Abstract
Chloride salts can prevent roads from freezing in cold regions, together with severe corrosion on steel constructions. To develop an ecological and low-corrosive antifreeze, di-sodium hydrogenphosphate (Na2HPO4) was chosen as the additive into chloride salts. The addition of Na2HPO4 into either of the antifreezes of natural salt (N/S), NaCl, CaCl2 or MgCl2 can suppress the corrosion reaction of mild steel in the 3.0% antifreeze solution. Moreover, the addition of CaCl2 or MgCl2 into the antifreeze containing NaCl and 2.0% Na2HPO4 significantly decreased the corrosion rate. According to polarisation and XPS analyses, it is evident that the obtained low corrosion rates on mild steel are related to the suppressed cathodic and anodic reactions and the formation of a protective film containing Fe, P, O, Ca or Mg.
This paper is part of a supplementary issue from the 17th Asia-Pacific Corrosion Control Conference (APCCC-17).
Introduction
To protect roads from freezing of water in cold winter regions, antifreezes, such as NaCl and CaCl2, are usually sprayed on roads to depress the freezing point of water or to melt ice and snow [1–5]. However, the chloride ions dissolved in water generally cause corrosion of metallic structures on/in roads as well as running automobiles. It is known as the salt damage of corrosion [4–9]. Various anticorrosive components, such as phosphates, chromates, nitrites, magnesium salts, aluminium salts, acetates, methylglucoside, triethanolamine, organozinc compounds, have been utilised as corrosion inhibitors into the traditional antifreezes of NaCl or CaCl2 [1,2]. The use of non-toxic phosphates, particularly the safe hydrogenphosphate in the food industry had attracted the interest of researchers in suppression of the corrosion of steels [1,3,10]. Till date, it had been confirmed from a continuous immersion corrosion test that the addition of Na2HPO4 to NaCl really promoted rather than suppressed the corrosion of steel [1]. On the other hand, the corrosion inhibition effect was confirmed when adding the Na2HPO4 together with CaCl2 or MgCl2 to the NaCl solution.
However, the authors considered that it should be more prudent to assess the result of the addition of Na2HPO4 to NaCl before drawing final conclusion. Furthermore, the wet and dry cyclic corrosion test, which is closer to the real corrosion environment, should be carried out, and the polarisation measurement as well as the surface composition analysis should be utilised to clarify the mechanism. Although the addition of K2HPO4 in antifreeze had appeared in opened patents [1], Na2HPO4, which is safely used as food additive and has higher antifreeze ability, has not been tried. With the aim of developing low cost, ecological, low-corrosive and mass-produced antifreezes, it is worthy to know the effect of adding Na2HPO4 to NaCl, MgCl2 or CaCl2 on the corrosion behaviour and understand the mechanism. Based on the above considerations, the authors had developed an ecological and low-corrosive antifreeze easily mass-produced antifreeze by adding di-sodium hydrogen phosphate (Na2HPO4) into a natural salt (N/S) [3]. In the N/S, mainly the NaCl and a little of impurities of CaCl2 and MgCl2 are contained. However, even if the low corrosiveness of the antifreeze on mild steel had been confirmed in the wet and dry cyclic test, the influence of each component in the N/S and the amount of Na2HPO4 on the corrosion behaviour of the steel should be assessed.
Therefore, here in this work, several types antifreezes containing NaCl, MgCl2, CaCl2 and Na2HPO4, were produced and their aqueous solutions were used in the wet and dry cyclic corrosion test to get the corrosion rate of mild steel. Furthermore, based on the polarisation and X-ray photoelectron spectroscopy (XPS) analysis, the mechanism of the corrosion behaviour of mild steel in the aqueous solution of the antifreezes is discussed.
