Abstract
Finding efficient ways to decrease wood decay caused by fungi is an important issue in the timber construction. A possible way to avoid wood decay by fungi is by reducing the water content of wood, since water is a primary condition for fungal growth. Bulking of the wood cell wall by chemical reagents occupies the space where water normally occurs. This also improves the dimensional stability of the modified wood. The aim of the work was to react non-toxic reagents using a Maillard type of reaction in the wood cell wall. Wood was soaked in different aqueous solutions with a primary amine and a sugar as the main constituents. The wood was thereafter cured in an oven at 120°C. The preliminary results showed that the use of the Maillard reaction for wood modification is a promising method and is worth further research.
Keywords
Introduction
Wood is susceptible to degradation by fungi and exhibits dimensional instability when exposed to changing atmospheric conditions, which restricts its use in some situations like outdoor exposure, its use in bathrooms or basements, etc. To prevent degradation, non-durable wood products need to be treated when used in applications where they are susceptible to deterioration.
Currently, biocides are most commonly used for wood preservation. When they come into contact with water, these preservatives could potentially be leached from the wood, contaminating the environment (Hingston et al. 2001), or they present potential environmental hazards at the end of the lifetime of the timber product. Owing to environmental concerns and governmental regulations, preservatives are moving towards non-metallic compounds but potential leaching in extreme environments, high costs of the chemicals or degradation of the preservatives is still a problem (Schultz et al. 2007). Wood also exhibits dimensional instability in service which can result in problems such as windows and doors not functioning as intended. Treatment of wood using biocides does not affect the dimensional instability of the material, but an impregnation modification with a cell wall bulking agent will result in reduced shrinking and swelling of the wood.
The protection mechanism behind the decay resistance of an impregnation wood modification is a reduction of the cell wall moisture content, which limits the ability of fungal degradative agents to penetrate the cell wall and ensures that most fungal species cannot degrade wood that has a low moisture content (Papadopoulos and Hill 2002, Rowell 2006). In order to control the cell wall moisture content, it is necessary that the wood is treated with chemical solutions which diffuse into the wood cell wall and can be fixed in place. The presence of these chemicals will cause bulking of the cell wall, decreasing the cell wall volume accessible to water (Rowell and Banks 1985). An effective impregnation modification of this type must be non-toxic under service conditions and, furthermore, there should be no release of any toxic substances during its service life, or after disposal or recycling of the modified wood (Hill 2006), requiring that the polymeric network formed in the cell wall should react with the wood polymers or become entangled with them. Furthermore, the modification polymer should not display hygroscopic behaviour.
In this article, we report on investigations where the intention was to bulk the wood cell wall with chemical compounds, based on the Maillard reaction as an impregnation modification. This type of reaction is well-known in food chemistry, where it is responsible for the browning in many foods during baking (Ames 1998; Manzocco et al. 2001). The Maillard reaction is a reaction between a reducing sugar and a primary amino acid induced by heat. Loss of water in the initial stage produces an imine that is able to cyclize, resulting in the formation of N glycoside (a sugar attached to an NR2 group). Instead of cyclization an Amadori rearrangement may take place. Glycosylamines and Amadori products are only initial intermediates formed during the course of the Maillard reaction. Subsequently, a range of reactions takes place, including cyclization, dehydration, retroaldolization, rearrangements, isomerization and further condensations which will first form low molecular weight Maillard reaction products but in the later stages generate melanoidins, which are predominantly high molecular weight compounds (Echavarría et al. 2012). Melanoidins are heterogeneous, nitrogen-containing brown pigments and are predominantly responsible for the characteristic brown colour of foods such as coffee, cocoa, bread, malt and honey (Lindenmeier et al. 2002). The composition of these chemical structures is relatively unknown, due to the complexity of the products that are generated in the reaction (Kim and Lee 2009). The advantage of this reaction is that it is an aqueous process and initiated by heat only, making it relatively straightforward to apply to wood in a commercial process. In addition, the reaction does not require the use of strong acids or bases, which could degrade the wood structure.
