Abstract
The adsorption tower made of type 316L stainless steel (SS) in Multi-nuclide Removal Equipment (Advanced Liquid Processing System) which uses Ag-impregnated activated carbon (Ag AC) as an adsorbent experienced crevice corrosion. The influence of Ag AC on the crevice corrosion susceptibility and Esp of 316L SS was investigated by performing electrochemical experiments. Crevice corrosion was observed in the specimen in contact with the Ag AC. On the other hand, there was no crevice corrosion without the Ag AC in both pH 7.4 and pH 12 solutions. Clear ennoblement of spontaneous potential (Esp) by in contact with activated carbon was observed and that was clearly higher than the repassivation potential for crevice corrosion (ER,CREV). Thus, the presence of the AC notably increased Esp of 316L SS and this resulted in increased crevice corrosion susceptibility by the galvanic effect.
This paper is part of a supplementary issue from the 17th Asia-Pacific Corrosion Control Conference (APCCC-17).
Introduction
In the Fukushima Daiichi Nuclear Power Plant, Multi-nuclide Removal Equipment (Advanced Liquid Processing System, ALPS) is used to remove all the 62 types of radioactive nuclides, except tritium, from contaminated water that is used in the cooling cycle. Figure 1 shows a schematic of the cooling cycle in the Fukushima Daiichi Nuclear Power Plant. In the first process, Cs is removed by the Cs removal equipment (KURION, SURRY). In the second process, the solution that was treated by the Cs removal equipment is sent to the desalination equipment. In the final process, 62 types of radioactive nuclides are removed from reverse osmosis (RO) concentrated salt water that was passed through the desalination equipment of the ALPS. ALPS consists of 16 adsorption towers that have 8 types of adsorbents [1].
Schematic of the cooling cycle in the Fukushima Daiichi Nuclear Power Plant [1].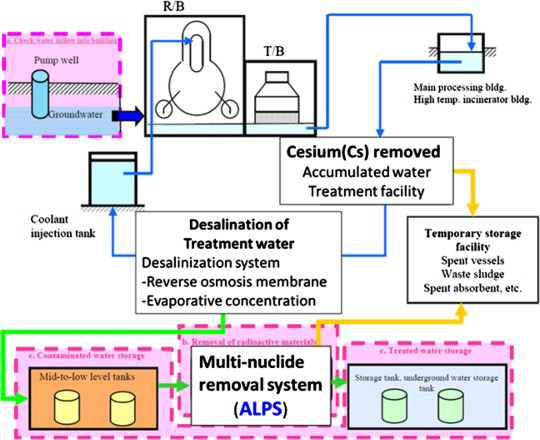
The Cl− concentration of salt water is increased by RO treatment, and this results in an increase in the susceptibility to localised corrosion. In adsorption towers of ALPS, crevice corrosion of 316L stainless steel (SS) that is used in piping flange occurred in Ag-impregnated activated carbon (Ag AC) adsorption towers where neutral and alkaline solutions are present (see Figure 2). The high Cl− concentrations and Ag AC promote crevice corrosion [2]. It is known that crevice corrosion can occur when the spontaneous potential (E sp) is higher than the critical potential for crevice corrosion initiation. In this study, the influence of AC on E sp and the crevice corrosion susceptibility of 316L SS were investigated by performing electrochemical experiments.
Examples of crevice corrosion observed in Ag AC adsorption tower [2].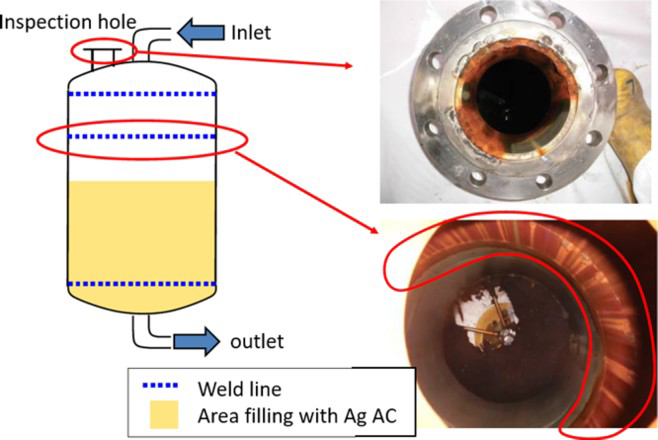
Experimental
Materials and specimens
Chemical composition of the 316L SS (wt-%).

