Abstract
Oxalate precipitation is an attractive method for selective recovery of metal cations from leach solutions and for wastewater treatment. In this paper, the experimental results are presented as an oxalate precipitation diagram. The precipitation experiments were carried out on a synthetic sulphate solution containing selected metal cations. At the end of each precipitation step, the precipitates and filtrates were analysed by XRD and AAS, respectively. The experimental results showed that the oxalate precipitation tendency decreased in the following order:
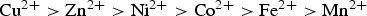 which was in good agreement with the calculated precipitation diagram based on solubility product data. Using this diagram, the selective precipitation of any metal cation can be predicted relative to the other cations in the solution. It can be concluded that oxalate precipitation can be employed as an effective process for selective precipitation of metal cations.
which was in good agreement with the calculated precipitation diagram based on solubility product data. Using this diagram, the selective precipitation of any metal cation can be predicted relative to the other cations in the solution. It can be concluded that oxalate precipitation can be employed as an effective process for selective precipitation of metal cations.
Keywords
Get full access to this article
View all access options for this article.
