Abstract
As part of Accident Tolerant Fuel initiative for light water reactors, uranium silicide and silicide-nitride are being considered as fuels that can be combined with a more robust cladding such as a ferritic (FeCrAl) alloy. Although these materials have been studied in the past, uncertainties remain concerning the fundamental behaviour of these systems. In this study, four compositions between U3Si5 and USi2 were fabricated by arc-melting. Additionally, in an effort to understand the UN–U3Si2 fuel system, an unidentified U–Si–N ternary was observed in samples fabricated by arc-melting U3Si2 in the presence of varied concentrations of N2 in Ar. The analyses of compositions prepared to investigate these systems include structural characterisation using X-ray diffraction and compositional analysis employing scanning electron microscopy with energy dispersive spectrometry. Initial values of the Gibbs energy parameters via the CALPHAD (CALculation of PHAse Diagrams) method have been developed for the ternary phases observed upon U3Si2–FeCrAl interactions.
Introduction
The push to develop new fuel types for use in current light water reactors (LWRs) is driven by the need for new ‘accident tolerant’ fuel (ATF) that exhibits improvement in operational and transient/accident behaviour [1]. Uranium silicides (U3Si2 and to a lesser extent U3Si5) and uranium silicide-nitride (UN–U3Si2, UN–U3Si5) composites are currently among the fuel types being considered due to their increased U-density and higher thermal conductivity over conventional UO2 [2-4]. The primary advantage of increased thermal conductivity is less stored energy in the core that must be mitigated in the event of an accident [5]. Different cladding options are simultaneously being explored to address the issue of a Loss of Coolant Accident (LOCA), which can lead to significant damage of the fuel from rapid steam oxidation of the zirconium-based alloys used currently and the resultant large heat input from oxidation of the zirconium and production of hydrogen [1]. New cladding candidates include ferritic alloys such as FeCrAl(Y) and SiC–SiC composites that reduce the heat generated and hydrogen produced, thus delaying cladding failure.
The candidate fuel U3Si2 has been the subject of studies detailing its thermophysical properties [2,6-8] and a comprehensive review can be found in the U3Si2 handbook published by Los Alamos National Laboratory (LANL) [9]. The uranium-silicion system has been thermodynamically assessed by Berche et al. [10] and Wang et al. [11], however some discrepancies within the phase diagram persist [1214]. A comprehensive understanding of U–Si phase equilibria and thermochemical behaviour will, therefore, be necessary for understanding fuel performance and to support licensing efforts. A compositional region requiring further study is the silicon-rich, 60–66.6 at.-% Si (Si/U = 1.60 to Si/U = 2) region where phase formation may occur during burn-up as uranium is lost to fission and fission products may not form sufficient silicides to take up the liberated element. The phase equilibria in this compositional range is still poorly understood and further experimental efforts have been suggested [12,13]. This paper explores the compositional range to clarify phase equilibria and expand current thermodynamic models. Additionally, this work aims to confirm the ternary U–Si–N phase formation that was observed in high-temperature sintered UN–U3Si2 composites [3,15] and to identify the phase(s).
Ferritic alloys of the composition, Fe (10–13%) Cr (3–5%) Al (0.03–0.07%) Y [16-19] have been identified as candidate cladding materials. In general, FeCrAl alloys exhibit excellent oxidation resistance through the formation of a protective alumina layer [20] where the addition of Cr induces a third element effect and allows for better oxidation kinetics of the Al2O3 surface layer, [21] with yttrium doping providing for better adhesion of the oxide [22]. A commonly mentioned drawback for the use of these materials in LWR's is their neutron penalty due to the higher constituent thermal neutron absorption cross-sections compared to zirconium, however coupling this cladding material with the high U-density U3Si2 fuel could overcome this disadvantage. Therefore, understanding U3Si2–FeCrAl(Y) compatibility, including determining possible low melting eutectics and secondary phase formation is a necessity. Recent efforts at Idaho National Laboratory studying diffusion couples of U3Si2 and a FeCrAl alloy at 500–1000°C [23] revealed the formation of a major ternary U–Fe–Si phase. While U–Fe–Si phases have been known for some time [24], no CALPHAD type (CALculation of PHAse Diagrams) thermodynamic assessment is reported. Gibbs energy parameters for U–Fe–Si ternary phases pertinent to fuel-cladding interactions are thus being addressed in this work.