Two types of specimens were prepared for the experiments; crevice-free specimen and specimen with crevice. The size of the specimen was 20 × 20 × 4 mm. A crevice was formed using an acrylic plate. The Ag AC (coconut husk + 0.1% AgCl, KURARAY COAL) is made using coconut husk and Ag impregnation is realised using silver chloride. Although the powder particle size had a wide size distribution, 250-500 μm particles accounted for around 90% of particles. Figure 3 shows the scanning electron microscope (SEM) images of the surface structure of Ag AC: its surface was found to be porous and several mesopores were observed inside the macropore. The specific surface area of Ag AC measured by the N2 gas adsorption method was 1122 m2 g−1. The results show the specific surface area is very large, and it is almost the same as the typical specific surface area (i.e.1036 m2 g−1) of AC made from coconut husk [3].
SEM images of the surface structure of Ag AC.
Tests to evaluate crevice corrosion susceptibility
The effect of Ag AC on the susceptibility to crevice corrosion was investigated by performing crevice corrosion tests in the open-circuit condition. Figure 4 shows schematic of experimental equipment and specimen used in this study. Both crevice-free specimen and specimen with crevice were used [4]. Modified artificial sea water (40°C, Cl− concentration = 13,000 ppm, pH = 7.6, and 12.0, atmosphere saturation) was used as the test solution. Open-circuit potential of the specimen was measured during the experiments using a potentiostat in two conditions: in contact with and without Ag AC. After the experiments, the specimen surfaces were observed using an optical microscope. The measurements were conducted in an open-circuit condition for almost 40 h (in the neutral solution) and 24 h (in the alkaline solution).
Schematic of experimental equipment and specimen.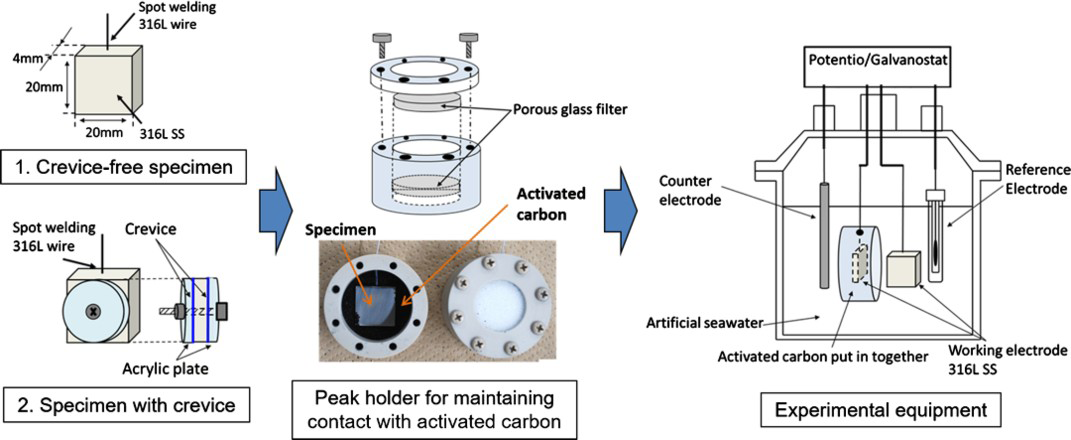
Cathodic polarisation measurement
A crevice-free specimen was used. A modified artificial sea water (40 ± 1°C, Cl− concentration = 200 ppm, pH = 7.8, and atmosphere saturation) was used as the test solution. The cathodic polarisation was measured at a 100 mV min−1 sweep rate using the electrochemical equipment.