Experimental
Preparation of solution with dissolved antifreeze and the mild steel specimen
Sodium chloride (NaCl), calcium chloride (CaCl2) and magnesium chloride (MgCl2) were considered as antifreezes and the latter two (CaCl2, MgCl2) were also treated as additives into NaCl. In addition, the natural salt (N/S) containing about 96.0% NaCl and 0.23% CaCl2 as well as 0.19% MgCl2 was also used as one of the antifreezes. On the other hand, di-sodium hydrogen phosphate (Na2HPO4) was used as the corrosion inhibitor to be added into the above antifreezes. It is the reagent of MgCl2·6H2O but not that of MgCl2 was used in this work. Therefore, the ratio of MgCl2·6H2O rather than that of MgCl2 was described in the antifreeze. The 3.0 wt-% antifreeze solution containing several types of the above salts (N/S, NaCl, CaCl2, MgCl2·6H2O or Na2HPO4) was used in the wet and dry cyclic corrosion test and the polarisation measurement. In the following description, the ratios of each type of salt (N/S, NaCl, CaCl2, MgCl2·6H2O or Na2HPO4) in the antifreeze are noted rather than their real concentrations in the solution.
A mild steel plate (JIS SPCC of composition (C < 0.15%, Mn < 0.60%, P < 0.100%, S < 0.035%)) with size of 67 mm × 50 mm × 1 mm was used as the specimen to evaluate the corrosiveness of the antifreezes. The specimens were ultrasonically cleaned in water and acetone and the weight change was measured using a precise balance.
Wet and dry cyclic corrosion test
The specimens were kept in 3.0 wt-% solution of antifreeze in a 300-mL glass beaker. The solution was kept in a chamber with constant temperature (296 ± 2 K) and humidity (50 ± 2%RH) for 7 days, with the cyclically wet immersion for 1 day and then dried for another 1 day in the same chamber. Air was continuously pumped into the chamber during the test. After that, the rust formed on the specimen was carefully removed by a soft brush without damaging the uncorded steel in a solution of 1.0% propargyl alcohol (C3H4O) and 5.0% hydrogen chloride (HCl), following which they were cleaned in water and acetone. From the weight change before and after corrosion test, the corrosion rate of steel was calculated in the unit of mdd (mg/(dm2·day)).
Polarisation measurement
The specimen surface was masked with silicone sealant, leaving only a central test zone of 10 mm × 10 mm as the polarisation area. The polarisation curve was measured with the three-electrode configuration in the above solutions at 303 ± 2 K with a platinum sheet counter electrode and Ag/AgCl (S.S.E.) reference electrode using an HAB151 potentiostat of HOKUTO DENKO. In the polarisation measurement, potential was swept with a rate of 0.33 mV s−1 from the cathodic potential of –600 mV to the anodic side to clarify the current variation on both the cathodic and the anodic reactions. The test solution was bubbled with air for 1.8 ks prior to polarisation.
XPS analysis
After the wet and dry cyclic test, several selected specimen surfaces were analysed by XPS (Shimadzu Co.: AXIS ULTRA). The X-ray was the Kα spectrum from Mg target, generated at the voltage of 15 kV and the anodic current of 10 mA. The air pressure in the vacuum chamber was lower than 5 × 10−7 Pa and the analysis area was 2 mm × 1 mm. The analysed binding energy ranges from 1100 to 0 eV with a scanning step of 0.2 eV per 300 ms without pre-etching the surface. From the obtained spectra, the components on the surface were determined and discussed in relation to the corrosion behaviour of the steel.
Ice melting capacity
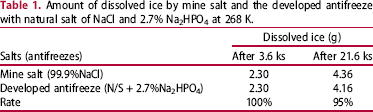
A glass dish with 6.70 g distilled water was first frozen in a cold chamber with a constant temperature of 268 K. Next, 0.35 g salt particles (99.9% NaCl) or 0.35 g of the developed antifreeze (97.3% N/S, 2.7% Na2HPO4) was sprayed on the ice. The dish was then sealed with a glass cover and kept in the above cold chamber for 3.6 or 21.6 ks. The dissolved water on the ice was removed by a tissue paper and the weight change of the tissue paper was measured to obtain the amount of dissolved ice.