U–Si binary system
Reported state of U–Si phase equilibria
The present U–Si phase diagram in Figure 1 is based on investigations by Kaufmann et al. [25] who determined the phase equilibria using thermal analysis, metallography and X-ray diffraction (XRD) and from Vaugoyeau et al. [26] who investigated the phase diagram from U3Si2 to USi3 using thermal analysis to determine liquidus temperatures. Seven identified intermetallic compounds; USi3, USi2, USi1.88, U3Si5, USi, U3Si2 and U3Si describe the U–Si binary system.
Calculated U–Si phase diagram utilising values of Berche et al. [10]. Red lines indicate the boundaries for the region of interest 0.6 to 0.66-mole fraction Si (60–66 at.-%Si).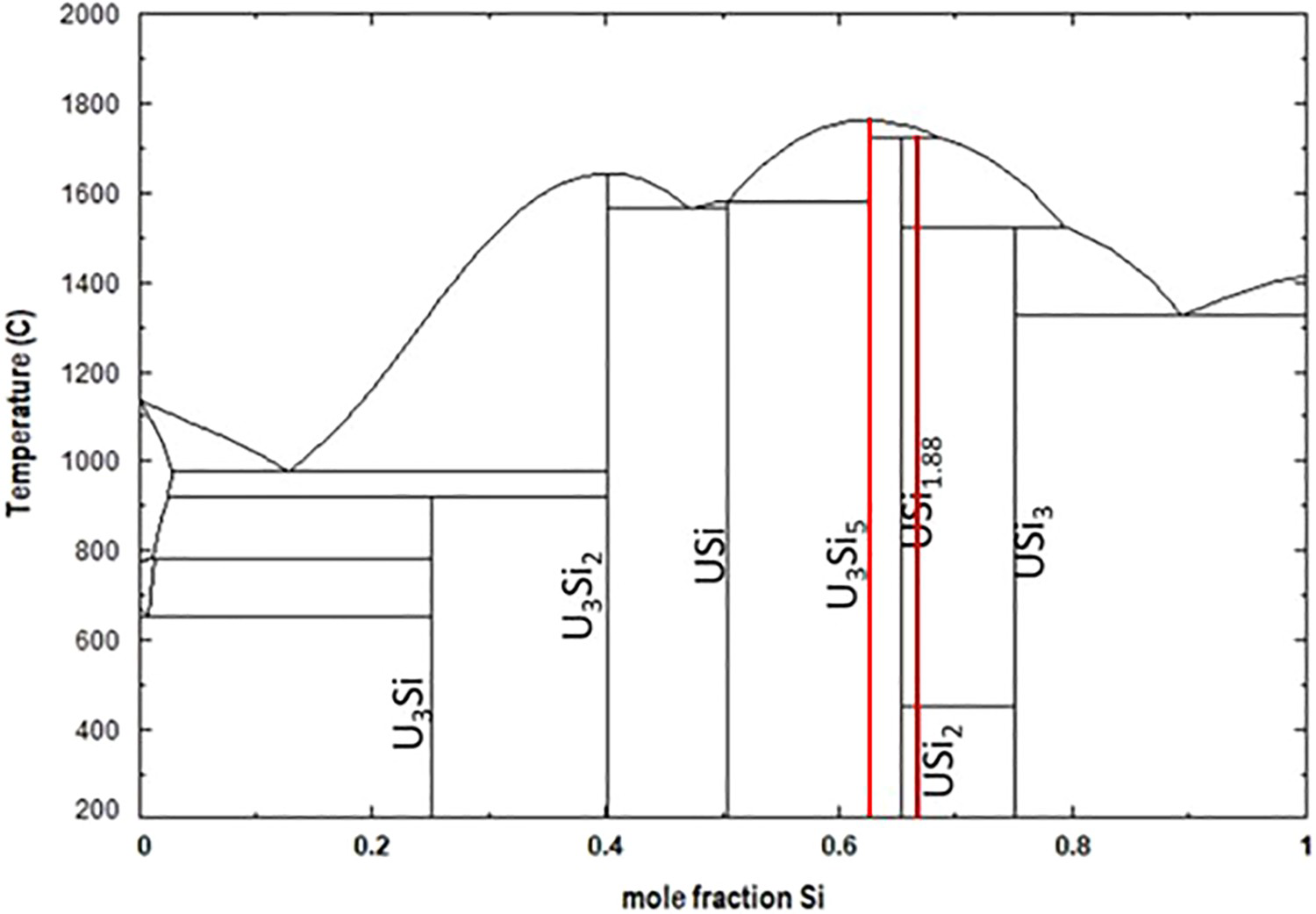
The U-rich portion of the diagram up to U3Si2 is well established and experimental data are in relatively good agreement [13,25,26]. Middleburgh et al. [12] reported DFT studies indicate U3Si2 has a narrow homogeneity range (