Open-circuit potential and repassivation potential measurement
A crevice-free specimen was used. The effect of Ag AC on the E sp was investigated by the open-circuit potential of the specimens measurement in both conditions: in contact with AC (with Ag/without Ag) and without AC. Artificial sea water (40°C, Cl− concentrations = 200, 20,000 ppm, pH = 8.0, and 12.0, atmosphere saturation, N2 deaeration) was used as test solution. The measurements were conducted in an open-circuit condition for almost 60 h (both in the neutral and alkaline solutions).
A critical potential for the initiation of corrosion and repassivation in steels is known to exist, but measuring the actual critical potential of crevice corrosion by potentiostatic testing is very time-consuming. In addition, it has been reported that the initiation potential measured in short-term potentiodynamic tests is not equal to the actual initiation potential. Consequently, the repassivation potential for crevice corrosion, E R,CREV, has generally been used as a substitute, as this provides a suitably conservative value when compared to the actual initiation potential[5,6]. The method for determining the E R,CREV is defined by JIS G 0592[7]. This standard is grounded in the ideal that the repassivation potential of early crevice corrosion is equal to the actual initiation potential.
Results and discussion
The effects of activated carbon on crevice corrosion susceptibility
The surface morphology of the specimens after the experiments was observed using an optical microscope and is shown in Figure 5. Crevice corrosion was not observed in the specimen that was not in contact with the Ag AC both in the neutral and alkaline solutions. On the other hand, crevice corrosion occurred on the specimen in contact with Ag AC both in the neutral and alkaline test solutions. These results clearly indicate that crevice corrosion susceptibility increased when the specimen was in contact with Ag AC. In the case of the neutral test solution, the maximum corrosion depth was 130 μm and crevice corrosion propagated to cover almost all the creviced area of the specimen. In case of the alkaline test solution, the maximum corrosion depth was 25 μm and the corroded area was much smaller than that in the case of the neutral solution. These results which show that the corroded area and depth are greater in the case of the neutral solution are indicative of the actual corrosion occurring in the adsorption towers of ALPS. The reason for the corroded area and depth being smaller in the case of the alkaline solution was considered to be the fact that the dissolution rate of Fe(OH)2 decreased in the alkaline solution and this resulted in a decreased rate of corrosion [8]. Basically, the decrease of pH to depassivation pH at an anode site is required for crevice corrosion initiation and propagation. Considering this aspect, the difference in crevice corrosion behaviour can be also explained.
Surface morphology of the specimen after the crevice corrosion experiment. (a) without Ag-impregnated activated carbon, (b) with Ag AC (in the neutral solution), and (c) with Ag AC (in the alkaline solution).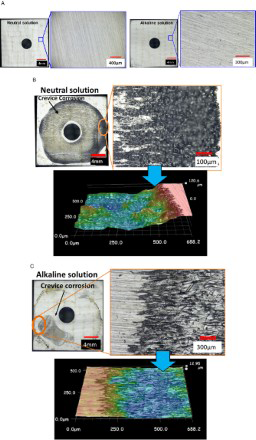
The variation in open-circuit potential of the specimen with time both in the neutral and alkaline solutions are shown in Figure 6. Open-circuit potential of the specimen in contact with Ag AC was noble at the very early stage and then decreased drastically at the beginning of experiments both in the neutral and alkaline solutions. From these results, it was considered that crevice corrosion occurred at the beginning of the experiment with a decrease in open-circuit potential when the specimen was in contact with Ag AC. The results indicate ‘galvanic effect’ as a primary reason for the severe crevice corrosion.
