Abstract
In order to improve both the hardness and corrosion resistance properties of AISI 430F stainless steel, plasma nitriding (PN) and nitrocarburising processes were carried out at different temperatures ranging from 350 to 500°C for 4 h. After PN, the nitrided layer was found to be thicker compared to that obtained by plasma nitrocarburising process. There was an increase in microhardness values by a factor of six to seven compared to the plasma nitrided and nitrocarburised specimens respectively, treated at 500°C. The electrochemical corrosion behaviour of the plasma nitrided and nitrocarburised AISI 430F specimens show that the plasma nitrided and nitrocarburised specimens treated at 400°C for 4 h showed better corrosion resistance and higher surface hardness than the untreated AISI 430F stainless steel specimens. This is mainly attributed to the presence of nitrogen in the modified layer existing as a solid solution in the ferrite phase.
Introduction
Ferritic stainless steels are iron–chromium alloys containing 16-30 wt-% Cr along with alloying elements like Mo and Si, which are added to stabilise the ferrite phase for a body centred cubic structure at all temperatures as well as to improve properties such as corrosion resistance [1]. Ferritic stainless steels are being increasingly used in car exhaust pipes, interior and exterior decoration, kitchen utensils, food industry, automotive industry, petrochemical and chemical industries and other functional applications, due to their low cost, workability and corrosion resistant properties [2]. These steels are not frequently used in conditions requiring resistance to wear because they are softer relative to austenitic and martensitic stainless steel grades.
The wear resistance of these stainless steels can be improved by modifying the surface using different surface modification techniques. In the past few decades, plasma nitriding (PN) and nitrocarburising processes have been widely studied by several authors [3
4
5
-6] to improve the surface performance, such as hardness, wear as well as corrosion resistance of austenitic and martensitic stainless steels. Compared to PN where only nitrogen is used, plasma nitrocarburising (PNC) involves a simultaneous introduction of nitrogen and carbon into the steel. The carbon atmosphere mostly employed in PNC is methane or acetylene. However, only a few published works are available on PN and PNC of ferritic stainless steels like AISI 439, AISI 405, AISI 441 and AISI 430 [7,8]. It was reported by Oliveira et al. [9] and Tuckart et al. [10] that PN of AISI 439 and AISI 405 ferritic stainless steels at 500°C produced thick uniform layers consisting of iron and chromium nitride phases compared to that of austenitic stainless steels due to the higher diffusivity of nitrogen in the ferrite network than in the austenite phase. Another study on PN of AISI 409L by Gontijo et al. [11] at temperatures lower than 450°C indicated that along with iron nitrides,
Furthermore, there seem to be no published results as per the author's knowledge on PN and nitrocarburising of AISI 430F stainless steel, which is a nickel free ferritic stainless steel containing sulphur. Hence, in the present investigation PN and nitrocarburising are carried out on AISI 430F ferritic stainless steel at various temperatures in order to obtain high surface hardness along with corrosion resistance.
Experimental
Material
Chemical composition of the alloy in wt-%.

Surface modification techniques
Both PN and PNC processes were conducted in a PN setup as described elsewhere [12]. The specimens were cleaned with acetone before placing them on the specimen holder present in the vacuum chamber. The specimen holder acted as a cathode and the grounded vacuum chamber wall acted as an anode. Initially, the vacuum chamber was evacuated to a base pressure of 8 Pa with the help a mechanical rotary pump. Before plasma processing, sputter cleaning was done for 1 h using hydrogen gas at 100 Pa pressure and a temperature of 250°C to get rid of the surface oxide layer. For PN, a gas mixture consisting of 80% N2 and 20% H2 was used whereas; a gas mixture of 78% N2, 20% H2 and 2% C2H2 was employed for PNC process. In both the processes a constant pressure of 500 Pa was maintained. During PN and PNC, under the influence of voltage bias, nitrogen, hydrogen and acetylene gases get ionised and accelerate to impinge on the specimens. The kinetic energy of these ions is converted into thermal energy during this ion bombardment process, which in turn heats up the specimens. Hence, to get the substrate temperatures of 350, 400, 450 and 500°C, the bias voltage was adjusted from 400 to 550 V. The temperature of the specimens was measured by a J-type thermocouple with an accuracy of ±2°C of the set value. The treatment duration was kept constant in both the processes for 4 h. After these processes, the bias voltage was reduced to zero and the specimens were cooled down to room temperature in a nitrogen gas atmosphere to avoid oxidation before removing them from the vacuum chamber.