Results and discussion
Wet and dry cyclic corrosion test
Solutions containing NaCl, Na2HPO4, CaCl2 and MgCl2
Figure 1(a,b) shows the appearances of the solutions of 3.0 wt-% N/S, NaCl, CaCl2 or MgCl2·6H2O before and after the wet and dry cyclic corrosion test. Figure 1(c) shows the mild steel surface after the test without removing the rust. No appreciable difference can be found from both the solutions and the specimen surfaces. According to the following description (Figures 2, 8, and 10), it is evident that the corrosion rates in the CaCl2 and MgCl2 solution are a slightly greater than those in the N/S and NaCl solutions.
Appearance of solutions of 3.0% N/S, NaCl, CaCl2 and MgCl2·6H2O before (a) and after (b) wet and dry cyclic corrosion and (c) specimens after wet and dry cyclic corrosion without the removal of rust.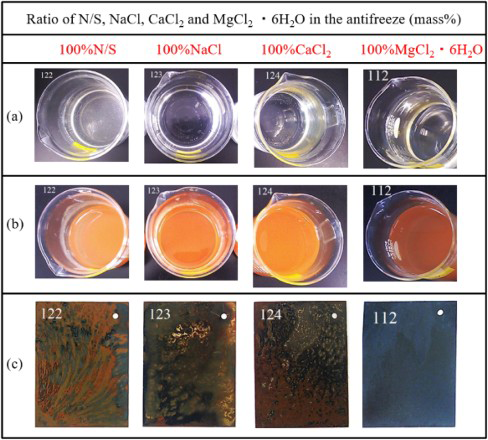
Figure 2 shows the corrosion rates of the mild steel obtained from the wet and dry cyclic corrosion test in 3.0% (NaCl + x% Na2HPO4) or 3.0% (N/S + x% Na2HPO4) solutions, where the x% represents the ratio of Na2HPO4 in the antifreeze of (NaCl + Na2HPO4) or (N/S + Na2HPO4). The corrosion rates of steel in 3.0% NaCl or 3.0% N/S are almost the same as 20.6 mdd. The value decreased to 16.5 mdd in the NaCl solution when 0.5% Na2HPO4 was added into antifreeze and remained same even when increasing the ratio of Na2HPO4 in the antifreeze to 20.0%. The value decreased to 10.3 mdd when the Na2HPO4 ratio of 30.0% and a value of 0.4 mdd was obtained at the Na2HPO4 ratio of 100% (solution concentration: 3.0% Na2HPO4). On the other hand, comparing to the NaCl solution, the decrease in corrosion rate in the N/S solution is much larger when adding Na2HPO4. The value decreased to 6.2 mdd at the ratio of 0.5% Na2HPO4, 0.59 mdd at the ratio of 2.0% Na2HPO4. The value remained almost the same low level when the Na2HPO4 ratio is 2.0% and above. This means that the Na2HPO4 really has an ability to inhibit the corrosion progress in both NaCl and N/S solution, and the suppression effect is much larger in the N/S solution than in the NaCl solution. The result in the NaCl solution is different from that reported literature [1], where a continuous immersion test was carried out. Since the antifreeze has 96.0% NaCl and a little of CaCl2 as well as MgCl2 in the N/S, the Ca2+ and Mg2+ should have played important roles with the Corrosion rates obtained in 3.0% (NaCl + x% Na2HPO4) or in 3.0% (N/S + x% Na2HPO4) solutions. to inhibit the corrosion reaction. Perhaps, an insoluble film had been formed on the steel surface to decelerate the corrosion reaction. Since a much small corrosion rate was obtained in the 3.0% (N/S + 2.0% Na2HPO4) solution, it should be important to note the role of CaCl2 and MgCl2 in (NaCl + 2.0% Na2HPO4) antifreeze in the following experiments.
to inhibit the corrosion reaction. Perhaps, an insoluble film had been formed on the steel surface to decelerate the corrosion reaction. Since a much small corrosion rate was obtained in the 3.0% (N/S + 2.0% Na2HPO4) solution, it should be important to note the role of CaCl2 and MgCl2 in (NaCl + 2.0% Na2HPO4) antifreeze in the following experiments.
