Abstract
New lead volatilisation rate and activity data in copper mattes containing low levels of Pb were determined through high-temperature experiments. Measurements on volatilisation rate of lead from mattes (Cu-Fe-S) were carried out at temperatures from 1200 to 1570°C by injection of nitrogen into shallow baths. The concentration of lead in the melts covered a range of 200–6 mg.kg−1. The rate data exhibited first order behaviour with respect to concentration of Pb. The deduced vapour pressure of Pb in the gas was found to be dependent on the concentration of the Pb in melts and in accord with the equilibrium data on mattes at similar temperatures. Increasing temperature had a strong influence on volatilisation rate and total partial pressure of Pb in the gas phase. Matte grade (45–83 wt%) does not have a strong influence on volatilisation rate, but increasing sulphur potential does have a marked impact in supressing the volatilisation rate.
Introduction
Removing impurities from copper matte during smelting is of considerable importance and has been considered thermodynamically in many studies. Yazawa and Azakami (1969) considered this via two approaches, during smelting by slagging and by volatilisation. They reported that impurity elements like As, Bi, Pb and Sb in mattes were not removed by oxidation into the slag, but by volatilisation. Allaire and Harris (1989), considered removal of these elements by vacuum technologies, and Villarroel (1999) showed that it was possible to remove these elements from solid concentrates at temperatures between 900°C and 950°C under vacuum. Sohn et al. (2004), studied the removal of the same group of elements (As, Bi, Pb, Sb) by injecting Ar and Ar-O2 gas mixtures into copper matte. They observed that the removal rate of lead was higher under pure Ar than Ar-O2 mixtures and increased with temperature. The arsenic removal rate was not affected by the oxygen content in the gas. Antimony removal by volatilisation was low. Roine bubbled gas through copper matte for a fixed period. From the volatility of the elements the activity coefficient of As, Bi. Pb and Sb in the matte was calculated (Roine and Jalkanen 1985; Roine 1987a, 1987b). The transportation technique has also been applied by Zhong and Lynch (Zhong and Lynch 1998, 2000, 2001; Lynch and Zhong 2005) and also by Tran et al. (2013), for determining the activity coefficient of lead when it was at the parts per million basis in copper matte.
Removal of arsenic and lead by the smelting gasses during copper converting is regarded as difficult due to their high affinity with copper and hence low activity coefficient (Yazawa and Azakami 1969). Once blister copper is produced in the converter, only a small amount of these elements are removed in the gas phase. The proportion of volatile elements that are collected in the converter dusts are primarily from the slag blow of the converting process. Optimum removal of volatile elements is therefore more likely in the smelting stage, or possibly by introducing a purging stage in the converter prior to starting the slag blow, when no or minimal slag is present.
The present study was aimed at studying the removal of lead at relatively low concentrations in copper matte, and the likely conditions that will influence the deportment between gas and matte.
Experimental
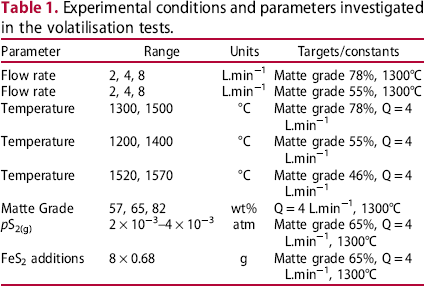
Experimental conditions and parameters investigated in the volatilisation tests.
Materials
The mattes were prepared from a mixture of technical grade FeS2 supplied as sticks, and technical grade Cu2S as powder. These reagents were analysed for Pb to ensure they were suitable for use in the current study and the results of analysis by ICP-MS showed that the FeS2 contained 7 mg.kg−1 of Pb and the Cu2S about 1 mg.kg−1 of Pb. Laboratory reagent grade PbS was used to dope the mattes to the desired Pb concentration.
Furnace and experimental procedures
A 400 kHz induction furnace was used for heating. A photograph of the 400 kHz induction furnace and heating station with the crucible in place is shown in Figure 1. A schematic of the heating station and bag house is shown in Figure 2.
The induction furnace and kilogram scale heating station with the crucible and gas ducting in place for an experiment. Schematic of the heating station for smelting experiments (no slag in this work) shown in Figure 1.
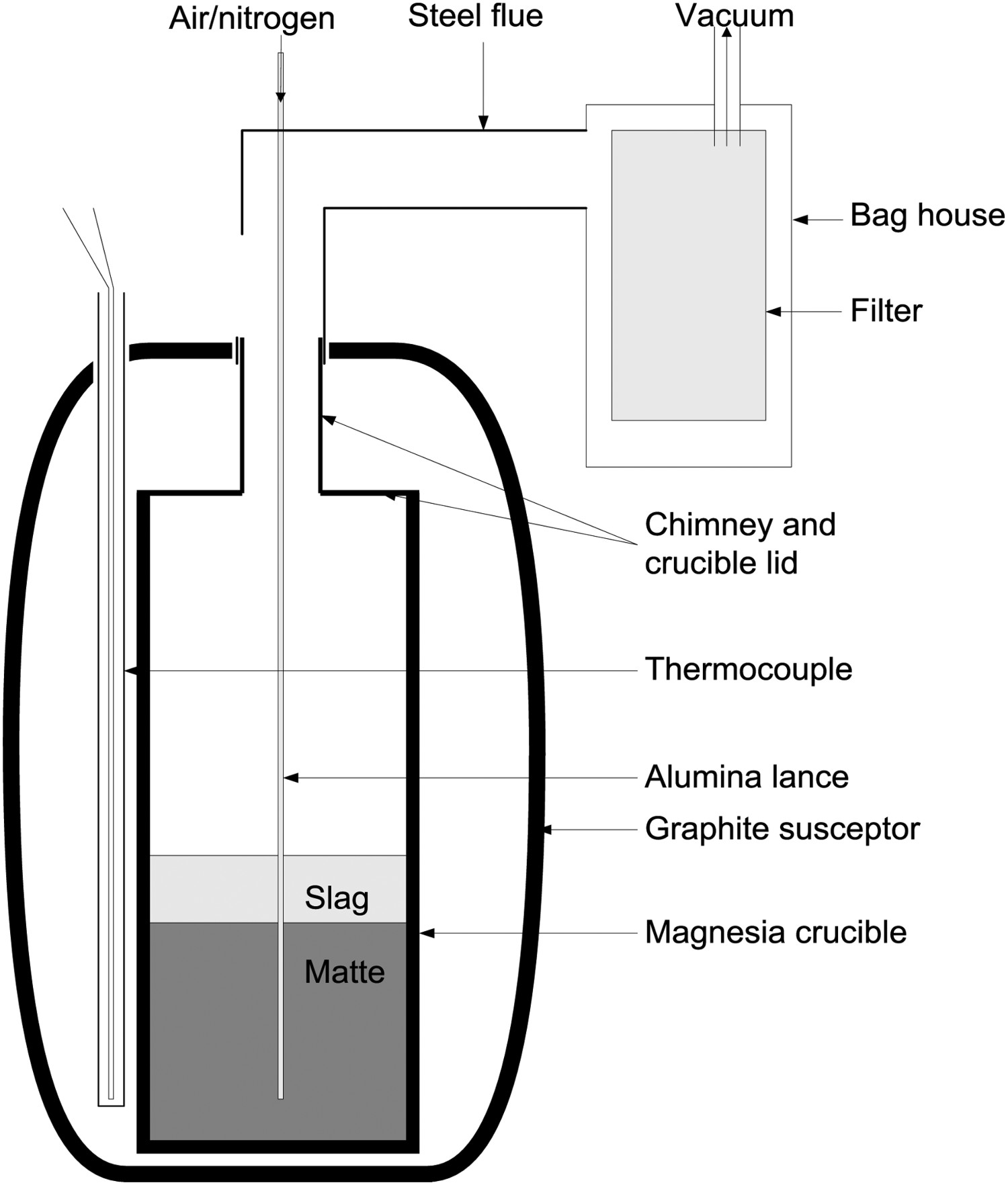
Straight walled magnesia crucibles with a diameter of 60 mm and a height of 300 mm with a capacity of 600 cm3 were used in this study. The crucible had a nominal capacity of 1 kg. A magnesia lid with a central hole for a flue was cemented to the top of the crucible, with a Pythagoras tube (30 mm O.D. and 60 mm long) cemented over the hole for the flue. The flue allowed a lance to be passed down into the crucible and into the matte and let exhaust gasses exit and be collected in the gas off-take. An alumina sheathed thermocouple was located alongside the crucible. The temperature measured is this manor was the same as that measured by a similarly sheathed thermocouple immersed in the matte. The crucible was heated by two susceptors, one inverted over the other, which were heated by the radio frequency induction. A programmable PID controller was used control the furnace power and hence temperature.
