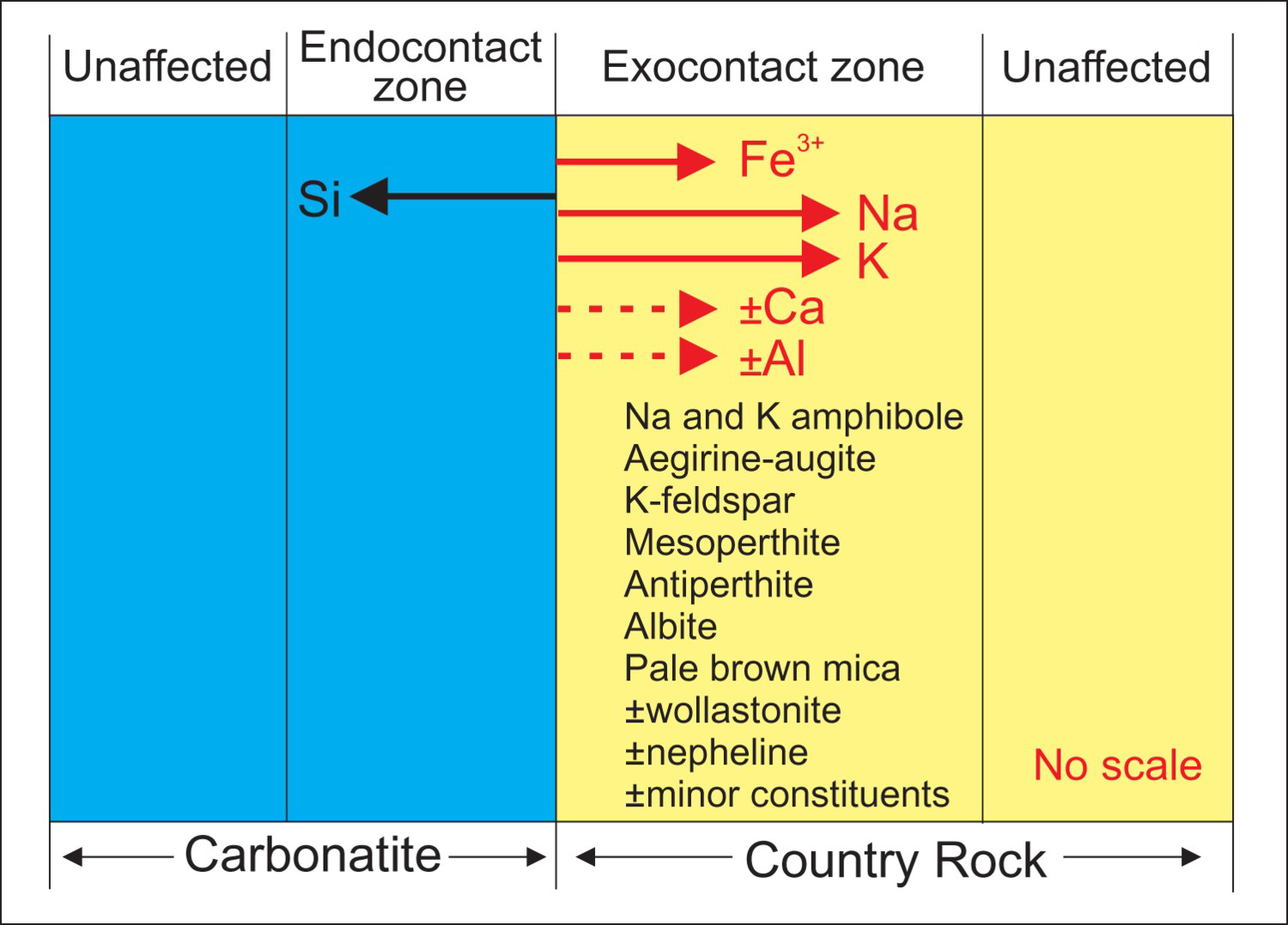



As with carbonatite-associated silicate rocks, recognising fenitisation-type metasomatic halos during exploration significantly increases the footprint of the carbonatite or alkaline-carbonatite complex (Figure 8). The documented thickness of fenitisation-type metasomatc zones varies from a few centimetres to several kilometres and their shape is influenced to a large extent by the geometry of permeable zones open to metasomatic fluids.
Estimates of surface areas affected by fenitisation related to carbonatites or alkaline-carbonatite complexes based on maps in scientific and government publications are contained in Table 1. This data demonstrates that in some cases the fenitisation-type metasomatic aureole substantially increases the footprint of the carbonatite or alkaline-carbonatite complex. For example, at Panda Hill, Naantali, Callander Bay, Kangankkunde, and Ipanema, fenitisation represents more than 80% of the complex in terms of surface area.
For many alkaline-carbonatite complexes, it is difficult or impossible to determine if fenitisation-type metasomatism is related directly to the carbonatite or to the spatially associated ijolite rocks (Morogan 1994). Where more than one source of magmatic or carbohydrothermal fluids is involved, several fenitisation-type metasomatic patterns may overlap, complicating the vectoring procedure. For the purpose of this paper, we adopt the classification developed by Heinrich (1985) and popularised by Le Bas (2008), which divides fenites into ‘sodic’, ‘potassic’, and ‘sodic-potassic’ (an intermediate between the two end-members). Sodic fenites are characterised by the presence of Na-rich amphiboles and pyroxenes, and K- and Na-feldspars. In extreme cases of Na fenitisation, albitite rock will develop (Denaeyer 1966; Le Bas 2008). Within carbonatite complexes, sodic fenites are believed to form earlier, at higher temperatures, and at deeper levels than potassic fenites (Le Bas 2008). Potassic fenites consist mainly of K-rich feldspars (orthoclase or microcline), and in some cases may contain low-alumina phlogopite or biotite. Potassic fenites most commonly develop adjacent to or above the upper levels of intrusive calcite and dolomite carbonatites. These fenites plot in the leucite solidus field on the quartz-nepheline-kalsilite diagram (Le Bas 2008). Historically, it has been suggested that within alkaline-carbonatite complexes, early calcite carbonatites are accompanied by strong Na-K fenitisation-type metasomatism, and that there is no fenitisation-type metasomatism related to ferrocarbonatites (Le Bas 1987); however, there are a number of cases where ferrocarbonatite-related fenitisation-type metasomatism has been recognised, such as the Swartbooisdrift complex, Namibia (Drüppel et al. 2005) and the Gifford Creek ferrocarbonatite complex, Western Australia (Pirajno et al. 2014).
Morphology and geometry of alkaline-carbonatite complexes
This section contrasts the morphologies of undeformed carbonatites in unmetamorphosed intracratonic settings to those that are strongly deformed (pre-metamorphic) in high grade metamorphic terranes. In the latter case, there may be a need to use microscopy, geochemistry, stable isotopes, and rock associations to distinguish between carbonatites and carbonate metasediments.
Undeformed carbonatites in extensional settings
The classic carbonatite model (Figure 13(a)) proposed by Garson and Smith (1958) was popularised by Heinrich (1980) and Bowden (1985), and is still in use (e.g. Pirajno 2015). This model, based largely on field observations from Chilva alkaline province in southern Malawi, fits many complexes from Eastern Africa and elsewhere. More recent models (Figure 13(b, c)) have been proposed by Le Bas (1977, 1987), and Sage and Watkinson (1991). The model of Le Bas (1987) displays well the age relationships between lithological units and highlights fenitisation-type overprints (Figure 13b). The model produced by Sage and Watkinson (1991) displays a limited number of lithologies (Figure 8c) relative to the Garson and Smith (1958) model; however, it better depicts the relationship between the volcanic edifice and crater facies. The erosional level shown in this model (Figure 13(c)) corresponds to the current level of exposure displayed by carbonatites in southwest Quebec and Ontario, Canada.