Increasing the dimensional stability by bulking the cell wall in wood is a very well-known strategy (e.g. Hill and Jones 1996), but the use of the Maillard reaction to bulk the cell wall has not been studied to any significant extent. Hauptmann et al. (2015) treated wood with tricine at 103°C to induce the Maillard reaction between amine of the tricine and reducing sugar of the cell wall components (cellulose and hemicellulose). Their main aim was to react the tricine with the wood cell components or strengthen the cell wall via formation of tricine crystals. Despite the high amount of leaching and low weight percentage gain (WPG), some of the expected reaction occurred between hemicellulose and tricine. The treatment also increased the tensile strength and hardness of the wood, as well as decreased the equilibrium moisture content. Larnøy et al. (2007) treated wood with low (18 kDa) and high molecular weight (58 kDa) chitosan at 100°C, supposedly inducing the Maillard reaction to occur between amine of the chitosan (which is an amino-sugar) and reducing sugar of the wood cell components. Results showed that heat-modified, chitosan-treated wood showed similar properties to chitosan-treated wood, except for brownish colouration, enhanced hydrophobation, and slightly reduced antifungal properties. The modulus of rupture and hardness showed little or no change. The modulus of elasticity of the heat-modified, chitosan-treated wood increased by 27% compared with untreated wood. No data were shown about the dimensional stability, leachability and fixation of the chitosan into the wood. It is highly unlikely that chitosan was introduced into the cell wall, due to its high molecular weight. Umemura et al. (2010) evaluated bonding properties of the Maillard-reacted chitosan in three-ply plywood by a tensile shear test. They found that the addition of glucose to low-molecular-weight chitosan and the subsequent Maillard reaction are useful for the development of good bonding properties. The Maillard reaction was also already used successfully in certain applications as resole binders for composite wood and wood particle boards (Jackson et al. 2008; Jackson et al. 2010; Hand 2017).
Materials and Method
Small (20 × 20 × 5 mm) and defect-free sapwood specimens (beech – Fagus sylvatica L. and pine – Pinus sylvestris L.) were used for the modification studies. Samples were cut such that growth rings were parallel to the tangential face, to prevent diamonding during the leaching tests. This size of sample was selected in order to ensure even penetration of reactants into the wood sample and allow for good access of the reagents to the cell wall.
An analytical balance (minimum reading: 1 mg) was used to weigh the wood specimens. The size of the wood samples was determined with a precision of ±0.001 mm The size in each direction and weight was only measured once per sample. In this way, the contact with air was minimized as much as possible, which is necessary since weight and volume increase due to the moisture present in air. Wood samples were dried in an oven and left to cool down in a desiccator. Air was removed from the wood sample during the aqueous impregnation step at ambient temperature in an autoclave.
For the reactions we used a primary amine: lysine (CAS Number: 56–87–1) or glycine (CAS Number: 56–40–6); a sugar: xylose (CAS Number: 58–86–6) or glucose (CAS Number: 50–99–7); glucosamine (CAS Number: 66–84–2) which is an amino-sugar and can react as an amine as well as a sugar; and an extra reagent to facilitate the reaction: Magnesium chloride (MgCl2, CAS Number: 7786–30–3), maleic acid (CAS Number: 110–16–7) or citric acid (CAS Number 77–92–9). Zee et al. (1998) found that catalysts like MgCl2 and a temperature of 100–150°C markedly improved the dimensional stabilization, while carboxylic acids like maleic and citric acid are known crosslinkers to react with hydroxyl groups of cellulose. All chemicals were purchased from Sigma Aldrich.
