Abstract
The health effects of low-dose radiation (LDR) have been the concern of the academic spheres, regulatory bodies, governments, and the public. Among these effects, the most important is carcinogenesis. In view of the importance of immune surveillance in cancer control, the dose-response relationship of the changes in different cell types of the immune system after whole-body irradiation is analyzed on the basis of systemic data from the author's laboratory in combination with recent reports in the literature. For T lymphocytes J- or inverted J-shaped curves are usually demonstrated after irradiation, while for macrophages dose-response curves of chiefly stimulation with irregular patterns are often observed. The intercellular reactions between the antigen presenting cell (APC) and T lymphocyte (TLC) in the immunologic synapse via expression of surface molecules and secretion of cytokines by the two cell types after different doses of radiation are illustrated. The different pathways of signal transduction thus facilitated in the T lymphocyte by different doses of radiation are analyzed to explain the mechanism of the phenomenon of low-dose stimulation and high-dose suppression of immunity. Experimental and clinical data are cited to show that LDR retards tumor growth, reduces metastasis, increases the efficacy of conventional radiotherapy and chemotherapy as well as alleviates the suppression of immunity due to tumor burden. The incidence of thymic lymphoma after high-dose radiation is lowered by preexposure to low-dose radiation, and its mechanism is supposed to be related to the stimulation of anticancer immunity induced by low-dose radiation. Recent reports on lowering of standardized cancer mortality rate and all cause death rate of cohorts occupationally exposed to low-dose radiation from the US, UK, and Canada are cited.
INTRODUCTION
Currently, the linear no-threshold (LNT) hypothesis is adopted for cancer risk assessment in radiation protection, which is based on observations from the carcinogenic effect of high-doses of ionizing radiation (UNSCEAR 1994; NCRP 2001). However, data from inhabitants in areas of high natural radioactivity and workers in nuclear industry have not shown increase in cancer risk after exposure to low-dose radiation. The LNT hypothesis emphasizes the stochastic nature of DNA damage caused by ionizing radiation. However, it should be emphasized that the defense and adaptive reactions in the body are often activated by low-dose radiation and down-regulated by high-dose radiation. Among these reactions antioxidants, DNA repair, apoptosis of damaged cells, adaptive response in mutagenesis, and immune surveillance are important ones (Yamaoka et al. 1991; Feinendegen et al. 1995; Kondo 1993; Zhou et al. 1993; Rigaud and Moustacchi 1996). The present paper concentrates on the analysis of the dose-response relationship in the immune system after whole-body irradiation (WBI).
Establishment of dose-response relationship of biological parameters is one of the key topics in radiobiology research. Due to the importance of immune surveillance against cancer and infection, radiation effect on the immune system has been one of the chief research fields in radiation biology and radiation protection. However, in most previous studies only the changes in immunity after exposure to doses above 0.2 Gy were observed, thus usually giving rise to a linear or quadratic dose-response curve (Liu and Liu 1990; Liu 1996; Yamaoka et al. 1991). When the effect of doses of 0.2 Gy and lower are studied, usually a J-or inverted J-shaped curve can be demonstrated for parameters of the lymphocytes, and curves showing chiefly stimulation with irregular patterns are often observed for macrophages. Elucidation of the characteristics of these dose-response curves is of importance in understanding the manifestations and mechanisms of immunologic changes induced by different doses of ionizing radiation.
Since the 1980s of the last century radiation effect on the immune system has been a constituent part of many important international conferences concerning the biological effects of low-dose radiation (Sagan 1987; Sugahara et al. 1992; Liu 1994; Yamada et al. 2000; Burkart et al. 2002), and there have been many peer-reviewed papers discussing the stimulatory effect of low-dose radiation on immunity in international journals (Troup and Anderson 1982; Liu et al. 1987; James and Makinodan 1988; Makinodan and James 1990; Anderson 1992; Liu 1992, 1995; Hattori 1994). However, not many of these involve the dose-response relationship of ionizing radiation-induced immune changes. In 1994 in its report to the UN General Assembly, UNSCEAR for the first time recognized the adaptive response induced by low-dose radiation (UNSCEAR Report 1994 to the General Assembly of United Nations). In this document stimulation of immunity induced by low-dose radiation was included in adaptive responses. However, its mechanism and influence on cancer risks were not fully analyzed. Since the publication of UNSCEAR 1994 report, more scientific data have been accumulated in international literature, making it possible to give an analysis of the topic in more depth. In the present paper three aspects of the effect of ionizing radiation on the immune system will be analyzed, that is, dose-response relationship of immunologic parameters, mechanistic studies on low-dose stimulation of immunity, and the implications in relation to cancer risks.
DOSE-RESPONSE RELATIONSHIP OF RADIATION INDUCED IMMUMOLOGICAL CHANGES
The dose-response relationship of immunological parameters following exposure to ionizing radiation is affected by a number of factors. Most important among these are the target cells under observation and the experimental design. When the lymphocytes and related functions are studied, a J- or inverted J-shaped dose-response curve is usually observed. If the macrophages and related functions are under study, chiefly stimulation with irregular patterns may appear over the usual dose range within 10 Gy. In order to confirm the existence of stimulation by low-dose radiation in animal models, experimental design may be a critical factor. Dose range, dose spacing, and dose rate as well as temporal relationship of the changes are crucial in the experimental design. Usually three orders of magnitude have to be chosen for the dose range, that is, from 0.01 to 10 Gy and within this range 3∼4 doses are necessary for each of the dose ranges of 0.01∼0.1 Gy, 0.1∼1 Gy, and 1∼10 Gy with 10 or more dose points. In the author's laboratory, for whole-body irradiation (WBI) with X-rays in Kunming mice usually 0.025, 0.05, 0.075, 0.1, 0.2, 0.5, 1.0, 2.0, 4.0, and 6.0 Gy and sham-irradiated control are used to construct a dose-response curve in most immunological studies evaluating the response of lymphocytes to whole-body irradiation (Liu 1992, 1995; Liu et al. 1992). For more radioresistant parameters, such as NK activity and ADCC activity, more doses in the higher end are needed, e.g., extending to 10, 12, and 14 Gy, in order to reveal the suppressive effect (Liu et al. 1992; Fan and Liu 1989).
