Abstract
Carbon steel is the candidate material of the canister in high level radioactive nuclear waste disposal repository in China. The corrosion behaviour of X65 grade low carbon steel exposed to bentonite thermally aged at 363.15 K after exposure to 0 and 2.98 kGy h−1 γ-irradiation for 1007 h was studied using optical microscopy, scanning electron microscopy, energy dispersive X-ray spectroscopy, X-ray diffraction, micro-Raman spectroscopy (μ-Raman), weight loss measurements and white-light interferometry. The results show that 2.98 kGy h−1 γ-irradiation causes the formation of new phases of siderite and maghemite, and accelerates the corrosion rate by ∼33%.
This paper is part of a supplement on the 6th International Workshop on Long-Term Prediction of Corrosion Damage in Nuclear Waste Systems.
Introduction
In recent years, the nuclear industry has developed rapidly in China. Thirty-seven nuclear power units have been in service with a total installed capacity of 34.62 GW as of March 2017 [1], and the total installed capacity is expected to reach 70 GW by 2020. Consequently, a large amount of high level radioactive nuclear waste (HLW) has been produced during the operation of nuclear power plants, which is projected to reach 8,011.8 t of spent fuel [2] by 2020. The disposal of HLW is a significant issue, which is related to the safety and health of the public. Geological disposal has been selected as the strategy for handling the HLW in China, and the repository would be a multiple-barrier system. In brief, HLW would be enclosed in carbon steel canisters and then buried in granite at a depth of 500 m. The space between the canisters and the granite will be filled with bentonite. Carbon steel is one of the candidate materials for the construction of the disposal canisters. Its corrosion behaviour is directly related to the safety of the repository.
The corrosion behaviour of carbon steel in bentonite can be influenced by many factors [3], such as the type, density, and pore water composition of bentonite, and the irradiation coming from the decay of radionuclides. To accurately predict the long-term corrosion rate of carbon steel, every factor should be studied in detail. Limited research on the effect of irradiation on the corrosion behaviour of carbon steel in bentonite has been reported in the literature. Many researchers have investigated the effect in groundwater, and the results show that γ-irradiation accelerated the corrosion rate [4,5] and altered the phase of corrosion scales [6]. However, the conclusions acquired from solutions cannot be used to predict the real reactions occurring in bentonite. This paper focuses on the effect of γ-irradiation on the corrosion behaviour of carbon steel exposed to aerobic Gaomiaozi bentonite containing 17 wt-% Beishan groundwater and thermally aged at 363.15 K for simulating the conditions in the preliminary stage of the Beishan repository. A γ-irradiation dose rate of 2.98 kGy h−1, which is 30 times [7] of the maximum dose rate expected at the surface of waste packages, was used to amplify the effects of γ-irradiation.
The surface morphology of corrosion scales was characterised by optical microscopy (OM) and scanning electron microscopy (SEM) equipped with energy dispersive X-ray spectroscopy (EDS); the phases of the corrosion scales were detected by X-ray diffraction (XRD) and micro-Raman spectroscopy (μ-Raman). The corrosion rates were obtained by weight loss measurements and the surface morphology of samples after weight loss was observed by white-light interferometry (WLI).
