Abstract
Healthy rats had been treated for 2 or 6 weeks with 1.0 mL of 10−8 and 10−16 mg/mL of cisplatin. After 2 weeks of treatment, a significant increase in leukocyte and erythrocyte count and also in hematocrit was observed. Among leukocytes the number of neutrophils and eosinophils significantly increased. Biochemical analyses indicated a decrease in the glycogen content in the liver and kidneys after 2 weeks of treatment with low doses of cisplatin but at the end of the experiment (8th week of experiment) the stores of glycogen increased significantly. Biochemical analyses concerning the activity of some enzymes in the liver revealed a significant increase of peroxidase and acid phosphatase as well as catalase activities after 2 weeks of treatment. However, catalase was induced by a very low concentration of cisplatin, 10−16 mg/mL. After the cessation of cisplatin treatment the activity of enzymes returned to normal values.
Human lung carcinoma cell line A549 (ECACC No 86012804) was also studied after treatment with the same doses of cisplatin and inhibition of its growth was observed. The results of these experiments strongly indicated that low doses of cisplatin could be stimulating for healthy cells but cytostatic for tumor cells.
Possible mechanisms involved in the biological activity of very low cisplatin concentrations are discussed.
INTRODUCTION
Cisplatin is an antineoplastic agent that is used commonly in human and veterinary medicine to treat a variety of malignant tumors of testicles, prostate, ovaries, and lungs. Cisplatin is one of the most active agents in lung cancer with a high response rate in small cell lung carcinoma (Arbaye et al. 1993; Ogilvie et al. 1992). In the doses used for anti-tumor therapy, cisplatin is a very toxic substance that induces nephrotoxicosis, bone marrow injury with anemia, trombocytopenia, and other adverse effects such as blindness, deafness, severe nausea, vomiting, hearing loss, and allergic reactions.
Because of such high toxicity of cisplatin, it was interesting to examine whether low doses of this cytostatic drug could inhibit tumor cell growth in vitro and whether a long-term treatment of healthy animals with very low cisplatin concentrations could exert a toxic effect. According to hormesis, we expected a decrease in organism intoxication as well as an increase in its vitality under the influence of very low doses of the same substance, which is known to exert toxic effects.
Wagner et al. (1988) examined the influence of the broad concentration range of cytotoxic and such cytostatic agents as emetin, plumbagin, azatioprine, colchicine, cyclophosphamide, fluorouracil, methotrexate, and vinblastine on human granulocytes and lymphocytes in vitro. Most of the examined compounds showed a dose-dependent biphasic effect: immunosuppressive activity at a concentration higher than 100 ng/mL and immunostimulatory activity at a very low concentration below 100 pg/mL.
Hormesis together with the inverse effect of high and low doses of toxic substances can explain this phenomenon. There are several papers in the literature concerning this problem (Calabrese 2001; Calabrese and Baldwin 2001). Wagner et al. (1988) showed that cytostatic agents inhibit the growth of lymphocytes at concentrations ranging from 100 mg/mL to 10 μg/mL, but stimulate the growth at picogram and femtogram concentrations. A similar dose-dependent effect on phagocytic activity of human granulocytes was observed with emetin and plumbagin, which were toxic at high concentrations, but at very low concentrations they stimulated the phagocytosis of granulocytes. According to the paper by Davenas et al. (1987), silica particles, which are very toxic for phagocytes, can stimulate macrophage function measured by increased amounts of platelet activating factor (PAF) released when mice were injected with very low silica doses (1000 silica particles or less per mouse).
The blood cells are constantly renewed during an individual's lifetime. Hemopoiesis is a very complex process, which is regulated at different levels and by various factors. The most important factors for the regulation of hemopoiesis were recognized as IL-3, CSF-1, G-CSF, GM-CSF, and erythropoietin. They are involved in growth regulation and the differentiation of blood cell precursors (Bastide et al. 1987).