Open-circuit potential variation of the specimen with time during crevice corrosion experiments both in the neutral and alkaline solutions. (a) neutral solution and (b) alkaline solution.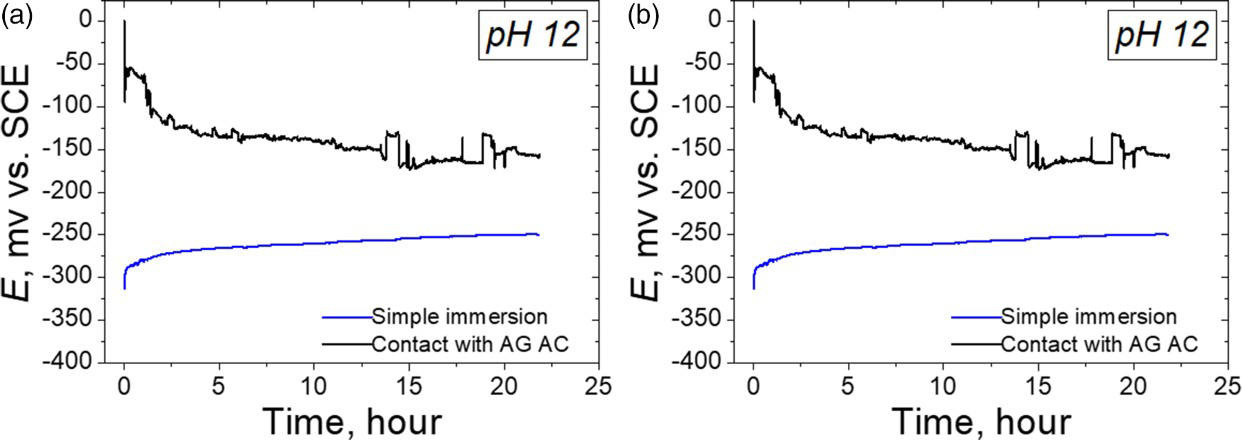
Effect of Ag AC on cathodic reaction of SS
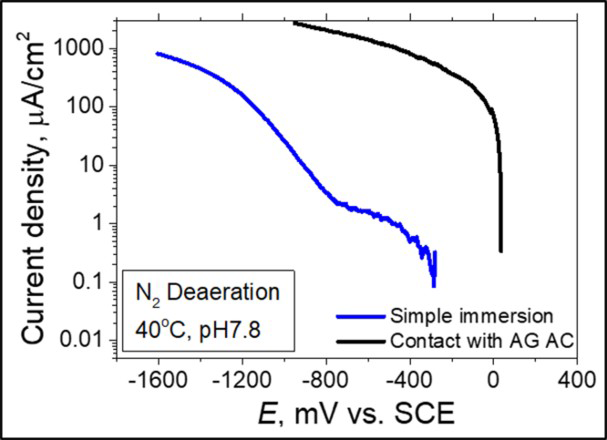
If galvanic corrosion occurred in the above experiments, the surface of Ag AC should work as cathode site. Figure 7 shows the measured cathodic polarisation curve. At the beginning of the measurement, the current density of the SS in contact with Ag AC was significantly higher than that of the specimen that was not in contact with AC. From the results, it was concluded that most of the cathodic current came from the cathodic reaction on the surface of the Ag AC. Similar finding was reported in the cathodic polarisation measurements results for 13Cr SS and graphite specimens by Yamamoto et al. [9]. When the surface area of 13Cr SS and graphite specimens were the same (1 cm2), observed cathodic currents for both specimens showed the same behaviour. On the other hand, when the area of the graphite specimen was 150 cm2, the cathodic current increased drastically. This result shows that the cathodic reaction occurring on the surface of the SS and graphite specimen was the same ( Cathodic polarisation curves in the neutral solution.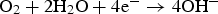 ) [9]. Since Ag AC is the same carbon material as graphite, it was considered that the same cathodic reaction occurred on its surface. In addition, the Ag AC has a large specific surface area (1122 m2 g−1) over which the cathodic reaction occurs. From these results, it was considered that the cathodic reaction of the 316L SS was promoted because of the additional cathodic reaction on the large surface area of Ag AC.
) [9]. Since Ag AC is the same carbon material as graphite, it was considered that the same cathodic reaction occurred on its surface. In addition, the Ag AC has a large specific surface area (1122 m2 g−1) over which the cathodic reaction occurs. From these results, it was considered that the cathodic reaction of the 316L SS was promoted because of the additional cathodic reaction on the large surface area of Ag AC.