Experimental design
For the purpose of this research, X65 grade low carbon steel was used. Its chemical composition (wt-%) is as follows: C 0.065, Si 0.14, Mn 1.58, P 0.008, S 0.0016, Al 0.042, Cr 0.22, Ni 0.01, Cu 0.02, V 0.05, Ti 0.015, Nb 0.063, Ca 0.0025 and Fe Bal. Eight carbon steel samples (10 mm × 10 mm × 1 mm) were cut from a hot-rolled pipeline. Prior to testing, the samples were mechanically polished with silicon carbide emery paper down to 2000 grits, and dried by air blower. For convenience, the eight samples were numbered as a1, a2, a3, a4, b1, b2, b3 and b4, respectively. Samples a1, a2, a3, b1, b2 and b3 were used as weight loss samples, and samples a4 and b4 were used for corrosion scales characterisation. Gaomiaozi bentonite containing 17 wt-% Beishan groundwater was used as the immersion medium. Gaomiaozi bentonite comprises 63.77-80.92% montmorillonite ((Na,Ca)0.3(Al,Mg)2Si4O10(OH)2·nH2O) and Beishan groundwater is Na–Cl–SO4 type. At first, all samples were buried in bentonite and exposed to air. Samples a1–a4 were loaded into canister A with bentonite and samples b1–b4 were loaded into canister B, also with bentonite. Following loading, both canisters were sealed. Canister B was irradiated by a Co-60 source at an absorption dose rate of 2.98 kGy h−1 for 1007 h to obtain a cumulative dose of 3 MGy, while canister A was kept at room temperature as a blank control. Finally, both canisters were thermally aged at 363.15 K for 2880 h to standardise test conditions.
Following the immersion experiments, the surface morphology and cross-sectional morphology of the corrosion scales formed on samples a4 and b4 were studied using OM, SEM and EDS. The nature of the corrosion products formed on samples a4 and b4 was examined by XRD and μ-Raman. The weight loss of samples a1, a2, a3, b1, b2 and b3 was measured after chemically cleaning in aqueous solution of HCl (15wt-%) + 3.5 g L−1 C6H12N4. The surface morphology of the samples after chemically removing the corrosion products was observed by SEM and WLI.
Results and discussion
Surface morphology
Figure 1 shows an OM view of the surface morphology of samples a4 and b4. It shows that less corrosion products were formed on the surface of a4 when compared with those on the surface of b4, which indicates that irradiation accelerates the corrosion rate of carbon steel. A higher magnification image of the corrosion products formed on the surface of the two samples (SEM) is shown in Figure 2. These SEM micrographs show that the entire surface of sample b4 was covered with corrosion products, while large areas of the surface of sample a4 remained pristine, which confirms the accelerating effect of γ-irradiation.
OM images of the surface of X65 grade low carbon steel samples exposed to bentonite and thermally aged at 363.15 K for 2880 h after exposure to (a) 0 kGy h−1 and (b) 2.98 kGy h−1 γ-irradiation for 1007 h. SEM images of the surface of X65 grade low carbon steel samples exposed to bentonite and thermally aged at 363.15 K for 2880 h after exposure to (a) 0 kGy h−1 and (b) 2.98 kGy h−1 γ-irradiation for 1007 h.

Cross-sectional analysis
The cross-sectional morphology of samples a4 and b4 were studied. As illustrated in Figure 3, the corrosion scales formed on the surface of a4 and b4 are both non-uniform. The corrosion depth ranged from several micrometres to tens of micrometres, which suggests that pitting is the main corrosion type. Figure 4 reveals the SEM analysis of the surface of the samples after chemically removing the corrosion products. It shows a specific ‘cauliflower-shape’ morphology, which corresponds to pitting corrosion. In addition, the thicker scales observed in the cross-section of b4 indicate its high corrosion rates.
SEM images of the cross-sections of X65 grade low carbon steel samples exposed to bentonite and thermally aged at 363.15 K for 2880 h after exposure to (a) 0 kGy h−1 and (b) 2.98 kGy h−1 γ-irradiation for 1007 h. SEM micrographs of the surface of the samples (a) 0 kGy h−1 and (b) 2.98 kGy h−1 after chemically removing the corrosion products.

Chemical composition (at.-%) of the corrosion scales on the cross-section of X65 grade low carbon steel samples exposed to bentonite and thermally aged at 363.15 K for 2880 h after exposure to 0 and 2.98 kGy h−1 γ-irradiation for 1007 h.
