Abstract
In Japan, low-level radioactive waste including aluminium alloys is generated from nuclear power plants. It is well known that aluminium reacts with both alkaline cement and water, generating hydrogen gas. For the safe management of radioactive waste disposal facilities, it is necessary to evaluate corrosion and hydrogen generation behaviours of aluminium corresponding to changing environmental conditions. In the present study, the corrosion behaviour of aluminium in alkaline solutions at 15 °C was evaluated.
This paper is part of a supplement on the 6th International Workshop on Long-Term Prediction of Corrosion Damage in Nuclear Waste Systems.
Introduction
In Japan, low-level radioactive waste (LLW) generated in nuclear facilities is disposed of by either near-surface trench disposal, near-surface pit disposal or intermediate depth disposal [1]. Near-surface pit disposal involves the placement of waste packages in near-surface disposal facilities with engineered barriers. Solid waste, such as metal, is stored in drums and then solidified as a mixture using cement-based fillers. Aluminium remaining in the solid waste reacts with the alkaline cement or ground-water, generating hydrogen gas by the following corrosion reaction:
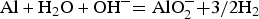
An increase in the internal pressure caused by hydrogen gas generation may affect the performance of engineered barriers. For the safe management of radioactive waste disposal facilities, it is necessary to consider the corrosion and hydrogen generation behaviour corresponding to the changes in the environmental conditions [2].
Previous research has suggested that the corrosion of aluminium accompanied by H2 gas evolution is reduced after a month due to the protective effect of corrosion products such as gibbsite and calcium aluminate hydrate [3–5]. In the early stage of disposal, surface of aluminium will be covered with corrosion products that were formed during the manufacture of the waste packages. At this stage, the inside of the waste packages will be dry. In this state, aluminium will not react with alkaline water to generate H2 gas. However, the corrosion inhibition mechanism of the corrosion products has not been sufficiently studied in wet conditions, which could arise due to the penetration of ground-water into the waste packages. Due to the likelihood of these waste packages coming in contact with ground-water, we propose that the stability of corrosion products should be investigated in wet condition. In the present study, the corrosion behaviour of aluminium covered with corrosion products was evaluated in alkaline solutions at 288 K (15 °C).
Experimental
Composition of equilibrium solutions of OPC (g m−3).

Figure 1 shows the test procedure. The specimens were immersed in equilibrium solution at 288 K for about 3000 h under reducing conditions (Run 0) to form a corrosion product (or film) on the surface. The specimens were ultrasonically cleaned in acetone after Run 0, and were dried in vacuum before the subsequent immersion test at 288 K (Runs 1a–3a).
(a) Assumed environmental conditions in this study and (b) procedure of immersion test.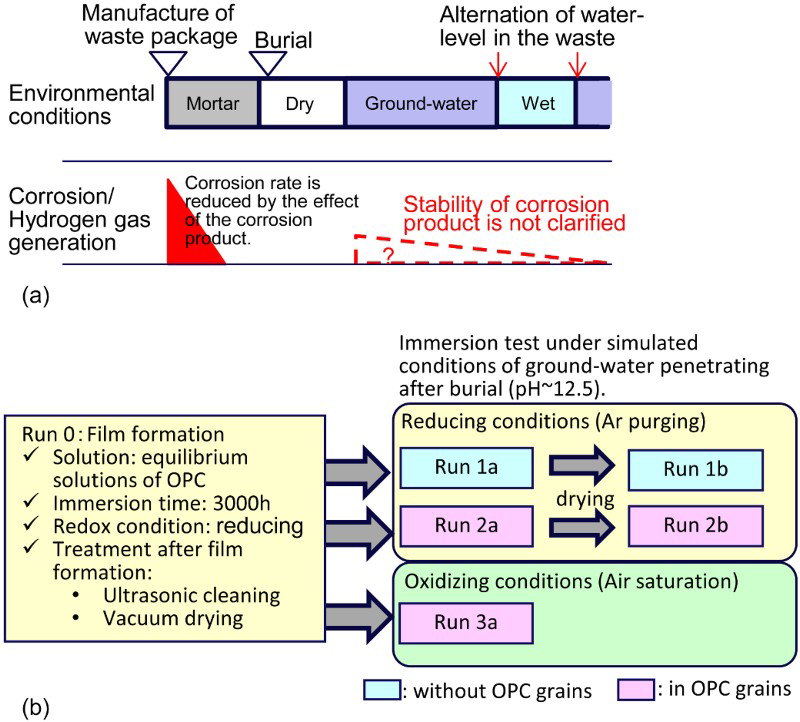
Figure 2(a) shows the apparatus used for the immersion test. In Runs 1a and 1b, specimens were installed in a glass flask with a perforated beaker. The immersion tests were carried out without OPC grains. By contrast, Runs 2a, 2b and 3a were carried out with the specimens embedded in OPC grains as shown in Figure 2(b). The drying treatment after Run 2a was carried out while keeping the specimens embedded in the OPC grains.
