Abstract
This study aims to investigate the mechanical performance and bioactive potential of polyetheretherketone (PEEK)-based composites. PEEK powder is mixed with up to 20 wt-% zinc doped nano-hydroxyapatite (ZnHA) powder to extrude filaments. Subsequently, 3D-printed samples are prepared via fused deposition modelling (FDM). Micro-Computed Tomography (µ-CT) analysis shows the distribution of ZnHA particles in extruded filaments. Fourier transform infrared spectroscopy (FTIR) confirms the functional groups in 3D-printed samples. The tensile testing results showed that at higher concentration of ZnHA, the strength and strain at failure decrease, while the elastic modulus increases within the acceptable range of human cortical bone. Bioactivity tests in simulated body fluid (SBF) for up to 14 days illustrate the formation of apatite precipitates on the composite samples’ surface compared to pure PEEK. Based on the results, it can be concluded that FDM 3D-printing of PEEK/ZnHA composites can be used effectively in manufacturing customised craniofacial/orthopedic implants.
Keywords
Introduction
In the past few decades, a wide range of biomaterials have been tested for orthopedic applications. The research has been focused on the mechanical properties and biocompatibility of different material systems such as metals, ceramics, polymers and composites [1]. Bioactive ceramics such as hydroxyapatite (HA) is considered as a good choice because of its spontaneous integration with natural bone by rapidly making a bone-like apatite on its surface. However, due to their high modulus and low fracture toughness, alone they have limited usage as a biomaterial [2]. It is often used to make polymer-ceramic composites which have better properties than metals, polymers or ceramics alone [3]. Recently, polyetheretherketone (PEEK) has attracted researchers as a load-bearing material for orthopedic applications due to its excellent properties such as biocompatibility, mechanical strength, chemically inert, non-toxicity, radiolucency and stability in sterilisation process. However, the only major drawback associated with PEEK is its lack of osteointegration ability with surrounding tissue [4-6].
Extensive research has been conducted to enhance the bioactivity of PEEK. Primarily, HA or doped HA has been used for that purpose in the form of either coating or as a filler material [7-10]. Like other minerals present in the body, zinc is an essential mineral that is required for normal skeletal growth and several cellular processes in the body [11]. It also enhances the bone remineralisation process by activating various enzymes in the body. It has been reported that zinc doped hydroxyapatite (ZnHA) stimulates bone formation by promoting osteoblast activity and inhibiting osteoclast activity [12]. Additionally, HA does not inhibit bacteria from adhering to its surface which can lead to infection/inflammation and can cause surgical operation failure after the implantation process due to the infection. Since, ZnHA has broadly demonstrated antibacterial properties and thus reduces the risk of infection or inflammation after implantation [13].
Previously, PEEK and its composites have been processed by conventional technologies such as injection moulding, extrusion and so on [14-18]. These technologies inherit some limitations such as expensive, lack of customisation and complexity, etc, [19]. On the other hand, additive manufacturing also known as 3D printing is a recent technology that can produce complex parts with customisation [20, 21]. Fused deposition modelling (FDM) and selective laser sintering (SLS) are its common types. In comparison, a molten polymer is deposited in layer after layer to produce FDM 3D printed object while high-energy laser beam is used in SLS to sinter consecutive layers of powder and produce final object. In a few aspects, FDM has clear advantages over SLS such as lower capital cost, lower production cost and high usage of materials [22]. In order to reduce the risk of warping in SLS, both the part and unsintered powder are kept at high temperature which causes thermal degradation and makes it unsuitable for reusage [23]. However, in the FDM process there is almost negligible wastage of material in either producing filaments or 3D printing.
Hence, in this study, PEEK/ZnHA nanocomposite filaments are extruded and then 3D-printed via fused deposition modelling (FDM). The samples are characterised by scanning electron microscopy (SEM), energy dispersive X-ray (EDX) spectroscopy, Fourier Transform infrared (FTIR) spectroscopy, µ-CT analysis, mechanical testing and bioactivity testing in simulated body fluid (SBF).
Materials and methods
Extrusion and 3D printing temperatures.