Surface characterisation
Scanning electron microscopy (SEM) was used for studying the layer thickness and the surface morphologies after PN and nitrocarburising processes. Further, Energy dispersive spectrometry (EDS) was used to measure the elemental composition of the treated surfaces. After PN and PNC, the phases were identified using a glancing incidence XRD (3000 PTS X-ray diffractometer) with Cu–Kα radiation (
Electrochemical studies
Corrosion behaviour of the untreated, plasma nitrided and plasma nitrocarburised 430F stainless steel specimens were studied using potentiodynamic polarisation technique. The setup consisted of a Reference 600 Gamry potentiostat driven by a software. The electrochemical tests were carried out in a flat electrochemical glass cell having graphite as a counter electrode, a saturated calomel electrode (SCE) as a reference electrode and the specimen as the working electrode. A specimen area of 2.85 cm2 was exposed to the solution. The electrochemical studies were carried out at ambient temperature (28°C). The tests were performed in a freely exposed 3.5 wt-% NaCl solution, which was open to the atmosphere. After stabilising the open circuit potential (OCP) for 30 min, when the drift in the potential became negligible, the potentiodynamic polarisation scan was started, by setting a potential 500 mV below the OCP. Each experiment was repeated at least three times to check for the reproducibility.
Results and discussion
Phase analysis
Glancing incidence X-ray diffraction (GIXRD) patterns obtained with an incident angle of 2° are shown in Figure 1 for the untreated along with the 350, 400, 450 and 500°C plasma nitrided and plasma nitrocarburised ferritic stainless steel specimens.
GIXRD patterns of the (a) plasma nitrided and (b) plasma nitrocarburised specimens treated at different temperatures.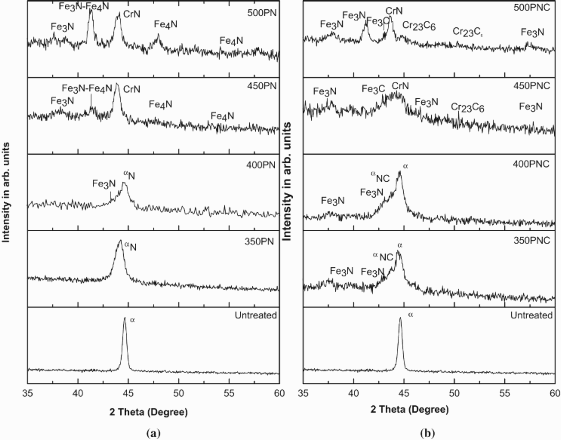
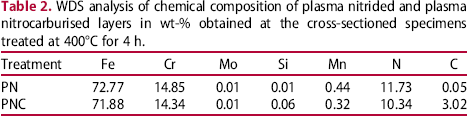
WDS analysis of chemical composition of plasma nitrided and plasma nitrocarburised layers in wt-% obtained at the cross-sectioned specimens treated at 400°C for 4 h.
Further, it is interesting to note the presence of ferrite peaks in the XRD pattern of the specimen along with the ones corresponding to
Effect of temperature on layer thickness
Images of cross-sectioned plasma nitrided and plasma nitrocarburised specimens treated at 400 and 500°C are shown in Figure 2.
Scanning electron microscope images of the cross-sectioned specimen obtained after PN at (a) 400°C and (b) 500°C and PNC at (c) 400°C and (d) 500°C.