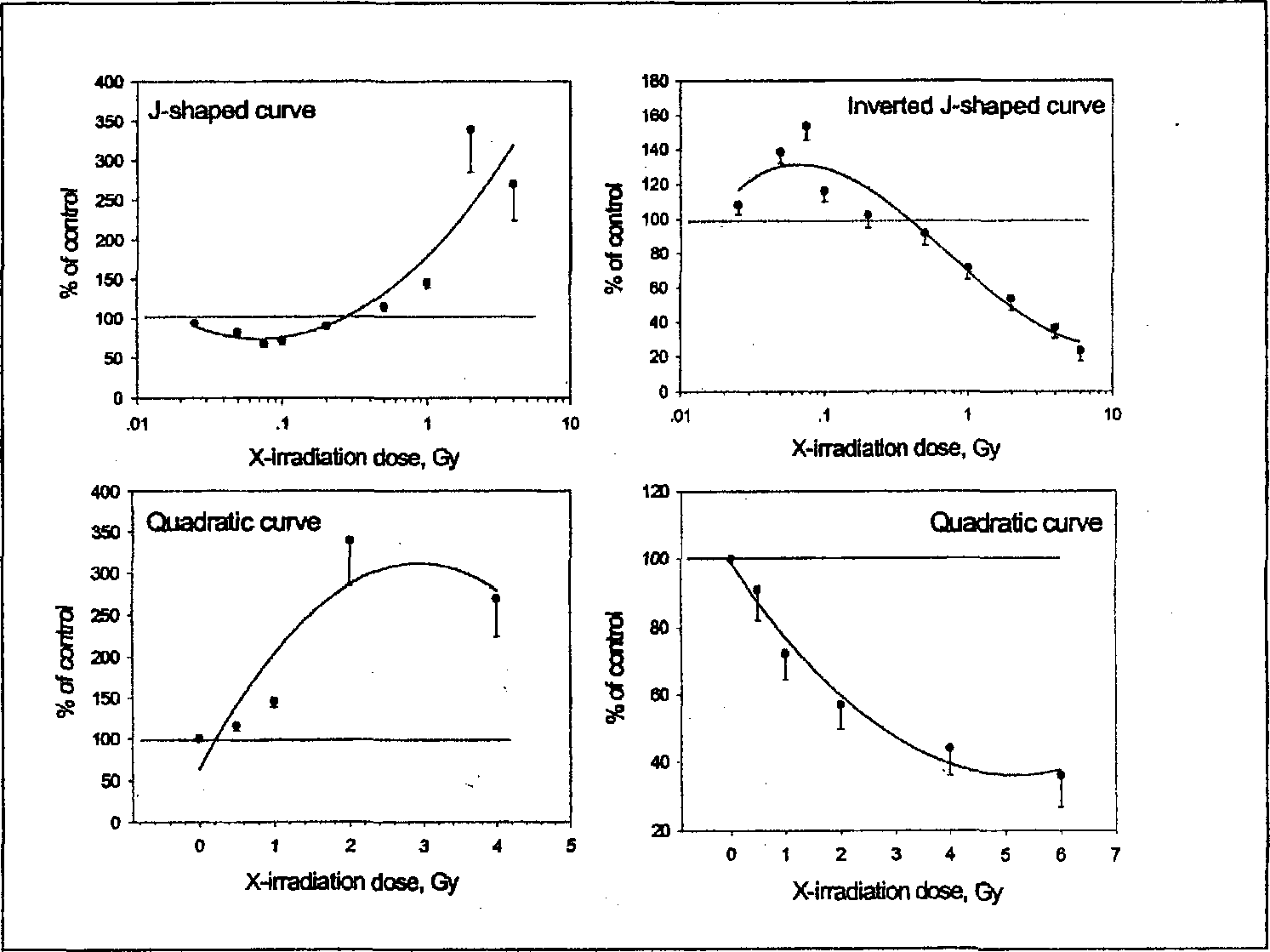
The stimulatory effect of low-dose radiation on immunity has been documented (Troup and Anderson 1982; Liu et al. 1987; James and Makinodan 1988; Makinodan and James 1990; Anderson 1992; Liu 1992, 1995, 1996; Hattori 1994). However, the dose-response relationship of the immunologic changes, especially in a coordinated manner synthesizing various effects at different levels, has not been reported. In the present paper 52 immunologic parameters in two categories are pooled to construct dose-effect curves. The dose-effect relationship of one category consisting of 20 parameters shows a J-shaped curve (left upper panel of Figure 1). These parameters include: (1) cell activities, such as cell cycle control, cell survival (apoptosis); (2) surface molecules, such as CTLA-4; (3) cytokines, such as IL-10 and IL-4 synthesis, (4) signal molecules, such as cAMP and cAMP to cGMP ratio, protein kinase A and phospholipase A2, glucocorticoid receptor (GCR). Up-regulation of all these parameters in the immune organs would lead to decreased immune activity. They are down-regulated after LDR, and that of the second category consisting of 32 parameters shows an inverted J-shaped curve (right upper panel of Figure 1). These parameters include: (1) cell number, such as thymocyte count, splenocyte count, peripheral blood WBC count; (2) cell activity, such as spontaneous 3H-TdR incorporation into thymocytes, proliferative reaction of splenocytes to Con A and LPS, PFC reaction of spleen, NK and ADCC activity of splenocytes; (3) cytokine secretion, such as, IL-2 and IFN-gamma secretion, (4) surface molecule expression, such as: CD25 (IL-2Rα), CD71 (transferring receptor), CD28, CD2, CD48; (5) DNA repair, such as UDS, ribonucleotide reductase, DNA polymerase (total and β activity); (6) signal molecules, such as free calcium ion concentration, cGMP content, p38MAPK. Up-regulation of these parameters in the immune organs would lead to increased immune activity. They are up-regulated after low-dose radiation. These curves were constructed each with 10∼11 doses ranging from 0 to 10 Gy (Liu 1996; Liu et al. 1994a, 1996, 2000, 2001; Liu and Xie 2000; Liu et al. 2001a, 2001b; Sun and Liu 2000a). In many studies in the literature doses below 0.2 Gy were often neglected, thus the effect of low-dose radiation would be missed. This is shown in the lower panels of Figure 1 in which data with doses of 0.025,0.05,0.075, 0.1, and 0.2 Gy were omitted, thus giving rise to quadratic curves instead of the J- or inverted J-shaped curves.