The surface morphologies of ZnHA nanoparticles, PEEK powder and 3D printed samples were analysed by SEM (HITACHI Japan) and compositional analysis was performed via energy dispersive X-ray (EDX; Oxford Instruments) equipped with SEM. The internal structures of the filaments were determined by micro-computed tomography (µ-CT; Microtomograph SkyScan 1275 Bruker, USA) with source voltage 40 kV, source current 25 µA and pixel size 10 µm. FTIR spectra of the samples were collected with scan range 550–4000 cm−1, at resolution 8 cm−1 and 128 total scans by Nicolet iD5 instrument in Attenuated Total Reflection (ATR) mode. The samples for tensile testing were prepared according to ISO 527–2 type VA and tested with crosshead speed 5 mm min–1 with load cell 5 kN on INSTRON equipment.
Bioactivities of the samples were tested by simulated body fluid (SBF) immersion test. The SBF was prepared according to Kokubo's method [25]. After preparation, the samples were immersed in simulated body fluid (SBF) for 7 and 14 days to analyse their bioactivities. Following immersion, the top surfaces of dried samples were observed under SEM for apatite layer formation.
Results and discussion
The morphology and particle size of ZnHA and PEEK powders were observed via SEM, as shown in Figure 1. It was observed that the particle size of ZnHA and PEEK were in the range of 30–80 nm and 1–2 micrometres, respectively. Furthermore, it was observed that the few ZnHA particles were fused due to sintering at 900°C. EDX analysis of PEEK and ZnHA powders showed that there was not any major impurity present, and it also confirmed the presence of zinc along with calcium, phosphorous and oxygen in ZnHA powder while only carbon and oxygen were detected in PEEK.