An alumina tube (5 mm O.D. and 3 mm I.D.) was used for injecting nitrogen and stirring the matte. Nitrogen was delivered from a bulk supply and the rate was maintained by an electronic mass flow controller.
A stainless steel cover over the crucible and steel ducting above the crucible mouth under negative pressure collected fumes and hot gases from the crucible. Any fumes were drawn through the ducting and collected on large air sampling filter papers.
A 600 g bath of matte with grades between 45% Cu and 80% Cu were prepared by adding powdered mixtures of FeS2 and Cu2S in the desired proportions down the chimney (flue) of the crucible assembly, then heating the crucible over 2–3 h to melt the contents. A blanket cover of nitrogen gas was maintained over the crucible to prevent oxidation of the matte. When the crucible was at 1250°C, the lance was lowered to within 5 mm of the crucible base with a low flow of nitrogen to gently stir and homogenise the matte. When the experimental temperature was attained, the lance was then lifted above the matte and lead sulphide was added to yield a lead content between 0.02 and 0.01 wt%. The lance was then lowered and the bath gently stirred for 30 s. The lance was then lifted out of the crucible and a matte sample collected by drawing a quantity into a 4 mm OD quartz tube. When the matte sample had cooled, a gram quantity was retained for analysis, and the rest returned to the bath.
After the initial sample was collected, the timer clock was then started and nitrogen at the desired rate was injected into the bath. After 15 min of injection, the lance was raised from the bath, the clock stopped, another matte sample was collected, quenched and a small quantity retained for chemical analysis, and excess returned to the matte bath. This process was repeated for two hours. The experimental conditions were then changed by addition of a small amount of lead sulphide to re-adjust the lead content of the matte to around 0.02–0.01 wt%, and a new experimental condition (temperature or injection rate) adopted. The new condition was also studied for 2 h, and then possibly a third condition was studied with more lead sulphide added to the matte, so that the total blowing time may have been up to 6 h.
After the final sample, the furnace was shut off, the lance lifted and the crucible was allowed to cool under a blanket cover of nitrogen. The bag house was weighed to determine the mass of fume collected. The steel ducting was swept clean and the fume collected. The crucible was weighed and separated from the matte. The matte was weighed and a representative sample collected for chemical analysis. In some tests where the final matte grade was at the white metal composition, a layer of copper was observed underneath the matte. The copper was separated from the matte and slag, weighed and a sample collected for chemical analysis. Copper metal was often collected in the matte samples as large droplets or ‘wires’ during sampling, confirming that copper metal was present during the experiment.
To study the effect of sulphur potential on lead volatilisation rate from mattes, a sulphur hot bath was assembled to generate a gas saturated with sulphur containing species (S8, S7, S6, S5, etc) by bubbling dry nitrogen into liquid sulphur. Preheated nitrogen gas was bubbled through liquid sulphur and then the sulphur containing gases passed through heated gas lines and injected into the matte. The effect of sulphur gas injection was investigated at two sulphur partial pressures with the sulphur bath being at a temperature of 160 or 175°C.
Conditions for the lead volatilisation experiments. Each purging cycle lasted for 120 min.
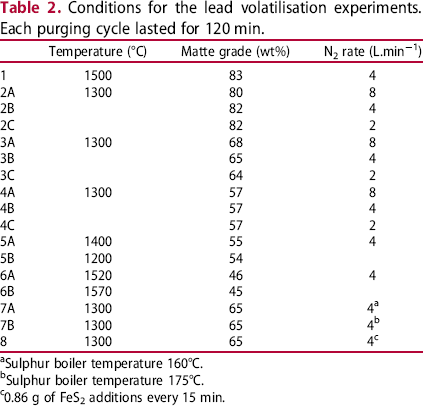
Sulphur boiler temperature 160°C.
Sulphur boiler temperature 175°C.
0.86 g of FeS2 additions every 15 min.
Sample preparation for chemical analysis
In some of the solid cylinders of matte samples collected, metallic copper was observed as small droplets or rods in the sample. For these, the metal was separated from the matte by gently crushing the matte and picking out the copper with tweezers. This was usually sufficient to remove the contaminating copper. The copper from the matte sample was bagged and given the same time stamp as the matte and submitted for chemical analysis. Copper that escaped detection usually smeared into flat platelets during pulverising and was removed from the matte sample with a fine screen.
The samples were pulverised in a small tungsten carbide ring mill and between each sample, the bowl and ring were wiped clean using an ethanol saturated chemical wipe. The bowl was allowed to dry and then the next sample was pulverised. To minimise possible cross contamination The matte samples collected were pulverised in reverse sample (i.e. time) order, from lower to higher lead content samples.