Morphology of carbonatite complexes as proposed by: (a) Garson and Smith (1958); (b) Le Bas (1987); and (c) Slightly modified from Sage and Watkinson (1991) to show convex and concave nature of ring dikes and cone sheets, respectively. None of these models provides a complete picture; however, in combination these models provide a useful summary for exploration geologists.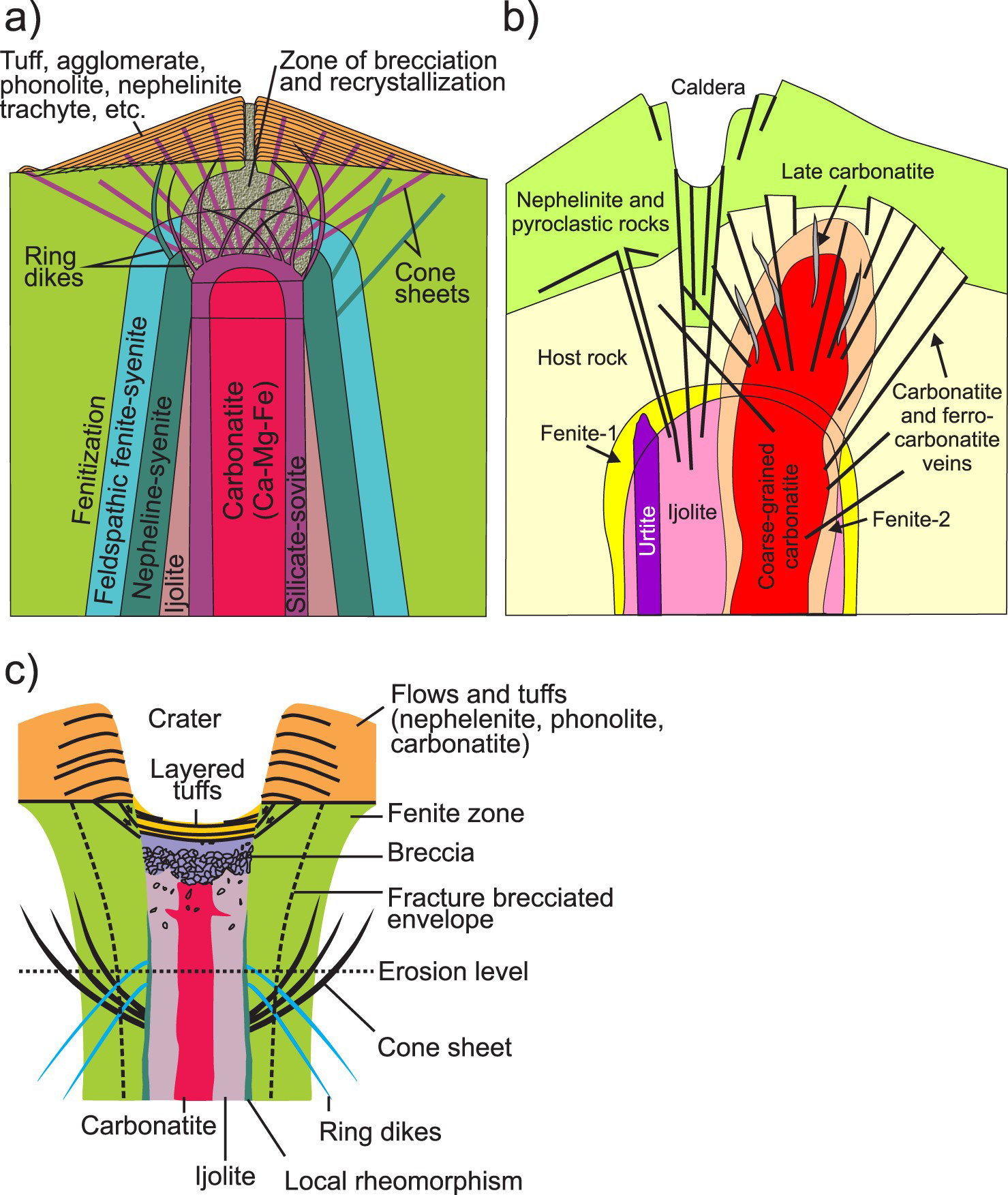
No model depicts all of the possible rock associations encountered in alkaline-carbonatite complexes, or is universally applicable. At deep erosion levels, carbonatites are commonly spatially associated with ultramafic rocks. At moderate levels, they are spatially associated with pyroxenites and jacupirangites, and with ijolites and nepheline syenites at progressively shallower levels (Garson and Smith 1958).
Regardless of the model that we adopt, subvolcanic (hypabyssal) rocks in carbonatite complexes commonly form radial dikes, cone sheets, and ring dikes (Figure 14). Examples include Salpeterkop structure, South Africa (Verwoerd 1990); Alnö, Sweden (Eckermann 1942, 1948, 1966; Kresten 1980); Gardiner complex, Greenland (Nielsen 1980); and Oka, Canada (Gold et al. 1967). These dikes and cone sheets may consist of nephelinites, alkali-rich mafic rocks, melilitolites, phonolites, trachytes, lamprophyres, ijolites, or carbonatites. The emplacement of a magma chamber (carbonatite or otherwise) and its related radial dikes, cone sheets, ring dikes, and doming is fundamentally understood based on a combination of geological field observations, laboratory experiments, and numerical modelling (Koide and Bhattacharji 1975; Kresten 1980; Walter and Troll 2001; Klausen 2004; Bistacchi et al. 2012; Andersson et al. 2013). Where anisotropic regional (tectonic) stress exceeds intrusion-related stresses, steeply dipping or subvertical dike swarms are likely to form. Where local stresses related to a shallow magma chamber predominate, subvertical radial dikes; subvertical to outward dipping, semicircular to crescent shaped, nearly concentric ring dikes; and inward dipping, circular, oval, or crescent shaped, concentric cone sheets are expected to form (Figure 14). These intrusion-related features can be used to vector towards mineralisation. Radial dikes are expected to intersect at the vertical projection of the intrusive centre. Because ring dikes are subvertical to steeply dipping, the outermost of these roughly coincide with the horizontal extent of the magma chamber. Cone sheets dip toward the intrusive centre. In most older models, the projections of the visible portions of cone sheets are interpreted to meet near a single point coinciding with an explosion focus, such as at Chilwa, Malawi (Garson and Smith 1958); Alnö, Sweden (Eckermann 1966); and Homa Mountain, Kenya (Le Bas 1977). In such models, the cone sheets farthest away from the magma chamber will have the shallowest dip; however, modelling by Bistacchi et al. (2012), shows that at least in some cases cone sheets may be coaxial with the same apical angle. At the levels where magma pressure exceeds lithostratigraphic pressure, zones of brecciation develop and doming takes place. The volcanic edifices of carbonatites and related intrusions commonly consist of tuffs, agglomerates, and lava flows consisting of nephelinites, mellilite-bearing rocks, and, in some cases, phonolites and trachytes (Garson and Smith 1958).
Schematic three-dimensional presentation of relationship between magmatic chamber, ring dikes, radial dikes, cone sheets, and volcanic neck of a carbonatite complex.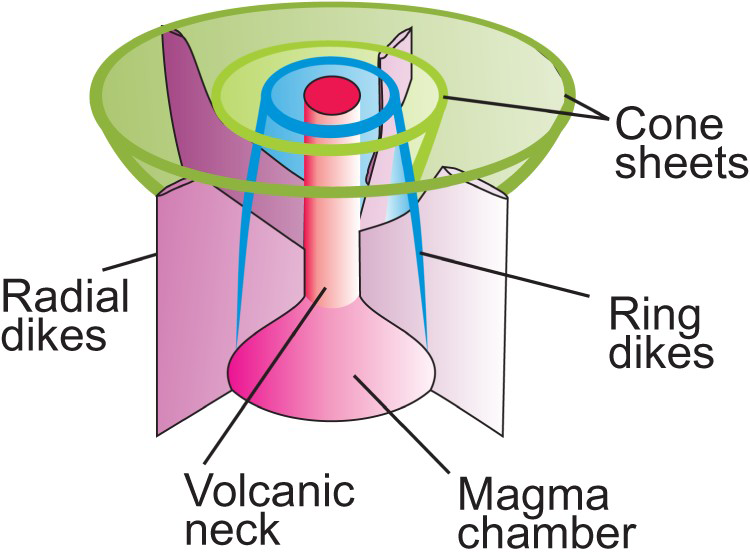
Distinction between carbonatites and sedimentary carbonate rocks affected by high grade metamorphism and tectonic activity
Distinction between well exposed intrusive carbonatites and carbonate rocks of sedimentary origin does not present a problem in unmetamorphosed intracontinental extensional settings because the morphology of lithological units, cross-cutting relationships (Figure 13(a, b, c); Figure 14), primary magmatic and carbohydrothermal textures, and mineralogical compositions are preserved (e.g. Barker 1993). Post-emplacement deformation significantly changes the shape of a carbonatite and related mineralised zones, and commonly associated metamorphism modifies the mineralogical, chemical, and textural characteristics of the ore and gangue minerals. This is especially true where dynamothermal metamorphism reaches upper amphibolite or granulite facies, at which point many distinguishing features are partially or completely obliterated. Examples include Dahomeyide suture zone, West Africa (Attoh et al. 2007); carbonatite-marble dikes of Abyan Province, Yemen Republic (Le Bas et al. 2004); metacarbonates from Paleoproterozoic collision zone of the Borborema province, NE Brazil (EJ dos Santos et al. 2013; RV Santos et al. 2013); and some carbonatites of the British Columbia alkaline province, Canada (Figure 15a, b; Kulla and Hardy 2015). Both marbles derived from carbonatites and those from sedimentary carbonates may appear concordant with country rocks, be repeated by folding (Figure 16) or faulting, display metamorphic layering, and develop identical granoblastic, porphyroblastic, lepidoblastic, and mylonitic textures.