The aim of this paper is to collect and compare physiological data of healthy animals and tumor cell cultures treated with very low doses of cisplatin. Proper levels of enzymes in the liver and proper hematological parameters are very important markers of good health. For this reason the main hematologic parameters were determined in the blood of rats, which had been treated per os with low doses of cisplatin. At the same time, the activities of catalase, peroxidase, as well as alkaline and acid phospahatases were determined in livers because of their physiological function in organism detoxication. The results of these experiments were compared with the reaction of tumor cells growing in vitro and treated with identical dilutions of cisplatin as those used in animal analyses.
MATERIALS AND METHODS
Preparation of Cisplatin Dilutions
One mg of cisplatin (Ciba Geigy) was dissolved in 1 ml of double distilled water and 100-fold dilutions from 10−2 to 10−16 mg/mL were prepared serially and were vigorously mixed after each stage. The dilutions 10−8 (as a concentration of cisplatin that can be detected by sensitive methods) and 10−16 mg/mL (as a concentration approaching Avogadro's number) were chosen and used in all experiments.
Treatment of Tumor Cells
Human lung carcinoma cell line A549 (European Collection ECACC No 86012804) was grown at 37°C in an atmosphere containing 5% CO2 in Eagle's MEM supplemented with 10% fetal calf serum and antibiotics (100 U/mL penicillin and 100 μg/mL streptomycin). All the experiments were carried out on cells that after trypsinization were seeded at the density of 1 × 105 cells/mL on plastic Petri dishes (82 mm in diameter). After the adhesion of the cells, the medium was removed and 0.9 mL of fresh medium and 0.1 mL of cisplatin dilution were added to the respective dishes. Cell controls were treated with 0.1 mL of distilled water instead of cisplatin. Four plastic plates were used for each dilution and control. The experiments were repeated three times. Cell cultures were incubated at 37°C for 72 h. The culture medium was discarded, the cells were rinsed in phosphate-buffered saline (PBS), and 1 mL of 0.025% solution of trypsin in PBS was added. The cells suspended in trypsin solution were counted under the microscope in a hemacytometer and cell viability was determined by standard trypan blue exclusion test (Szalewicz et al. 1992). The protein level in the cells was determined with the Lowry et al. (1951) method after washing the cells in PBS. Absorbance at 660 nm was measured by spectrophotometry (Shimazu Gmbh, Japan UV-1202).
Animal Treatment
Forty-eight 5-week-old Lewis rats were divided into three groups, 8 male and 8 female in each group. They were fed on a standard laboratory solid diet supplemented with raw potatoes and carrots as a source of vitamin C and A. During the experiment, every morning at 8 a.m., each animal received per os one mL of cisplatin dilution, either 10−8 (10 picograms/mL) or 10−16 (10 attograms/mL) mg/mL, instead of drinking water. After that they had a free access to water only during the day but not during the night. Control rats received an equivalent volume of distilled water. Drinking water was provided in new glass bottles with rubber stoppers and glass tubing. Food consumption, behavior, skin lesions, diarrhea, and other external abnormalities were recorded every day. During the 2 weeks of treatment with dilution 10−8mg/mL of cisplatin, each rat received a total of 140 picograms of the drug. Rats treated with dilution 10−16 mg/mL received a total of 140 attograms of cisplatin per animal. After 6 weeks of treatment rats received, respectively, 420 picograms or 420 attograms per animal. After 6 weeks of treatment with cisplatin hematological and enzymatic parameters of 16 rats were also estimated 2 weeks after cessation of drug treatment (8th week of experiment). During those additional 2 weeks rats had a free access to water and food.
Hematological Variables
In the second week and the sixth week, as well as 2 weeks after the sixth week of treatment (8th week of the experiment) the blood samples were collected under deep ether anesthesia. Before blood collection, the animals had a free access to drinking water for 24 h to prevent dehydratation. Differential blood counts were performed using routine methods. The numbers of red and white cells were counted in a hemocytometer. Hematocrit was established in a hematocrit centrifuge and hemoglobin content was measured by routine cyanomethemoglobin method. Differential blood counts were performed on Giemsa stained blood smears by counting the minimum of 200 white blood cells per each smear.