Current efforts in the 60–66.6 at.-% Si (U3Si5 to USi2) region of the U–Si system
Four samples with Si/U ratios of 2.01, 1.87, 1.71 and 1.65 were prepared by arc-melting depleted uranium (dU) and silicon metal under an atmosphere of high purity gettered argon, and characterised using scanning electron microscopy (SEM), XRD and differential scanning calorimetry (DSC). To examine structures that may be thermodynamically stable in the region of interest, cluster expansion (CE) calculations were used to evaluate the convex hull of the USi–USi2 phase space, using the hexagonal and tetragonal USi2 structures as one of the end members. Molecular Dynamics (MD) using the U–Si EAM pair-potential was used to relax the structures generated by CE. The red lines in Figure 1 are bounding the region of interest for this work.
XRD analysis of the samples with Si/U ratios of 1.65 and 1.71 in the as-cast condition revealed two high-temperature phases that are isostructural forms of U3Si5. For the silicon-rich composition, Si/U = 1.71, two U3Si5 phases are observed, having different Si site defect arrangements (Figure 2), which appear to correspond to the structures determined by Dwight [27] and Remschnig et al. [13]. At the uranium-rich composition, Si/U = 1.65, these two phases were obtained together with USi.
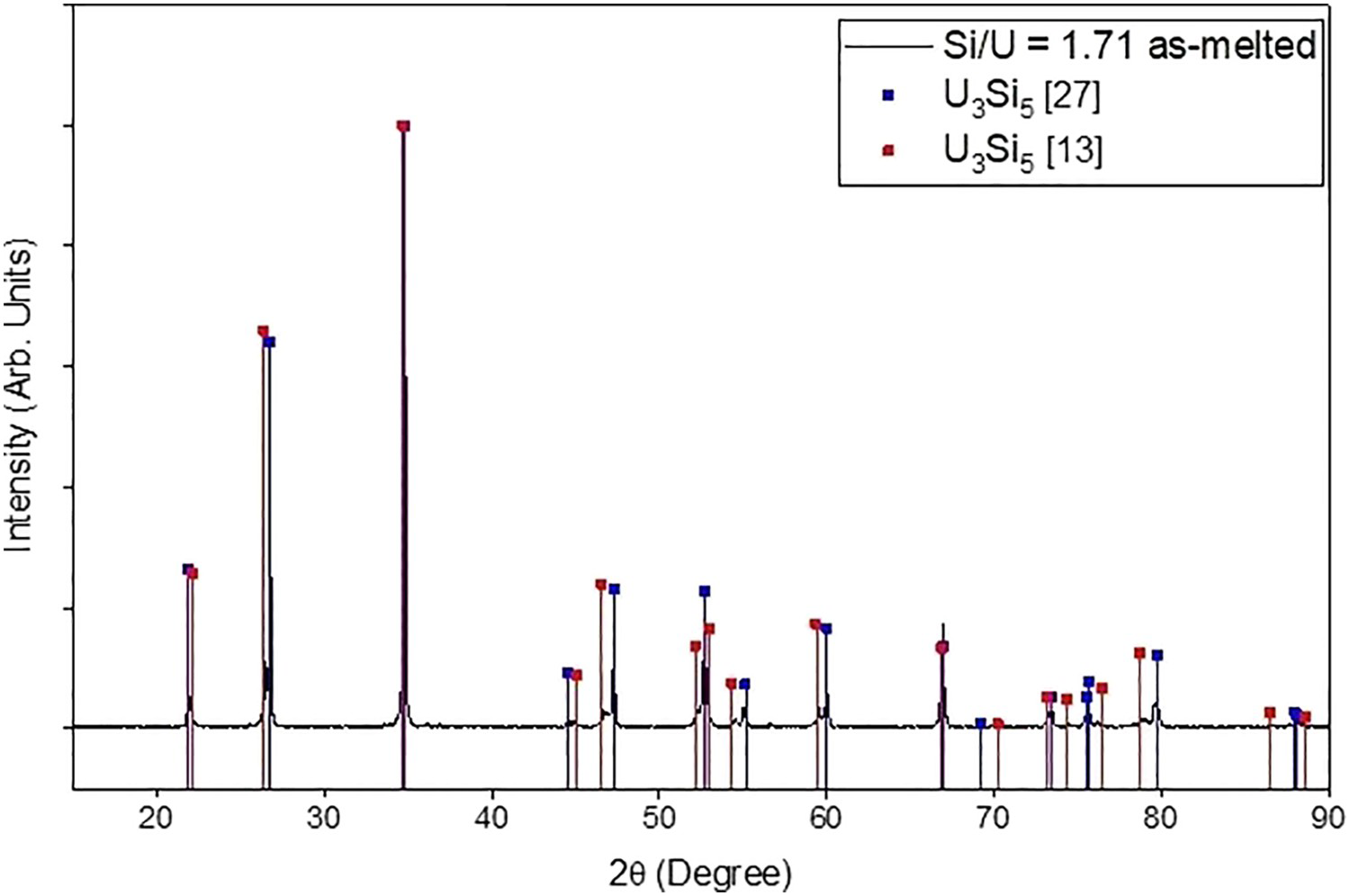
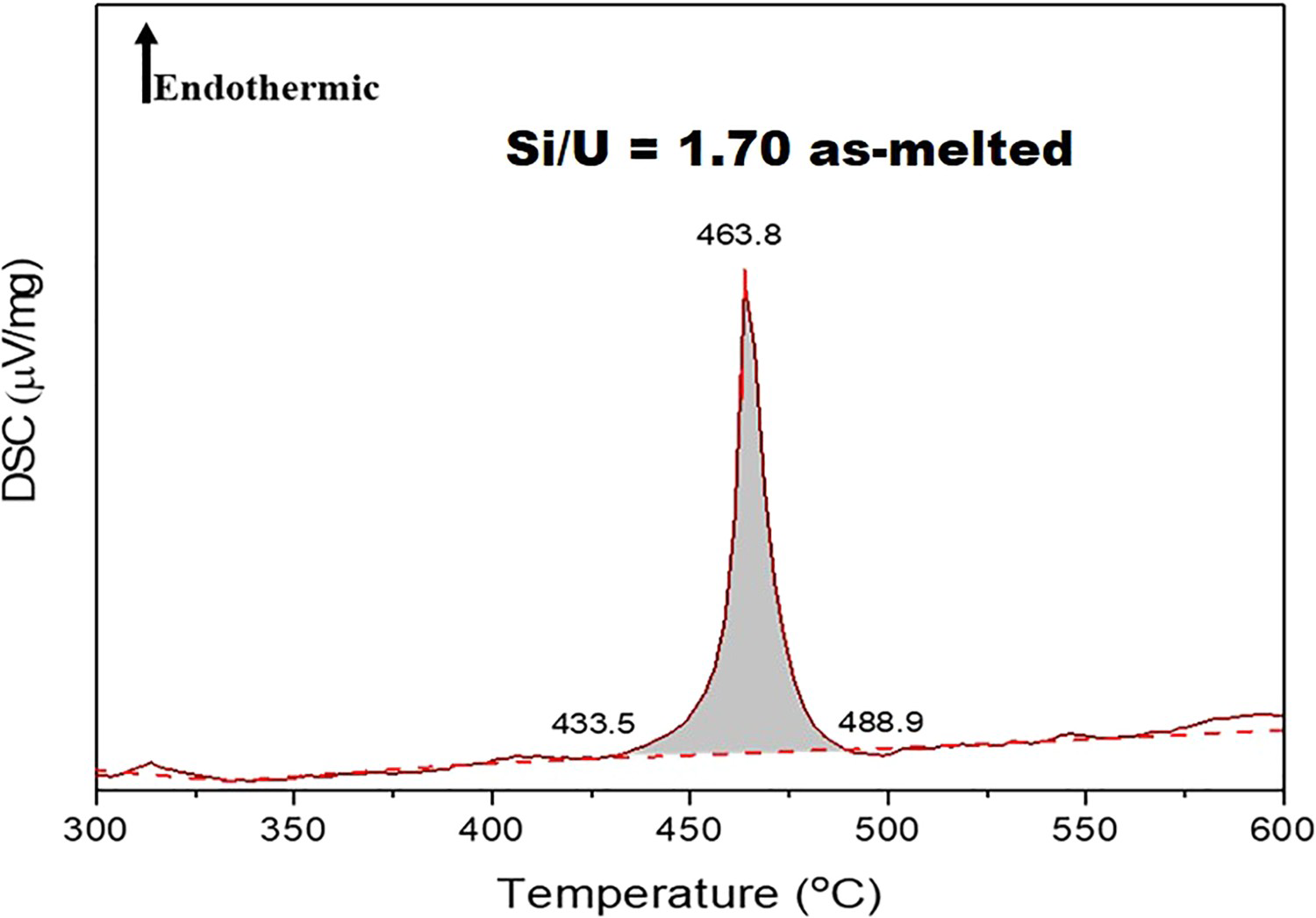
The computed convex hull in Figure 3 shows three structures proximate to the Si/U ratios of 1.5, 1.75 and 1.95. It has been reported that U3Si5 exhibits a homogeneity range to some degree [26,27]. However, in this work, because the relative fractions of the two phases are observed to vary with composition with their lattice parameters remaining constant, it appears that there is no extended solubility for U3Si5. The formation of the two isostructural phases at high temperature appear to