Abstract
The corrosion behaviour of 2101 duplex stainless steel (DSS) in NaCl solution was studied and compared with that of 2205 DSS. The effects of chloride concentration and solution temperature on pitting corrosion behaviour were focused. The relative sensitivity to pitting corrosion of the constituent phases in both 2101 and 2205 DSSs was also explored. Pitting corrosion susceptibility was evaluated by conducting cyclic polarisation curve measurement. The corrosion morphology was examined by a scanning electron microscope equipped with energy dispersive spectrometer. The experimental results showed that the pitting corrosion resistance of 2101 DSS was inferior to that of 2205 DSS by exhibiting a lower threshold chloride concentration and a lower critical pitting temperature. For both 2101 and 2205 DSSs, the ferrite phase was more susceptible than austenite phase to pitting corrosion.
This paper is part of a supplementary issue from the 17th Asia-Pacific Corrosion Control Conference (APCCC-17).
Keywords
Introduction
Duplex stainless steels (DSSs) contain approximately equal volume fractions of austenite (γ) and ferrite (α) phase. Due to their good corrosion resistance, they are widely used in many applications such as condenser, desalinator, heat exchanger, etc. [1]. Since the price of nickel is not stable and fluctuates unpredictably, therefore, the development of DSSs has followed lean route after the introduction of the modern 22%Cr duplex grades. In lean duplex grades, Ni is partially replaced by Mn and N, while Cr and Mo contents are also slightly reduced to reach economical purposes. Among the lean grade of DSSs, 2101 DSS, comprising less than 2% Ni, is one of the attractive alloys recently. Zhang et al. [2] had addressed that 2101 DSS has better pitting corrosion resistance than 304 austenitic SS. Gao et al. [3] had also pointed out that it has lower susceptibility to form precipitates as compared with the famous 2205 DSS. However, the studies on corrosion behaviours of 2101 DSS are still limited, especially its pitting corrosion resistance in chloride (Cl−) ion-containing environments. It is well known that pitting corrosion susceptibility of stainless steels depends very much on the environmental conditions in service [4]. A few studies have been conducted to determine the critical pitting temperature of 2101 DSS in NaCl solution at certain concentrations [2,5,6]. However, the roles of the constituent phases in 2101 DSS on its pitting corrosion behaviour are seldom explored. In this study, hence, the relative susceptibility between austenite and ferrite phase to pitting corrosion nucleation is focused. In the meantime, the effects of chloride concentration and solution temperature on pitting corrosion behaviour of 2101 DSS are also studied, in comparison with those of 2205 DSS.
Experimental
Materials and microstructures
Chemical compositions of 2101and 2205 DSS bulk and the average concentrations of major alloying elements in each constituent phase (wt-%).
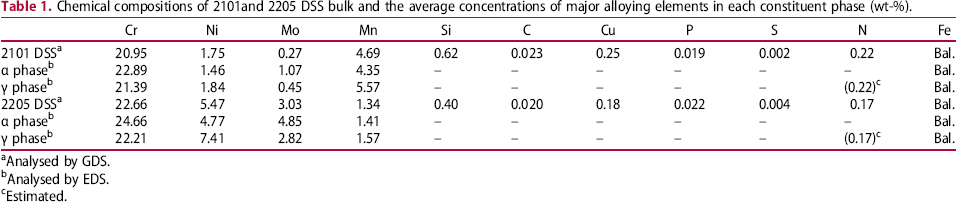
aAnalysed by GDS.
bAnalysed by EDS.
cEstimated.
Electrochemical test
Cyclic polarisation curve measurements were conducted in deaerated NaCl solutions with different concentrations (250, 2500, and 21300 ppm, respectively) and at various temperatures (ranging from 10 to 75°C). The solution annealed sample as mentioned above was embedded in epoxy resin with the plane perpendicular to the rolling direction (L plane) exposed. The exposed surface was successively ground with SiC papers to a grit of #1000 and then polished with 1 µm diamond paste. This sample was used as the working electrode, while a platinum foil and a saturated calomel electrode (SCE) were used as the counter and the reference electrode, respectively. All the potentials reported in this paper are referred with respect to SCE. The testing solution was purged with nitrogen gas for 40 min before and throughout conducting potentiodynamic polarisation test. The potential was scanned from −600 mV vs. open circuit potential towards anodic direction at a scan rate of 1 mV/sec. When the current density was up to 1 mA/cm2 or the potential reached at + 1500 mV, the potential was scanned back until intersecting the original curve.
Results and discussion
Microstructures
Three-dimensional metallographs of both 2101 and 2205 DSS are shown in Figure 1.