Another factor is the strain of animal used. When the irradiation conditions remain the same, different strains of mice may react differently to low-dose radiation. Take the proliferative reaction of lymphocytes to Con A as an example, BALB/c mice and C57BL/6 mice may react in an opposite direction (Shankar B et al. 1999). When C57BL/6 and BALB/c mice were exposed to fractionated low-dose radiation (0.04 Gy/day, 5 days/week, total dose 0.2 Gy) and subsequently stimulated with Con A, the proliferation of spleen cells in response to Con A as measured by 3H-TdR incorporation was significantly higher in 0.2 Gy-irradiated C57BL/6 mice when compared with that in the Con A-stimulated cells from sham-irradiated controls, but the same response was suppressed by 0.2 Gy in BALB/c mice. The augmentation of response to Con A in C57BL/6 mice was marked by the decrease in the proportion of cells expressing p53 as estimated by flow cytometry. A reduction in expression of p53 was accompanied by reduced apoptosis in the Con A-stimulated spleen cells of irradiated C57BL/6 mice when compared with that in the sham-treated controls. However, the splenic cells of BALB/c mice showed exactly opposite profiles in this respect. The Kunming strain and C57BL/6 strains of mice react to low-dose radiation similarly (Liu 1995, 1996, 1998). Thus, alteration in the immune response following low-dose radiation is evidently affected by the strain of mice used. It has been reported that BALB/c mice are more sensitive to cancer induction by radiation than C57BL/6 mice due to the genetic instability induced by radiation in the former strain of mice (Ponnaiya et al. 1997).
For BALB/c mice the stimulatory effect of the low-dose radiation may be conditioned by the dose rate used, e.g., a dose rate of 15 mGy/min or lower would usually be effective in eliciting a stimulating effect with total doses of 77 and 106 mGy, resulting in doubling the proliferation rate to that of sham-irradiated control, but a dose rate of 200 mGy/min with the same total dose may not be effective in eliciting stimulation of proliferation in BALB/c mice (Su et al. 1997). In this experiment the proliferative response of splenocytes to Con A was observed in BALB/c mice measured by 3H-TdR incorporation after whole-body X-irradiation (Table 1).
Dose rate effect of T cell stimulation by low-dose radiation

P<0.01 vs sham-irradiated control
The temporal changes of the biological parameter under study may sometimes also influence the shape of the dose-response curve. It is especially important when “overcompensation stimulation” is concerned in the mechanism of the hormetic effect (Calabrese 2001). As listed in this article, the hormetic effect elicited by a number of chemical agents showed an inverted J-shaped curve only at a certain time point after the administration of the chemical agent. The same may be true for some of radiation-induced effects. Take apoptosis as an example, a J-shaped dose-response curve was demonstrated both for thymocytes 24 h after WBI of mice (measuring DNA fragmentation with fluorescence staining using DAPI) and for EL-4 cells 24 h after in vitro X-irradiation (measuring apoptotic bodies with flow cytometry) (Liu et al. 1996). In these two cases a statistically significant decrease in apoptosis rate occurred at doses of 0.05 and 0.075 Gy, but the decrease was not statistically significant at the dose of 0.025 Gy. A further study measuring apoptosis in frozen sections of different immune organs, including thymus, spleen, mesenteric lymph nodes and Peyer's patches with TUNEL (terminal deoxynuleotidyl transferasemediated dUTP nick end-labeling) also demonstrated J-shaped curves of apoptosis 12 h after WBI (Liu et al. 2000). Again, a statistically significant decrease in apoptosis rate was observed after doses of 0.05 and 0.075 Gy, but not after 0.025 Gy. Time course studies revealed a brief increase in apoptosis rate 1 h after irradiation with these doses, but the magnitude was greater after 0.05 and 0.075 Gy than that after 0.025 Gy. It was supposed that a certain magnitude of initial DNA damage triggering an increase in apoptosis was required in order to stimulate a compensatory reaction that would be able to clear the apoptotic cells to a level significantly below the control base line in the course of 12 h. An example is given in Table 2 showing the time course of the changes in apoptosis rate in the cortex of thymus assayed with TUNEL.