correspond to the endothermic transition at 450°C first reported by White et al. [30] and observed in our DSC measurements of samples with Si/U = 1.65 and 1.71 (Figure 4), suggesting a possible miscibility gap.
Formation energies, ΔEf, of USi DSC scan for sample Si/U = 1.71.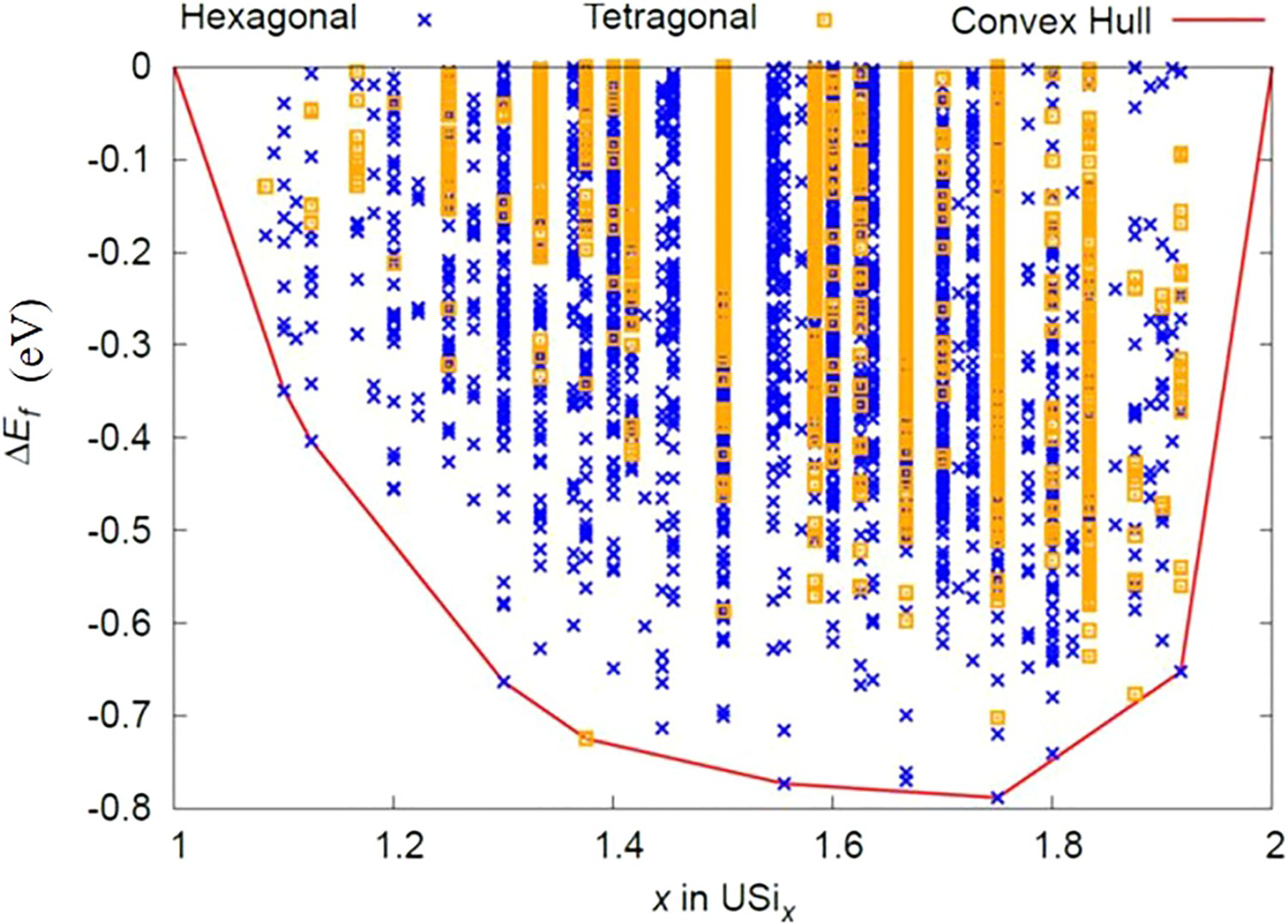
Based on the currently accepted U–Si phase diagram in Figure 1, the USi1.88 phase (tetragonal structure) is formed from the peritectic reaction, U3Si5+L ↔ USi1.88. For the sample Si/U = 1.87, in the as-cast condition we observed that the major phase had a hexagonal structure. However for the sample with Si/U = 2.01 the tetragonal structure was observed, which confirms the existence of this structure type, however, the composition of the phase cannot be confirmed as USi1.88. It is possible that the cooling rate of the arc-melted sample may have been fast enough to prevent the formation of the tetragonal structure USi1.88, whereas annealing could result in its formation. Fully ordered stoichiometric USi2 was not observed in the as-melted samples, in agreement with Remschnig et al. [13], and corroborated by CE calculations which do not have the structure located on the convex hull.
UN–U3Si2 composite
Previous UN–U3Si2 composite observations
Only three groups have prepared UN–U3Si2 composites [3,15,32-34]. Johnson et al. [15] fabricated U3Si2 by conventional arc-melting techniques and prepared UN powder by hydriding-nitriding methods; the composite was formed by mixing ground silicide particles with the UN powder and spark plasma sintered (SPS) at 1500°C. Liquid phase sintering of UN–U3Si2 fuel has been patented [32] as a means to improve densities, with sintering at temperatures close to the melting point of U3Si2, and where >90% theoretical density was achieved. Work by Ortega et al. [34] and White et al. [3] used this technique, sintering at temperatures above 1600°C.