The matte and copper samples were analysed by ICP-OES. Approximately 100 mg portions of sample were treated with bromine and nitric acid solution to quantitatively oxidise the sulphides present prior to decomposition with mixed HF, HNO3 and HClO4 solution. Samples were taken to near dryness before redissolving in nitric acid solution and being made to 100 mL. These solutions were diluted by a factor of 20 to bring elements into a concentration range suitable for the ICP-OES. Low level sulphur results were analysed and quantified using a matrix matching method.
For low level lead analysis, the samples of matte or copper were digested using hot-block digestion tubes to eliminate any contamination issues from the Teflon beakers. A sample of 0.05 g was digested in 2 ml nitric and hydrofluoric acid and taken to dryness before being made up in 2% HNO3. The samples were then diluted to the acceptable concentration range for ICP-MS analysis.
Results
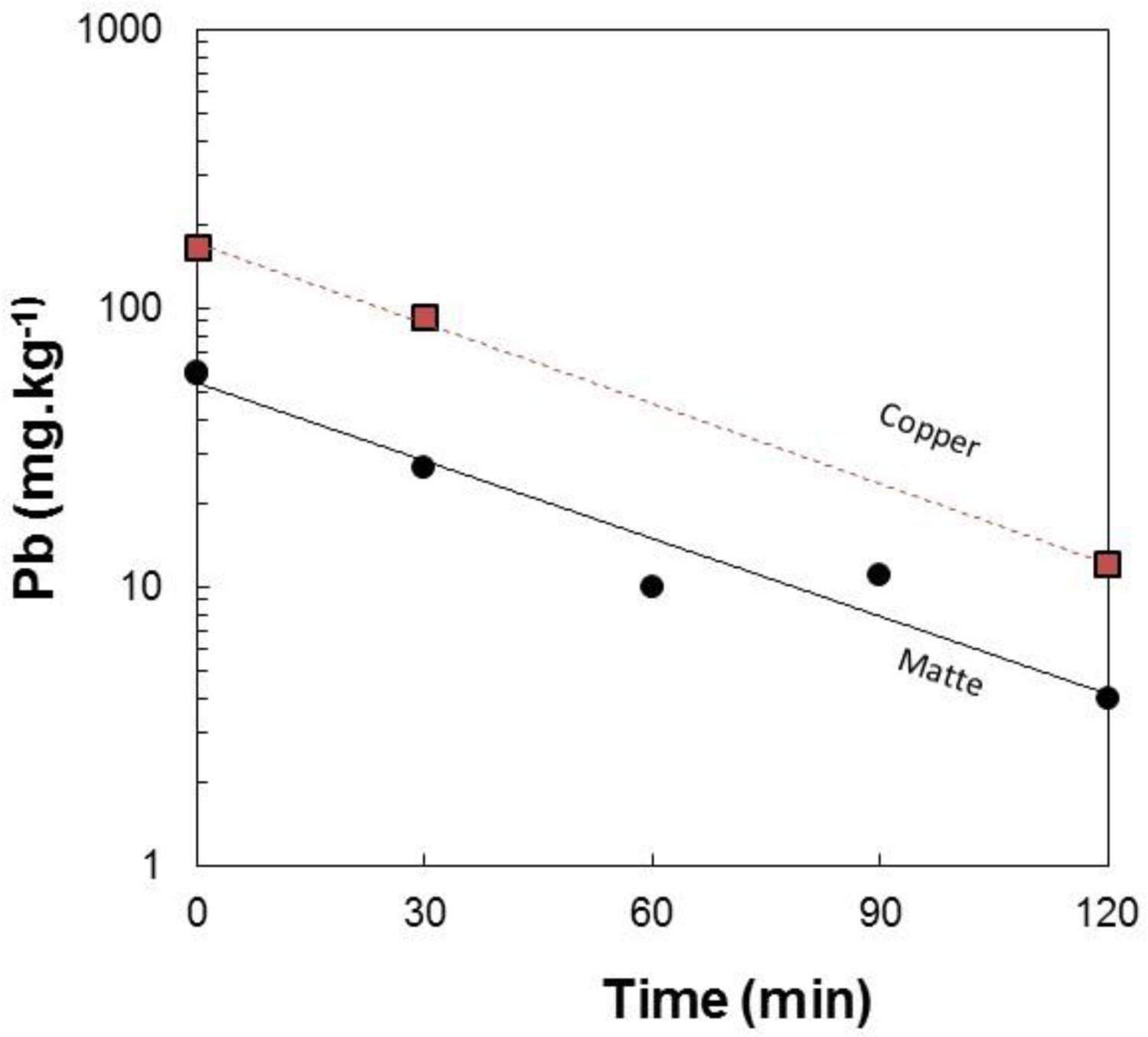
The chemical analysis of the samples collected during the nitrogen injection experiments are given in Table 3. The bulk matte chemistry was usually only determined for the initial and final samples of each test. Chemical analysis shows that within the analytical uncertainties, the matte composition with respect to Cu, Fe and S did not change during a test. The results for very high matte grade; i.e. white metal containing about 80 wt% Cu indicate that the samples had entrained copper droplets that resulted in total copper contents of up to 82 wt%. Typical results for the changes in lead concentration in the matte (and copper if separated from the synthetic matte on melting) are shown in Figure 3. These and other results obtained show that the change in concentration of lead in the matte and copper phase follows a first order relationship where the concentration,
Variation of Pb in matte and copper collected from nitrogen injection at a rate of 4 L.min−1 into molten bath with composition close to white metal at 1500°C. Chemical analysis of samples from the lead purging experiments. {} and [] denote dissolved in matte and copper, respectively. calculated using MPE package [Zhang et al. (2002)].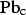 at time
at time
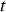 is of the form:
is of the form:
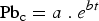
 is equal to the initial lead content of matte or copper in mg.kg−1 and
is equal to the initial lead content of matte or copper in mg.kg−1 and
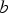 is an exponential term (a first order reaction rate constant) and t is time in minutes.
is an exponential term (a first order reaction rate constant) and t is time in minutes.