Morphology of the Upper Fir carbonatite, part of the Blue River carbonatite cluster, British Columbia, Canada shown in (a) vertical longitudinal section and (b) vertical cross-section perpendicular to longitudinal section [blue vertical line in (a)]. Metamorphosed carbonatites commonly appear concordant with lithological contacts and metamorphic layering within the country rock and are repeated by folding (simplified from Kulla and Hardy 2015). Folds within dolomite carbonatite highlighted by white apatite-rich layers; Aley Carbonatite, British Columbia, Canada.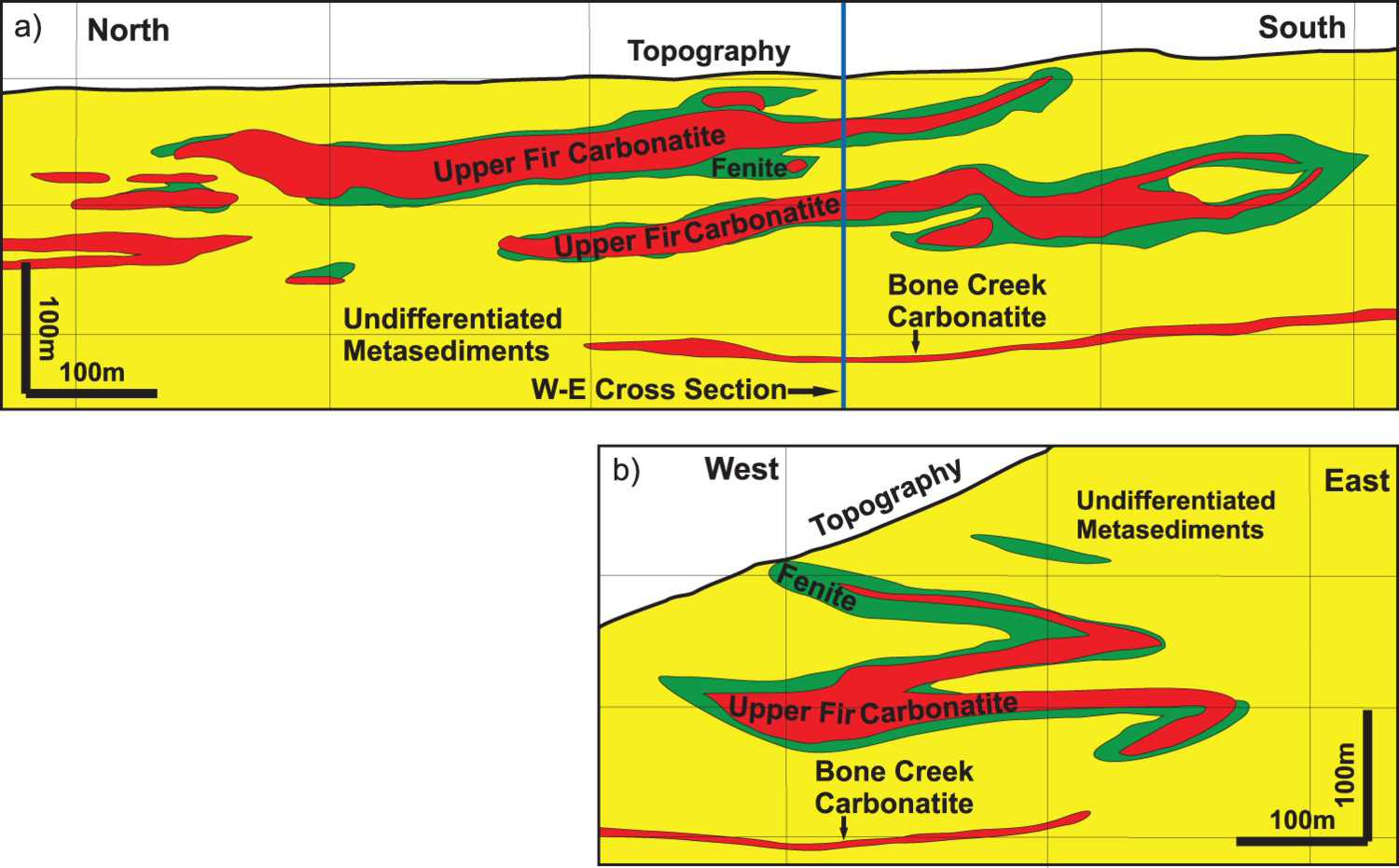

Metacarbonates (marbles) of uncertain origin that are spatially related to alkaline silicate gneiss, blue–green amphibolite (preserved fenitisation zone), or pyroxenite should be suspected to have originated from a carbonatite protolith. Shoshonitic igneous rocks or their metamorphic equivalents commonly indicate a transition in tectonic regimes involving block faulting and uplift within island arcs (Morrison 1980) and have been reported in association with post-collisional carbonatites by Hou et al. (2006), Chakhmouradian et al. (2008), and Woodard and Hetherington (2014). The presence of blue–green Na- and K- amphiboles, perovskite, pyrochlore, columbite-(Fe), and fersmite in marble is evidence of a carbonatite origin. Le Bas et al. (2004) note that the presence of aluminous minerals such as anorthite, scapolite, and ‘fassaitic’ pyroxene are (subsilicic aluminium ferrian diopside; Ca(Mg, Fe3+,Al)(Si, Al)2O6) indicative of a sedimentary origin; however, relying on these minerals to be diagnostic of a non-carbonatite origin in metamorphosed rocks may be misleading, as carbonatites can incorporate Al-rich country rock xenoliths or xenocrysts, and volcanic carbonatites may have been in contact with an Al-rich regolith or interbedded with Al-rich sediments prior to metamorphism.
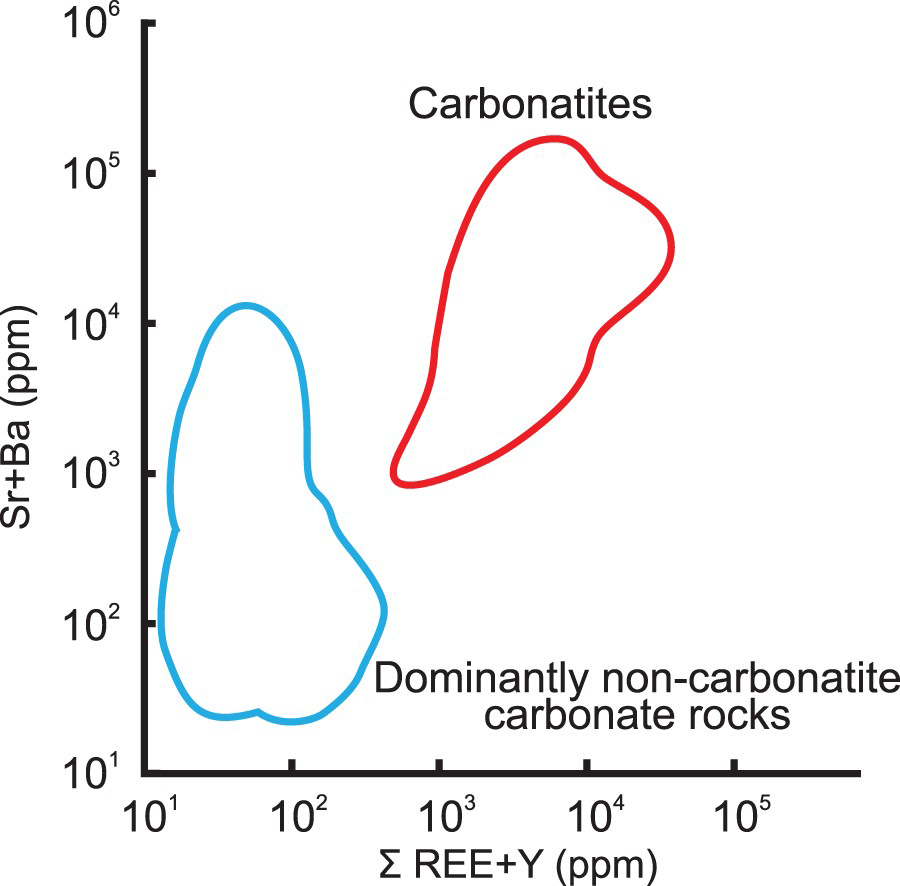
If diagnostic textural and mineralogical criteria are missing, geochemical analysis of REE, Nb, Sr, and Ba concentrations is recommended. Carbonatites are strongly enriched in light rare earth elements (LREE) and are characterised by steep chondrite-normalised REE patterns, without a Eu anomaly (Figure 17). The [Sr + Ba]-[REE + Y] discrimination diagram (Figure 18) proposed by Samoilov (1991) can also be used to discriminate between marble of sedimentary and carbonatitic origins.