(a) Apparatus used for immersion test and (b) specimen embedded in OPC grains.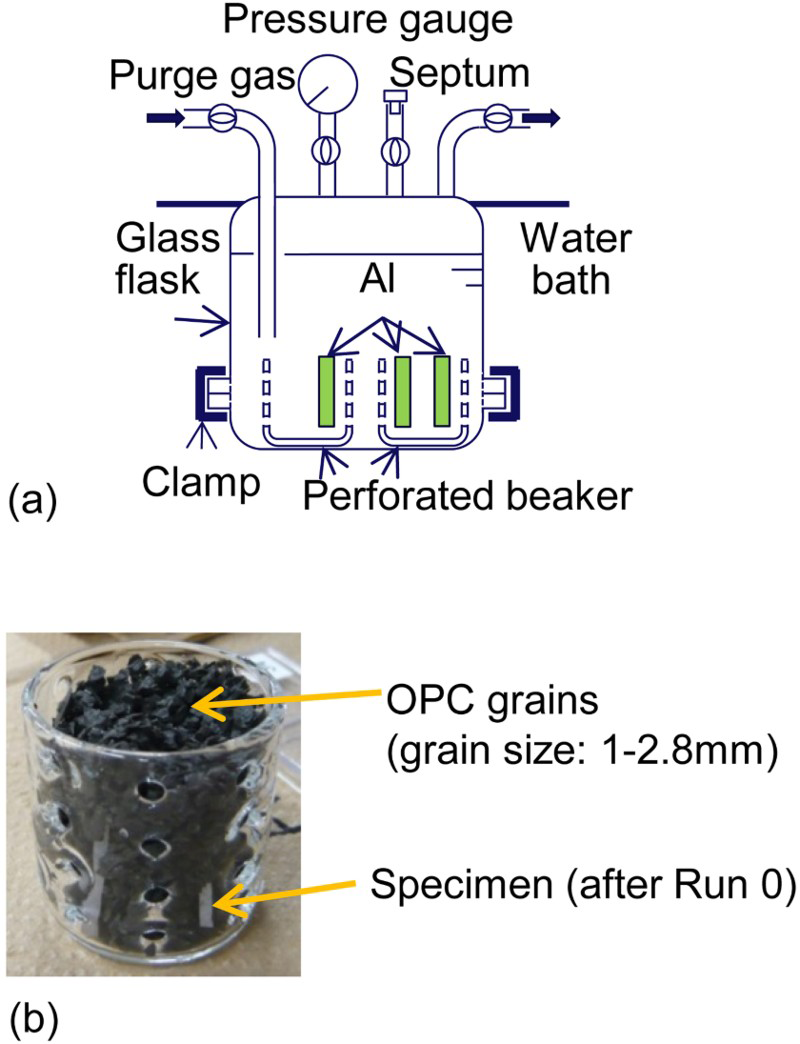
During each immersion test, the glass flask was sealed periodically and the concentration of hydrogen gas in the vapour phase was measured using gas chromatography. The corrosion rate of the specimen was calculated using the following equations:
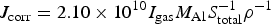
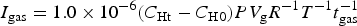
Here, J corr is the corrosion rate (mm/y). I gas is the outgassing rate of hydrogen gas (mol s−1). S total is the surface area of the specimens (m2). ρ is the density of Al (g m−3). M Al is the atomic weight of Al (g mol−1). T, P and V g are the temperature (K), pressure (Pa) and volume (m3) of the vapour phase, respectively. C H0 and C Ht is the hydrogen concentration (ppm) in the vapour before and after the collection of hydrogen. t gas is the time (s) of collection. R is the gas constant.