To determine the size and weight of unmodified wood samples, the wood samples were oven-dried for 16 h at 103°C to remove the moisture. Once removed from the oven, samples were transferred to a vacuum desiccator to cool to ambient temperature over silica gel, immediately thereafter the weight and volume of the wood samples were determined. Thereafter, samples were treated in an aqueous solution containing 0.1 g mL−1 of a primary amine: glucosamine, lysine or glycine; 0.1 g mL−1 of a sugar: glucosamine, xylose or glucose; and 0.05 g mL−1 of a reagent to facilitate the reaction: MgCl2, citric acid or maleic acid. These concentrations were chosen to obtain high quantities of reagents that are completely soluble in water, while still having a solution density and viscosity that allows for easy penetration into the wood. 50 mL of solution was prepared for each different combination of reagents, and both pine and beech samples (simultaneously) were added into each one. Each wood species was present in five replicates. Control samples were treated with water only. The wood was placed into the amine-sugar solution, then transferred for 1 h into a vacuum chamber to make sure all the air was removed from the wood lumen. The wood was subsequently soaked for 24 h in the solution to allow for diffusion of reagent into the cell wall. The wood samples were then removed from the solution and put into an oven for 72 h at 120°C. After this, the wood samples were removed from the oven and cooled down to ambient temperature in a vacuum desiccator over silica gel and the volume and weight of the oven-dried wood was again determined.
Experimental set-up: There were two experimental rounds. In the first round, the amino acids lysine and glycine were combined with the two sugars (xylose and glucose) and MgCl2 and maleic acid. Glucosamine was seen as a chemical compound that has the features to react as a sugar, as well as a primary amine. Therefore, glucosamine was reacted by itself, in combination with MgCl2 and maleic acid. Since the results of the first round showed that maleic acid gave much better results than MgCl2, we tested in a second round of experiments citric acid, which is an acid with one carboxylic group more than maleic acid. Because the experiments with glucosamine alone were not favourable we decided in the next experiments to combine glucosamine with amino acids. So, in the second round of experiments the amino acids lysine and glycine were combined with the two sugars (xylose and glucose) and citric acid. Glucosamine was combined lysine and glycine, with and without citric acid. During the discussion of the results, data from both experiments are compared in the same table.
Leaching tests on the wood samples in water were performed in deionized water. A leaching cycle length of two days was chosen, and the deionised water in the leaching chambers was changed three times a day. After each leaching cycle, the wood was dried in an oven for 16 h at 103°C, cooled down to ambient temperature in a vacuum desiccator over silica gel, and the volume and weight gain of the oven-dried wood was determined. It was observed that three leaching cycles were necessary to get a stable weight and dimension of the samples, upon which the leaching was stopped.
The volumetric swelling coefficients (S) were calculated according to the formula (Stamm 1964):
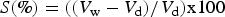
The WPG and volume change (VC) were calculated according to the formulas:
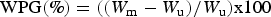
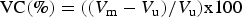
Results and discussion
As can be seen from Figure 1, most of the wood pieces turned to a darker brown colour after the wood modification procedure.
After the wood modification procedure, most of the (A) beech and (B) pine wood samples turned to a darker brown colour.
Weight per cent gain (WPG) and VC of oven-dry beech and pine after wood modification.
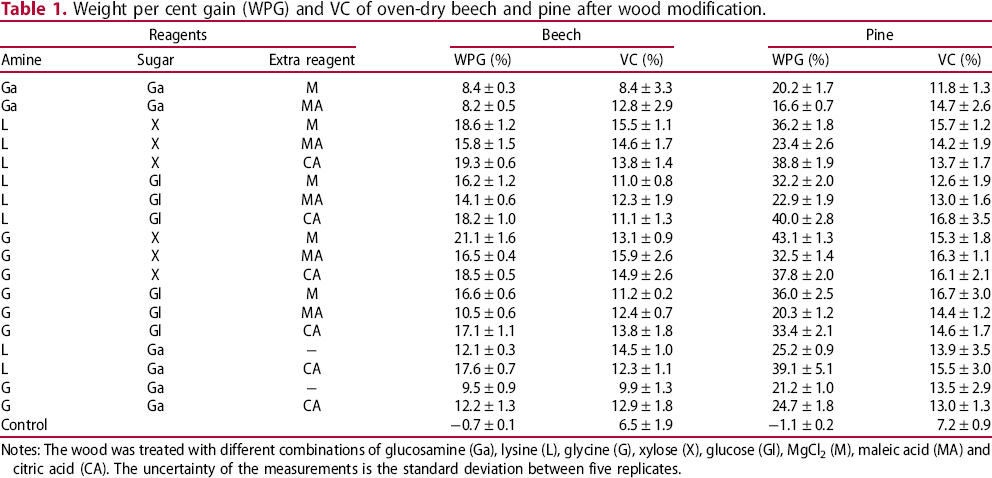
Notes: The wood was treated with different combinations of glucosamine (Ga), lysine (L), glycine (G), xylose (X), glucose (Gl), MgCl2 (M), maleic acid (MA) and citric acid (CA). The uncertainty of the measurements is the standard deviation between five replicates.