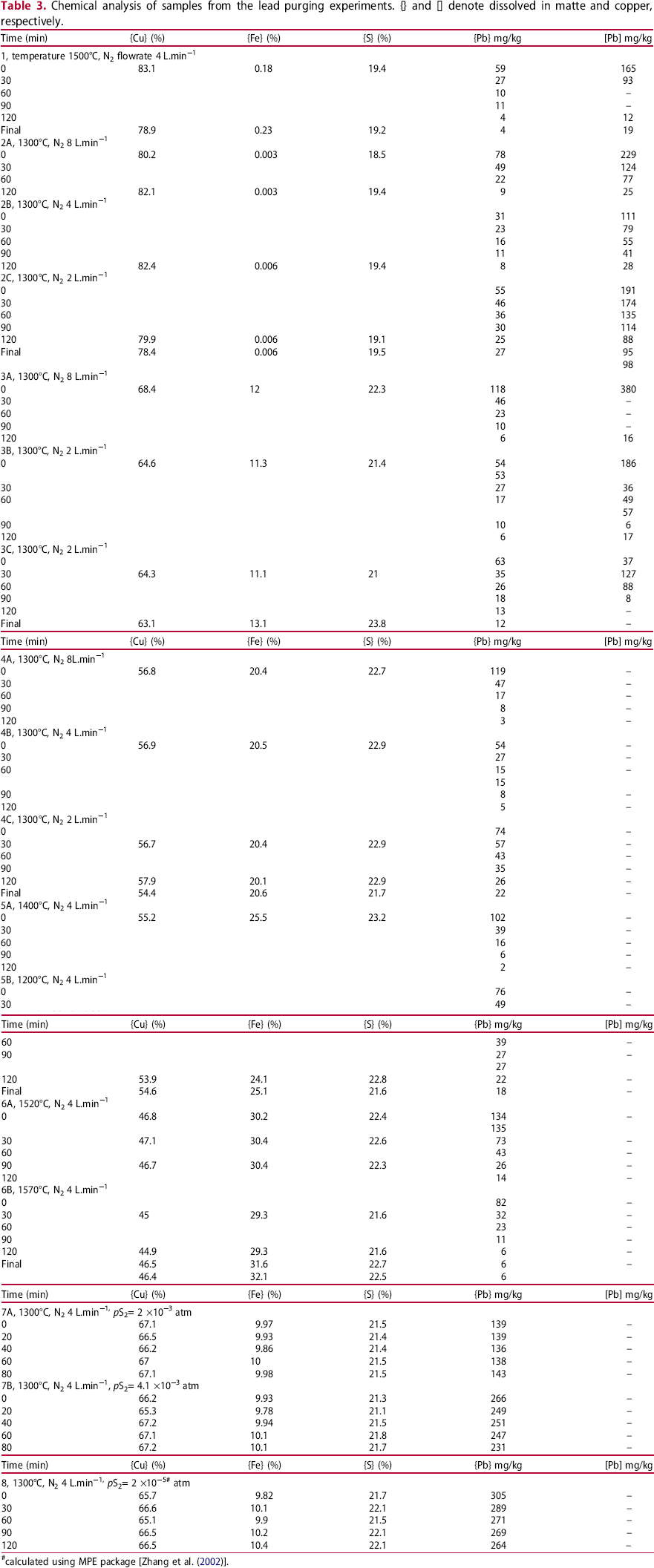
Summary of nitrogen injection experiments with the deduced first order rate parameters and calculated deportment of Pb between phases. {Cu}I and {Cu}f denote initial and final concentration of copper in matte.
Discussion
Effect of nitrogen flow rate on lead removal
The effect of nitrogen flow rate on lead removal was investigated at flows of 2, 4 and 8 L.min−1 (STP) at three matte grades covering a range of 58% Cu to white metal (∼80% Cu) at 1300°C. The removal of Pb from white metal at the flow rates studied are shown in Figure 4(a). The removal of Pb from Cu sampled from the white metal experiment is also shown in Figure 4(a). It is apparent that the rate of lead removal from the copper has the same slope as that of the matte. Even though the lead removal rate varies with flow rate, the lead distribution ratio between matte and copper (Lmatte/copper) was constant with respect to flow rate and time as shown in Figure 4(b). The observed value for the distribution of lead between matte and copper was 0.30 ± 0.04, which compares well with equilibrium studies where white metal-copper or slag-white metal-copper were equilibrated and the ratio Lmatte/copper was between 0.26 and 0.31 were obtained at 1300°C (Sinha et al. 1984).
Removal of lead from (a) matte and copper/matte, and (b) the distribution ratio of lead between matte and copper at 1300°C as a function of time at the gas injection rates studied. The initial lead content of the matte {Pb}i was between 35 and 80 mg.kg−1.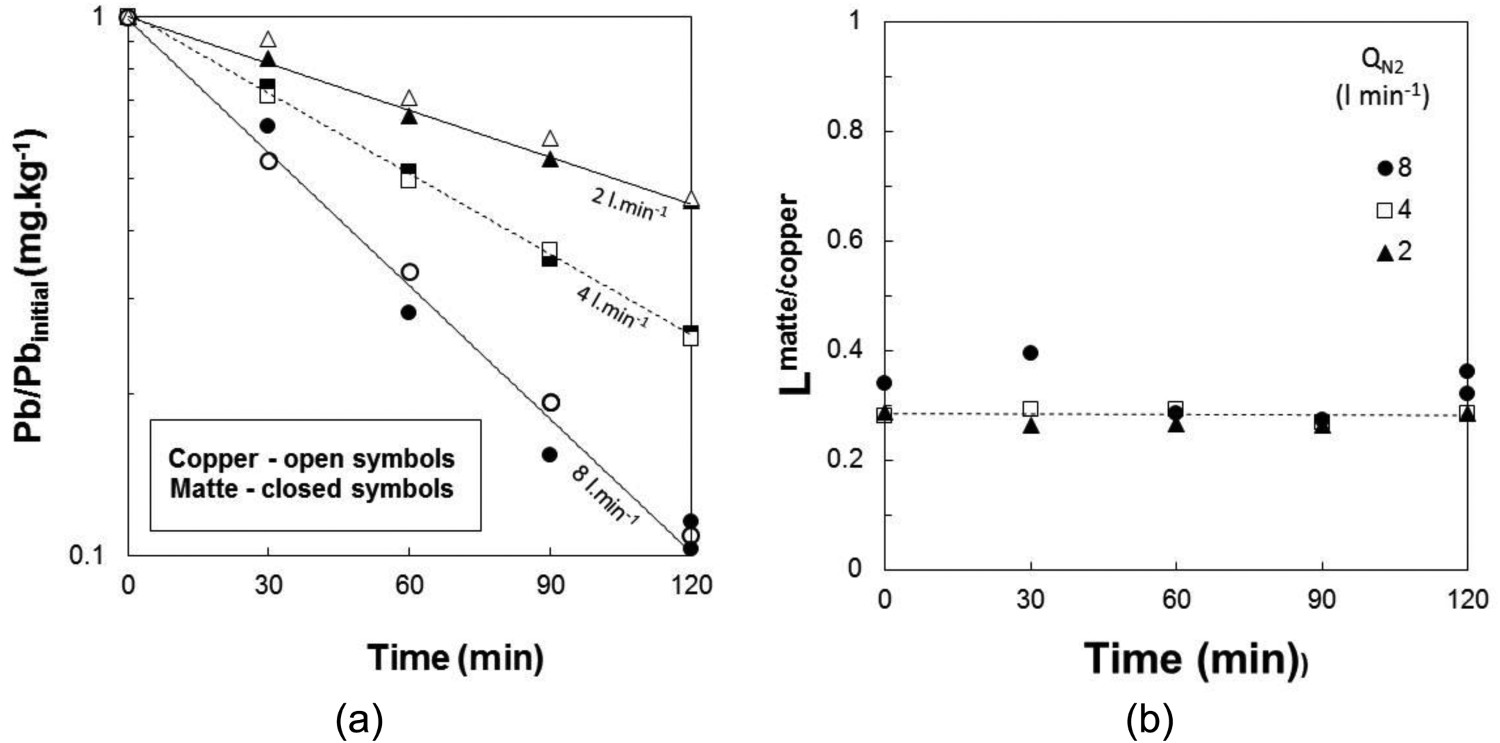
Differentiation of Equation [1] with respect to time will allow calculation of the rate of Pb removal at any specific injection time t, in terms of mg.kg−1.min−1, i.e.