Time course of apoptosis rate in thymic cortex measured with TUNEL after whole-body X-irradiation with low-doses
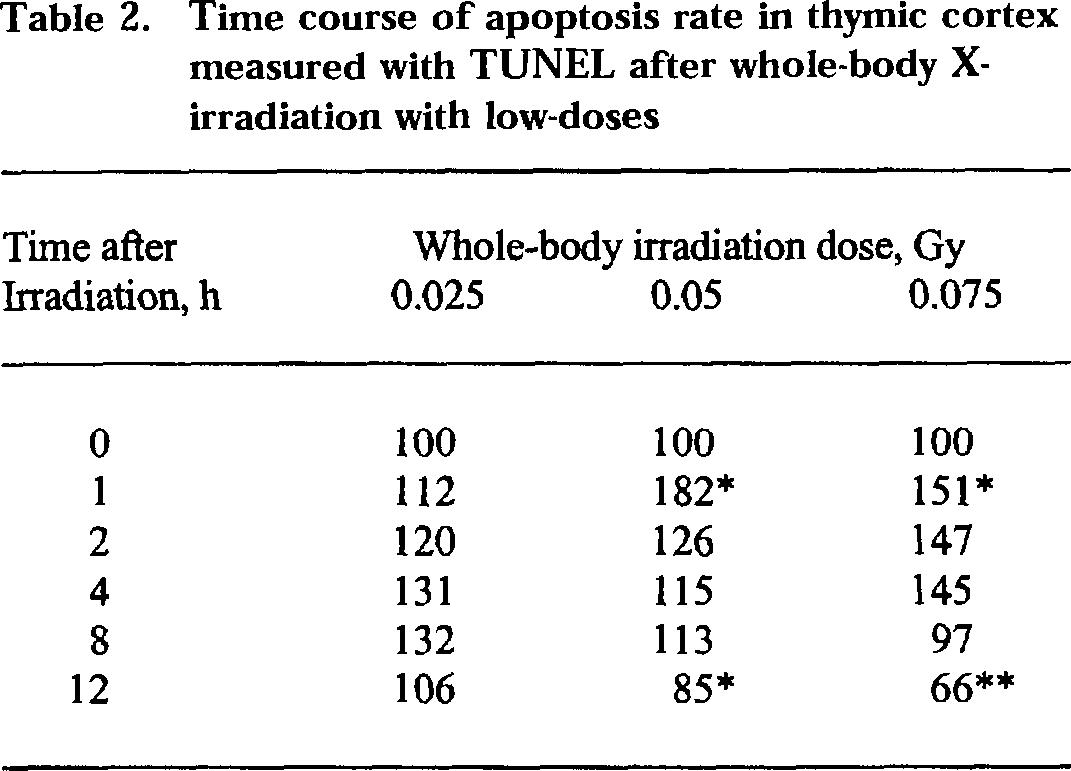
Each figure in the table is the average from counting 15 microscope fields (1000–1200 cells scored in each field) of 3 tissue sections from 3 mice, expressed as % of sham-irradiated control
P<0.05
P<0.01 vs control.
It is seen in this table that a higher abrupt early increases in apoptosis rate (>150%) in the first hour after 0.05 and 0.075 Gy was followed by an “overcompensation” at 12 h with the apoptosis rate going down to below control. The mechanism of the “overcompensation” may of course consist of a number of defense reactions, including increased DNA repair elicited by low-dose radiation.
However, in many other cases direct stimulation may be the mechanism of the hormetic effect. Take the dose-effect curve of thymocyte cAMP/cGMP ratio and the activity of its downstream molecule protein kinase A (PKA) as an example, a J-shaped curve was demonstrated 24 h after whole-body irradiation as shown in Figure 2 (Liu 1998; Liu and Xie 2000).

Dose-response curves of cAMP/cGMP ratio (left panel) and PKA activity (right panel) in mouse thymocytes 24 h after WBI of Kunming mice.
The time course studies showed that there was statistically significant drop in cAMP/cGMP ratio beginning from 8 h after whole-body irradiation with 0.075 Gy, reaching its nadir at 24 h, and no rise above control was observed even as early as 30 min after irradiation. This change in cAMP/cGMP ratio is due to both up-regulation of cGMP and down-regulation of cAMP after low-dose radiation. The opposite is true for the time course after a higher dose (2 Gy). The same pattern of the time course of PKA was also demonstrated after low and high-doses of radiation. These data may speak for a direct effect of low-dose radiation but not due to “overcompensation”.
The dose-response relationship of changes in macrophages after exposure to ionizing radiation is apparently different from that of the T lymphocytes. Most of the parameters studied showed a pattern of chiefly stimulation from low to high-doses of ionizing radiation. Examples are given in Figures 3 and 4. Figure 3 shows the changes in expression of surface molecules B7-1 (CD80) and B7-2 (CD86) on peritoneal macrophages after whole-body irradiation and on J774 A. 1 cells (a mouse macrophage cell line) after in vitro irradiation (Liu et al. 2001). The changes in secretion of cytokines are given in Figure 4 in which the upper left and right panels show, respectively, the secretion of IL-1β and TNFα after WBI, and the lower left and right panels show, respectively, the secretion of IL-12p70 by LPS-stimulated peritoneal macrophages after whole-body irradiation and the expression of CD48 on J774A.1 cells after in vitro irradiation. Here only the CD48 expression showed an inverted J-shaped dose-response curve, while the other seven parameters in the two figures all demonstrated chiefly stimulation (except a suppression of TNFα at the dose of 4 and 6 Gy) (Sun and Liu 1998, 2000a).

Dose-response curves of CD80 (upper panel) and CD86 (lower panel) on peritoneal macrophages after WBI of Kunming mice and on J774A.1 cells after In vitro irradiation.