From the results in Table 1, it can be seen that pine after modification has a higher WPG than beech wood, but only a slightly higher volume increase. The WPG of pine is greater than that of beech because of its lower density and thus higher porosity. The bigger lumenar volume makes it easier for the treatment solutions to penetrate. Volume increase of wood occurs only when the reagents penetrate into the cell wall via pores and the results indicate that the beech and pine cell wall are equally readily penetrated by the reagents. The reasons for this may be linked to the cell wall structure and composition at the molecular scale, e.g. differences in hemicellulose and lignin composition; in addition, there are microstructural factors, such as the restraining effect of the middle lamella that should be taken into account. From the table, it can be seen that the control sample exhibited a VC of approximately 7%. In all cases where the Maillard reaction modification was used, the VC exceeded 7%, showing that there was cell wall penetration of the reagent in all cases beech impregnated with glucosamine and magnesium chloride, or glycine with glucosamine exhibited little volume increase after modification, as well as relatively low weight percentage gains after the reaction. With beech, the VC after impregnation and curing was generally in the range 11–16%, whereas it was found that the water-saturated average VC for beech was around 27%, which shows that cell wall bulking was relatively low and that much of the material was located outside of the cell wall. With pine, the VC ranged from approximately 12–17%, which at the higher end of this range indicated that there was a high level of cell wall filling/bulking, given that the average water-saturated VC for pine was found to be about 20%.
Weight per cent gain (WPG) and VC of oven-dry beech and pine after wood modification and leaching.
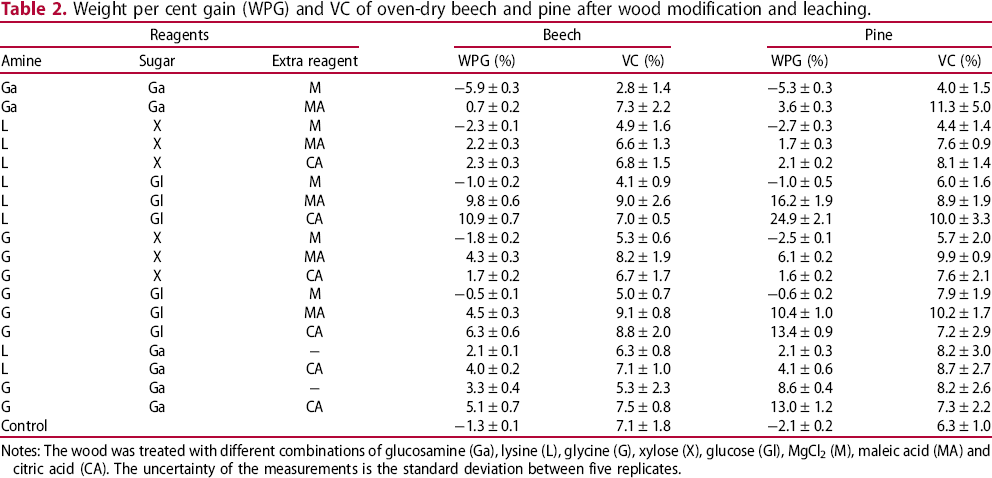
Notes: The wood was treated with different combinations of glucosamine (Ga), lysine (L), glycine (G), xylose (X), glucose (Gl), MgCl2 (M), maleic acid (MA) and citric acid (CA). The uncertainty of the measurements is the standard deviation between five replicates.