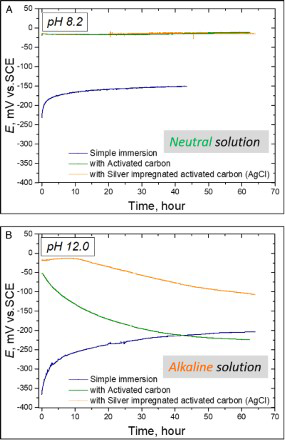
Effects of activated carbon on spontaneous potential and its relationship with the repassivation potential
There was no corrosion on the specimen during open-circuit potential measurement both in the neutral and alkaline solutions. The time variation of the open-circuit potential under the different test conditions is shown in Figures 8. In the neutral solution, E sp of the specimen without AC was evaluated to be −150 mVSCE. E sp of the specimen in contact with both Ag AC and AC (without Ag) were evaluated to be the same −20 mVSCE. Since E sp of the specimen which contacted with AC (Ag/without Ag) was over 100 mV higher than that of specimen alone. In the alkaline solution, E sp of the specimen without AC was −200 m VSCE, while those of the specimen in contact with Ag AC and AC (without Ag) was higher than −100 mVSCE at the beginning, and then decreased with time; the reason for this eventual decrease with time was considered to be the fact that the equilibrium rate of the cathodic reaction ( Open-circuit potential variation of the specimen with time both in the neutral and alkaline solutions. (a) neutral solution and (b): alkaline solution.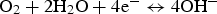 ) shifted to the negative direction with concentration of hydrogen peroxide that was adsorbed on the AC surface [10].
) shifted to the negative direction with concentration of hydrogen peroxide that was adsorbed on the AC surface [10].
Figures 9 shows the comparison of E sp with the repassivation potential for crevice corrosion (E R,CREV) with different Cl− concentrations. E sp of the specimen without Ag AC is lower than E R,CREV, on the other hand, that of the specimen with Ag AC is higher than E R,CREV regardless of the Cl− concentrations. Therefore, it can be concluded that crevice corrosion at the adsorption tower due to activated carbon.
Comparison of E sp with repassivation potential for crevice corrosion (E R,CREV) with different Cl− concentration.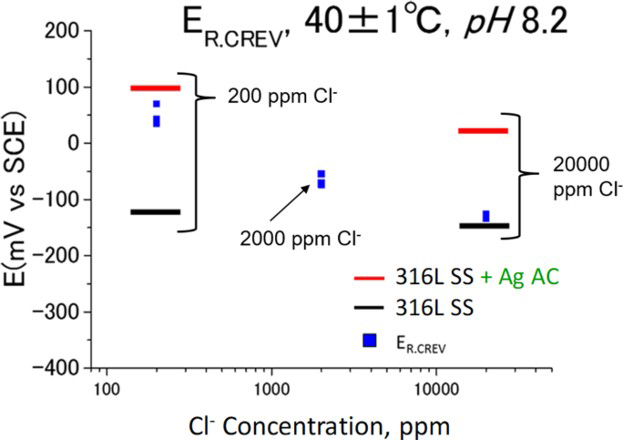
Conclusions
The influence of Ag AC on the crevice corrosion susceptibility and E sp of 316L SS was investigated by performing electrochemical experiments. Crevice corrosion was observed in the specimen in contact with the Ag AC. On the other hand, there was no crevice corrosion without the Ag AC in both pH 7.4 and pH 12 solutions. Clear ennoblement of E sp by in contact with activated carbon was observed and that was clearly higher than the E R,CREV. Thus, crevice corrosion at the adsorption tower due to activated carbon is clearly explained; the presence of the AC notably increased E sp of 316L SS and this resulted in increased crevice corrosion susceptibility by the galvanic effect.
Footnotes
Disclosure statement
No potential conflict of interest was reported by the authors.