Biochemical Determinations
Livers were taken from rats after blood sampling, washed in phosphate-buffered saline, and homogenized in a glass homogenizer in cold buffer and centrifuged. Enzyme activities were determined in the supernatant. Also, kidneys were taken from rats after the last blood sampling, washed in phosphate-buffered saline, homogenized in a glass homogenizer in cold buffer, centrifuged, and the level of glycogen was measured.
Catalase activity was measured according to the Hamilton et al. (1961) method. Two mL of the supernatant were mixed with 7 mL of distilled water and 2 mL of 1% H2O2. The rate of H2O2 degradation was measured at 240 nm. One unit of the activity was considered as the amount of the enzyme, which degraded one mg of H2O2.
Peroxidase activity was measured in accordance with Clairborne and Fridovich (1979), the method based on o-dianisidine oxidation in the presence of 0.003% H2O2in 0.1 M acetic buffer, pH 5.5. The relative activity was calculated on the basis of the absorbance coefficient = 11.3.
Alkaline and acid phosphatase activities were calculated on the basis of the rate p-nitrophenol release from 2.5 mM nitrophenyl phosphate at pH 5 or pH 8.6, respectively, according to Szalewicz et al. (1992). The hydrolysis of one μM of the substrate per minute was considered as the activity unit.
The level of glycogen in livers and in kidneys of the tested animals was measured according to the Babcock and Cardell method (1974) and calculated in μg per mg of tissue dry weight.
The Student's t test was used for statistical analysis of the results.
RESULTS
Cell Cultures
As shown in Figure 1, cisplatin tested at concentrations of 1 μg/mL inhibited the growth of A549 cells, when compared with control cultures treated with water. Cell numbers and protein levels were lower in the cultures treated with cisplatin, compared with control cultures treated with water. Compared with control where few dead cells were present and the viability of cells was 99%, the number of viable cells after a treatment with cisplatin concentration of 1 μg/mL decreased to 78%. Moreover, low concentrations of cisplatin equal to 10 pg and 0.1 ag/mL were also active and inhibited cell growth. Especially the concentration of cisplatin 0.1 ag/mL significantly decreased cell numbers and protein concentration. This dilution of cisplatin was cytostatic rather than cytotoxic, because the viability of cells in cultures was between 95% and 97%.

Observations of Animals Treated with Cisplatin
All tested animals were observed during the treatment with low cisplatin doses. No significant statistical differences were observed between control and experimental animals concerning food consumption and behavior; no weight loss, skin lesions, diarrhea or other specific abnormalities were noticed.
Hematological Measurements
The influence of low doses of cisplatin on some hematological parameters in rats was examined. Two dilutions of cisplatin were chosen for experiments: 10−8 mg/mL and 10−16 mg/mL. Rats treated with both dilutions of cisplatin had increased total erythrocyte and leukocyte count, and a higher hematocrit and hemoglobin concentration (Table 1). The differences were especially visible and statistically significant in comparison with control after treating rats with picogram doses. After 6 week of treatments this dilution was still more active than the attogram doses; however, the differences in comparison with control were not statistically significant. Differential leukocyte counts revealed increased numbers of neutrophils and eosinophils in the blood (Table 2). It should be stressed that all hematological parameters were still within the range considered as normal in rats. These parameters were also evaluated 2 weeks after the cessation of drug treatment. As shown in Table 1 and Table 2, they returned to normal and no statistically significant differences between control and experimental animals could be observed. Blood cell counts, hematocrit index, and the number of neutrophils and eosinophils returned to the values observed in control animals.
The levels of leukocyte, erythrocyte, hematocrit and hemoglobin in the blood of rats after 2, 6, and 8 weeks (2 weeks after 6 weeks of cisplatin treatment) (*statistically significant difference in comparison to control at p<0.05). The results are presented as a mean ± SD.

- 140 picograms
-140 attograms
- statistically significant difference at p< 0.05 in comparison to control.
The influence of cisplatin treatment on differential leukocyte count in rats after 2, 6, and 8 weeks (2 weeks after the 6-week treatment) of pico- and atto-grams/mL of cisplatin (* statistically significant difference at p < 0.05 in comparison to control). The results are presented as a mean ± SD.