From the data in Table 2 can be seen that pine after leaching has a much higher WPG than beech wood in the cases where not all reagents and reaction products are leached out, while the VC of pine is in most cases only slightly higher than of beech wood. Treatments where MgCl2 was used, were considered unsuccessful, in that the weight percentage gains were negative (as much as −5 to −6% WPG in the case of glucosamine with magnesium chloride) indicating removal of cell wall components as the result of the reaction (Shimada et al. 2007). From all combinations that were tested in the experiments, the two most successful (i.e. relatively high WPG and VC) modifications used a lysine – glucose – maleic acid solution and in a lysine – glucose – citric acid solution.
Since maleic acid and citric acid were also necessary to get a polymeric structure, we can conclude that their carboxylic groups react with the amino groups and/or less substituted alcohol groups from the sugars or wood (Reddy and Yang, 2010; Feng et al. 2014). Feng et al. (2014) found that wood treated with citric acid alone can already lead to an improvement of the dimensional stability due to reaction with the wood cell wall polymers.
An even better indicator than VC for potentially useful impregnation modifications can be determined by comparison of the swelling coefficients of modified wood with unmodified control samples. The swelling coefficient compares the volume of modified wood with the volume of water-saturated wood. The smaller the difference and thus the coefficient, the higher the swelling. An increase in WPG but with relatively low VC and a high swelling coefficient, means that reaction products are present in the cell lumen, but not in the cell wall. From Table 2 and Figure 2 it can be seen that the treatments have a high deviation between the different replicates.
Graphs of the WPG versus the volumetric swelling coefficient (S) for (A) beech and (B) pine. The samples depicted in the graphs are the ones showing a high increase in weight in comparison with unmodified wood and/or high decrease in the swelling coefficient in comparison with the control samples. The reagents were glucosamine (Ga), lysine (L), glycine (G), xylose (X), glucose (Gl), MgCl2 (M), maleic acid (MA) and citric acid (CA).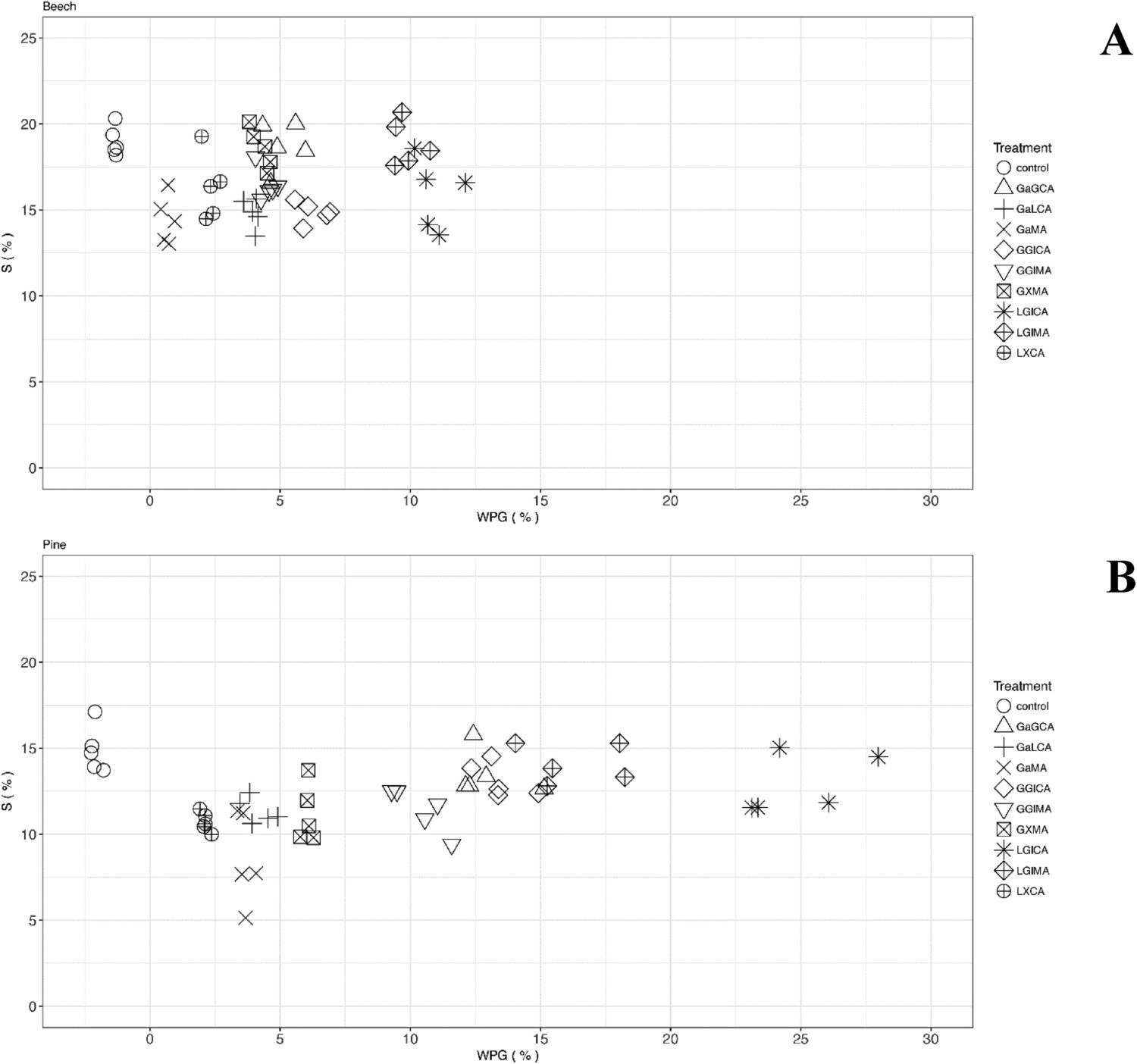