The composition of the corrosion products formed on the aluminium specimen during the immersion test Run 2a was analysed using scanning electron microscopy (SEM), energy dispersive X-ray spectrometry (EDX), and Fourier transform infra-red spectroscopy (FT-IR). In order to obtain the cross-sectional structure of the corrosion products, the specimens were dried following the immersion tests and embedded in epoxy resin without being removed from the OPC grain.
Results and discussion
Corrosion behaviour in equilibrium solutions under reducing conditions
Figure 3 shows the corrosion behaviour of the specimens in the equilibrium solution under reducing conditions. In Run 0, although the corrosion rate of aluminium was initially on the order of mm/y, it decreased with time to 1.0 × 10−6 m/y or less. This behaviour is consistent with previous findings [2–4]. When the specimens were reimmersed after Run 0, a maximum corrosion rate of more than 1.0 × 10−4 m/y occurred in the early stages (Run 1a). To determine the cause of this behaviour, following Run 1a, the specimens were treated via three different drying methods; ultrasonic cleaning followed by vacuum drying, vacuum drying alone, and desiccator storage (atmospheric drying). During the subsequent immersion tests (Run 1b), the maximum corrosion rates were observed for all three drying methods. It was predicted that the drying itself would affect the properties of the surface film on aluminium.
Corrosion behaviour of specimens in the equilibrium solutions under reducing conditions, (a) Run 0, (b) Run 1a and (c) Run 1b.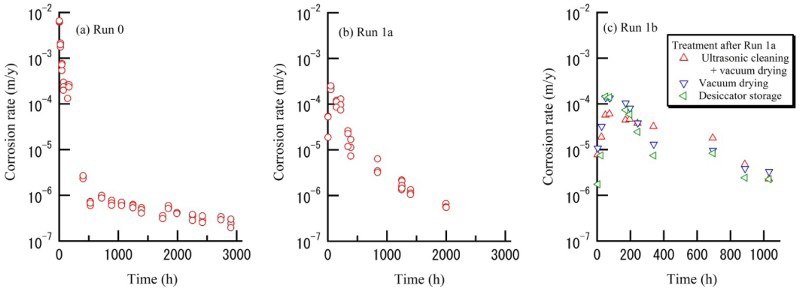
Corrosion behaviour in OPC grains under reducing and oxidising conditions
Following Run 0 the specimens were embedded in OPC grains, and the immersion tests, denoted by Runs 2a, 2b and 3a, were carried out to determine the effect of drying conditions in an environment similar to that in which aluminium-containing low-level waste is stored. Figure 4(a,b) shows the corrosion behaviour of specimens during Runs 2a and 2b, respectively. During Run 2a, the first immersion test carried out under embedding conditions, the maximum corrosion rate was observed in the early stages then decreased to less than 1.0 × 10−6 m/y after 8000 h. It is noteworthy that the maximum corrosion rate was not observed during the subsequent test (Run 2b). OPC grains are proposed to have inhibited the degradation of the film caused by drying.
Corrosion behaviour of specimens in OPC grains under reducing conditions, (a) Run 2a, (b) Run 2b. (c) Comparison between behaviour under oxidising conditions (Run 3a) and reducing conditions (Run 2a).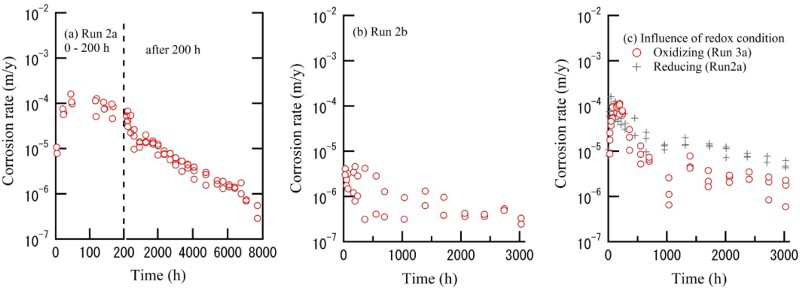
Figure 4(c) shows a comparison of the corrosion behaviour between the oxidising conditions (Run 3a) and reducing conditions (Run 2a). It was determined that the redox condition has little effect on the corrosion behaviour.