- 140 picograms
- 140 attograms; N - neutrophils, L - lymphocytes, E - eosinophils, M - monocytes, B - basophils
- statistically significant difference at p< 0.05 in comparison to control.
Enzymatic Activity Detection
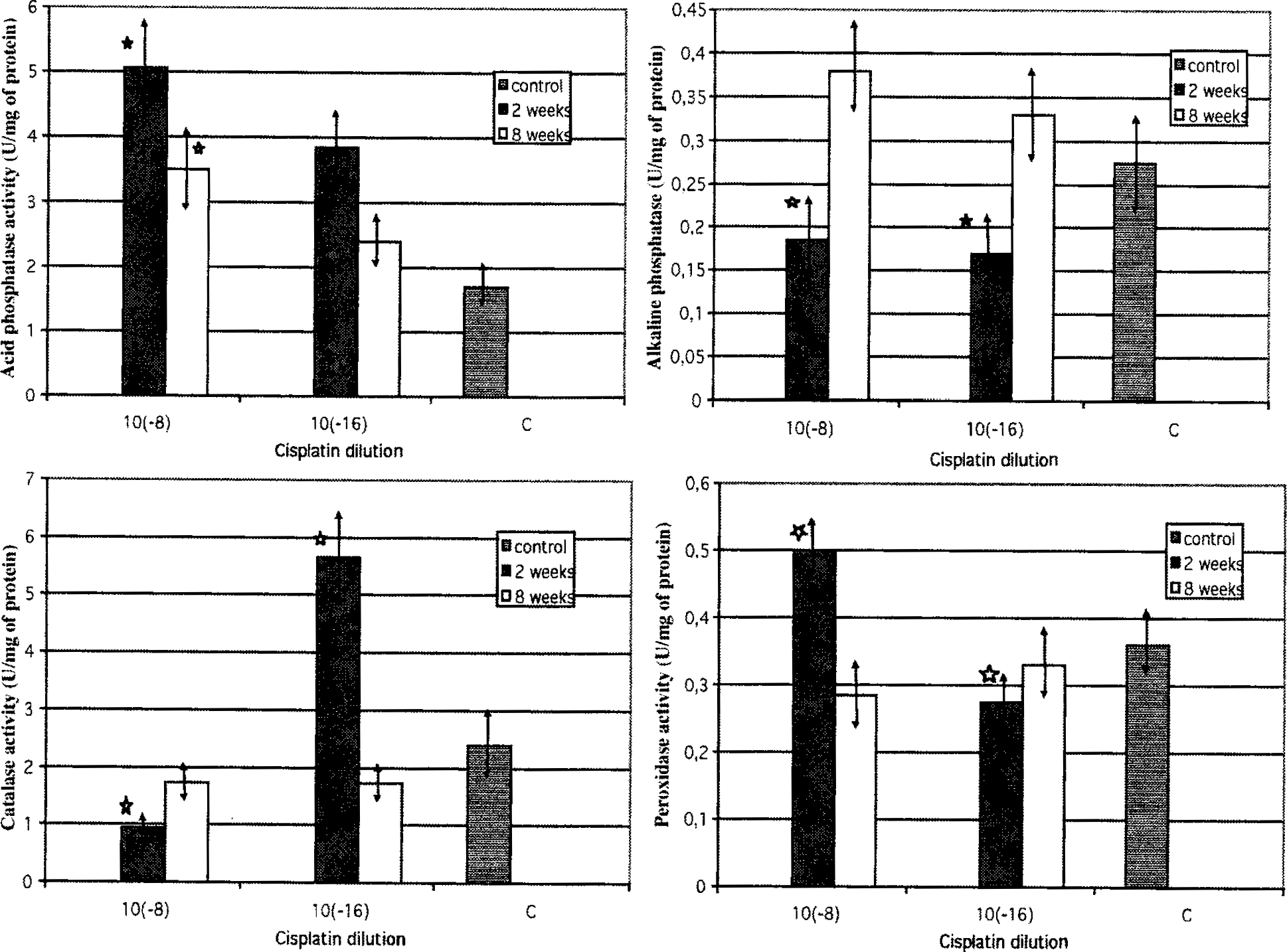
The level of activity of four enzymes: catalase, peroxidase, and two phosphatases were measured in the liver homogenates after 2 and 8 weeks of treatment of rats with low doses of cisplatin. Figure 2 shows the results of enzymatic experiments. The patterns of every enzyme activity were different. When rats were treated with pico concentrations of cisplatin, peroxidase and acid phosphatase were significantly higher than in control, but they decreased after 8 weeks (6-week treatment + 2 weeks of interval). Compared with the enzymes mentioned above, alkaline phosphatase activity was higher at the end of the experiment rather than at the beginning. After 2 weeks of treatment, the activity of catalase was stimulated with a low concentration of cisplatin (10−16 mg/mL). It is worth mentioning that the levels of acid and alkaline phosphatases, catalase, and peroxidase at the end of the experiment, 2 weeks after cisplatin cessation, were more similar to controls than at the beginning. However, they never fell below the level observed in control.