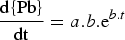
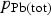 ) in the gas during injection was calculated using the estimated mass of matte (
) in the gas during injection was calculated using the estimated mass of matte (
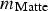 ) and copper (
) and copper (
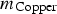 ) in grams, nitrogen gas flow rate (
) in grams, nitrogen gas flow rate (
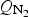 ) in L.min−1 at STP and the following equation:
) in L.min−1 at STP and the following equation:
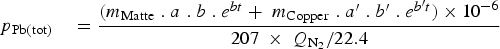
The values of
 and
and
 listed in Table 4 were used for these calculations, with the condition that
listed in Table 4 were used for these calculations, with the condition that
 and
and
 have the same value. We assumed that the lead is volatilised as monomeric species Pb and PbS; thus
have the same value. We assumed that the lead is volatilised as monomeric species Pb and PbS; thus
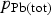 is the sum of
is the sum of
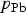 and
and
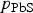 .
.
The calculated partial pressure as a function of the lead concentration in the matte is shown in Figure 5(a) for high-grade matte and Figure 5(b) compares the lead partial pressure above the lower matte grades studied at 1300°C. As no metal was present in the lower grade mattes, the uncertainties due to the calculation assumptions are lower compared to the medium and high-grade mattes where copper was present, the most significant assumption being that the initial mass of copper is inferred from the final mass of copper plus the mass of samples collected. The results show that at flow rates of 2 and 4 L.min−1, at the respective matte grades studied, the lead partial pressure was constant with respect to gas flow rate. At the flowrate of 8 L.min−1 the results show lower total partial pressures of Pb than 2 or 4 L.min−1. This suggests that at higher flowrate the gas did not fully reach equilibrium with the matte due to insufficient residence time in the crucible and/or contact with the melt. This trend is typical of transport studies where the rate of species transported is proportional to the gas flow rate, and then decreases as the rate limiting step is approached. The data used to calculate equilibrium partial pressures are from the flow rate region where the transportation rate was proportional to the gas injection rate.
Calculated total lead partial pressure above; (a) high-grade matte and (b) low-grade matte as a function of lead content in the matte at 1300°C.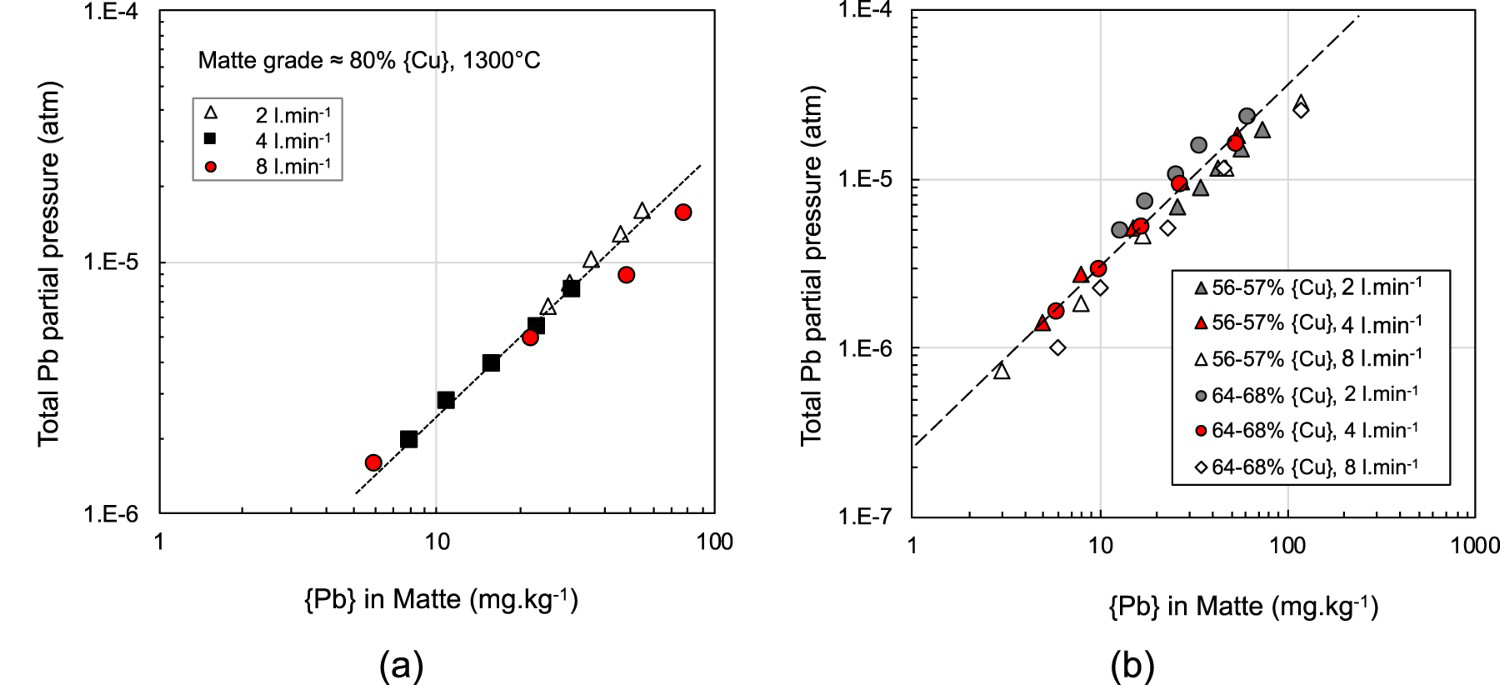
In Figure 6, the results from the present study are compared with those of Tran et al. (2013) for matte grades containing 50–56% Cu at 1300°C. There is good agreement at the lower gas flow rates of the Tran et al. study with the much higher injection rates used in this study. The results of Tran et al. at the higher gas rates (>0.15 L.min−1) were affected by splash and loss of matte into the cold zone of the tube furnace where it solidified, resulting in a smaller matte bath and lower calculated lead partial pressure. The enclosed heating chamber in the induction coil maximised the hot zone and was not affected by splash. Comparison of these two studies, performed under very different injection conditions suggest that equilibrium between the gas and liquid phases with respect to lead was achieved over the flow rates studied. Figure 6 also shows results obtained by Zhong and Lynch (1998) for a matte grade of 56% Cu. Their data follows the same slope but is systematically lower.