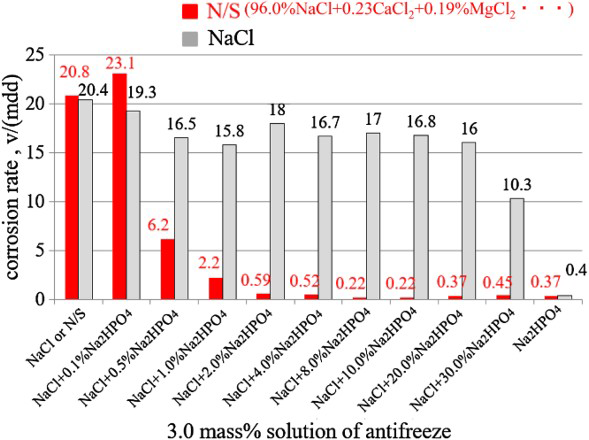
Figure 3 shows the corroded specimen surfaces with rust. Brighter surfaces can be seen in case of lower corrosion rates. Slight corrosion occurred in the N/S solution when the Na2HPO4 ratio is over 2.0%, leaving a bright non-corroded surface where a protective film might have been formed. On the other hand, the occurrence of the bright surface in NaCl solution is delayed only when the Na2HPO4 ratio is over 30.0%.
Specimen surfaces after wet and dry cyclic corrosion in 3.0% (NaCl + x% Na2HPO4) or in 3.0% (N/S + x% Na2HPO4) solution, without the removal of rust.
Figure 4 shows the corrosion rates of the steel obtained in 3.0 wt-% (NaCl + 2.0% Na2HPO4 + x% CaCl2) solutions, where the x% means the ratio of CaCl2 in the antifreeze of (NaCl + Na2HPO4 + CaCl2). From this figure, it can be seen that the corrosion rate largely decreased from 20.4 to 3.5 mdd when adding 0.5% CaCl2 into the antifreeze of (NaCl + 2.0% Na2HPO4). The rate then gradually increased to 7.2 mdd with the increase in addition of CaCl2 ratio to 3.0% and kept almost the value in the case of 10.0% CaCl2. Figure 5 shows the solution appearance before and after the test as well as the corroded specimen surfaces with rust. The appearance of the solutions before the test is also shown as inset in Figure 4. No large difference could be found on the specimen surface. On the other hand, it can be seen from the solution pictures that the white precipitation appeared just when the ratio of Na2HPO4 is more than 1.0%, which is well corresponding to the variation of the corrosion rate. The precipitated matter is considered as CaHPO4, which has only a solubility of 0.02 g in 100 mL water at 298 K. Too many Ca2+ ions promoted the formation of slightly soluble CaHPO4 at much early stage before the corrosion test, which decreased the effective inhibitor species of Corrosion rates obtained in solution with the concentration of 3.0% (NaCl + 2.0% Na2HP04 + x% CaCl2). Appearance of solutions of 3.0% (NaCl + 2.0% Na2HPO4 + x% CaCl2) before (a) and after (b) wet and dry cyclic corrosion and (c) specimens after wet and dry cyclic corrosion without removing rust. in the solution. On the other hand, it also indicates that the condensation and protective film containing Ca and P should have also slowly formed during the corrosion reaction, which directly inhibited the corrosion reaction. However, the much more quickly formed CaHPO4 before corrosion test is porous and would adhere on the surface, which might act as obstacle for formation of protective film.
in the solution. On the other hand, it also indicates that the condensation and protective film containing Ca and P should have also slowly formed during the corrosion reaction, which directly inhibited the corrosion reaction. However, the much more quickly formed CaHPO4 before corrosion test is porous and would adhere on the surface, which might act as obstacle for formation of protective film.