Chondrite-normalised REE plot of representative samples from Aley cabonatite (Nb-deposit) and Wicheeda carbonatite (REE-deposit), British Columbia, Canada, and field of unmineralised carbonatites from Hornig-Kjarsgaard (1998). The [Sr + Ba] to [REE + Y] discriminative bivariate plot is one of the criteria recommended to distinguish carbonatites from marbles derived from carbonate sedimentary rocks (Samoilov 1991).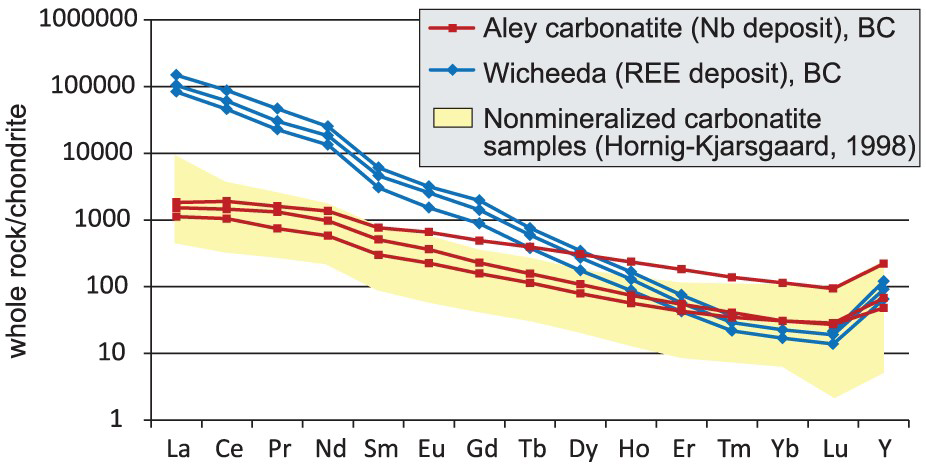
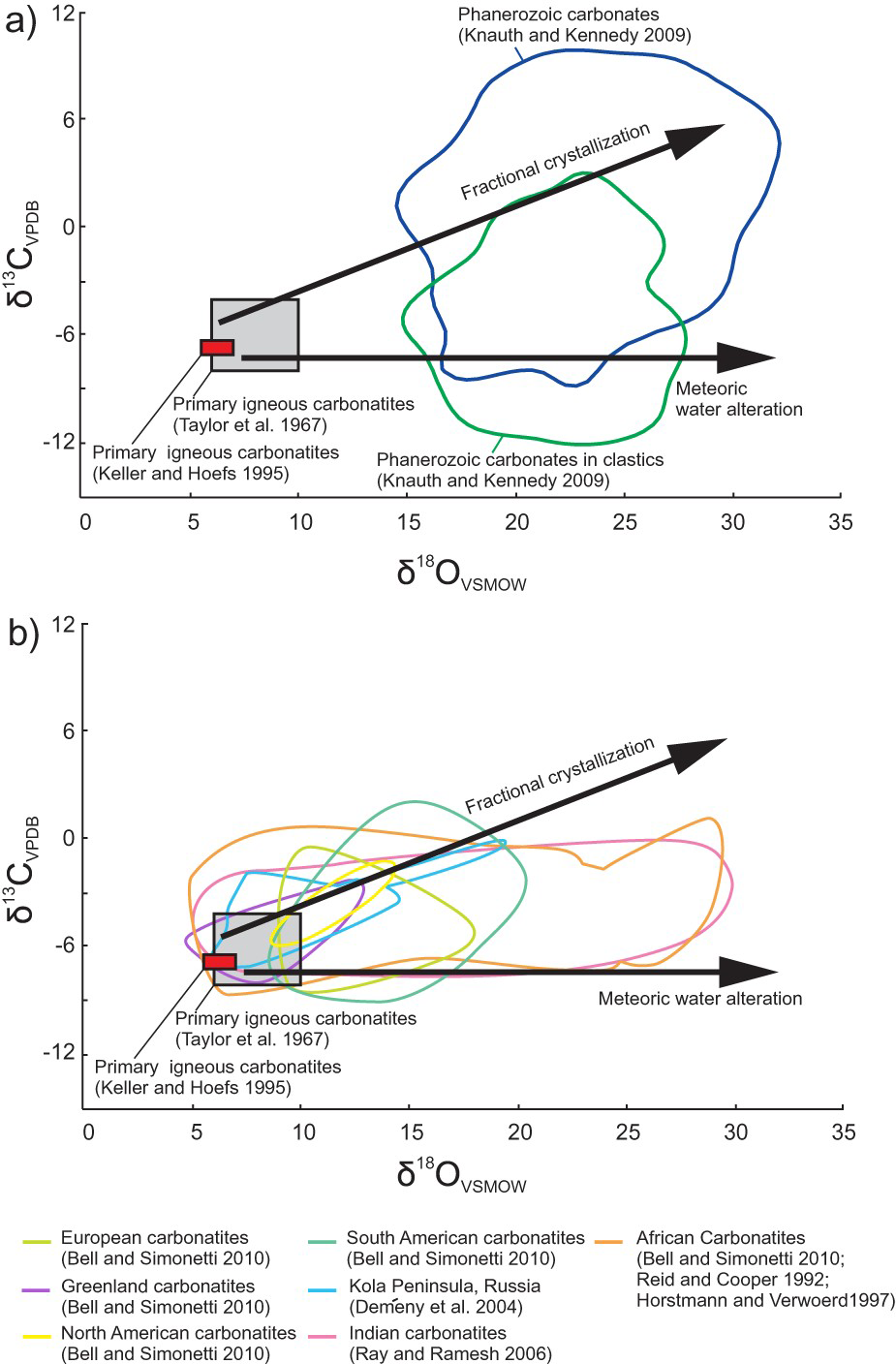
Isotopic composition can also be used to differentiate carbonatites from marbles of sedimentary origins. Ideally, magmatic carbonatites have well constrained isotopic compositions and on δ18O and δ13C diagrams plot within the primary carbonatite fields (Figure 19(a)) defined by Taylor et al. (1967) and Keller and Hoefs (1995), easily distinguishing them from carbonates of sedimentary origin. However, carbonatites that are interpreted as primarily based on mineralogical criteria may plot outside of these fields, as illustrated on Figure 19(b) by the data from Indian carbonatites from Ray and Ramesh (2006). The horizontal trend (no change in δ13C), indicates the effects of alteration by meteoric water, whereas an increase in both δ18O and δ13C is commonly attributed to fractional crystallisation (Figure 19(a)). If a marble plots within or near the primary carbonatite fields (Figure 19(a, b)) or if the data form a trend originating from them, then this marble is likely derived from a carbonatite protolith, as is the case of several carbonatites metamorphosed to upper amphibolite facies in the British Columbia Alkaline province, Canada. If a marble has higher than expected δ18O and plots along the fractional crystallisation trend, it could have been derived from (1) a carbonatite protolith that interacted with late carbohydrothermal or metamorphic fluids; (2) metamorphism of carbonate-rich sedimentary rocks; or (3) metamorphism of hydrothermal carbonate bodies. Distinguishing metamorphosed carbonatites and carbohydrothermal carbonates from metacarbonates of sedimentary origin may be impossible if they plot within the Phanerozoic carbonates fields (e.g. African and Indian carbonatites; Figure 19(a, b)).
δ18O and δ13C plots showing: (a) Composition of primary igneous carbonatites, fractional crystallisation and meteoric water alteration trends, and composition of Phanerozoic sedimentary carbonates and carbonates in clastic sedimentary rocks; and (b) Compositional fields of European, Greenland, South American, Indian, African, and Kola peninsula carbonatites. Compositional fields corresponding to African and Indian carbonatites are elongated along the trend representing alteration by interaction with meteoric water. The Kola Peninsula carbonatite field follows the trend representing fractional crystallisation. Chondrite value from McDonough and Sun (1995
).
Carbonatite-related ore deposits
Carbonatites and alkaline-carbonatite complexes and, in some cases, associated fenitisation-type metasomatic zones and overlying regolith (including zones of supergene enrichment) are favourable hosts of metallic and industrial mineral deposits (Figure 20). Reviews and compilations of carbonatite-related mineralisation are provided by Deans (1966), Heinrich (1980), Mariano (1989a, b), Pell (1996), Richardson and Birkett (1996a, 1996b), Birkett and Simandl (1999), Woolley and Kjarsgaard (2008a), Berger et al. (2009), Simandl (2014), and Mackay and Simandl (2014a, 2015).