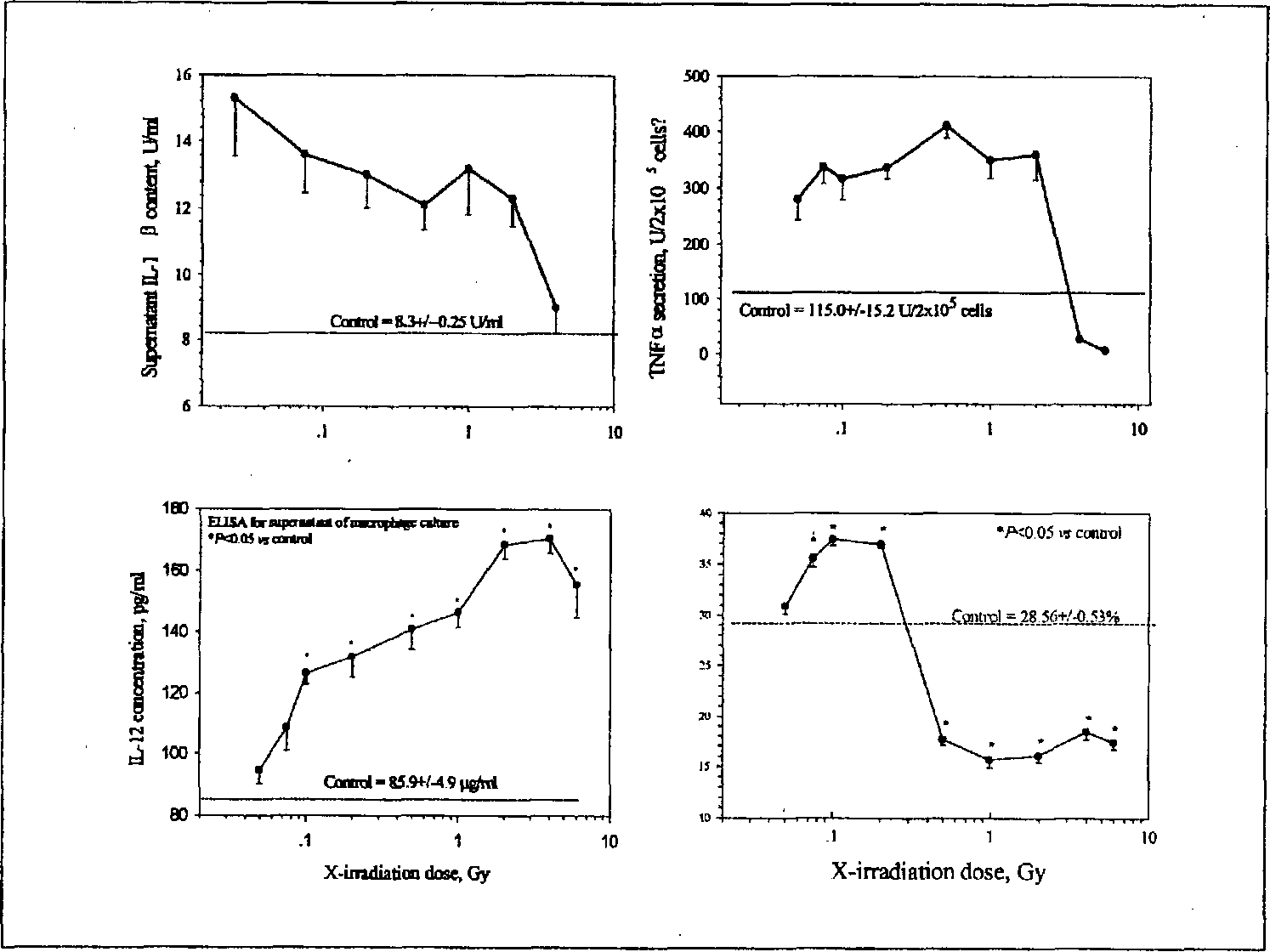
Dose-response curves of IL-1β and TNFα secretion (upper left and right panels, respectively) as well as secretion of IL-12p70 (lower left panel) by peritoneal macrophages after WBI of Kunming mice and expression of CD48 on J774A.1 cells (lower right panel) after in vitro irradiation.
MECHANISTIC STUDIES ON LOW DOSE STIMULATION OF IMMUNITY
The stimulation of immunity by low-dose radiation is a complex process concerning intercellular reactions within the immune system (Liu 1995, 1998; Liu et al. 2001) and signal transduction in the immune cells (Weichselbaum 1991; Hallahan et al. 1993; Liu et al. 1994a, b; He and Liu 2000; He et al. 2000, 2001; Pajonk and McBride 2001). Changes in the expression of surface molecules and the secretion of cytokines are accompanied with the induction of relevant genes and the expression of corresponding proteins (Liu et al. 2000, 2001; Sun and Liu 1998, 2000a; Jin et al. 2001; Liu et al. 2001a, b). It has been demonstrated in mice that these molecular changes induced by low-dose radiation constitute a unique pattern distinctly different from those caused by high-dose radiation (Liu 1995, 1998; Liu et al. 1996, 2000; Liu et al. 2001a). Low-dose radiation acts on both the APCs and T lymphocytes, facilitating the intercellular reactions within the immunmologic synapse (Malissen 1999) formed between these two categories of immune cells (Figure 5). It stimulates the expression of a number of surface molecules and modulates the secretion of certain cytokines (Liu et al. 2001; Sun and Liu 1998, 2000a; Jin et al. 2001; Liu et al. 2001a, b). Low-dose radiation stimulates the expression of B7-1 (CD80) and B7-2 (CD86) molecules on and secretion of IL-12p70 by the macrophages as well as the expression of TCR/CD3 (Liu et al. 1995) and CD2 (Sambani et al. 1996), CD4 and CD28 (Liu et al. 2001) on the lymphocytes and down-regulates IL-10 synthesis by T cells together with decreased CTLA-4 expression, and high-dose radiation exerts opposite effects on these parameters of the T cells (Liu et al. 2001; Liu et al. 2001a, b).

Schematic diagram showing the intercellular reactions between an antigen presenting cell and a T lymphocyte via the surface molecules. (See text for explanations).
As shown in Figures 3 and 4, there is a significant increase in secretion of TNFα, IL-1β, and IL-12p70 by the macrophages in response to whole-body irradiation of mice (Sun and Liu 1998, 2000a). In the immunological synapse, in addition to CD80/86 and CD28/CTLA-4, CD48 of macrophages and CD2 of lymphocytes are also important costimulating molecules regulating the immune response. It has been reported that low-dose radiation strongly enhanced the expression of CD2, both in PHA-stimulated and in resting human T lymphocytes, as demonstrated by a rosette assay or by immunofluorescence (Sambanmi et al. 1996). Flow cytometric analysis showed that in vitro irradiation of EL-4 (mouse lymphoma) cells resulted in enhanced expression of CD2 after low-dose radiation and its down-regulation after high-dose radiation, giving rise to an inverted J-shaped dose-response curve and expression of CD48 on J774A. 1 cells responded similarly (unpublished data). It is seen that both low and high-doses of radiation caused up-regulation of surface molecules on the macrophages (except CD48) and increased secretion of IL-12. Thus, the differential response of the immune reactions to low- vs. high-dose radiation chiefly depends on distinct changes of the T lymphocytes. So far, as the surface molecules CD80/86 and CD28/CTLA-4 as well the cytokines IL-12/IL-10 are concerned, this opposite relationship of the changes in surface molecule expression on and cytokine secretion by the lymphocytes after low and high-doses of radiation and its impact on the final outcome of the immune response are illustrated in the diagram in Figure 6 (Liu et al. 2001). It is shown in this diagram that low-dose radiation up-regulates the T cell activity, especially in favor of TH1.