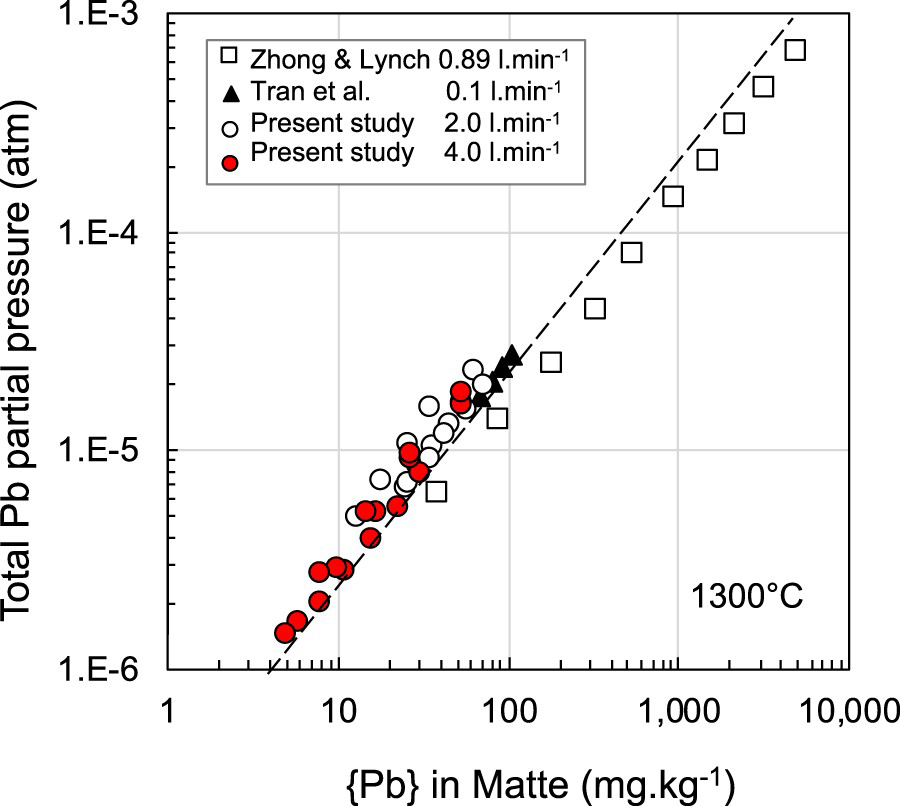
Effect of matte grade
Figure 7 compares the calculated Pb partial pressure at 1300°C for the range of matte grades and flow rates studied as a function of the lead content in the matte. There appears to be very little effect of matte grade on the lead partial pressure and hence, if the gas and matte are close to equilibrium, the activity coefficients of Pb and PbS are independent of matte grade; a similar finding to that of Tran et al. (2013) who observed a slight decrease in total lead partial pressure as matte grade increased from 50% to 78% for mattes containing around 100 mg.kg−1 of lead. Tran et al. (2013) reported that the activity coefficient of Pb in the matte was nearly constant at matte grades above 62%Cu. Both Roine (Roine and Jalkanen 1985; Roine 1987a) and Zhong and Lynch (1998), found that apart from Cu/Fe ratio, the sulphur deficiency in mattes has strong effect on the activity coefficient of Pb in the matte and hence partial pressure of Pb above the melt.
Calculated total Pb partial pressure above 56–78% Cu matte as a function of the lead content in the matte for different nitrogen injection rates used in this study at 1300°C.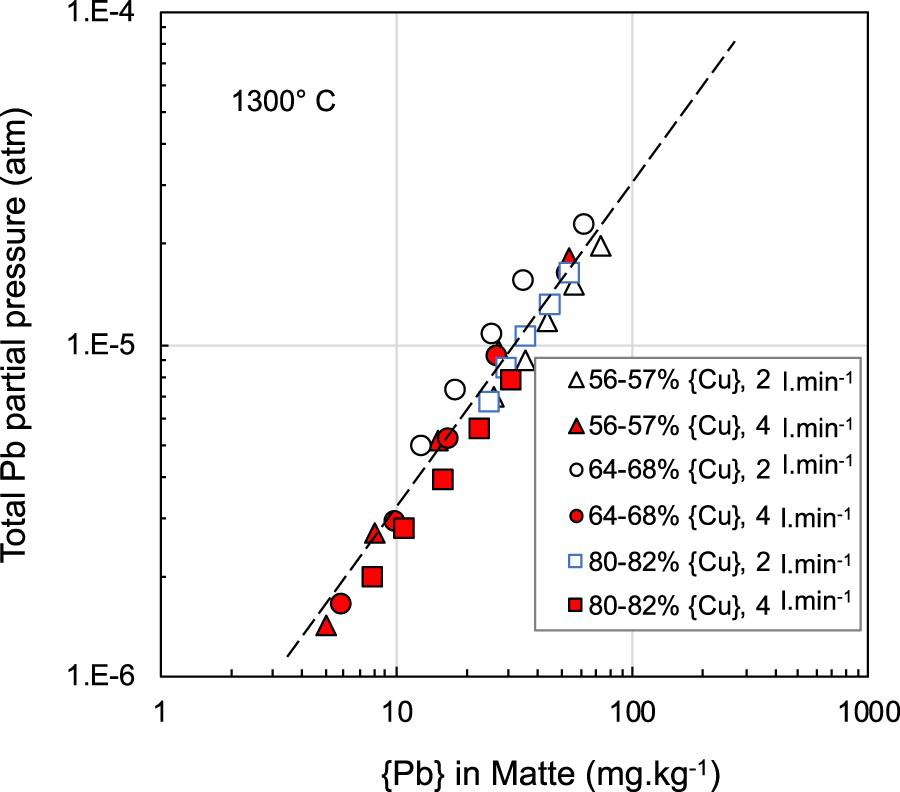
Effect of temperature
The results obtained from tests with nitrogen flow rate of 4 L.min−1 at temperatures between 1200°C and 1570°C are shown in Figure 8. Temperature has a strong effect on both the total partial pressure of lead and volatilisation rate of Pb from the copper mattes studied.