Vertical section of a hypothetical carbonatite mineralising system displaying the relationship between metallic and industrial mineral deposits relative to lithological units and geological contacts. The ‘distal’ carbo-hydrothermal fluid-related mineralisation or hydrothermally remobilised mineralisation (away from alkaline-carbonatite complex) and residual deposits within weathered crust above the carbonatite complex are also highlighted. Bi – biotite, Px- pyroxene. Modified from Laznicka (2006).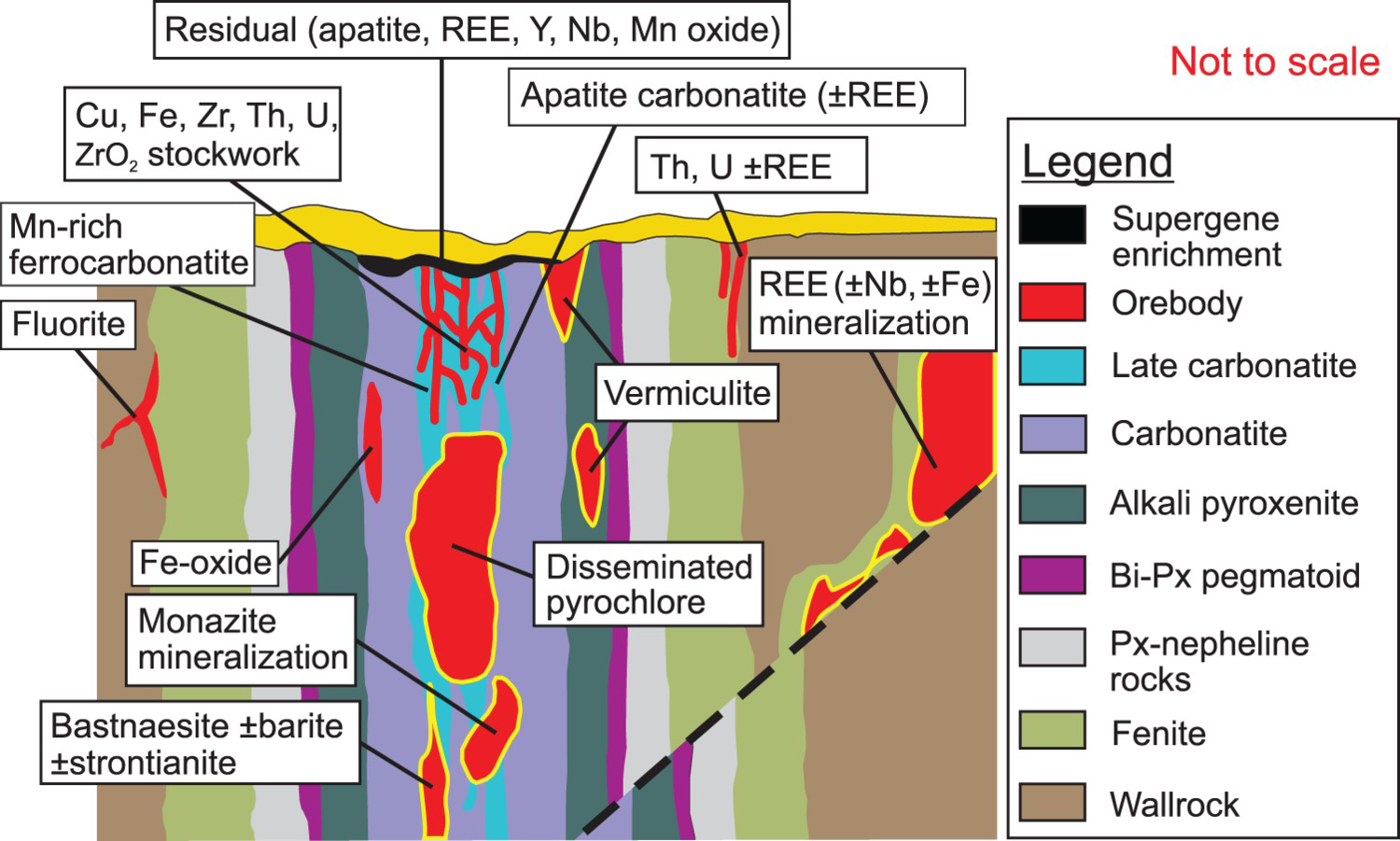
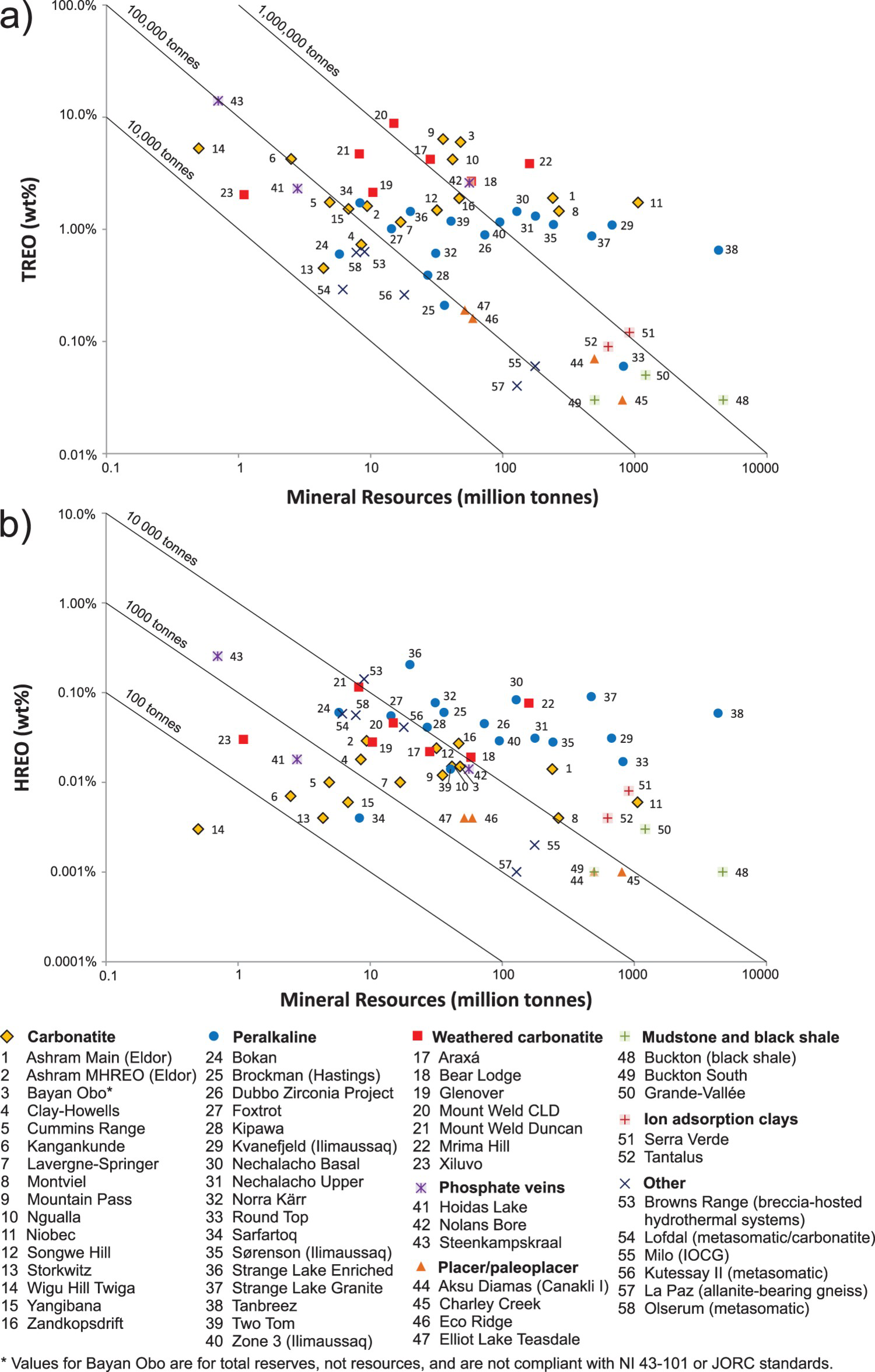
Alkaline-carbonatite complex-related ore deposits represent large resources in terms of REE (Figures 21(a, b) and 22(a), e.g. Bayan Obo, China; Maoniuping, China; Mountain Pass, USA; and Mount Weld, Australia). Most of these deposits are strongly enriched in LREE and have a high LREE/REE(total) ratio relative to peralkaline intrusion-related, and ion adsorption clay deposits (Mariano 1989a, Simandl 2014; Verplank et al. 2016); however, they also contain significant resources of heavy rare earth elements (HREE; Figure 22(b)). Under current market conditions, concentrates of REE-bearing minerals from typical alkaline-carbonatite complex related deposits are the main source of LREE. The ion adsorption clay deposits are low grade relative to alkaline–carbonatite intrusion-related- and peralkaline intrusion-related deposits and can't compete effectively against high-grade-carbonatite-related deposits as a source of LREE (Figure 22(a, b)). However, because peralkaline complex-related REE deposits are metallurgically challenging (Verbaan et al. 2015), and carbonatite concentrates have a high LREE/REE(total) ratio, the REE-bearing ion adsorption deposits which benefit from simple and cost-effective metallurgy are currently the main source of HREE (Simandl 2014). With the exception of the LREE-enriched, loparite and eudialyte–bearing, nepheline-feldspar-aegirine pegmatite at Lovozero (Russia), there is currently no production of REE from peralkaline intrusion-hosted deposits.
Examples of main REE and Nb minerals and their concentrates; (a) Mixture of monazite [(Ce, La,Nd, Th)PO4] and bastnaesite [(Ce, La)(CO3)F] forms orange-brown fracture fillings in dolomite carbonatite; pen for scale; Wicheeda Lake prospect, British Columbia, Canada. (b) Bastnaesite concentrate containing 60–65 wt.% rare earth element oxides (REO), Mountain Pass carbonatite, California, USA; scale in millimetres. (c) Pyrochlore crystal [(Na, Ca)2Nb2O6(OH, F)] from the Upper Fir carbonatite, British Columbia, Canada; scale in millimetres (d) Pyrochlore concentrate, Niobec Mine, Quebec, Canada; scale in millimetres. Grade and tonnage of REE-bearing deposits associated with carbonatite complexes in terms of (a) Total REO (TREO) and (b) Heavy REO (HREO) relative to pegmatites, peralkaline complexes, and rare element granites. Updated from Simandl (2014).
Alkaline-carbonatite complex-related REE mineralisation may consist of unweathered carbonatite rock such as at Mountain Pass, USA, (Castor 2008) and St-Honoré, Canada, (Lafleur and Ayad 2012) or corresponds to overlying regolith such as at Mount Weld, Australia (Lottermoser 1990) and Araxá, Brazil (Neumann and Medeiros 2015). The REE mineralisation tends to be concentrated in late carbonatite pulses forming central breccia zones, ring dikes, or cone sheets, and fracture and open space fillings within these features. The REE are probably concentrated in carbohydrothermal fluids and melts produced by fractional crystallisation from carbonatitic melts (Richardson and Birkett 1996a; Song et al. 2016). In several cases, the REE mineralisation is interpreted as distal to carbonatite or alkaline-carbonatite complex; however, in most of these cases the relation of REE mineralisation to the ultimate source of fluids from which it formed is not clear or is controversial (e.g. the famous Bayan Obo REE-Nb-Fe deposit in Central Mongolia; Smith et al. 2015; Lai et al. 2016; Liu et al. 2018). Based on the close spatial relationship of many REE deposits to faults, fractures, suture zones, breccias, and other paleo-permeable zones, in combination with paragenetic studies of ore minerals, and in agreement with supporting experimental evidence indicating that REE, Nb, and Ta can be transported by hydrothermal or carbo-hydrothermal fluids (e.g. Williams-Jones et al. 2012; Timofiev et al. 2015, 2017, Migdisov et al. 2016; Trofanenko et al. 2016), it is reasonable to expect the presence of REE (± Nb) deposits exploration targets distal to the alkaline-carbonatite complexes (Figure 20).