Three-dimensional metallographs of (a) 2101 and (b) 2205 DSS after solution annealing at 1100°C.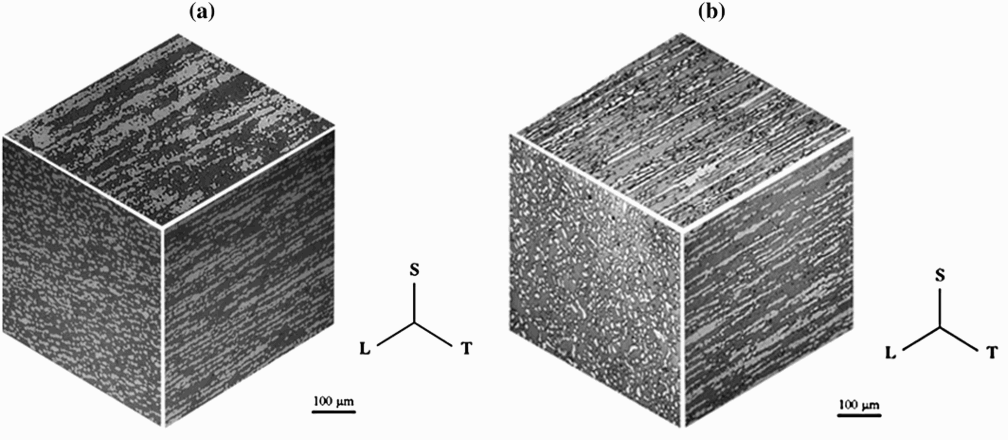
The volume percentages of austenite (γ) and ferrite (α) phase in three orientations (S, T and L planes) of 2101 and 2205 DSS.
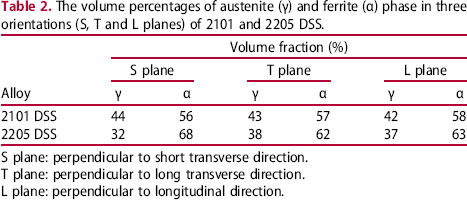
S plane: perpendicular to short transverse direction.
T plane: perpendicular to long transverse direction.
L plane: perpendicular to longitudinal direction.
Effects of temperature and chloride concentration on pitting corrosion behaviour
The cyclic polarisation curves of 2101 DSS determined in the deaerated solution containing 250 ppm Cl− ion at different temperatures are shown in Figure 2.
Results of cyclic polarisation curve measurement of 2101 DSS in deaerated 250 ppm Cl− ion contained NaCl solution at different temperatures.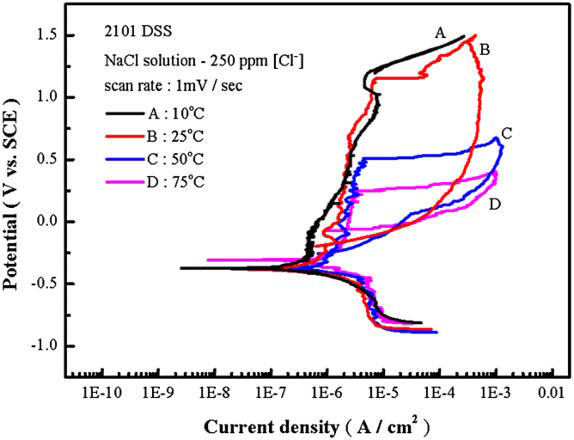
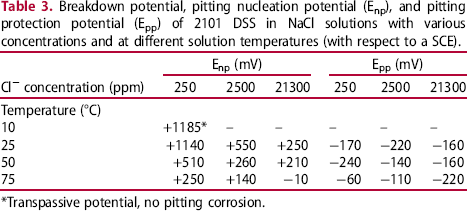
As shown in this figure, no hysteresis loop was observed at 10°C, indicating the absence of pitting corrosion in this solution up to a potential as high as +1500 mV. A sharp increase in current density at + 1185 mV actually corresponded to the transpassive behaviour of the passive film formed on the surface of 2101 DSS, not associated with pitting corrosion. As the solution temperature was raised to 25°C and above, a hysteresis loop was seen at each testing temperature, revealing pitting corrosion characteristics. The pitting corrosion nucleation potential (Enp) was about + 1140 mV, while the pitting corrosion protection potential (Epp) was about −170 mV. As the temperature was raised to 50°C, a significant decrease of Enp (+510 mV) was noted, indicating the increasing susceptibility to pitting corrosion. The temperature-dependent pitting corrosion behaviour was similar to others DSSs as reported elsewhere [7]. However, the value of Epp was only slightly lowered to −240 mV. At 75°C, the values of Enp and Epp for 2101 DSS in 250 ppm chloride-containing solution were + 250 and −60 mV, respectively.