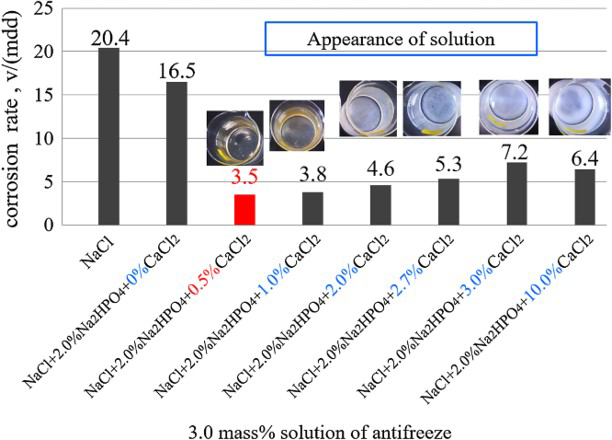

Figure 6 shows the corrosion rates of the steel obtained in 3.0% (NaCl + 2.0% Na2HPO4 + x% MgCl2·6H2O) solutions, where the x% means the ratio of MgCl2·6H2O in the antifreeze of (NaCl + Na2HPO4 + MgCl2·6H2O). Different from the results in Figure 4, the corrosion rate gradually decreased with the increase in the ratio of MgCl2·6H2O in the antifreeze and the smallest value was obtained at the MgCl2·6H2O ratio of 10.0%.
Corrosion rates obtained in solutions with the concentration of 3.0% (NaCl + 2.0% Na2HPO4 + x% MgCl2·6H2O).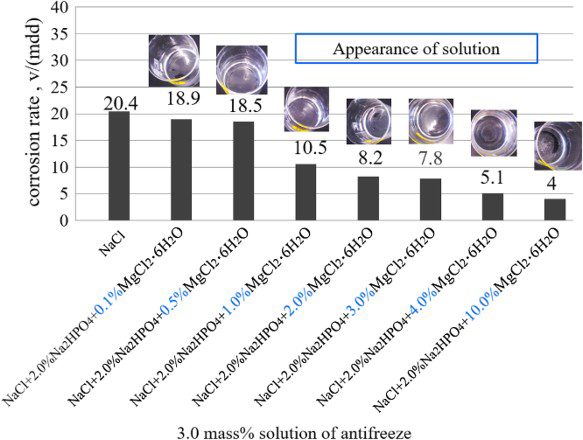
Figure 7 shows the solution appearance before and after the test as well as the corroded specimen surfaces with rust. The appearance of the solution before the immersion of the steel is also shown as inset in Figure 6. According to the observation of the specimen, lesser corrosion had occurred when the ratio of MgCl2·6H2O is 3.0% and above. According to these observations, no precipitation can be found from all the conditions with various MgCl2·6H2O ratios (0.1-10.0%). It suggests that the solubility of MgHPO4 in water is large enough to avoid precipitation in this experiment.
Appearance of solutions of 3.0% NaCl + 2.0%Na2HPO4 + x%MgCl2·6H2O) before (a) and after (b) wet and dry cyclic corrosion and specimens after wet and dry cyclic corrosion without removing rust.
Solutions containing CaCl2, MgCl2 and Na2HPO4
Figure 8 shows the corrosion rates obtained in 3.0 wt-% (CaCl2 + x% Na2HPO4) solutions, where x% represents the ratio of Na2HPO4 in the antifreeze of (CaCl2 + Na2HPO4). When the ratio of Na2HPO4 increased from 0.1 to 1.0%, the corrosion rate gradually decreased. However, the value increased again with the increase in the Na2HPO4 from 4.0 to 30.0%. The smallest corrosion rate was about 5.5 mdd, which was obtained at the Na2HPO4 ratio of 1.0-4.0%. Figure 9 shows the solution appearance before and after the test as well as the corroded specimen surfaces with rust. From the specimen surface observation and the solutions after corrosion test, the corrosion in the solution with Na2HPO4 ratio more than 1.0% is slighter. According to the solution observation (Figure 8: inset; Figure 9), it is found that precipitation occurred when the Na2HPO4 ratio is over 1.0%. This tendency is very near to that described in Figure 4, where CaCl2 was added into the solution with NaCl and Na2HPO4. Therefore, the white precipitation should be also CaHPO4, which decreased the effective component of Ca2+ ions in the solution in this case. Of course, the dissolved Corrosion rates obtained in solutions with the concentration of 3.0% (CaCl2 + x% Na2HPO4) solution. Appearance of solutions of 3.0% (CaCl2 + x%Na2HPO4) before (a) and after (b) wet and dry cyclic corrosion and (c) specimens after wet and dry cyclic corrosion without removing rust.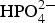 ions with the Ca2+ ions in the solution should have helped to form a protective film when it was formed during corrosion.
ions with the Ca2+ ions in the solution should have helped to form a protective film when it was formed during corrosion.