The microstructure of the layer significantly changed when the treatment temperature was increased from 400 to 500°C. That is, the plasma nitrided and nitrocarburised specimens treated at 400°C showed a layer which did not have significant features mainly due to the presence of Measured thickness of plasma nitrided and nitrocarburised layers under different process temperatures.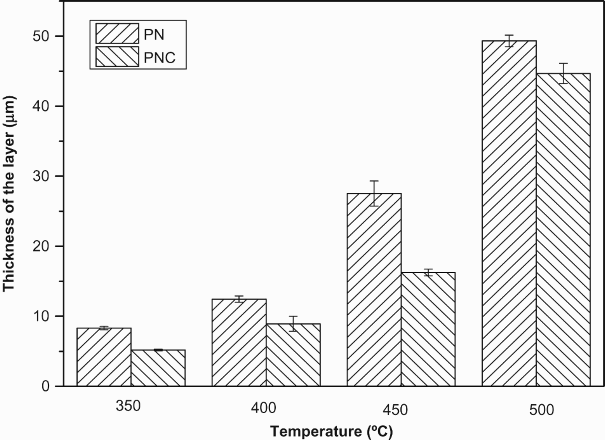
It is seen that the layer thicknesses increase with the treatment temperature. Another point worth noting is that the thickness of the plasma nitrided specimens was higher as compared to the plasma nitrocarburised specimens. The reason why plasma nitrided specimen exhibited higher thickness than the plasma nitrocarburised layer at a given temperature is due to faster diffusion of nitrogen than carbon in ferrite, (BCC lattice) phase [14]. This is because of the fact that the BCC structure is more open [15]. On the contrary, during PNC, in presence of carbon, the thickness of the layer decreases. For this very reason, ferrite peaks were visible in the XRD patterns of 350 and 400°C as shown in Figure 1(b). de Sousa et al. [16] also reported a similar observation and attributed this to the dissimilar nitrogen and carbon diffusion rates in ferrite matrix after PNC process.
Microhardness
The microhardness of the untreated AISI 430F stainless steel was measured to be 200 HV0.05. The microhardness values of plasma nitrided and plasma nitrocarburised specimens treated at different temperatures are shown in Figure 4.
Microhardness values obtained on plasma nitrided and plasma nitrocarburised specimens treated at different temperatures.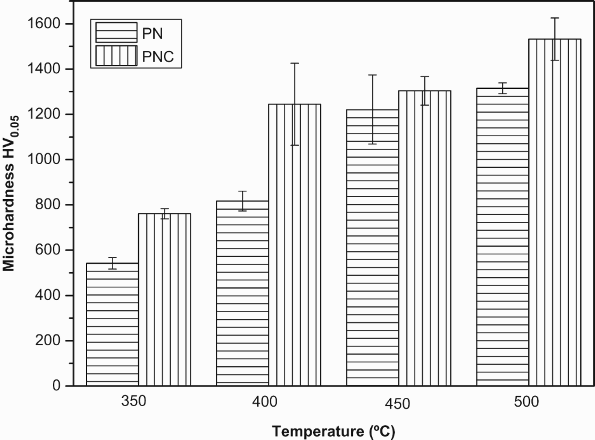
An increase in hardness with temperature was found on all the treated specimens. The hardness values of the 500°C plasma nitrided and nitrocarburised surface were found to be highest among all the cases. They were found to be 1315 HV0.05 and 1532 HV0.05 respectively; which are about 6 and 7 times as hard as the untreated material. This steep increase in the surface hardness was due to the presence of chromium nitride precipitates [17]. However, between the two treatments PNC has yielded higher hardness values at all these temperatures. It is interesting to note that in spite of the thin modified layer present in 350 and 400°C plasma nitrocarburised specimens; there was an increase in the microhardness values in comparison to the plasma nitrided specimens. This is attributed to the presence of iron nitrides along with the carbon and nitrogen saturated expanded ferrite phases. Whereas, the higher hardness of 450 and 500°C plasma nitrocarburised specimens is due to the presence of carbides and nitrides of iron and chromium present in the modified layer [18].