The effects of Cl− ion concentration and a solution temperature of the changes of cyclic polarisation curves of 2101 DSS were also explored (see Figure 3).
Cyclic polarisation curves of 2101 DSS in deaerated NaCl solutions with different Cl− ion concentrations and at (a) 10°C, (b) 25°C, (c) 50°C, and (d) 75°C.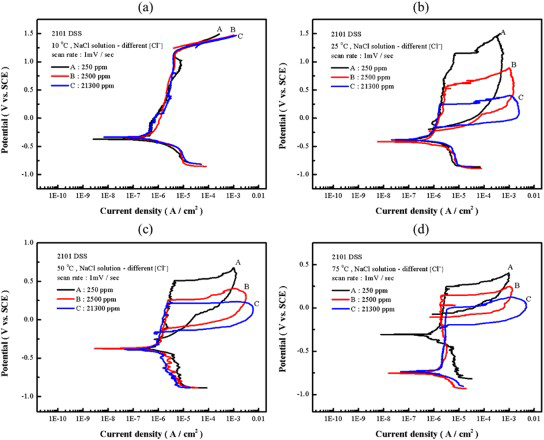
At 10°C, as can be seen in Figure 3(a), there were no hysteresis loops observed in all the solutions with different Cl− ion concentrations tested. These results indicated that 2101 DSS was resistant to pitting corrosion at and below 10°C even at a very high Cl− ion concentration. The effect of Cl− ion concentration on the change of polarisation curve at 25°C is manifested in Figure 3(b). All cyclic curves exhibited hysteresis loops, showing the susceptibility to pitting corrosion in all Cl− ion-containing solution at this temperature. The value of Enp decreased with increasing Cl− ion concentration; however, the value of Epp was almost the same (in the range of +380 ∼ +410 mV). The results suggested that nucleation of pitting corrosion was more sensitive to Cl− ion concentration while pitting protection was nearly independent of it. The concentration-dependent pitting corrosion susceptibility was similar to that of austenitic stainless steels [8,9]. As the solution temperatures were increased to 50 and 75°C, the value of Enp continued to decrease with increasing solution temperature and Cl− ion concentration, as shown in Figure 3(c) and (d), respectively. The dependences of Enp and Epp on solution temperature and Cl− ion concentration are listed and summarised in Table 3. The variations of the characteristic potentials, namely, transpassive potential, pitting nucleation potential, and pitting protection potential with Cl− ion concentration and temperature are more clearly presented in Figure 4.
Effects of Cl− ion concentration and solution temperature on the characteristic potentials of 2101 DSS determined by cyclic polarisation tests in NaCl solutions. Breakdown potential, pitting nucleation potential (Enp), and pitting protection potential (Epp) of 2101 DSS in NaCl solutions with various concentrations and at different solution temperatures (with respect to a SCE). *Transpassive potential, no pitting corrosion.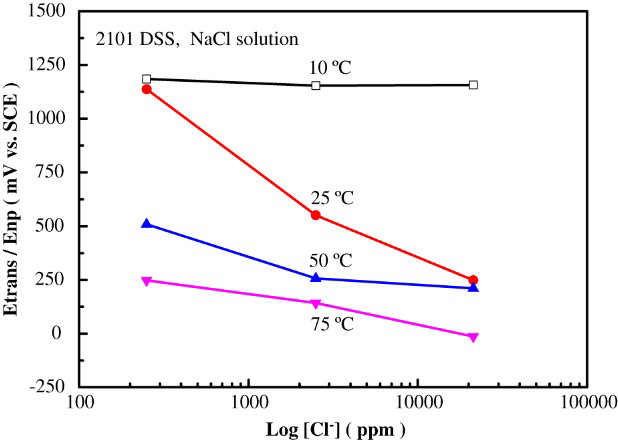
After potentiodynamic polarisation test, the surface morphology of each sample was examined by SEM. Each SEM micrograph shown in Figure 5 demonstrates the surface appearance of 2101 DSS after polarisation test at a specific condition. As shown in this figure, there was no corrosion pit found on the surface at 10°C in the solution containing Cl− ion at a concentration even as high as 21300 ppm. When the temperature was increased to 25°C and above, pitting corrosion was seen in all the Cl− ion-containing solution. The size of the pit seemed to increase with increasing temperature and Cl− ion concentration. More importantly, close examination of the SEM micrograph, pits preferentially appeared in the grains of α phase. Whether the chemical composition or the crystal structure played a more important role on pitting corrosion deserves further investigation in the future.