Corrosion products
Figure 5(a) shows a cross-section SEM image of the corrosion products (film) formed on a specimen after Run 2a. It was confirmed that the corrosion products consist of a two-layer film. Figure 5(b,c) shows the composition of the layers obtained by EDX and FT-IR spectra, respectively. The inner layer consists mainly of aluminium hydroxides and the outer layer consists of Ca- and Al-containing oxides and hydroxides. The thickness of the inner layer is about 5 × 10−5 m which is much smaller than the outer layer (about 3 × 10−4 m). Many cracks were observed in both the inner and outer layers. Although these cracks may have formed at the resin embedding stage in the specimen which were visualised using SEM, it is known that cracks arises relatively readily in these films.
Cross-sectional analysis of corrosion products (film) formed on specimen after Run 2a. (a) SEM image, (b) composition, (c) FT-IR spectra.
Proposed corrosion inhibition mechanism
When a bare aluminium alloy was immersed in the equilibrium solution, the corrosion rate decreased with time and the formation of a surface film was observed. This is in agreement with the studies by Hironaga and Mahara [3], Walton et al. [4] and Takatani et al. [5]. It is reasonable to conclude that the surface film inhibits the corrosion of aluminium in an alkaline environment. It is hypothesised that Ca in a solution influences the corrosion behaviour of Al. The results of cross-sectional analysis revealed the existence of film with inner and outer layers. Run 1b showed that the method of drying the specimen would affect the properties of the inner and/or outer layer. X-ray diffraction (XRD) patterns of the specimens showed that hydroxides, such as gibbsite (γ-Al(OH)3) and bayerite (α-Al(OH)3), existed in the inner layer. The hypothesis that a protective effect is caused by the crystalline oxide alone cannot explain the influence of the drying which was observed in this study. Therefore, it is proposed that an unidentified compound with low crystallinity, e.g. aluminium oxyhydroxide, exists within both the inner and outer layer.
The present study also revealed that using drying treatment after film formation resulted in the maximum corrosion rate and that the presence of OPC grains mitigated the degradation of the film. Figure 6 shows the proposed corrosion mechanism. The corrosion reaction proceeds through cracks in the inner and outer layer. The corrosion rate decreases gradually with the growth of the both layer. It is hypothesised that the growth of the inner layer was induced by the formation of the outer layer. If the inner and/or outer layer is dried, the cracks will expand due to the shrinkage of hydroxide or oxyhydroxide particles and the corrosion rate will increase in the early stage of re-immersion. When the specimens were embedded in OPC grains, it is proposed that the surface film did not dry completely due to the moisture from the OPC grains and consequently the expansion of the cracks was reduced.
Proposed corrosion mechanism of aluminium in mortar solution.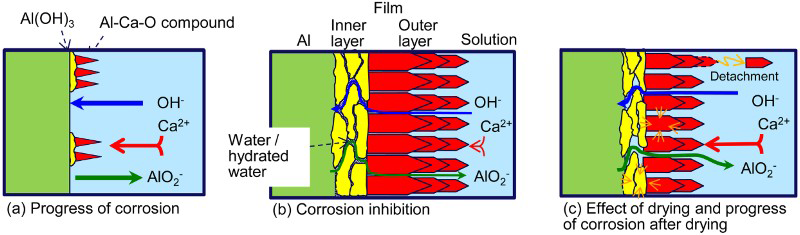
Conclusion
The corrosion behaviour of aluminium was evaluated in conditions that mirror the environment after ground-water permeates into nuclear waste packages. The following results were obtained.
Although a maximum corrosion rate of more than 1.0 × 10−4 m/y was observed for specimens after drying, the corrosion rate decreased to 1.0 × 10−6 m/y or less after 8000 h. A surface film, which was found to have a two-layer structure, was formed on the specimens. The corrosion behaviour of aluminium will be affected by the stability of the film.
Footnotes
Acknowledgements
The authors are grateful to Mr Naoji Mitani (Hokuriku Electric Power Company), Mr Takayuki Koashi (Japan Atomic Power Company) and Mr Mamoru Kumagai (Japan Nuclear Fuel Limited) for their help in the research program. We particularly acknowledge the help of Dr Keiji Watanabe (Central Research Institute of Electric Power Industry) and Dr Hideaki Takahashi (Professor Emeritus, Hokkaido University), who provided experimental knowledge used in these studies.
Disclosure statement
No potential conflict of interest was reported by the authors.