Electrochemical corrosion studies in 3.5 wt-% NaCl
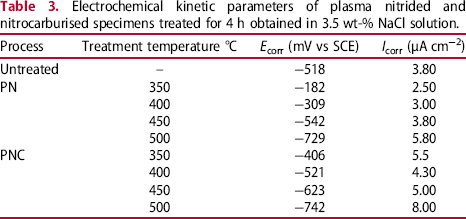
Cathodic and anodic polarisation curves of the untreated and treated specimens obtained from 3.5 wt-% NaCl are shown in Figure 5. Table 3 summarises the electrochemical corrosion parameters such as corrosion potentials ( Potentiodynamic curves of the untreated and plasma nitrided AISI 430F ferritic stainless steels treated at different temperatures in 3.5 wt-% NaCl solution. Electrochemical kinetic parameters of plasma nitrided and nitrocarburised specimens treated for 4 h obtained in 3.5 wt-% NaCl solution.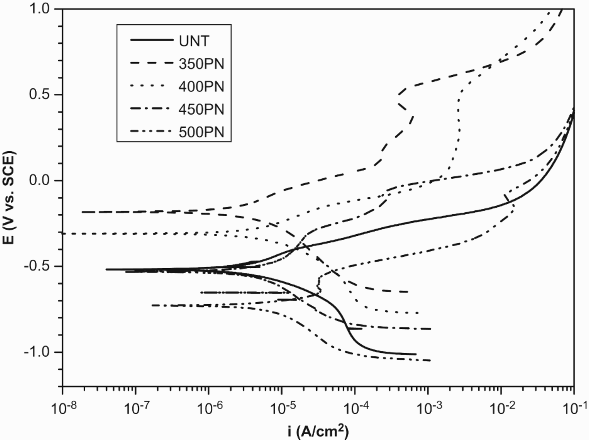
As seen from Figure 5, the anodic polarisation curve of the untreated AISI 430F stainless steel obtained from 3.5 wt-% NaCl solution did not show any passive region and very low corrosion potential (−518 mV vs SCE). The reason for the poor corrosion resistance of AISI 430F can be suggested to be due to the presence of MnS inclusions. It is reported that MnS inclusion promotes localised corrosion in steel as they are known to be the initiation sites of pitting corrosion [19]. After PN of AISI 430F, it is seen from Figure 5 and Table 3 that the
However, it decreased on further increasing the treatment temperature to 500°C. It was observed that the corrosion current densities for the specimens treated at 350 and 400°C were lower than the untreated specimens. Such observations were also found in 350 and 400°C plasma nitrided AISI 409 specimens [11].
The above results with respect to
It was observed from Figure 6 and Table 3 that 350 and 400°C plasma nitrocarburised AISI 430F ferritic stainless steel behaves similarly to that of plasma nitrided ones at similar temperatures with respect to the untreated specimens.
Potentiodynamic curves in 3.5 wt-% NaCl solution of the untreated and plasma nitrocarburised AISI 430F ferritic stainless steel at different temperatures.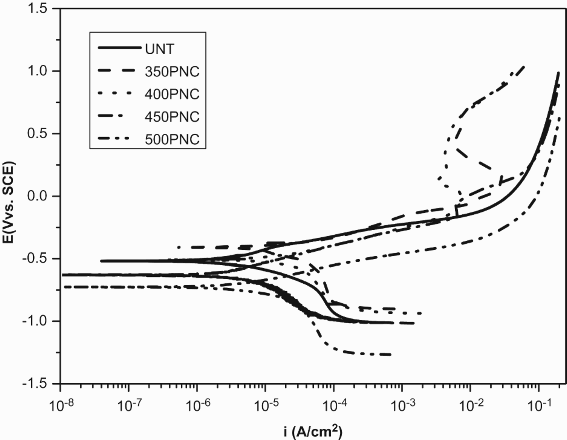
Thus, the
Typical SEM images of the specimens of (a) untreated (b) plasma nitrided at 400°C and (c) plasma nitrocarburised at 400°C (d) plasma nitrided at 500°C (e) plasma nitrocarburised at 500°C after subjecting them to polarisation tests are shown in Figure 7. The micrographs of the untreated specimen, after polarisation test, show a large number of deep pits (about 25 µm) supporting the potentiodynamic data (Figure 7(a)). Compared to the untreated specimens, 400°C treated specimens had only a fewer pits of small sizes (about 5 µm) indicating improvement in pitting resistance (Figure 7(b) and (c)). This is mainly due to the presence of nitrogen in solid solution as revealed by WDS analysis shown in Table 2. The EDS analysis at a location near the pit of 400°C treated specimen showed chromium and iron contents to be Fe: 72.77 and Cr: 14.85 wt-% respectively for PN case and Fe: 71.88 and Cr: 14.85 wt-% respectively for PNC case indicating that there was only a slight decrease compared to the untreated specimen (Fe: 79.96 wt-% and Cr: 15.67 wt-%). At 500°C, the entire surface of the specimen was found to have suffered severe corrosion. The extensive uniform corrosion seen in Figure 7(d) and (e) could be attributed to the extensive loss of Cr in the matrix as observed from the EDS analysis (Cr: 13.35; and Fe: 67.53; in wt-%) in plasma nitrided and (Cr: 12.85; and Fe: 63.55 in wt-%) in plasma nitrocarburised specimens due to the formation of a large amount of iron and chromium nitrides in the former and their nitrides/carbides in the later as has been revealed by XRD patterns (Figure 1(a) and (b)).