Variation of the total partial pressure of Pb with lead content of the mattes at various temperatures.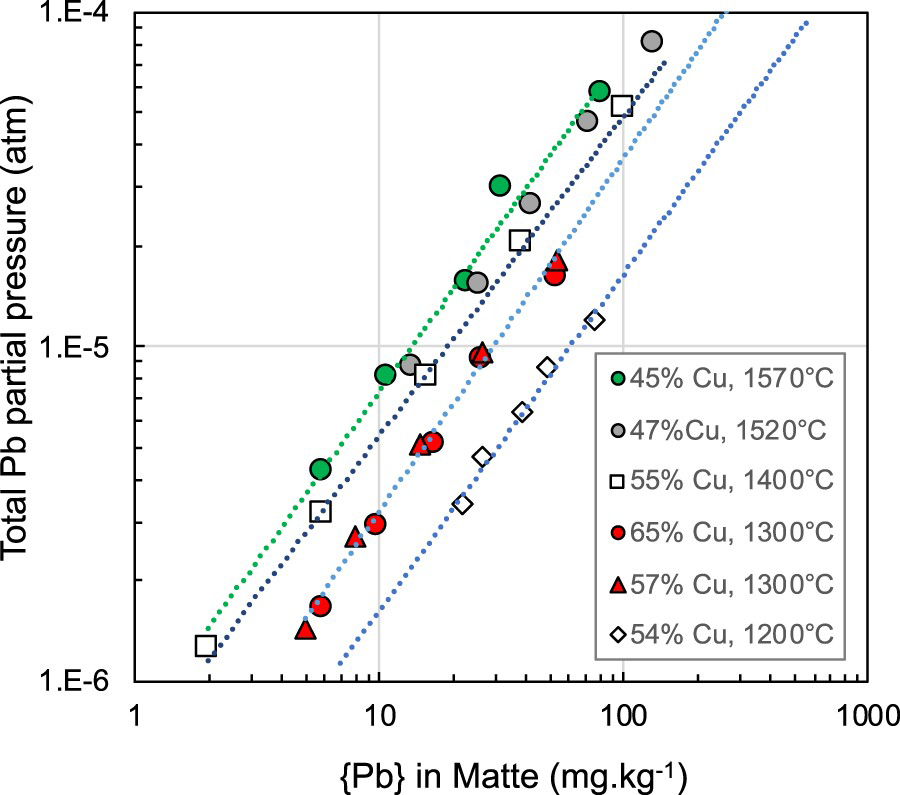
Estimated variation of the activity coefficient of Pb in mattes with temperature, standard state being pure molten lead.

Effect of sulphur pressure
Copper mattes could be sulphur deficient, and at a constant Fe/(Cu + Fe) ratio, the sulphur content of the matte generally increases as the sulphur pressure increases (Krivsky and Schuhmann 1957; Bale and Toguri 1976). The potential impact of sulphur on the transport of lead from the matte was examined by either injecting sulphur containing gas into the matte or adding a sulphidant such as pyrite to the matte.
The effect of sulphur gas injection was investigated at two sulphur partial pressures with the sulphur bath being at a temperature of 160 or 175°C. At these temperatures, the major sulphur species are S8 and S6 (Rau et al. 1973; Detry et al. 2011). The partial pressures of the species were calculated using the ThermoCalc software package (Andersson et al. 2002) and the S8 partial pressures were 4.0 × 10−4 and 8.1 × 10−3 atm (40 and 82 Pa) respectively, if the injected gas was at equilibrium. The sulphur species will then redistribute when injected into the matte phase, so that S2 will be the major sulphur species and the corresponding sulphur pressures are 2 × 10−3 and 4.1 × 10−3 atm (200 and 414 Pa). The corresponding sulphur partial pressure above the 65% Cu matte using the matte model of the CSIRO developed MPE thermodynamic software package is 1.0 × 10−5 atm (1 Pa) (Zhang et al. 2002). The changes in the concentration of Pb in the matte with sulphur gas injection are shown in Figure 9(a). The rates of Pb removal based a first order fitting of the data from Table 3 are listed in Table 4. The rate of lead removal from the matte was almost negligible with sulphur injection.
Changes in Pb content of a 65% Cu matte with time (a) when N2 + S2 at a fixed sulphur pressure was injected into the matte and (b) when FeS2 was added every 15 min. In both cases, the temperature was 1300°C and the nitrogen gas rate was 4 L.min−1.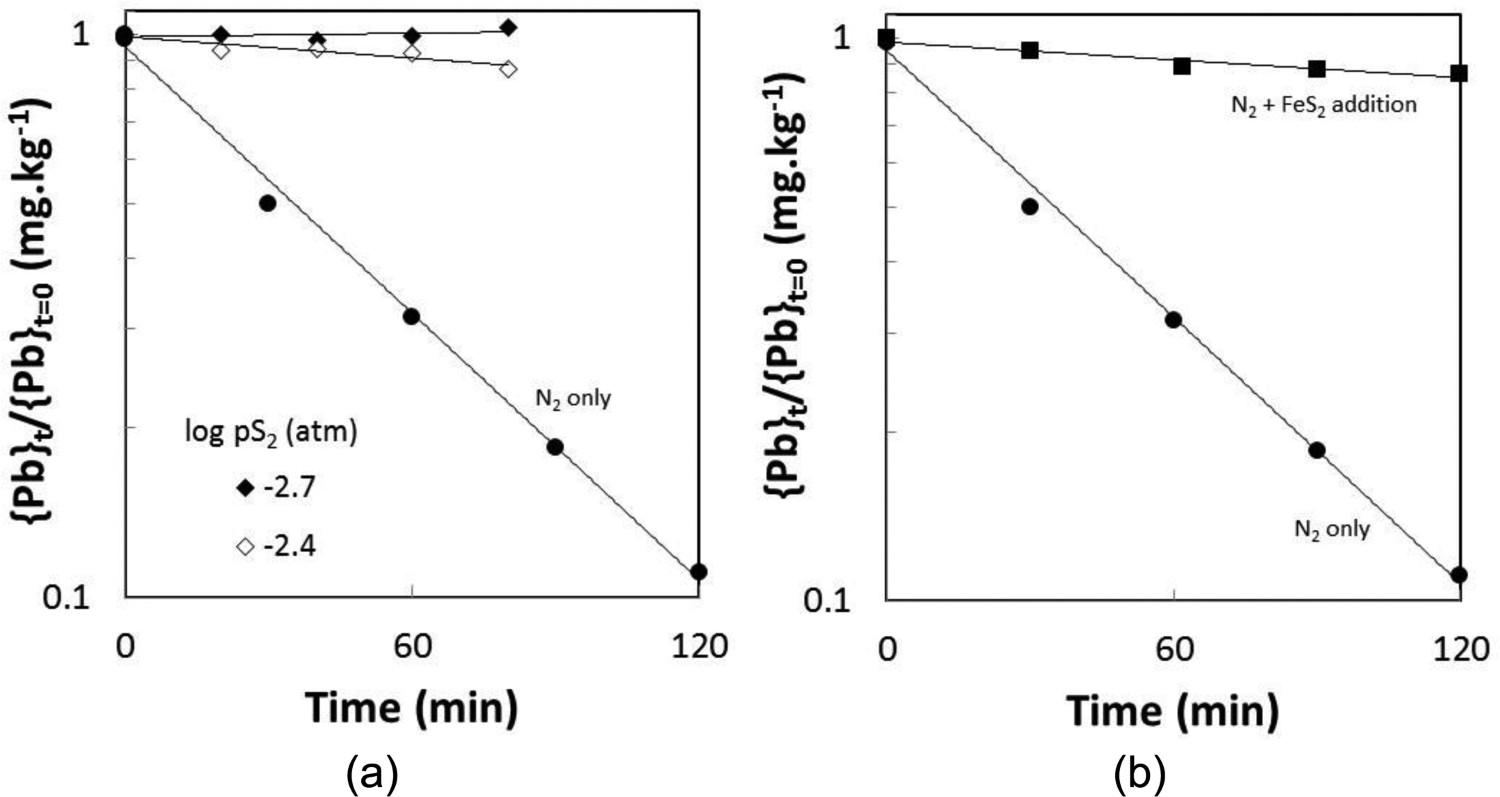
The effect of sulphur was also investigated by the addition of FeS2. The FeS2 would decompose in the bath to form FeS and S2(g) and increase the sulphur pressure above the bath. Using the matte model of the MPE (Zhang et al. 2002), the overall addition of 6.8 g of FeS2 to the 65%Cu matte would increase the pS2 to 2.0 × 10−5 atm (2 Pa). The calculations also predicted that the sulphur content of the matte would increase from a value of 21.8% to 22.2, a difference that is within the variation observed from the chemical analysis shown in Table 3. Compared to the corresponding test with only nitrogen gas injection, the rate of lead removal was decreased considerably, but not as much as that with sulphur gas addition.
The total lead vapour pressure above the sulphide matte is the sum of the vapour pressure of predominantly Pb(g) and PbS(g), with the two species inter-related by the reaction
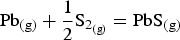
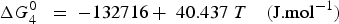
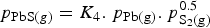
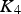 being the equilibrium constant and
being the equilibrium constant and
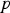 being the partial pressure of the vapour species. At 1300°C,
being the partial pressure of the vapour species. At 1300°C,
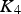 has a value of 197. At the lower rate of sulphur injection into the matte,
has a value of 197. At the lower rate of sulphur injection into the matte,
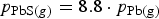 and at the higher sulphur rate,
and at the higher sulphur rate,
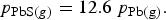 With only nitrogen injection, the corresponding relationship would be
With only nitrogen injection, the corresponding relationship would be
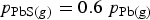 . So increasing
. So increasing