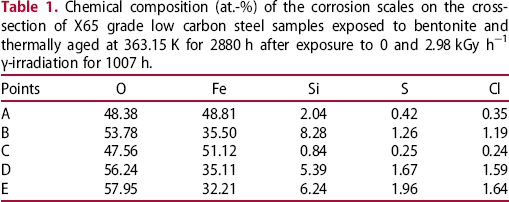
XRD spectra of the corrosion scales
Figure 5 shows the XRD spectra of the corrosion scales formed on a4 and b4. It can be seen that both patterns consist of peaks that correspond to iron, marked with solid triangles. The intensity of the iron peaks in the pattern of b4 is lower, which means that the corrosion scales formed on b4 are thicker, again confirming the accelerating function of γ-irradiation. Furthermore, both patterns also contain peaks that correspond to magnetite, marked with solid stars. In addition, the pattern of b4 comprises peaks that correspond to siderite, marked with hollow triangles. This indicates that the corrosion scales formed on carbon steel are mainly magnetite, and γ-irradiation accelerates the formation of a siderite phase.
XRD spectra of the corrosion scales formed on X65 grade low carbon steel samples exposed to bentonite and thermally aged at 363.15 K for 2880 h after exposure to 0 and 2.98 kGy h−1 γ-irradiation for 1007 h.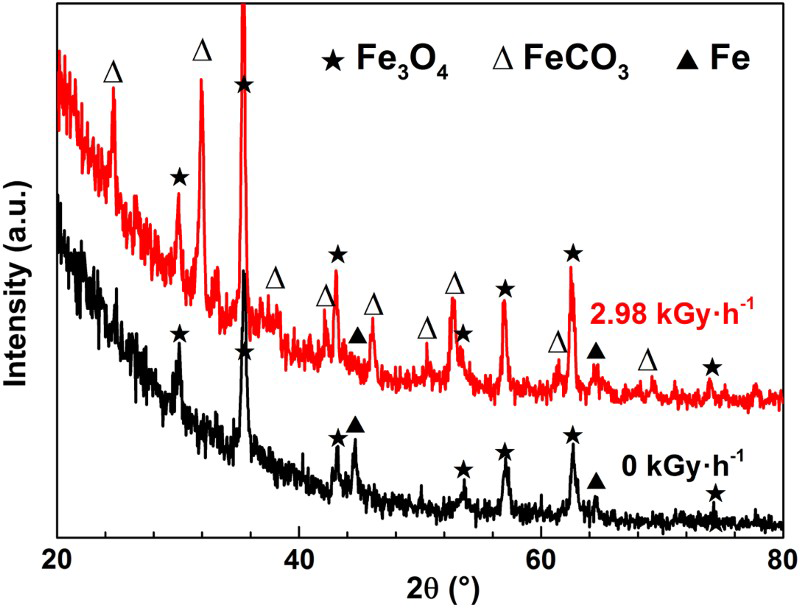
μ-Raman spectra of the corrosion scales
To confirm the distribution of magnetite and siderite, five areas, marked with A, B, C, D and E in Figure 3, were analysed using μ-Raman. Figure 6(a) displays the Raman spectrum of area A: the peaks at 229, 296, 414 and 1327 cm−1 correspond to haematite [8], and the sharp peak at 669 cm−1 corresponds to magnetite [8–10]; Figure 6(b) illustrates the Raman spectrum of area B: the peaks at 296, 393, 551 and 693 cm−1 correspond to goethite [8,9]. It means that the lighter scales on a4 are mainly composed of magnetite and haematite, while the darker scales on a4 are mainly composed of goethite.
µ-Raman spectra of the corrosion scales formed on X65 grade low carbon steel samples exposed to bentonite and thermally aged at 363.15 K for 2880 h after exposure to (a), (b) 0 kGy h−1 and (c), (d), (e) 2.98 kGy h−1 γ-irradiation for 1007 h.