SEM images of specimens obtained after the polarisation tests for (a) untreated (b) plasma nitrided at 400°C (c) plasma nitrocarburised at 400°C (d) plasma nitrided at 500°C (e) plasma nitrocarburised at 500°C conditions.
From these results, it is found that 400°C plasma nitrided and nitrocarburised specimens treated for 4 h exhibited higher surface hardness and better corrosion resistance among all the treated specimens. Hence, plasma nitrided and nitrocarburised specimens treated at 400°C for 4 h can be considered for applications demanding both wear and corrosion resistance properties.
Conclusions
PNC of AISI 430F ferritic stainless steels treated by PN and PNC at temperatures between 350 and 500°C for 4 h was systematically studied to optimise the process parameters to give higher hardness for the alloy along with corrosion resistance. The following are the salient conclusions.
Plasma nitrided specimens of AISI 430F alloy exhibited thicker layers than the plasma nitrocarburised specimens at any given temperatures, which have been attributed to a faster diffusion of nitrogen than carbon in ferrite phase. The 350 and 400°C plasma nitrided and nitrocarburised specimens revealed The surface hardness of AISI 430F ferritic stainless steel was higher on the plasma nitrocarburised specimen than on the PN specimen and the hardness was found to increase more with temperature in the former case than in the latter. PN and PNC significantly suppressed the pitting tendency of the alloy in 3.5 wt-% NaCl solution at all the treatment conditions. Significant reduction in anodic kinetics was observed for PN specimens treated at 350°C though PNC did not give rise to similar improvement, however, it did not cause further deterioration in relation to the untreated specimens. It appears that PNC gives rise to nitride phases even at low temperatures causing this problem. However, among the plasma nitrided and nitrocarburised specimens treated at 400°C, the corrosion resistance was better compared to the untreated specimens mainly due to the presence of nitrogen in solid solution as revealed by few pits of smaller sizes. At 450 and 500°C, plasma nitrided and nitrocarburised specimens treated for 4 h lost the passivity and this was mainly due to the chromium depletion in the alloy matrix. 400°C, 4 h treatment gave optimised hardness and corrosion resistance in both the treatments.
Footnotes
Acknowledgements
The authors acknowledge Dr S.L. Kamath of IIT Bombay (IIT B) and Mrs Purvi Dave (FCIPT) for carrying out SEM and EDS analysis. One of the authors J. Alphonsa expresses her gratitude to Mr G. Jhala for his assistance in conducting the experiments. The National Facility (OIM and Texture Lab), Department of Metallurgical Engineering and Materials Science, IITB extended its facility to carry out GIXRD studies. The authors would like to acknowledge Sophisticated Analytical Instrument Facility, IITB for WDS studies.
Disclosure statement
No potential conflict of interest was reported by the authors.