Diagrammatic illustration of changes in surface molecule expression on and cytokine secretion by antigen presenting cells (APC) and T lymphocytes (TLC) in response to low-dose radiation (LDR) and high-dose radiation (HDR) and their impact on the final outcome of the immune status. (See text for detailed explanation).
Only part of the intercellular changes in the immune system induced by low-dose radiation vs. high-dose radiation are included in this diagrammatic illustration. Changes in other surface molecules and cytokines, as described in previous sections of the present paper, also play important roles.
It is quite clear that research on the molecular basis of the changes in the intercellular reactions after different doses of ionizing radiation in recent years has promoted the understanding of the nature of hormetic effect of low-dose radiation on immunity. The signals from APCs are absolutely necessary for the activation of lymphocytes in response to low-dose radiation (Liu et al. 2001). The part played by the APCs (macrophages) is essential for the realization of low-dose radiation-induced immunolgic stimulation because depletion of these cells from the splenocyte suspension obtained from the low-dose-irradiated mice will lead to the disappearance of the stimulatory effect. The up-regulation of expression of surface molecules (CD48, CD80, CD86) on APCs and increase in secretion of cytokines (IL-12, IL-1 beta, TNF-alpha) by APCs after exposure to low-dose radiation are crucial factors for up-regulation of T cell activity. On the other hand, all these changes except the down-regulated expression of CD48 are also observed after exposure to high-dose radiation (2 Gy). Thus, T lymphocytes play an even more important role in determining the fate of the immune response since the expression of their surface molecules (TCR, CD2, CD28, CTLA-4) changes in opposite directions after low and high-doses of radiation, and, even more, the secretion of IL-10 is down-regulated after low-dose radiation and up-regulated after high-dose radiation. Despite the fact that the expression of CD80 and CD86 on and the secretion of IL-12 by the APCs are sustained at a high level after high-dose radiation, the down-regulated CD28 expression and up-regulated expression of CTLA-4 on and increased IL-10 secretion by the lymphocytes after high-dose radiation will ultimately lead to down-regulation of T cell activation. Therefore, it is these distinct dose-dependent changes in the lymphocytes that determine the final outcome of immune response.
The molecular changes in the immunologic synapse would apparently reinforce the facilitation of the signal transduction in the lymphocyte initiated by the action of low-dose radiation. There have been reports on the effect of ionizing radiation on lymphocyte signal transduction (Weichhselbaum 1991; Hallahan et al. 1993; Liu et al. 1994a, b; He and Liu 2000; He et al. 2000, 2001; Pajonk and McBride 2001). Low-dose radiation may facilitate the transduction in multiple signal pathways, including the protein kinase C/calcium ion (PKC/[Ca2+]i) system, cyclic adenosine monophosphate/protein kinase A (cAMP/PKA) system, phospholipase A2/prosdaglandin E2 (PLA2/PGE2) system, and others. The effect of low-dose radiation on these signal pathways may be schematically illustrated in Figure 7.

Comprehensive diagram of cellular and molecular basis of LDR-induced up-regulation of T cell reactions. (See text for explanations).
In Figure 7 it is shown that low-dose radiation may act on the two categories of immune cells (APC and TLC) directly or via the reactive oxidative species (ROS) to influence the expression of their surface molecules and secretion of relevant cytokines within the immunologic synapse as described in previous sections. On the left side in the middle part of Figure 7 three signal pathways and their interrelationship are illustrated. Among these the up-regulation of PKC/[Ca2+]i is chiefly due to the activation of phospholipase C/phosphatidylinositol bisphosphate (PLC/PIP2) (Liu 1992). The facilitation of the PKC/[Ca2+], pathway would lead to the activation of transcription factors (TFs), among which nuclear translocation of NF-κB has been observed (He and Liu 2000; He et al. 2000, 2001). Calphostin C (CPS in the figure), a PKC inhibitor, was found to be able to block low-dose radiation-induced increase in nuclear translocation of NF-κB (unpublished data). On the other hand, tetramethyl benzidine-8 (TMB-8), a calcium antagonist, could completely suppress the low-dose radiation-induced augmentation of thymocyte proliferation (Liu 1998; Liu and Xie 2000). Therefore, the facilitation of PKC/[Ca2+]i pathway may occupy a pivotal position in low-dose radiation-induced up-regulation of T cell activation. Low-dose radiation decreases cAMP content and lowers cAMP/cGMP ratio in thymocytes accompanied with decreased activity of its down-stream molecule PKA (refer to Figure 3). These changes are related to the up-regulation of expression of CD3/CD28 with activation of phosphodiesterase7 (PDE7) (Liu 1998; Liu and Xie 2000; Li et al. 1999). The decrease in cAMP content may be a direct result of the activation of Gαi (inhibitory α subunit of G protein), and the significance of the changes in this signal pathway has been tested by the addition of cholera toxin (CT) to the cultured thymocytes to activate Gs (stimulatory subunit of G protein). It was found that CT added to thymocytes obtained from low-dose-irradiated mice could partially alleviate the stimulation of thymocyte proliferation induced by low-dose radiation (Liu and Xie 2000). In addition, the lowering of the activity of phospholipase A2 (PLA2) in the thymocytes after low-dose radiation may further decrease the cAMP content via prostaglandin E2 (PGE2) (Mele et al. 2000; Ribardo et al. 2001; McHowat et al. 2001). The changes in these three signal pathways would converge on the activation of transcription factors, which would increase the transcription of genes related to immune response. In the diagram up-regulation of IFNγ and IL-2 genes are shown as representatives (Liu et al. 2000). It is shown on the right side of the diagram that increased secretion of IL-1β could positively feedback to stimulate the APC (Liu 1998), and the rise of nitric oxide (NO) and ROS induced by TNFα (Sun and Liu 2000b) may exert direct stimulation to the transcription factors (TFs). The ultimate result, as shown at the bottom of the diagram, is T lymphocyte activation and proliferation, which would lead to clonal expansion and potentiation of anticancer immune activity.