Alkaline-carbonatite complex-related deposits are also the main source of Nb (Figures 21(c, d) and 23(a); e.g. Catalão, Brazil; Lueshe, Democratic Republic of Congo; and St. Honoré, Oka, and Aley, Canada) as summarised by Verwoerd (1986), Mariano (1989b), Berger et al. (2009), Mackay and Simandl (2014a), and Simandl et al. (2018). The Nb mineralisation may be part of the carbonatite rock unit (e.g. St-Honoré, Canada; Tremblay et al. 2015), carbonatite-associated alkaline rock (e.g. Crevier deposit, Canada; Solgadi et al. 2015), fenitised zone, or overlying regolith (e.g. Catalão I, Brazil; Cordeiro et al. 2011; and Araxá, Brazil; Issa Filho et al. 2015). Although high Ta concentrations are expected mainly in pegmatites and peralkaline intrusion-related deposits (Mackay and Simandl 2014a), some deposits related to alkaline-carbonatite complexes have comparable Ta concentrations to pegmatites and contain significant resources of tantalum (e.g. Upper Fir carbonatite and Crevier alkaline-carbonatite complex, Canada; Figure 23(b)). Unfortunately, under current market conditions, low Ta2O5/[Ta2O5 + Nb2O5] ratio in concentrates makes carbonatites unattractive for Ta extraction using conventional technology (Simandl et al. 2018).
Grade and tonnage of (a) Nb and (b) Ta deposits associated with carbonatite complexes relative to pegmatites, peralkaline complexes, and rare element granites. Diagonal lines indicate tonnage of contained Nb2O5 and Ta2O5. From Simandl et al. (2018). Most grade and tonnage references are available in Mackay and Simandl (2014a). Abbreviations: (HR) hard rock ore, (W) weathered ore, (HR+W) hard rock and weathered ore combined.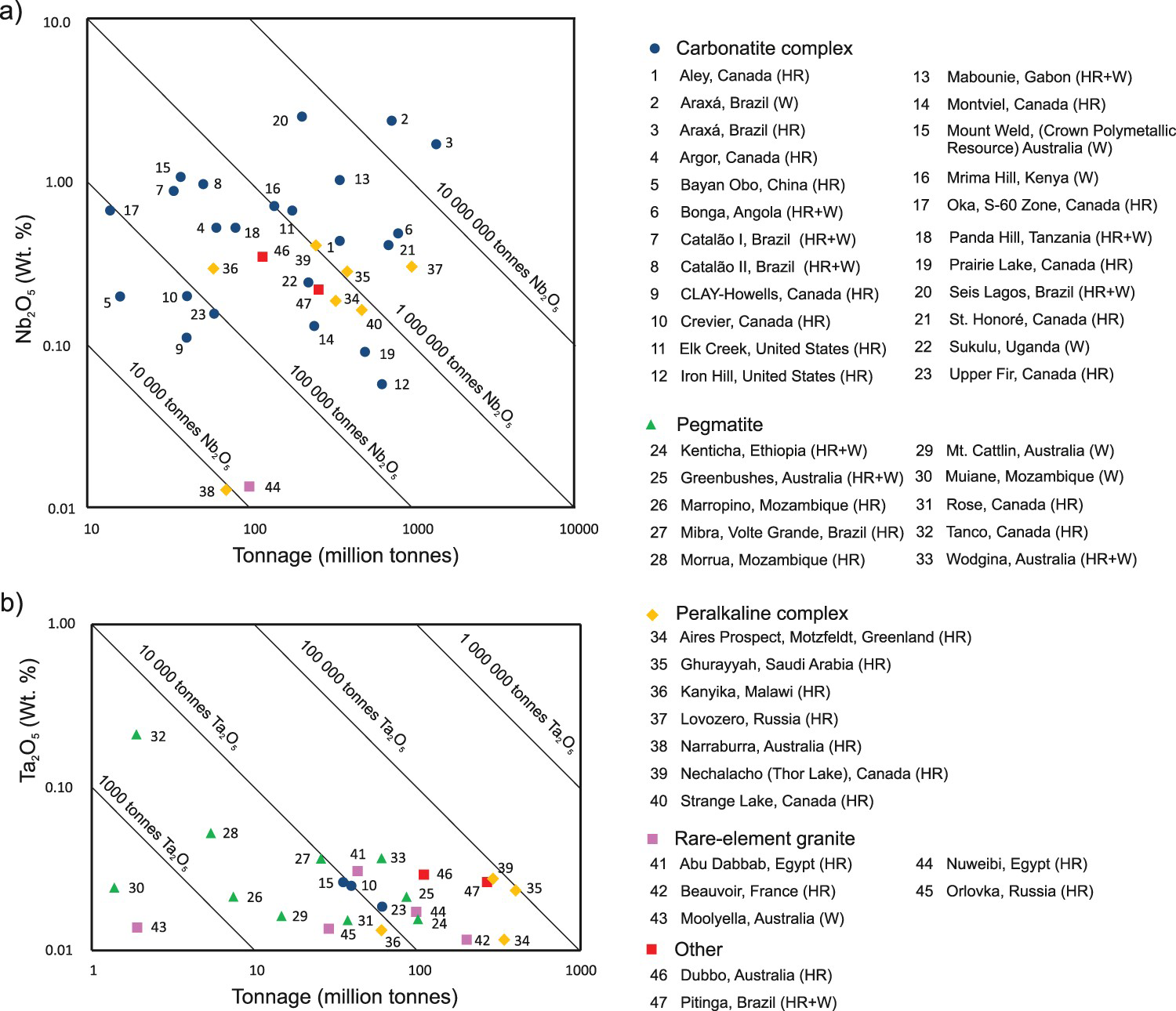
Vermiculite and phlogopite deposits are predominantly hosted by mafic or ultramafic rocks of the alkaline-carbonatite complex (e.g. Northern pyroxenite at Palabora, South Africa; Heinrich 1970; Fourie and De Jager 1986) near the contacts of carbonatites with these rocks, or within mafic country rocks (e.g. Upper Fir carbonatite, Canada; Simandl et al. 2010). Most carbonatite-related apatite deposits currently in production, such as Tapira, Brazil (Capponi et al. 2009); Ipanema, Brazil (Born 1986); Catalão I, Brazil (Carvalho and Bressan 1986); and Matongo, Burundi (Decree et al. 2015) were enriched by weathering. Examples of exceptions are the Siilinjärvi mine, Finland (O'Brien et al. 2015), and Cajati mine, Jacupiranaga Complex, Brazil (Alves 2008). Copper, U, Th, and baddeleyite (natural zirconia) were produced for decades from the Palabora carbonatite-phoscorite complex in South Africa (Heinrich 1970; Clarke 1981; Milani et al. 2017), but baddeleyite is currently produced only from the Kovdor deposit in Russia (Dickson 2015).
Other materials produced from carbonatites or related rocks are: iron (e.g. Kovdor, Russia; Dickson 2015; Bayan Obo, China; Smith et al. 2015; and Palabora, South Africa; Heinrich 1970); fluorite (e.g. Mato Preto, Brazil; Okorusu, Namibia; and Amba Dongar, India; Hagni 1999); carbonates for lime and cement production (e.g. Tororo, Uganda and Xiluvo, Mozambique; van Straaten 2002; and Jacupiranga, Brazil; Alves 2008); and sodalite for use as dimension, ornamental, and semi-precious stone (e.g. Swartboosdrift, Namibia; Menge 1986; and Cerro Sapo, Bolivia; Schultz et al. 2004). Some alkaline-carbonatite complexes, such as Tapira, Brazil (Swanepoel 2014) and Powder Horn, USA (resource of 350 million tonnes with an average grade of 11.5% TiO2; Van Gosen and Lowers 2007; Van Gosen 2009), contain titanium minerals, and were considered for future development. However, both projects stalled due to difficulties in producing market-acceptable TiO2 feedstock using conventional or alternative technologies.
In summary, carbonatites and alkaline-carbonatite complexes should be considered exceptional multi-commodity targets. Several of them host large deposits containing metals (Nb, Fe, Cu and a variety of noble metals as by-products), industrial minerals (e.g. apatite and vermiculite), construction materials required to build and maintain local infrastructure (crushed aggregate and dimension stone), and a variety of niche products such as badelleyite and sodalite, which is used as ornamental and semi-precious stone. Projects involving niche products (e.g. badelleyite and sodalite) are not ideal for large exploration and mining companies; however, such projects can sustain smaller communities and offer value-adding opportunities and related employment.
Exploration methods
Copper and Fe deposits within the Palabora alkaline-carbonatite complex were discovered and mined for copper and iron by African tribes about 1300 A.D. (Heinrich 1970), long before Bose (1884) first described a carbonatite occurrence and before A.G. Högbom recognised the magmatic origin of carbonate rocks at the Alnö carbonatite complex in Sweden near the end of the nineteenth century (Heinrich 1980) using traditional prospecting. Worldwide, a significant number of carbonatites and related deposits were discovered after the Second World War by traditional prospecting and during regional geological mapping. The rate of discovery increased significantly between 1956 and 1966 due to technical improvements in airborne radiometric and magnetic survey technology in combination with aerial photointerpretation. This mid-century surge in the rate of discovery is apparent from the numbers of known carbonatites reported in a series of consecutive review studies: Pecora (1956), Heinrich (1958), Heinrich (1966) and Woolley and Kjarsgaard (2008a), listed 32, 60, 320, and 527 known carbonatite occurrences, respectively. Selected examples of the use of well-established methods (radiometric and magnetic) and new promising exploration methods are described below.