Surface morphologies of 2101 DSS after cyclic polarisation test in deaerated NaCl solutions with various Cl− ion concentration and at different temperatures: (a) 250 ppm, 10°C; (b) 250 ppm, 25°C; (c) 250 ppm, 50°C; (d) 250 ppm, 75°C; (e) 2500 ppm, 10°C; (f) 2500 ppm, 25°C; (g) 2500 ppm, 50°C; (h) 2500 ppm, 75°C; (i) 21300 ppm, 10°C; (j) 21300 ppm, 25°C; (k) 21300 ppm, 50°C; (l) 21300 ppm, 75°C.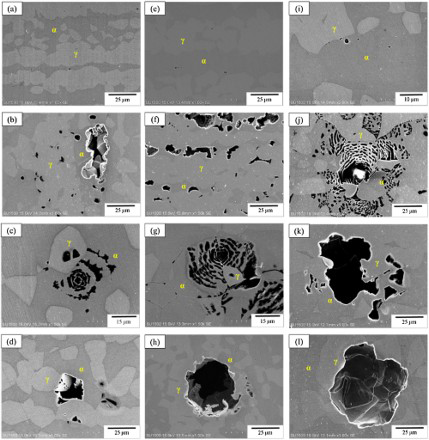
Similar investigation on pitting corrosion susceptibility of 2205 DSS was performed. The cyclic potentiodynamic polarisation curves obtained in NaCl solutions with various Cl− ion concentrations and at temperature ranging from 10 to 75°C are demonstrated in Figure 6.
Cyclic polarisation curves of 2205 DSS in deaerated NaCl solutions with different Cl− ion concentrations and at (a) 10°C, (b) 25°C, (c) 50°C, and (d) 75°C.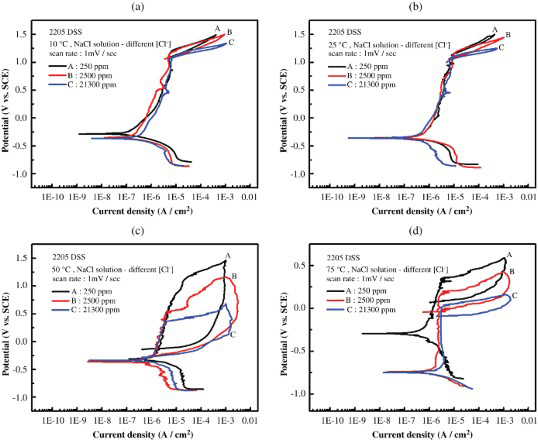
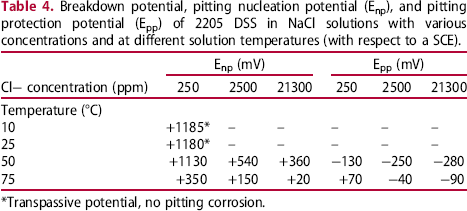
As shown in Figure 6(a) and (b), the hysteresis loops were absent at 10 and 25°C. Comparing with the results shown in Figure 3, the critical pitting temperature of 2205 DSS was higher than that of 2101 DSS, which was expected due to the higher Cr and the lower Mn contents in the former. At 50 and 75°C (see Figure 6(c) and (d)), the cyclic polarisation curves exhibited hysteresis loops, which are characteristics of pitting corrosion, in all the Cl− ion-containing solutions. The values of Enp and Epp obtained from the cyclic polarisation tests are summarised in Table 4 and Figure 7.
Effects of Cl− ion concentration and solution temperature on the characteristic potentials of 2205 DSS determined by cyclic polarisation tests in NaCl solutions. Breakdown potential, pitting nucleation potential (Enp), and pitting protection potential (Epp) of 2205 DSS in NaCl solutions with various concentrations and at different solution temperatures (with respect to a SCE). *Transpassive potential, no pitting corrosion.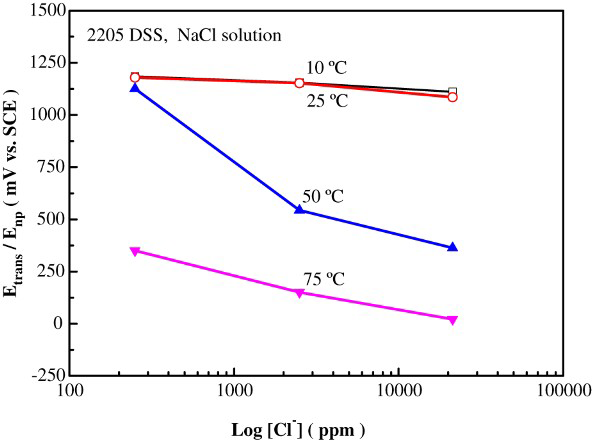
The corresponding SEM micrographs showing the surface morphologies of 2205 DSS after cyclic polarisation tests are demonstrated in Figure 8. The surfaces were clean when the solution temperatures were held at 10 and 25°C, in agreement with the polarisation curves shown in Figure 6(a) and (b). Raising the temperature to a 50 and 75°C, respectively, corrosion pits were formed on the surfaces as shown in Figure 8. Similar to those appeared in Figure 5 for 2101 DSS, α phase was more favourable than γ phase acting as the pit nucleation site. The lacy-type of pit morphology, with a large cavity underneath, was common for both 2101 and 2205 DSSs in the conditions investigated.