The time course of the changes in signal molecules after whole-body irradiation with 0.075 Gy is shown in Figure 8. It can be seen that all the signal molecules reach their peak or nadir at 12 to 24 h after whole-body irradiation. Among these 10 parameters [Ca2+]i, calcineurin, cGMP, PKC (α, β1, and β2 isoforms) are up-regulated after low-dose radiation, with the first three reaching their peak at 24 h, and the last three reaching their peak at 12 h, while cAMP, cAMP/cGMP ratio, PKA, and PLA2 are down-regulated after low-dose radiation, all reaching their nadir at 24 h (Liu 1998; Liu and Xie 2000). These data further demonstrate the influence of temporal factor in construction of the J- or inverted J-shaped curves because most of the changes usually take 12 to 24 h to come to their peak or nadir.
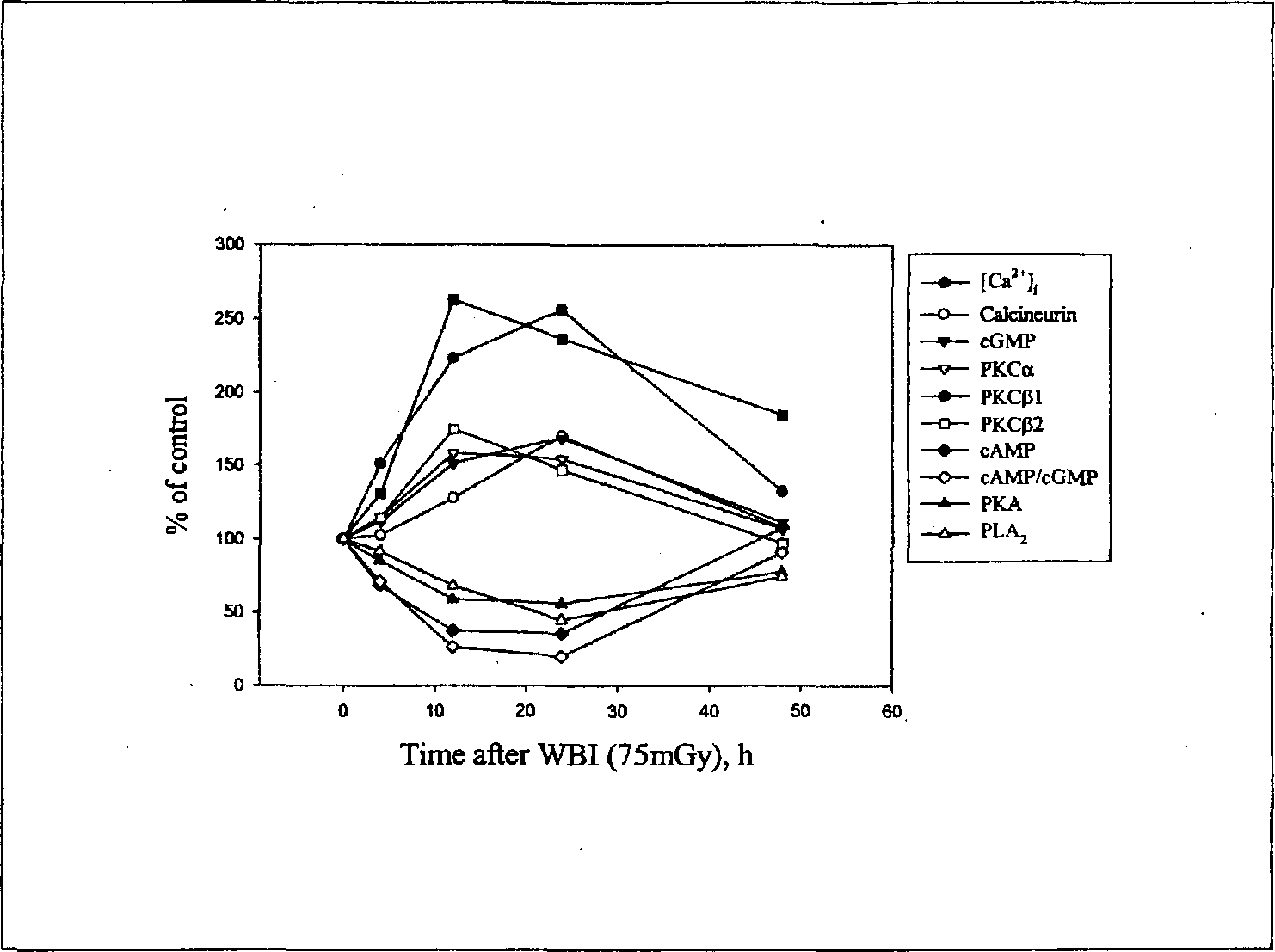
Time course of changes in expression of signal molecules in thymocytes after WBI of Kunming mice with 0.075 Gy.
Recent studies showed the interrelationship between cytokines and surface molecules, further supporting our analysis of the molecular mechanisms of immune responses evoked by different doses of radiation (Kubin et al. 1994; Dickensheets et al. 2000). It has also been demonstrated that other signal molecules may join the cohort of events evoked by low-dose radiation among which p38MAPK (mitogen-activated protein kinase) is an important one acting jointly with PLC (phospholipase C) and PKC (Shimizu et al. 1999).