Alkaline-carbonatite complexes which have not been severely deformed by post-intrusive tectonic activity have oval, circular, ring-shaped, and crescent-shaped configurations of diagnostic rock units with contrasting geophysical properties. These characteristic morphological features are commonly detected by ground and airborne magnetic and radiometric surveys, and in some cases gravity surveys (Satterly 1970; Thomas et al. 2011, 2016; Shives 2015). The Oka alkaline-carbonatite complex, located approximately 15 km west of Montreal, Canada (Figure 24(a)), is a particularly good example of applying these geophysical methods (Thomas et al. 2011). It is of Cretaceous age and is emplaced in the gneisses of the Canadian Shield exposed within an erosional window through flat lying Paleozoic sedimentary rock of the St. Lawrence Platform (Figure 24(a)). The complex aligns roughly with Cretaceous Monteregian intrusions (e.g. St. Bruno). The complex is oval-shaped, approximately 10 km in length, and consists of two distinct lobes and numerous concentric ring dikes (Figure 24(b); Gold et al. 1967). It hosted the St. Lawrence Columbium mine which operated from 1961 to 1977, and consisted of two open pits and underground workings. Significant Nb resources remain in the ground. The Bouguer gravity anomaly (Figure 24(c)), residual total magnetic field (Figure 24(d)), equivalent Th (Figure 24(e)), and the first vertical derivative of the magnetic field (Figure 24(f)) coincide closely with the outline of the complex.
Oka carbonatite complex, Quebec, Canada (a) Geological setting, (b) Internal compositional rings, (c) Bouger gravity anomaly, (d) Residual total magnetic field, (e) Equivalent thorium, and (f) First derivative of magnetic field. Modified from Thomas et al. (2016).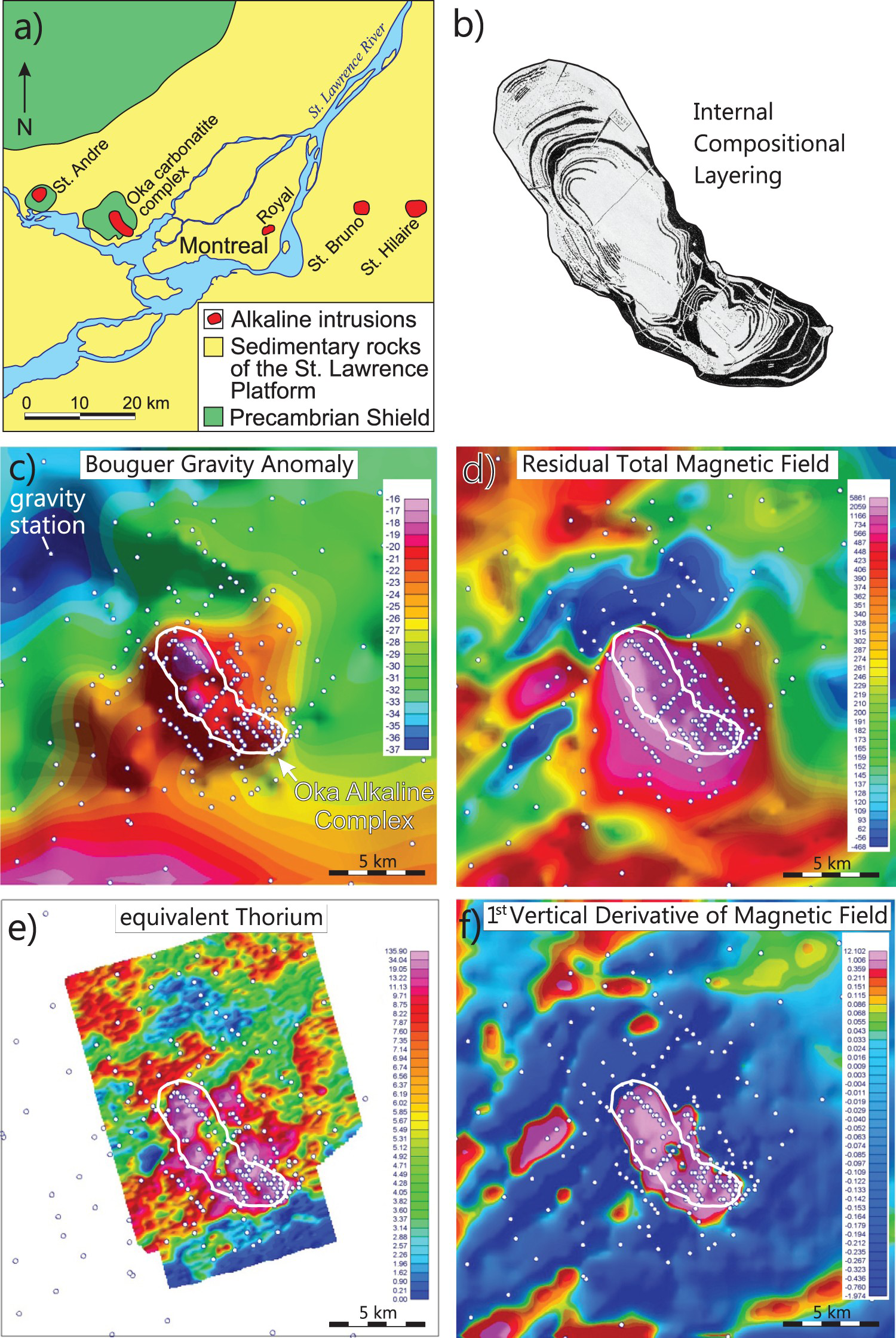
In some cases, glacial dispersal of ore minerals down-ice from the deposit enhances the size of both the geophysical and geochemical exploration target. For example, the Allan Lake carbonatite in Ontario, Canada, which was discovered by airborne gamma-ray spectrometry (5 km line spacing), is a small unexposed carbonatite body approximately 0.4 km2 in area, not including the fenite zone. The discovery of such a small intrusion was possible because carbonatite-derived material was incorporated into till, increasing concentrations of Ba, Nb, Th, Ce, La, Zn, Mn, and Fe 10 to 20 times, and Y, P, Cu, Pb, Mo, Co, and U 5 to 10 times those of background levels. The initial Th anomaly detected by an airborne gamma-ray survey coincided with the Th-rich till and large Th-bearing boulders that were dispersed down-ice for nearly 5 km from the deposit (Ford et al. 1988).
Examples of other carbonatites with distinctive geophysical signatures are the Elk Creek (Nebraska, USA) and Catalão I (Brazil) intrusive alkaline-carbonatite complexes. Elk Creek is a semi-circular (6×8 km) intrusive complex covered by a 200 m thick sequence of sedimentary rocks. It hosts the largest known Nb resource in the United States and significant REE mineralisation (Drenth 2014). This complex coincides with a roughly annular vertical gravity gradient high, a subdued central low, and a magnetic high surrounded by magnetic lows (Drenth 2014). The highest Nb concentrations are encountered within a dolomitic carbonatite, and REE are concentrated in a barite-dolomite carbonatite. Catalão I alkaline-carbonatite complex hosts important Nb and phosphate deposits, and has been covered by radiometric, magnetic, and gravimetric surveys. Inversions of gravity and magnetic data reveal very similar models in terms of volume and shape of the complex (Mantovani et al. 2016), and highlight the value of integrated studies.
Remote sensing involves acquiring, processing, and interpreting spectral and spatial data from space and airborne platforms resulting from the interaction between matter and electromagnetic energy (Sabins 1997). In general, multi- and hyperspectral instruments are able to map host rock mineralogy based on material specific absorptions by vibrational processes in the Short-Wave Infrared (SWIR) region and are promising techniques for delineating carbonatites and associated REE mineralisation (e.g. Rowan et al. 1986; Boesche et al. 2015).
Limitations of using ASTER and similar multispectral instruments over the Mountain Pass REE deposit, California, are provided by Rowan and Mars (2003). Due to limited spectral resolution, major mineralogy can be mapped, but minor differences (type of carbonate, accessory minerals) cannot. Hyperspectral remote sensing methods proved useful in outlining alkaline-carbonatite complexes that are not heavily covered by vegetation or glacial overburden such as the Sarfartoq carbonatite complex, southern West Greenland (Bedini 2009); Epembe, Namibia (Zimmermann et al. 2016); and the Khanneshin carbonatite, Afghanistan (Mars and Rowan 2011).
A review of the application of visible and shortwave infrared spectroscopy for the analysis of REE-bearing minerals is provided by Turner et al. (2015). The REE minerals exhibit characteristic absorptions, primarily in the Visible to Near Infrared (VNIR) region. However, as these absorptions are caused by transitional processes in the atom, they are sharp and a high spectral resolution is required. Several laboratory studies (e.g. Rowan et al. 1986; Turner et al. 2015; Neave et al. 2016) suggest that Nd is the key pathfinder element when exploring directly for REE deposits. Neave et al. (2016) indicate that hyperspectral instruments such as the Airborne Visible-Imaging Spectrometer (AVIRIS), HyMap, or Environmental Mapping and Analysis (EnMap) may provide the spectral resolution needed to detect targets containing more than 30, 000 ppm Nd if datasets are collected at metre-scale spatial resolution. However, since most carbonatite-hosted mineralisation is rich in La and Ce relative to Nd (Figure 17), and grades of carbonatite-related deposits do not normally exceed 10% REO(total) (Figure 22a), future technological refinements are required to make this approach adequate in exploration for REE deposits.