Compared with haematite and magnetite, goethite contains a higher percent of oxygen ions, which may be the reason why the darker scales in the cross-section of a4 are oxygen concentrated. However, haematite and goethite were not detected in the XRD spectrum of a4. This can be explained because the XRD spectra of haematite and magnetite cannot easily be distinguished. In addition, the darker scales located at the interface between the metal and the corrosion product layer (see Figure 3), tend to be covered with the lighter scales that are several tens of micrometres thick and hard to be penetrated by X-rays.
Figure 6(c) reveals the Raman spectrum of area C: the peak at 659 cm−1 corresponds to magnetite [8–10] and the peak at 1079 cm−1 corresponds to siderite [9,10]; Figure 6(d) illustrates the Raman spectrum of area D: the peaks at 301, 397, 557 and 693 cm−1 correspond to goethite [8,9]; Figure 6(e) shows the Raman spectrum of area E: the peaks at 281, 464 and 699 cm−1 correspond to maghemite [8] and the peak at 1072 cm−1 corresponds to siderite [9,10]. This reveals that the lighter scales on b4 are mainly composed of magnetite and siderite, and the darker scales on b4 are composed of, magnetite, siderite, goethite and maghemite. Besides, the peaks at 1327 cm−1 may be all attributed to a few amount of haematite [8], as shown in Figure 6(b–e).
In conclusion, the scales on a4 are mainly composed of magnetite, haematite and goethite, and the scales on b4 are mainly composed of magnetite, siderite, maghemite and goethite. Irradiation accelerates the formation of siderite and maghemite. The formation of these phases has been shown by many researchers in simulated geological disposal conditions [10,11] and archaeological iron samples [9,12,13].
Corrosion rates
Table 2 shows the weight loss (Δm), corrosion rates (ν) and standard deviations (σ and σ′) of the weight loss samples. Irradiation accelerates the average weight loss of carbon steel from (7.0 ± 1.7) × 10−2 kg m−2 to (9.9 ± 1.5) × 10−2 kg m−2. Consequently, irradiation caused a 33% increase of the average corrosion rate from (23.72 ± 5.77) μm·year−1 (0 kGy h−1) to (31.53 ± 4.71) μm·year−1 (2.98 kGy h−1).
To investigate the corrosion accelerating function of irradiation, the surface morphology of a1 and b1 was examined after weight loss measurements by WLI, as illustrated in Figure 7, showing the three-dimensional distribution of the corrosion sites. Differences in height of the surface are illustrated by various contrasting regions with the darker regions representing the lower sites. It is clear that the fluctuations on the surface of b1 are larger than those on the surface of a1, which is consistent with the measured corrosion rates.
WLI images of the surface of X65 grade low carbon steel samples exposed to bentonite and thermally aged at 363.15 K for 2880 h after exposure to 0 and 2.98 kGy h−1 γ-irradiation for 1007 h. Weight loss and corrosion rates of X65 grade low carbon steel samples exposed to bentonite and thermally aged at 363.15 K for 2880 h after exposure to 0 kGy h−1 (a1, a2 and a3) and 2.98 kGy h−1 (b1, b2 and b3) γ-irradiation for 1007 h.

Conclusions
In this work, the corrosion behaviour of X65 grade low carbon steel exposed to Gaomiaozi bentonite containing 17 wt-% Beishan groundwater and thermally aged at 363.15 K for 2880 h after exposure to 0 and 2.98 kGy h−1 γ-irradiation for 1007 h were studied by OM, SEM, EDS, XRD, μ-Raman, weight loss measurements and WLI. The following conclusions can be made:
The corrosion scales formed on non-irradiated X65 grade low carbon steel are mainly composed of magnetite, haematite and goethite, and 2.98 kGy h−1 γ-irradiation caused the formation of two new phases: siderite and maghemite; 2.98 kGy h−1 γ-irradiation accelerates the corrosion rate of X65 grade low carbon steel by ∼33%.
Footnotes
Disclosure statement
No potential conflict of interest was reported by the authors.