IMPLICATIONS IN CANCER RISKS
It is well known that most carcinogens are immunosupressants (Ball 1970; Urso and Gengozian 1982; Bebb et al. 2002; Laupeze et al. 2002). Ionizing radiation is one of these when delivered at moderate to high-doses. Immune surveillance is one of the most important defense mechanisms in the body monitoring the development of cancer (Dunn et al. 2000). As reviewed in the previous sections, low-dose radiation stimulates T cell activity. The significance of these immunologic changes in radiation-induced cancer is still a question of debate. In UNSCEAR 1994 report, it was stated that “it remains doubtful whether the immune system plays a significant role in any of the adaptive responses at low-doses.… the immune system may not play a major role in moderating human radiation oncogenesis, although immune function in certain organs may ensure that some early neoplastic cells are eliminated before they become established”. NCRP Report No. 136 (NCRP 2001) also briefly discussed the adaptive response induced by low-dose radiation. It is stated that “in the ultimate instance of cancer in vivo, the adaptive response may take the form of an immune surveillance of preneoplastic cells. This is certainly a possibility, but it is difficult to design laboratory experiments to test it”. In a recent commentary article on radiation hormesis it is recognized that “under appropriate conditions, exposure to X-rays or gamma rays at doses below those causing immunosuppressive effects has been observed to augment various immune responses in mice…” but “the mechanisms of the observed enhancement remain to be elucidated in full…” (Upton 2000). These statements accept the importance of experimental data on low-dose radiation-induced up-regulation of certain immune functions and immune surveillance of cancer formation on one hand and question its significance in the analysis of cancer risk assessment on the other. There have been accumulating data disclosing the cellular and molecular mechanisms of low-dose radiation-induced increase in antitumor immunity in animal models as reviewed in the present paper. Low-dose radiation increases cellular antioxidant activity, facilitates DNA damage repair, reduces cell mutagenesis, and stimulates immune surveillance. All these may contribute to the effect of low-dose radiation on the reduction of cancer incidence. There have been experimental studies illustrating the suppressive effect of low-dose radiation on tumor growth, metastasis, and carcinogenesis as well as increased anticancer immunity, including enhanced NK and CTL activity as well as increased IFNγ and IL-2 secretion, and all these data point to the significance of immune responses up-regulated by low-dose radiation in cancer control (Li et al. 1996, 1998; Fu et al. 1996, 1997; Zhang and Liu 1996; Jin et al. 1997; Zhang et al. 1997a, b, 1998a, b, 1999; Li et al. 1999a, b). It was shown that preirradiation with low-dose radiation decreased the growth rate and metastasis of implanted B16 melanoma and Lewis lung cancer in C57BL/6J mice. In a well-recognized model of thymic lymphoma induced by fractionated whole-body irradiation (1.75 Gy once a week for 4 consecutive weeks) in C57BL/6J mice, a low-dose radiation (0.075 Gy) 6–12 h preceding each fractionated high-dose radiation could reduce the lymphoma incidence from 43.3% in the control (exposed to only the high-dose radiation) to 15.1% (high-dose radiation being preceded by 0.075 Gy, with an interval of 6 h) and 17.6% (high-dose radiation being preceded by 0.075 Gy, with an interval of 12 h) in 6 months after the fractionated exposures (Li et al. 1998; Li et al. 1999a, b). It was found that the low-dose radiation-induced suppression of tumor growth, metastasis, and incidence of lymphoma were accompanied with enhancement of anticancer immune reactions (Zhang et al. 1998a; Li et al. 1998, 1999a). Figure 9 shows experimental data illustrating that low-dose radiation retards cancer growth, reduces metastasis, increases efficacy of chemotherapy of cancer, and alleviates immune suppression caused by tumor burden (Fu et al. 1996; Li et al. 1993; Zhang et al. 1997b, 1999). The clinical application of low-dose radiation in the treatment of cancer has shown enlightening results. When patients with non-Hodgkin's lymphoma under chemotherapy was given total body or half body (the thorax) low-dose radiation in a regimen of 0.1 Gy three times a week or 0.15 Gy two times a week for 5 consecutive weeks with a total dose of 1.5 Gy, the survival rate in 9 years rose to 84% from 50% of the control with chemotherapy alone (P<0.01). The low-dose radiation-treated patients showed enhanced anti-cancer immunity (Sakamoto et al. 1997).

Effect of LDR on cancer growth, metastasis, cancer therapy, and immune responses in tumor-bearing C57BL/6J mice.
Actually, J-shaped dose-incidence curves have been demonstrated for a number of cancers induced by exposure to different doses of radiation (Calabrese and Baldwin 2002). Human epidemiological surveys also showed decreased cancer incidence after exposure to low-dose radiation. Among these the U.S. nuclear shipyard worker study, the 100-year British radiologists observation and a Canadian occupational radiation dose registry analysis strongly speak for the possibility of existence of an hormetic effect of low-dose radiation in cancer incidence since the cohorts observed in these three reports would probably exclude the “healthy worker effect” (Matanoski 1991; Berrington et al. 2001; Sont et al. 2001; Baillargeon 2001). These reports would promote further research in this field, thus accumulating more scientific data giving impetus to the improvement of the present radiation protection paradigm for the benefit of health of the population and the development of the economy of the society.
CONCLUDING REMARKS
Stimulatory effects of radiation on anticancer immunity with exposure to very low-doses are becoming more evident with the accumulation of scientific data in the literature. This situation makes it possible to construct dose-response curves reflecting reality. However, full analysis of existing data is still limited, which may be due to inaccessibility or neglect of some of the scientific reports. The present paper tended to review the dose-response relationship of immune parameters after whole-body X-irradiation and the up-regulation of T cell activity induced by low-dose radiation with emphasis on its cellular and molecular mechanisms, a research field advancing along with the rapid progress in modern immunology. Just because of this situation the review may not be able to cope with the development in this rapidly advancing field. Further research using modern biochips and related technologies developed with the advancement of genomics (Diehn et ai. 2002) and proteomics (Zhang et al. 2002) would give great impetus to the understanding of the molecular mechanism of immune responses and their relation to cancer control in organisms exposed to different environmental agents including ionizing radiation.
Footnotes
ACKNOWLEDGMENT
This paper was supported by grants from NSFC (No. 3870855, No. 39270207, No. 39570188).