Limitations due to high detection limits of the above remote sensing methods in terms of total REE and Nd may be at least partially circumvented by taking advantage of recent advances in drone-borne technology. Booysen et al. (2017) were able to detect Nd absorptions and differentiate REE enriched dikes at Lofdal Farm, Namibia, with a small snapshot hyperspectral camera (attached to drone) from 40 m altitude.
Soil, till, and stream-sediment geochemical surveys are among geochemical methods used in exploration for carbonatite and related deposits. Soil geochemistry, where applicable, is favoured for its ability to delineate or extend mineralised zones prior to extensive trenching or drilling programmes, as shown by the results of surveys over the Upper Fir Carbonatite, British Columbia, Canada (Kulla and Hardy 2015). Typically, conventional sampling of the B soil horizon followed by an aggressive sample dissolution method (such as fusion with sodium peroxide, lithium meta-borate, or lithium tetra-borate) prior to inductively coupled plasma mass or emission spectroscopy (ICP-MS/ES) is recommended for analysis, especially if Nb, Ta, REE, and Zr are of interest, because fusion-ICP-MS/ES measures the near total concentration of an element in the sample. Fusion-ICP-MS/ES analyses ensure that Nb, Ta, REE, and Zr present in weathering-resistant minerals are detected. Without total dissolution, anomalies corresponding to weathering-resistant minerals dispersed by mechanical processes such as hydraulic and glacial transport (Winterburn 2015) are unlikely to be detected. Additionally, ‘Mobile Metal Ion’ (MMI), designed for detecting mineral deposits below glacial overburden (e.g. clay, till, and sand), is a promising carbonatite exploration technique as illustrated by an orientation survey aiming to detect mineralisation over the Fen alkaline-carbonatite complex, Norway (Lie and Østergaard 2011). This method is based on the premise that metal ions released from blind oxidising mineralisation rise vertically due to capillary rise and evaporation, and are adsorbed into clay particles in the top 10–25 cm of the soil cover, allowing detection in soil samples by MMI technology (Mann 2007; Turner et al. 2007).
Biogeochemical exploration is another promising approach; however, carbonatite-related cases employing this method are exceedingly rare in western scientific and government reports. Two examples that the authors are familiar with are the orientation surveys over the Allan Lake carbonatite, Ontario, Canada (Ford et al. 1988) and the Upper Fir carbonatite, British Columbia, Canada (Fajber et al. 2015). Barium, P, Sr, Co, and Cu in leaf tissue of sugar maple trees were analysed for pathfinder minerals at Allan Lake. Vegetation over the carbonatite showed concentrations of 13 times the background concentrations for Ba, and 21 times for Sr (Ford et al. 1988). Lanthanum, Ce, Pr, Nd, Sm, Dy, Fe, Nb, Ta, P, and Y were identified by Fajber et al. (2015) as preferred pathfinder elements in spruce and fir twigs and needles at the Upper Fir carbonatite. This study also showed that, in contrast to the absence of Eu anomalies in REE profiles of carbonatite rocks, the chondrite normalised REE profiles of vegetation and soil over the carbonatite may display strong negative Eu anomalies (Fajber et al. 2015). Although Nb is considered relatively immobile in surface environments, Nb and U anomalies in humus corresponding to major structural features within the Sokli alkaline complex, Finland, were documented by Vuotovesi et al. (1980).
Carbonatites and related mineralisation are known to have detectable signatures in till (e.g. Allan Lake, Ontario; Ford et al. 1988), glaciofluvial sediments (e.g. Sokli carbonatite complex, Finland; Perttunen and Vartiainen 1992), and stream-sediments (e.g. Aley carbonatite, British Columbia, Canada; Mackay and Simandl 2014b). An important development in recent years was the commercialisation of hand held XRF instruments and analytical protocols permitting the analysis of many key pathfinder elements such as La, Ce, Nd, Pr, Y, Nb, Th, Sr, and Ba in concentrations required for carbonatite exploration (Simandl et al. 2014a, 2014b, 2014c). Hand held XRF significantly reduces the costs of lithogeochemical, soil, and stream-sediment sample analyses, and increases exploration efficiency by providing real-time data in the field.
Recent studies demonstrated that high density (
The ‘direct indicator mineral’ concept combines low cost stream-sediment surveys and automated scanning electron microscopy to replace traditionally expensive indicator mineral surveys, eliminating the need for large samples and mineral hand-picking. When this approach is applied, only sediment samples anomalous in carbonatite pathfinder elements (detected using pXRF), with or without gravity pre-concentrating, need to be analysed by automated quantitative scanning electron microscopy to determine from which deposit-type direct indicator minerals were derived.
Successful exploration and development of carbonatite-related deposits: What are the odds?
The exploration industry estimates that less than one in 1000 discoveries become a profitable mine (Marshall 2012; Preston 2015). According to the Ontario Mining Association (2015), the odds are even lower: ‘Only about one mineral exploration project in ten is taken to the drill stage, and one drill programme in 1000 finds a viable mineral deposit’. Data compiled by Woolley and Kjarsgaard (2008a) indicates that approximately 6% of the 527 reported carbonatites and alkaline-carbonatite complexes host active mines, 3% hold historic mines, and 11% contain an established mineral resource. These numbers suggest that carbonatites have exceptional potential, and that a newly discovered alkaline-carbonatite complex has a 9 out of 100 probability of hosting a mine.
Conclusion
Carbonatite-related deposits are the main sources of Nb and LREE; significant sources of baddeleyite, Fe, Cu, vermiculite, phlogopite, fluorite, apatite, calcium carbonate, and sodalite; and have historically produced U and Th. Given that 9 out of 100 carbonatites and alkaline-carbonatite complexes contain currently producing or historic mines, carbonatite-related mineralising systems represent outstanding multi-commodity exploration targets.
Knowledge of tectonic setting, typical rock associations, and deposit morphology can assist explorationists seeking carbonatites and related deposits. Statistically, the most favourable exploration areas for carbonatites and related deposits are intracratonic rift (extensional) settings; however, most outcropping, or near-surface intrusive carbonatites and alkaline-carbonatite complexes in these settings are likely already discovered. The search for carbonatites in orogenic settings is not favoured by current statistics, but Bayan Obo, the world's largest REE-Fe-Nb deposit, is found in such a setting. Strongly metamorphosed and deformed carbonatites were historically difficult to identify; consequently, most new intrusive carbonatite discoveries will probably be in collisional settings. Identifying and incorporating carbonatite-related silicate igneous rocks and fenitisation-type metasomatic zones into carbonatite models significantly increase the size of exploration targets. Under ideal conditions, the intensity of fenitisation-type metasomatism may also be used to vector toward the source of metasomatising fluids. Knowledge of carbonatite morphology (spatial relationships between ring dikes, radial dikes, and cone sheets) permits vectoring toward the intrusive centre in undeformed geological settings; however, applications of these relationships in deformed terrains can be problematic at best. Age alone is not a valid parameter of prospectivity; however, within the same complex, the latest carbonatite pulses are commonly enriched in REE.
Although there is no universally applicable method used in exploration for carbonatites and related mineral deposits, on the regional or reconnaissance-scale, airborne geophysical methods (mainly radiometric and magnetic) are proven to be able to locate and delineate carbonatites. Ground radiometric and magnetic methods are useful for follow-up surveys. A variety of geochemical (e.g. stream-sediment, soil, and till chemistry; ‘direct indicator minerals’) and biochemical methods have been successfully used, tested, or customised for exploration and detection of carbonatites and related deposits.
Footnotes
Acknowledgements
This project was supported by the Targeted Geoscience Initiatives 4 (2010–2015) and 5 (2016–2020), a Natural Resources Canada programme. The Specialty Metal components of these initiatives are collaborative efforts between the Geological Survey of Canada and the British Columbia Geological Survey, Victoria, BC. Reviews and drafting by Pearce Luck, Michaela Neetz, and Carlee Akam from the British Columbia Geological Survey were essential for completion of this manuscript. Sections covering geochemical, geophysical and remote sensing exploration methods were reviewed for completeness by Ray Lett, Geochemical Consultant (Victoria), Michael Thomas from the Geological Survey of Canada (Ottawa), and Robert Zimmermann from Helmholtz Institute Freiberg for Resource Technology (Freiberg, Germany). The section on genesis benefited from constructive comments by Alexei Rukhlov, British Columbia Geological Survey, Victoria, BC, Canada. Encouragement from Alan Galley, Malleus Consulting Corp., Ottawa, ON., Mike Villeneuve, Geological Survey of Canada (Ottawa, ON), and David Lefebure formerly of the British Columbia Geological Survey and currently with Geologic Ltd. (Salt Spring Island, BC, Canada) were essential for initiating and completing this document. The final version of this manuscript benefited from suggestions and comments of three anonymous reviewers and handling by Simon Jowitt, co-editor of Applied Earth Science.
Disclosure statement
No potential conflict of interest was reported by the authors.