Only in samples sintered above the melting point of U3Si2 did White et al. [3] observe a U–Si–N ternary phase, although Johnson et al. [15] also produced a ternary phase in the lower temperature SPS samples. However, determination of local temperature within samples sintered via SPS is subject to larger uncertainty than conventionally sintered materials. It is commonly accepted that regions of pellets sintered using SPS will experience significantly higher temperatures than the bulk material [35,36]. It is, therefore, possible that regions of the UN–U3Si2 composites produced by Johnson et al. [15] where the ternary phase was observed exceeded the melt point of U3Si2. None report the composition of the U–Si–N phase.
Current efforts on U–Si–N
In our investigation, we have observed a U–Si–N ternary phase in arc-melted samples prepared under N2 partial pressure atmosphere. XRD and SEM-EDS analyses were conducted, however, EDS could not obtain quantitative values as the uranium electron density interferes with the measurement of the light element content. The XRD diffraction pattern revealed peaks not associated with U–Si or U–N phases at 2
Density functional theory (DFT) was used to identify potential ternary phases. It was assumed that the system was analogous to U–Si–C forming phases similar to U3Si2C2 and U20Si16C3 since the U–N and U–C systems are thermodynamically similar [37,38]. While both nitride analogues of the carbides were also computed to be stable, U20Si16N3 was favoured over U3Si2N2. The calculated XRD pattern for both U3Si2N2 and U20Si16N3 were compared to the indexed experimental peaks and a reasonable match was found for U20Si16N3, suggesting the nature of the ternary phase.
U3Si2–FeCrAl(Y) cladding compatibility
Observed interactions
A diffusion couple study performed at Idaho National Laboratory (INL) between U3Si2 and a FeCrAl composition found iron diffusing into the fuel and uranium diffusing into the cladding resulting in multicomponent phase formation, including an unidentified U–Fe–Si ternary composition [23]. The reported U–Fe–Si phases catalogued by Berthebaud et al. [24] include 11 structures, 9 of which are stoichiometric compounds, although thermodynamic values for the phases have not been reported. Since the results of the compatibility study at INL of Hoggan et al. [23] indicated ternary phases of potential nominal stoichiometries of UFeSi and U2Fe3Si, the focus is on these candidate phases. The phases identified in the diffusion layer [23] were confirmed by SEM-EDS and the UFeSi phase was additionally characterised by transmission electron microscopy (TEM), as it was the most abundant of the ternary phases.
First principles and thermodynamic modelling of U–Fe–Si phases
Coefficients for Gibbs energy parameters evaluated via Neumann-Kopp type methods and comparative enthalpies of formation from DFT.

Summary
The concept of ‘Accident Tolerant Fuel’ to improve efficiency and robustness for use in current LWR's during normal and off-normal conditions is explored by investigating uranium-silicide-nitride interactions and uranium-silicide compatibility with ferritic alloy cladding materials. In the study of the uranium silicide system, two isostructural forms of the U3Si5 phase were found. Furthermore, an endothermic transition at 450°C in the same region suggests a miscibility gap between the two structures. It is clear from the work done to date that the current phase diagram does not accurately predict the behaviour of the phases within the 60–66.6 at.-% Si (Si/U = 1.60 to Si/U= 2) region, therefore further work is required to accurately update the binary phase diagram. Additionally the identification of the likely U20Si16N3 ternary phase by more than one method demonstrates the relatively high thermodynamic stability of this phase. The Gibbs energy parameters for UFeSi and the U2Fe3Si were evaluated using the CALPHAD method and appear to correspond to phases observed in compatibility studies of ferritic cladding alloys with U3Si2.
Footnotes
Acknowledgement
This paper was originally presented at the Ultra-High Temperature Ceramics: Materials for Extreme Environments Applications IV Conference (Windsor, UK) and has subsequently been revised and extended before consideration by
Disclosure statement
No potential conflict of interest was reported by the authors.