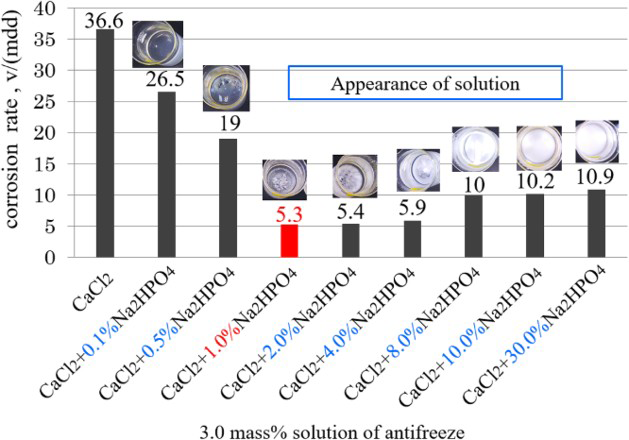

Figure 10 shows the corrosion rates obtained in 3.0 wt-% (MgCl2·6H2O + x% Na2HPO4) solutions, where x% represents the ratio of Na2HPO4 in the antifreeze of (MgCl2·6H2O + Na2HPO4). It is known that with the increase of Na2HPO4 addition the corrosion rate gradually decreased until to the ratio of Na2HPO4 of 3.0%. After that, the corrosion rate almost did not vary even when increasing the ratio to 30.0%.
Corrosion rates obtained in solutions with the concentration of 3.0% (MgCl2·6H2O + x% Na2HPO4) solution.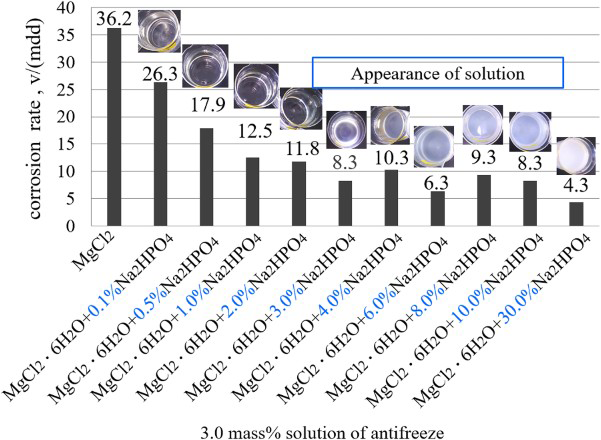
Figure 11 shows the solution appearance before and after the test as well as the corroded specimen surfaces with rust. From the solution observations, it is found that the solution kept transparent until the Na2HPO4 ratio of 3.0%. This means that the amount of MgHPO4 in the solution had been over its solubility when the Na2HPO4 ratio is more than 3.0%. However, though different from that obtained in Figures 4 and 8, the precipitation did not largely weaken the corrosion reaction.
Appearance of solutions of 3.0% (MgCl2·6H2O + x%Na2HPO4) before (a) and after (b) wet and dry cyclic corrosion, and specimens after wet and dry cyclic corrosion without removing rust.
According to the above results, high corrosion suppression effect was obtained in the 3.0% (NaCl + 2.0% Na2HPO4 + 0.5% CaCl2) and 3.0% (CaCl2 + 1.0% Na2HPO4). The corresponding solution concentrations of the possible CaHPO4 in the solutions are respectively about 0.018 and 0.028%, which are very near the solubility of CaHPO4 (0.02 g per 100 mL water at 298 K). Therefore, it is reasonable to consider that the best inhibition effect can be obtained at the saturated concentration of CaHPO4 and much more Ca2+ or 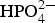 will cause the precipitation of CaHPO4 at the early stage. Such early precipitated CaHPO4 generally adheres on the steel surface in a porous state, which obstructs the formation of condensation and protective film. On the other hand, although the MgHPO4 was also as insoluble with almost the same solubility (0.025 g per 100 mL water at 298 K), the increased MgCl2 in solution might have helped to form protective film during the corrosion reaction.
will cause the precipitation of CaHPO4 at the early stage. Such early precipitated CaHPO4 generally adheres on the steel surface in a porous state, which obstructs the formation of condensation and protective film. On the other hand, although the MgHPO4 was also as insoluble with almost the same solubility (0.025 g per 100 mL water at 298 K), the increased MgCl2 in solution might have helped to form protective film during the corrosion reaction.