Surface morphologies of 2205 DSS after cyclic polarisation test in deaerated NaCl solutions with various Cl− ion concentration and at different temperatures: (a) 250 ppm, 10°C; (b) 250 ppm, 25°C; (c) 250 ppm, 50°C; (d) 250 ppm, 75°C; (e) 2500 ppm, 10°C; (f) 2500 ppm, 25°C; (g) 2500 ppm, 50°C; (h) 2500 ppm, 75°C; (i) 21300 ppm, 10°C; (j) 21300 ppm, 25°C; (k) 21300 ppm, 50°C; (l) 21300 ppm, 75°C.
Sites where pitting corrosion appeared on both 2101 and 2205 DSSs after cyclic polarisation tests in NaCl solutions at various temperatures.

x: No pit occurred.
Ο: Pit occurred.
Effect of chemical composition on pitting corrosion behaviour
The superior pitting corrosion resistance of 2205 DSS to that of 2101DSS is attributed to its higher pitting resistance equivalent (PRE) number [10–14]. The PRE value may be defined as:
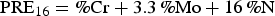
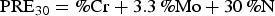
or,
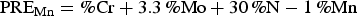
Since the S contents of 2101 and 2205 DSSs used in this study were low, Equation (2) might be applied for PRE value determination. Based on the chemical compositions listed in Table 1, the PRE values of 2101 DSS and 2205 DSS were 28.4 and 37.8, respectively. The lower Cr and Mo, even with a relatively higher N content, 2101DSS exhibited a lower PRE value, which gave rise to a lower pitting corrosion resistance. The results obtained from polarisation tests and morphological examinations were in agreement with the PRE values in evaluating the pitting corrosion resistance of these two DSSs.
For DSS, the constituent phases have different concentrations of the partitioning alloying elements. According to the above PRE equation and the chemical compositions of the constituent phases listed in Table 1, the PRE values for α and γ phases in 2101 DSS were 26.4 and 29.5 respectively. For 2205 DSS, they were 40.7 and 36.6, respectively. Since the EDS results were semi-quantitative, the PRE values of the constituent phases calculated according to the chemical compositions listed in Table 1 might differ slightly from the true values. In both DSSs, the PRE values only differed slightly between α and γ phases. Both Mn and N are austenite stabilising elements and enriched in γ phase. In these low S content DSSs, the detrimental effect of Mn might be ignored, as evidenced by the high pitting corrosion resistance of γ phase (Mn-rich) comparing with that of α phase (lower Mn content). The effect of N on the enhancement of passivation and pitting corrosion resistance of a stainless steel is well known. One of the beneficial effects of N in stainless steel is associated with the formation of NH3 and  , which can be easily adsorbed on the steel surface and promotes the passivation characteristics [16–19].
, which can be easily adsorbed on the steel surface and promotes the passivation characteristics [16–19].
Conclusions
Pitting corrosion resistance of both 2101 and 2205 DSSs decreased with increasing concentration of Cl− ion and solution temperature. The critical pitting temperature of 2101 DSS in the solution with a Cl− ion centration ranging from 250 to 21300 ppm was lower than that of 2205 DSS. The lower pitting corrosion resistance of 2101 DSS was attributed to its lower PRE as compared with that of 2205 DSS. SEM examination post cyclic polarisation test showed that the γ phase was more resistant to pitting corrosion than α phase. The high N content in the austenite phase played the key role in passivation against pitting corrosion of both DSSs investigated.
Footnotes
Acknowledgement
The supports from Bureau of Energy, Ministry of Economics of the Republic of China under contract of 104-D0303, and the Energy Research Center in National Cheng Kung University are greatly appreciated.