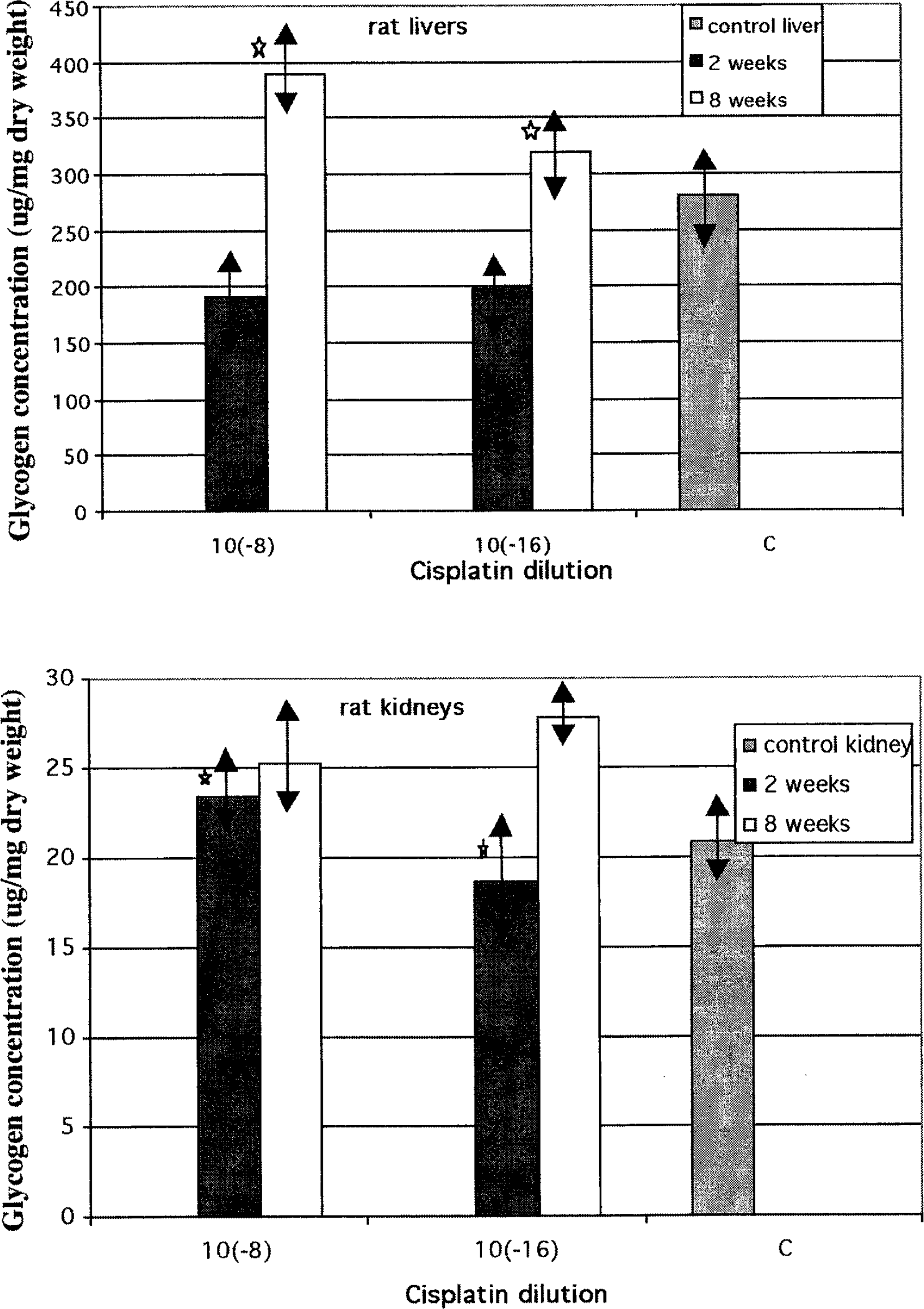
Figure 3 shows the glycogen levels in the liver and kidneys during and after cisplatin treatment of rats. The differences in the levels of glycogen during and after experiments with cisplatin exceeded no more than 10 μg in kidneys. In the liver, the level of glycogen was lower than control after 2 weeks of treatment and higher after finishing cisplatin administration.