Polarisation curves
Figure 12 shows several typical polarisation curves obtained in various solutions. The polarisation curves of the mild steel in NaCl and N/S are almost the same. In Figure 12(a), the corrosion potential became nobler when 2.0% Na2HPO4 is added into NaCl, and the corrosion current decreased due to the suppression of the anodic rather than the cathodic reaction. This result is different from that reported literature [1]. It indicates the adsorption of Polarisation curves of steel in different solutions with the total salt concentration of 3.0%.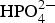 ions or the formation of compound containing iron and phosphorous on the anodic dissolution sites. The corrosion potential decreased further a little, when the ratio of Na2HPO4 increased, with slight increased suppression of cathodic reaction. In Figure 12(b), comparing to those in CaCl2 or MgCl2 solution, both corrosion potential and corrosion current largely decreased when adding Na2HPO4 into CaCl2 or MgCl2, with large suppression of cathodic reaction. These results indicate the formation of protective film containing phosphorous and calcium or magnesium to suppress cathodic reaction. According to Figure 12(c), the corrosion potential became nobler when adding MgCl2·6H2O to the (NaCl + 2.0% Na2HPO4) antifreeze, and the corrosion current decreased with the increase of MgCl2·6H2O due to both the suppression of cathodic and anodic reactions. When 0.5% CaCl2 was added to the antifreeze of (NaCl + 2.0% Na2HPO4), both the corrosion potential and the corrosion current became much lower, which is much similar with that obtained at (N/S + 2.0% Na2HPO4) in Figure 12(d). In the both cases of (N/S + 2.0% Na2HPO4) and (NaCl + 2.0% Na2HPO4 + 0.5% CaCl2), both their cathodic and anodic reactions, especially the cathodic reactions were suppressed. It indicates that the corrosion inhibition mechanism of the antifreeze of (N/S + 2.0% Na2HPO4) is due to the formation of a protective film containing P, Fe, Ca or Mg. The above polarisation curves have good correlation with the wet and dry cyclic test.
ions or the formation of compound containing iron and phosphorous on the anodic dissolution sites. The corrosion potential decreased further a little, when the ratio of Na2HPO4 increased, with slight increased suppression of cathodic reaction. In Figure 12(b), comparing to those in CaCl2 or MgCl2 solution, both corrosion potential and corrosion current largely decreased when adding Na2HPO4 into CaCl2 or MgCl2, with large suppression of cathodic reaction. These results indicate the formation of protective film containing phosphorous and calcium or magnesium to suppress cathodic reaction. According to Figure 12(c), the corrosion potential became nobler when adding MgCl2·6H2O to the (NaCl + 2.0% Na2HPO4) antifreeze, and the corrosion current decreased with the increase of MgCl2·6H2O due to both the suppression of cathodic and anodic reactions. When 0.5% CaCl2 was added to the antifreeze of (NaCl + 2.0% Na2HPO4), both the corrosion potential and the corrosion current became much lower, which is much similar with that obtained at (N/S + 2.0% Na2HPO4) in Figure 12(d). In the both cases of (N/S + 2.0% Na2HPO4) and (NaCl + 2.0% Na2HPO4 + 0.5% CaCl2), both their cathodic and anodic reactions, especially the cathodic reactions were suppressed. It indicates that the corrosion inhibition mechanism of the antifreeze of (N/S + 2.0% Na2HPO4) is due to the formation of a protective film containing P, Fe, Ca or Mg. The above polarisation curves have good correlation with the wet and dry cyclic test.