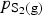 increases the proportion of PbS vaporising from the matte.
increases the proportion of PbS vaporising from the matte.
The other reaction to consider is the formation of PbS(g) from within the matte:
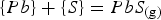
And the activity of Pb in the matte is related to
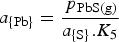
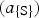 in the matte is increased (Bale and Toguri 1976), therefore the activity and hence the activity coefficient of Pb in the matte has decreased, and consequently the partial pressure of lead has also decreased by the relationship:
in the matte is increased (Bale and Toguri 1976), therefore the activity and hence the activity coefficient of Pb in the matte has decreased, and consequently the partial pressure of lead has also decreased by the relationship:
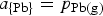
So increasing
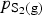 increases the proportion of PbS in the gas and decreases the activity of lead in the matte, so overall, decreased the rate of lead removal.
increases the proportion of PbS in the gas and decreases the activity of lead in the matte, so overall, decreased the rate of lead removal.
The considerable impact of sulphur potential on the activity coefficient and hence volatilisation of Pb from such mattes was also observed and reported by Roine (Roine and Jalkanen 1985; Roine 1987a), as well as Zhong and Lynch (1998). Roine showed over two orders of magnitude decrease in the values of γPb as the sulphur potential of the matte increased from 10−6 atm to about 10−2 atm.
The results of this study show that the rate of lead removal from both the matte and copper were identical under the same conditions of matte grade and temperature and gas injection. This means that in the converting furnace, the formation of copper should not slow the rate of lead removal from the liquid phases. The changing sulphur potential during converting has the greater impact on lead removal.
Conclusions
Volatilisation rate of lead from molten copper mattes containing very low concentration of Pb (5–100 mg.kg−1) was studied through submerged injection of nitrogen gas into a bath of matte. The measured rates followed the first-order behaviour with respect to Pb content of the bath and the rate increased with increasing injected gas flow rate.
The results obtained show the intimate contact between gas and matte resulted in a close approach to equilibrium at gas flow rates of 2 and 4 L.min−1. At high matte grades, where metallic copper co-existed with matte, the distribution of Pb between the copper and matte phase has reached equilibrium too. At a higher gas flow rate of 8 L.min−1, the residence time of the gas in the crucible may have been too short for achieving matte-gas equilibrium very closely.
The deduced total partial pressure of Pb increased with increasing concentration of Pb (2–100 mg.kg−1) in the matte. These results are in accord with other published thermodynamic studies where the concentration of Pb in matte was varied between 50 and 5000 mg.kg−1.
The total partial pressure showed a strong dependence on the temperature of the molten matte over the temperature range of 1200°C to 1570°C. Analysis of the effect of temperature, suggests that the activity coefficient of Pb in the matte decreases as temperature increases.
The effect of the matte grade (54–80% Cu) on lead volatility was too small to discern, but the effect of sulphur potential in the gas or matte has a very strong effect, virtually suppressing the Pb volatilisation from the matte at sulphur partial pressures of 10−2 atm. This strong effect of sulphur potential is at least partly due to the effect of sulphur on the activity coefficient of Pb in these mattes.
Footnotes
Acknowledgements
The authors wish to acknowledge; BHP and CSIRO Minerals Resources for financial support, and CSIRO's Analytical group for chemical analysis of samples.
Disclosure statement
No potential conflict of interest was reported by the author(s).