SEM micrographs of (A) PEEK powder, (B) EDX spectrum of PEEK powder, (C) ZnHA nanoparticles synthesised by wet chemical precipitataion method and sintered at 900°C and (D) EDX spectrum of ZnHA powder.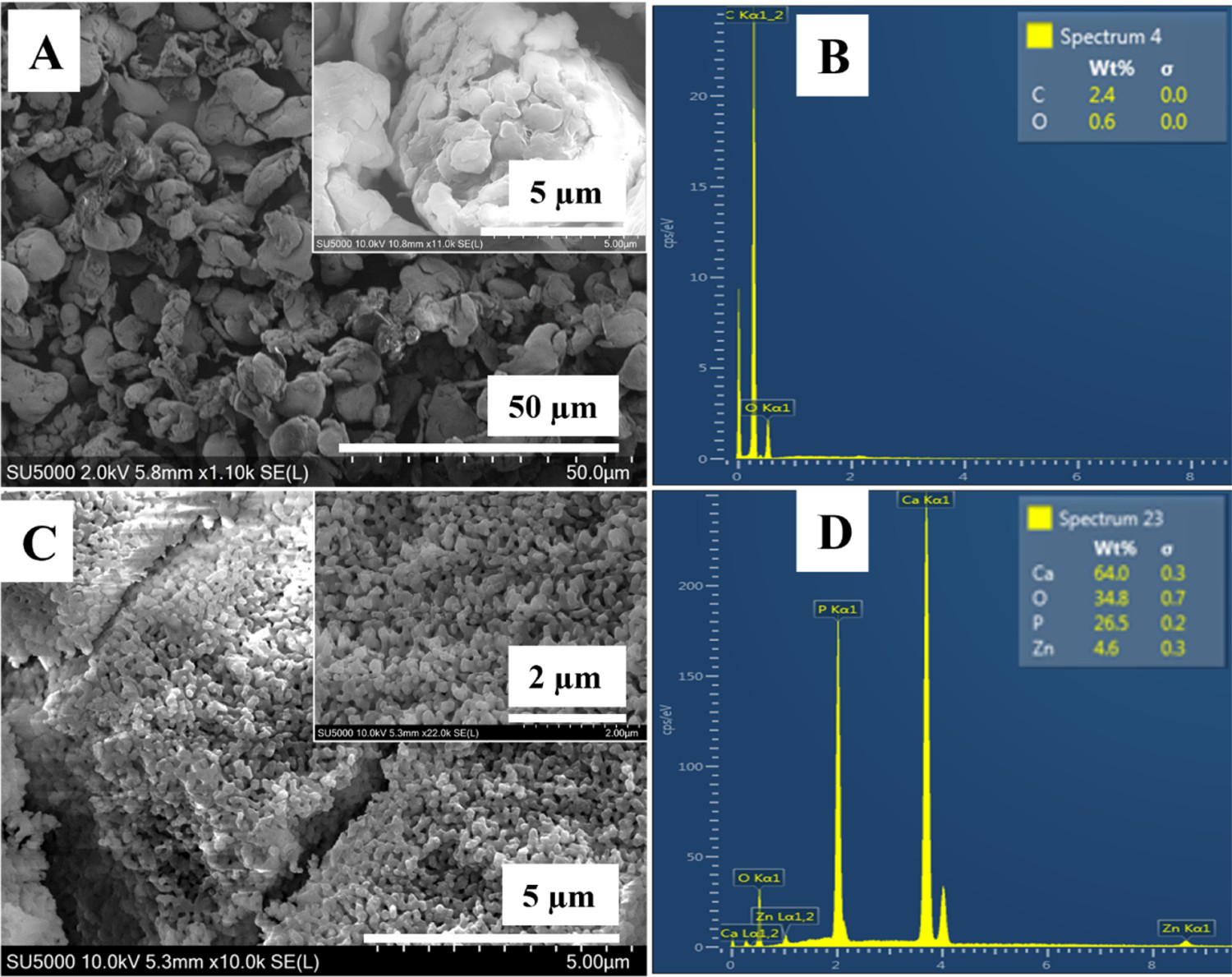
The ZnHA particle distribution in the filaments was examined via µ-CT. It was noticed that the ZnHA particles were homogeneously dispersed in the PEEK matrix. The tiny dots in Figure 2 are ZnHA particles that are dispersed throughout the PEEK matrix. However, for pure PEEK, the surface was smooth which showed the absence of particles, as expected.
Micro-CT analysis of filaments (A) PEEK, (B) PEEK/10ZnHA and (C) PEEK/20ZnHA.
SEM analysis was used to observe the surface structure and the dispersion of ZnHA particles in 3D printed samples as shown in Figure 3. It was noted that the ZnHA particles were distributed evenly without significant agglomeration and there were not any significant pores or gaps present on the surfaces of 3D printed samples. The surface of PEEK becomes bioactive due to the presence of these particles which also act as bioactive sites and attract biomolecules. Hence, makes PEEK a bioactive material [26].
SEM micrographs of 3D printed samples showing the distribution of ZnHA particles on top surfaces and their compositional analysis by EDX: (A) PEEK, (B) PEEK/10ZnHA and (C) PEEK/20ZnHA.