XPS analysis
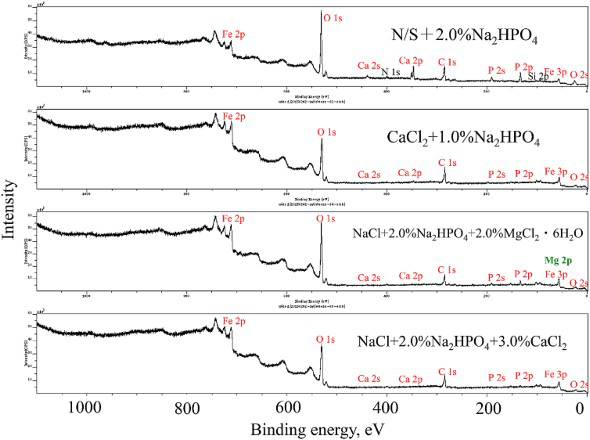
Figure 13 shows the XPS spectra of mild steel after the wet and dry test for 7 days in different solutions with the total salt concentration of 3.0%. The elements of Fe, Ca, P and O were detected from each of the surfaces. Comparing to other three, more Ca was detected on the surface tested in the solution containing (N/S + 2.0% Na2HPO4). The peak of Mg 2p and Fe 3p are overlapped on the surface corroded in the solution containing (NaCl + 2.0% Na2HPO4 + 2.0% MgCl2·6H2O). The main peak of P 2p lies at 131.8 eV, indicating the existence of XPS spectra of mild steel after the wet and dry test for 7 days in different solutions with the total salt concentration of 3.0%. on the surface. Therefore, it is evident that the elements of Fe, Ca, Mg, P and O have promoted the formation of protective film during corrosion.
on the surface. Therefore, it is evident that the elements of Fe, Ca, Mg, P and O have promoted the formation of protective film during corrosion.
Considering that FeHPO4, CaHPO4 and MgHPO4 have poor solubility in water, the isolate or the complex ones might have been formed as part of the protective film on the steel surface. Since direct and certain evidence of such compounds on steel surface had not been found, it is reasonable to consider that the suppression of corrosion should be due to the formation of a protective film containing Fe, O, P, Ca or Mg. Perhaps, the Fe, Ca or Mg atoms combined on steel can attract large  ions to the surface, which can reduce the adhesion of chloride ions.
ions to the surface, which can reduce the adhesion of chloride ions.
Capacity of dissolving ice
Amount of dissolved ice by mine salt and the developed antifreeze with natural salt of NaCl and 2.7% Na2HPO4 at 268 K.
Conclusions
To develop ecological and low-corrosive antifreezes, Na2HPO4 with different weight ratios was added to antifreezes of natural salt (N/S), NaCl, CaCl2 and MgCl2. The corrosion rate of mild steel was measured in the aqueous solution containing the antifreeze by a wet and dry cyclic test. The polarisation and XPS analyses were used to discuss the inhibition suppression mechanism. The following results are obtained:
The addition of Na2HPO4 into either of the antifreezes containing natural salt (N/S), NaCl, CaCl2 or MgCl2 can suppress the corrosion of mild steel (SPCC) in the 3.0 wt-% antifreeze aqueous solutions. The addition of CaCl2 or MgCl2 to the antifreeze of (NaCl + 2.0% Na2HPO4) significantly decreased the corrosion rate of steel. The polarisation shows that the
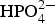 with Ca2+ or Mg2+ can suppress both the anodic and cathodic reactions but with larger effect on the cathodic reaction of the mild steel. The observation of solution and specimen as well as the XPS analysis indicates the formation of a protective film containing iron, phosphorous, oxygen and calcium or magnesium.
with Ca2+ or Mg2+ can suppress both the anodic and cathodic reactions but with larger effect on the cathodic reaction of the mild steel. The observation of solution and specimen as well as the XPS analysis indicates the formation of a protective film containing iron, phosphorous, oxygen and calcium or magnesium.
Footnotes
Acknowledgement
The authors are very grateful to JOY Co. Ltd for providing the natural salt. And the experiment was assisted by Mr T. Saito, N. Sakamura, A. Fujimori and N. Mae.
Disclosure statement
No potential conflict of interest was reported by the authors.