In Figure 2 the experiments are depicted, which have a low swelling coefficient and/or a high WPG. For most experiments, swelling coefficient values are close to the values exhibited by the control samples. It can be seen that in certain experiments, for the same treatment some replicates have values for the swelling coefficient, which are lower than the swelling coefficient of the control sample, while other replicates have values that are close to the swelling coefficient of the control sample. This means that it is possible to get some reaction products into the cell wall but the conditions should be optimized to be able to make it repeatable to all wood samples. This is currently the subject of more extensive investigations. Visualization of the data in this way did allow us to select the following promising wood modification systems, which are worthy of further investigation (containing the lowest swelling coefficient):
glucosamine with maleic acid glucosamine with lysine and citric acid lysine with xylose and citric acid glycine with xylose and maleic acid glycine with glucose and citric acid glycine with glucose and maleic acid
Conclusions
In this work, we described the preliminary results of experiments based on a Maillard type of reaction to induce wood cell wall buking. This wood modification procedure is water deliverable and thermally activated and uses non-toxic, low molecular weight reagents. Pine gave in general a higher WPG than in beech samples. Further research should still be performed to reduce the leaching of the samples by optimizing reaction conditions and using reagents that have less unreacted hydrophilic groups, so that water-insoluble reaction products will be formed. After the Maillard reaction, almost completely water-soluble reaction products were formed for most of the experiments. Better results were obtained when lysine and glucose were reacted with maleic acid or citric acid. After leaching tests, this gave about 10% WPG for beech and 20% WPG for pine. It was possible to get reagents to react in the cell wall but conditions should be optimised to be able to make it a repeatable procedure. Studies of diffusion of reagent into the cell wall are currently under way to understand this further. However, this preliminary screening reaction has shown that the Maillard reaction does show promise and is worthy of investigation as a potential new wood modification system.