DISCUSSION
It has been shown that cisplatin induces oxidative stress in the organism by generating oxygen free radicals in macrophages, platelets, and kidney cells and by inhibiting antioxidant enzyme activity, such as peroxidase and superoxide dismutase (Son and Kim 1995; Sugihara et al. 1987; Wachowicz and Szwarocka 1994; Wachowicz et al. 1995). It is well known that antioxidant enzymes play a fundamental role in protecting cells and tissues against oxidative damage (Andersen et al. 1997; Scandalios 1992; Batzer and Aggarwal 1986; Bishayi et al. 2002). The activity of these enzymes increases in the organism when reactive oxygen species are generated. As high concentrations of cisplatin can inhibit at least some of the antioxidant enzyme activity, it was important to examine if low concentrations of cisplatin can influence such activity. Two so-called “stress enzymes”, catalase and peroxidase, were chosen for experiments.
Mammalian catalase is abundant in the liver, where the enzyme resides mainly in peroxisomes and plays an important role in eliminating H2O2 generated during the dismutation of superoxide anion by SOD. Peroxidase works in a similar way, however, it acts when the amount of hydrogen peroxide is low. In our experiments picogram doses of cisplatin inhibited catalase activity in rat liver. However, lower attogram doses of cisplatin significantly stimulated its activity, especially after 2 weeks of treatment. When peroxidase activity was measured by oxidation of aromatic compounds such as o-dianisidine, a significant increase in its activity was detected in the liver of the rats which received the picogram doses of cisplatin. The attogram doses of cisplatin were not active. These observations suggest that cisplatin at very low doses can stimulate the activity of the antioxidant enzyme system in the liver. This phenomenon can be explained by the reverse activity of the high dose, which can inhibit the activity of cells and enzymes, and of the low dose, which stimulates it.
Hepatotoxicity is not considered as a dose-limiting toxicity for cisplatin, but liver toxicity can occur when the antineoplastic drug is administered at a high dose (Charalabopoulos et al. 2002; Cersosimo 1993; Bishayi et al. 2002; Zicca et al. 2002; Ueda et al. 1998). Alkaline phosphatase and especially its isoform present in the liver (LALP) are very often used as markers of hepatic subacute toxicity of antineoplastic agents (Pispirigos et al. 1993). It has been detected that ciplatin inhibits strongly LALP in the liver of dogs suffering from appendicular osteosarcoma (Garzotto et al. 2000) and also alkaline phosphatase and acid phosphatase in rat kidneys (Fenoglio et al. 2002). Therefore, we decided to measure the activity of two phosphatases, alkaline and acid isoforms, present in the liver. A significant increase in peroxidase activity was also accompanied by a significant increase in the activity of acid phosphatase. It should be pointed out that in our experiments the activity of alkaline phosphatase, which increases in the liver during infections and can also be induced by stress, was rather inhibited by low-dose cisplatin treatment and returned to normal level after cessation of drug treatment. It seems likely that different enzymes may be regulated differently by low doses of cisplatin.