FTIR spectra of synthesised ZnHA powder, PEEK powder and their 3D printed nanocomposites are shown in Figure 4. The peaks at 1205 and 905 cm−1 could be due to the stretching vibration of the P-O bond in ZnHA. The peaks observed between 1020 and 1090 cm−1 ( FTIR spectrum of ZnHA powder, PEEK powder and their 3D printed nanocomposites taken in ATR mode.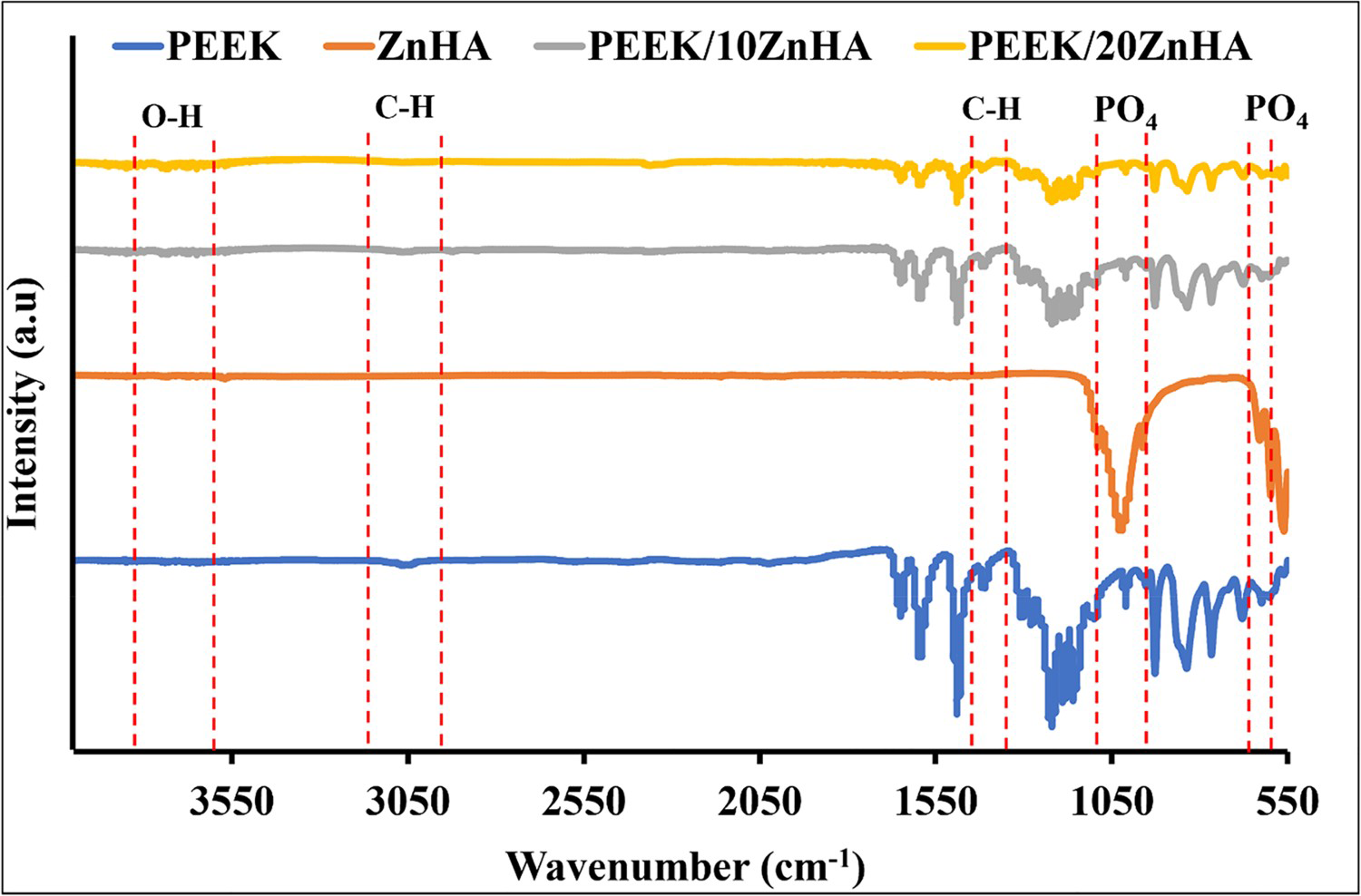
Figure 5 shows the graphs of tensile strength and Young's modulus of PEEK/ZnHA samples. The strength of pure PEEK was obtained as ∼75 MPa. The tensile strengths of the samples were decreased for 10 and 20 wt-% ZnHA by 12% and 19%, respectively. These strengths of the samples were still in the range of the strength of human cortical bone i.e. 50–150 MPa [34]. The decrease of tensile strength with the incorporation of ZnHA particles was due to the weak interaction between PEEK and ZnHA. When the load applied, the PEEK and ZnHA particles detached and produced a crack which led to the failure of the sample early as expected. However, the modulus increased in the presence of ZnHA particles, this was because the modulus of ZnHA was quite high as compared to the PEEK. Hence, the addition of ZnHA resulted in the higher modulus as compared to pure PEEK samples. Modulus of pure PEEK was measured as 2.8 GPa which was increased to 3.7 GPa and 4.2 GPa for 10 and 20 wt-% ZnHA, respectively. Hence, the samples produced by FDM 3D printing process suggests that the samples have adequate mechanical properties so that they can be used to manufacture patient-specific orthopedic implant.