According to the opposite activity of high and low doses of certain biologically active substances, we expected that cisplatin, an anticancer drug, would stimulate tumor cell growth in vitro. To our surprise, cisplatin at attogram concentrations inhibited tumor cell growth in vitro in comparison with untreated control. The activity of low cisplatin concentration was not as strong as cisplatin used at microgram concentrations; however, the effect measured by two different methods was detectable.
There arose a question whether the changes observed in the hematological parameters in the blood of experimental animals were the consequence of a direct action of cisplatin on progenitor cells in bone marrow or whether cisplatin is able to influence the production and action of several growth factors. The toxicity of cisplatin, at therapeutic doses, to bone marrow cells is evident and probably cisplatin can trigger apoptosis and affect cell cycle causing anemia and a decrease in leukocyte count. These effects are reversible after drug discontinuation (Holdack et al. 1992; Mazur et al. 2002; Sachs and Lotem 1994). In our experiments cisplatin at low doses acted as mild stimulator of bone marrow function that resulted in an increase of the total number of leukocytes and erythrocytes. Differential leukocyte counts revealed drat this increase was caused mainly by an increase in the number of neutrophils and eosinophils. The results of our experiments on rats are in agreement with the observations of Wagner et al. (1988), Holldack et al. (1992) and Sodhi and Pai (1992). They indicated that very low doses of cisplatin could stimulate bone marrow to produce red cells with higher hemoglobin content and high numbers of white blood cells.
The blood cells are constantly renewed during an individual's lifetime. Hemopoiesis is a very complex process that is regulated at different levels and by different factors, such as IL-3, M-CSF, G-CSF, GM-CSF, erythropoietin. These cytokines are involved in growth regulation and the differentiation of blood cell precursors (Sodhi and Pai 1992; Holldack et al. 1992). We can only speculate that low doses of cisplatin can influence cell cycles or cytokine production and in the final effect stimulate bone marrow activity.
It has been detected recently that, depending on the concentration, cytokines may exert opposite effects. For example, M-CSF at a high (5 U/mL) concentration activates the extracellular signal-regulated kinase (ERK) pathway, while a low (0.05 U/mL) concentration reduces ERK phosphorylation and affects cell proliferation (Rovida et al. 2002). Moreover, tumor necrosis factor (TNF) at a low concentration (1 U/mL) decreases myofibrillar protein content, while at relatively high concentrations (100 U/mL or more) increases protein content in muscle cell line (Alvares et al. 2001). Cisplatin, at high concentrations, possesses clastogenic activity in bone marrow; however, by modulation of cytokine production it may exert an opposite effect on bone marrow cells. It is worth mentioning that aflatoxin B, a natural cytotoxic substance, also exerts opposite effects: at high doses (280 ppb) stimulates bone marrow, while at a low dose (140 ppb) decreases total number of white blood cells in piglets (Marin et al. 2002).
All these results strongly indicate that low doses of toxic substances such as cisplatin could stimulate healthy cells but inhibit tumor cell growth. It is possible that these opposite reactions of animal cells to toxins used at low or high concentrations are the result of differential influence on the apoptotic mechanism in normal and pathological cells (Calabrese 2001).
The protective effect of high dilutions of metal on cultured cells was demonstrated by Delbancut et al. (1997). Cisplatin was used at a cytotoxic concentration (10−5 M), but protective doses of cisplatin or cadmium were used at high dilutions (10−10and higher). The protective index of cadmium against cisplatin cytotoxicity was weak, but it was more pronounced for the cells pretreated with low doses of cisplatin. It means that 10−5 M cisplatin killed only 63% of cisplatin-pretreated cells, while it killed 68.4% of cadmium-pretreated cells. These experiments confirmed the rule that the effectiveness of the pretreatment requires the use of the same substance for protecting cells against the toxic effect (Van Wijk and Wiegant 1997).
The results of the experiments, showing that very low doses of cisplatin are not dangerous to animal health but are toxic to tumor cells, opened the possibility of using low doses for protecting patients against adverse effects of anticancer therapy.