The graph shows the tensile strengths and moduli of PEEK and its nano-composites and 3D printed samples according to ISO 527-2 for tensile testing.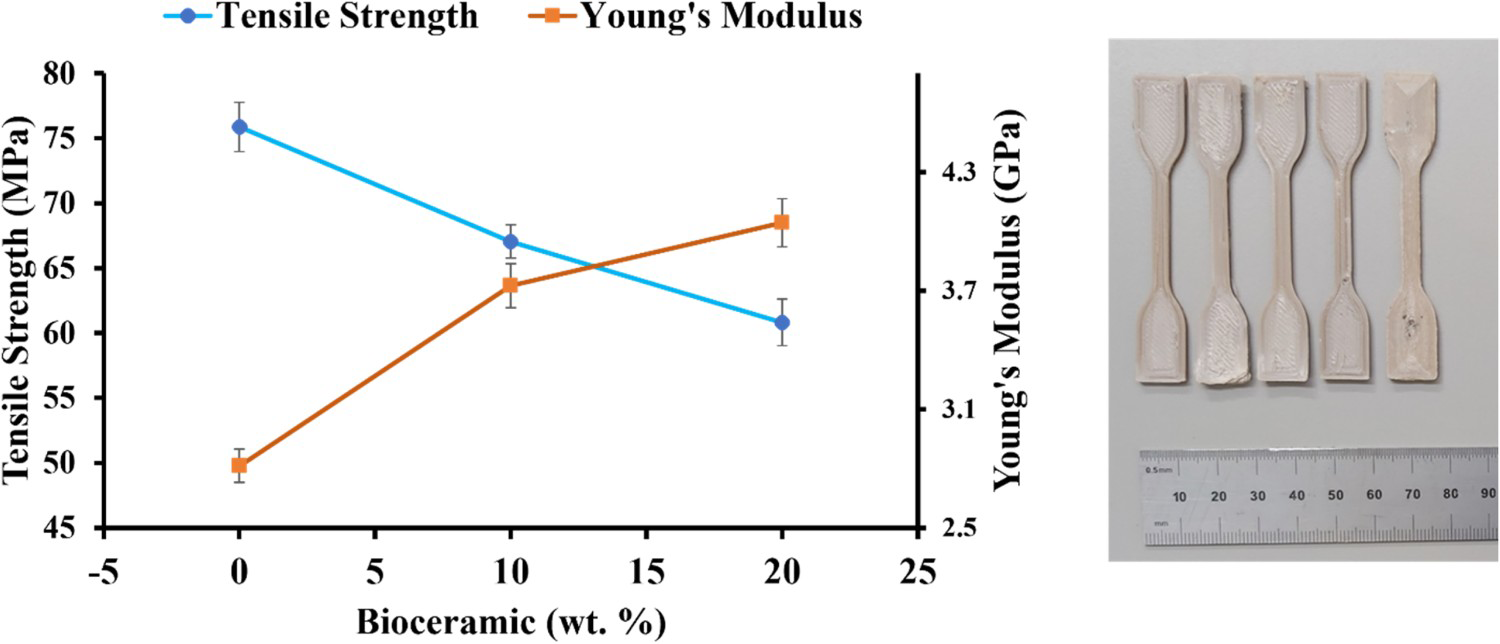
The SEM micrographs of the samples after immersion in SBF for 7 and 14 days were shown in Figure 6. The surface of pure PEEK 3D printed samples had not shown any apatite layer formation after 7 and 14 days of immersion. This confirmed the bioinert nature of the PEEK [14]. However, the apatite layer formation with characteristic morphology was observed on the samples with 10 and 20 wt-% ZnHA. It was observed that the quantity of apatite layer formation on the surface was dependent upon the immersion time and the amount of ZnHA particles in PEEK [35]. The apatite layer formation started even after 7 days of immersion. As the immersion time increased up to 14 days, the amount of apatite formation on the 3D printed surfaces of the samples also increased. Similarly, the samples containing 20 wt-% of ZnHA have shown more characteristic apatite particles on its surface as compared to the PEEK composite containing 10 wt-% ZnHA. This fact was observed after 7 and 14 days of immersion. These results showed that the bioactivity of the PEEK can be increased by the incorporation of ZnHA particles. Hence, the amount of particles can be carefully selected to obtain balance (optimum) between mechanical properties and bioactivity.
SEM micrographs of bioactivity tests showed the apatite layer formation of 3D printed samples on their surfaces after 7 and 14 days of immersion in SBF.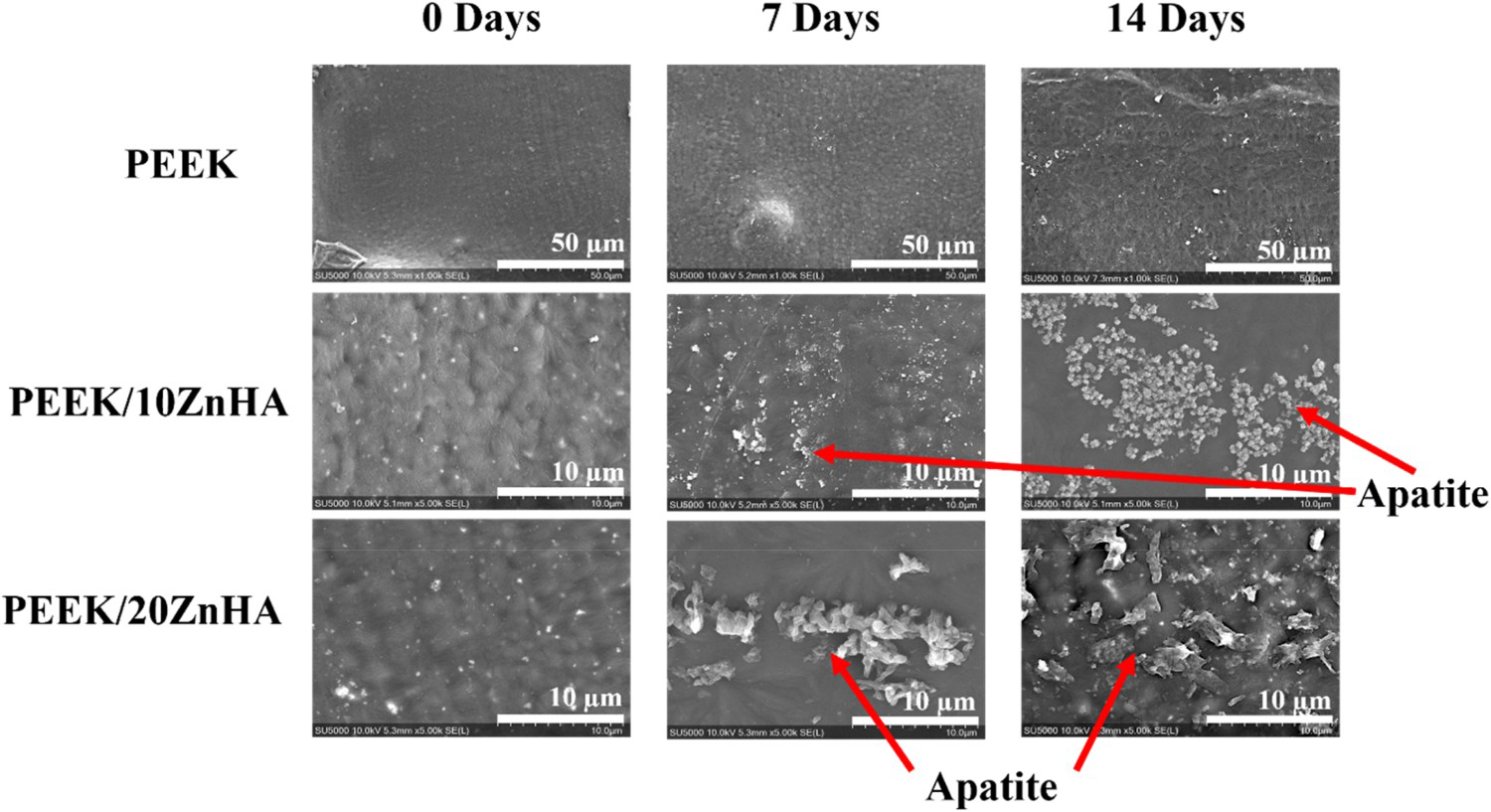
Conclusion
In this study, bioactive nanocomposites of PEEK with ZnHA were prepared with 0, 10 and 20 wt-% via FDM 3D printing. Raw materials such as PEEK and ZnHA powders were characterised and extruded into the filaments after physical mixing. µ-CT analysis and SEM analysis showed the uniform distribution of ZnHA particles in the extruded filament and on the surfaces of 3D printed samples, respectively. FTIR and EDX spectra showed that there was not any major impurity present after the processing of PEEK/ZnHA composites. In mechanical testing, it was observed that the tensile testing of the samples decreased while modulus increased but still the properties were in the range of cortical bone. SBF immersion test confirmed that the bioactivity of the PEEK increased due to the presence of ZnHA particles. It was observed that the apatite layer formation on the surface was enhanced as the immersion time increased and the amount of ZnHA particles increased. Hence, in the light of above results, PEEK/ZnHA nanocomposites have a great potential to be used as a material for manufacturing personalised orthopedic implants by FDM 3D printing technology.
Footnotes
Acknowledgements
The North-West Centre for Advanced Manufacturing (NW CAM) project is supported by the European Union's INTERREG VA Programme, managed by the Special EU Programmes Body (SEUPB). The views and opinions in this document do not necessarily reflect those of the European Commission or the Special EU Programmes Body (SEUPB).
Disclosure statement
No potential conflict of interest was reported by the author(s).
