Abstract
Electrophoretic deposition (EPD) is a powerful technique to assemble carbon nanotube (CNT) coatings and composite films with controlled architectures. This comprehensive review of the EPD of CNTs and CNT-containing composites focuses on achievements within the last 15 years and ongoing challenges. Stable CNT suspensions are a pre-requisite for successful EPD and have been prepared by a variety of strategies, discussed here. The resulting film microstructure is determined by the initial feedstock, the suspension, and the EPD approach applied, as well as a variety of EPD processing parameters. Nanocomposites can be prepared via co-deposition, sequential deposition, or post-deposition treatments, to introduce metallic, ceramic or polymeric phases. There are numerous potential applications for both homogeneous and patterned CNT films, including as structural reinforcements for composites, as field emission, energy storage and conversion devices, as well as in biomedical applications. The advantages and disadvantages of EPD processing in these contexts are discussed.
Introduction
For nearly three decades now, carbon nanotubes (CNTs) have attracted widespread scientific and technological interest. Formally, they are cylindrical allotropes of carbon either consisting of a single layer (single-walled CNTs, SWCNTs) or multiple concentrically arranged layers of graphene (multi-walled CNTs, MWCNTs). Along with a high aspect ratio and surface area, high-quality CNTs exhibit remarkable mechanical properties, high thermal and electrical conductivities and non-linear optical properties, which makes them attractive materials for a wide range of applications [1–6]. However, one of the challenges to practical implementation is the difficulty of manipulating and assembling individual CNTs to form constructs with an organised architecture. Today’s products mostly rely on poorly organised random or random planar arrangements, with significantly lower bulk properties than the individual constituent CNTs [7]. Depending on the intended use, controlled CNT networks or composites may be required over large areas, including on convoluted surfaces, or over large volumes, as 3D hierarchical structures [2,8]. Electrophoretic deposition (EPD) is a cost-effective method to manipulate CNTs for the ordered deposition of CNT coatings/films on flat or complex surfaces, as well as for shaping of bulk objects. As described in our previous review in 2006 [9], this simple and versatile colloidal processing technique enables the preparation of homogeneous microstructures with accurate control over coating thickness and CNT orientation, depending on the process parameters, and offers the possibility of upscaling to larger application-relevant dimensions. As a result, the EPD of CNTs, often in combination with other nanoparticles or molecules, has been widely investigated in recent years, as evidenced by a steady increase in publications each year, and associated citations. Developments have addressed a variety of applications, including carbon fibre-reinforced composite materials, field emission displays, catalyst supports, supercapacitors, solar cells and solid oxide fuel cells, hydrogen storage electrodes, biosensors and stress sensors, bioactive tissue scaffolds and other functional composites [8–17]. Here, we present an update on the EPD of CNTs, focussing on studies that have appeared in approximately the last 15 years, since our previous review. The Electrophoretic deposition of carbon nanotubes section addresses the fundamentals of EPD, including the preparation of stable CNT suspensions, as well as the variety of different EPD strategies and post-treatments. The EPD of CNT-based nanocomposites section considers the preparation of composite coatings combining CNTs with other components (metallic and ceramic particles, polymeric molecules). Finally, emerging applications of CNT-based materials prepared by EPD are highlighted in the Applications of electrophoretically deposited CNT films section.
Electrophoretic deposition of carbon nanotubes
General principle
EPD is essentially a two-step process during which colloidal charged particles or molecules in suspension migrate towards an oppositely charged electrode under the influence of an externally applied electric field (electrophoresis), followed by particle/molecule accumulation and coagulation to establish a compact deposit at the electrode surface (deposition) [18–22]. Depending on the surface charge of the particles in suspension, deposits can be obtained at the anode (anodic EPD) or at the cathode (cathodic EPD). In its simplest form, the EPD set-up consists of a power supply connected to two parallel electrodes, a working electrode and a counter electrode, immersed in a particle suspension (shown schematically in Figure 1); the process can also be applied on complex shape substrates, including porous substrates for infiltration purposes [20,23]. Conventional EPD, often from organic solvents, applies electric field generated by direct current (DC); more recently, modulated electric fields, generated, for example, by pulsed direct current (PDC) or alternating current (AC), have been considered, for their better compatibility with aqueous suspensions (see the Deposition of carbon nanotubes section) [24]. Schematic illustration of the anodic EPD process of carbon nanotubes. Electrodes are placed vertically in the deposition cell containing a (here negatively) charged carbon nanotube suspension. Under the influence of an externally applied electric field, the charged CNTs migrate towards the electrode of the opposite charge (here anode) accumulating to form a coherent deposit.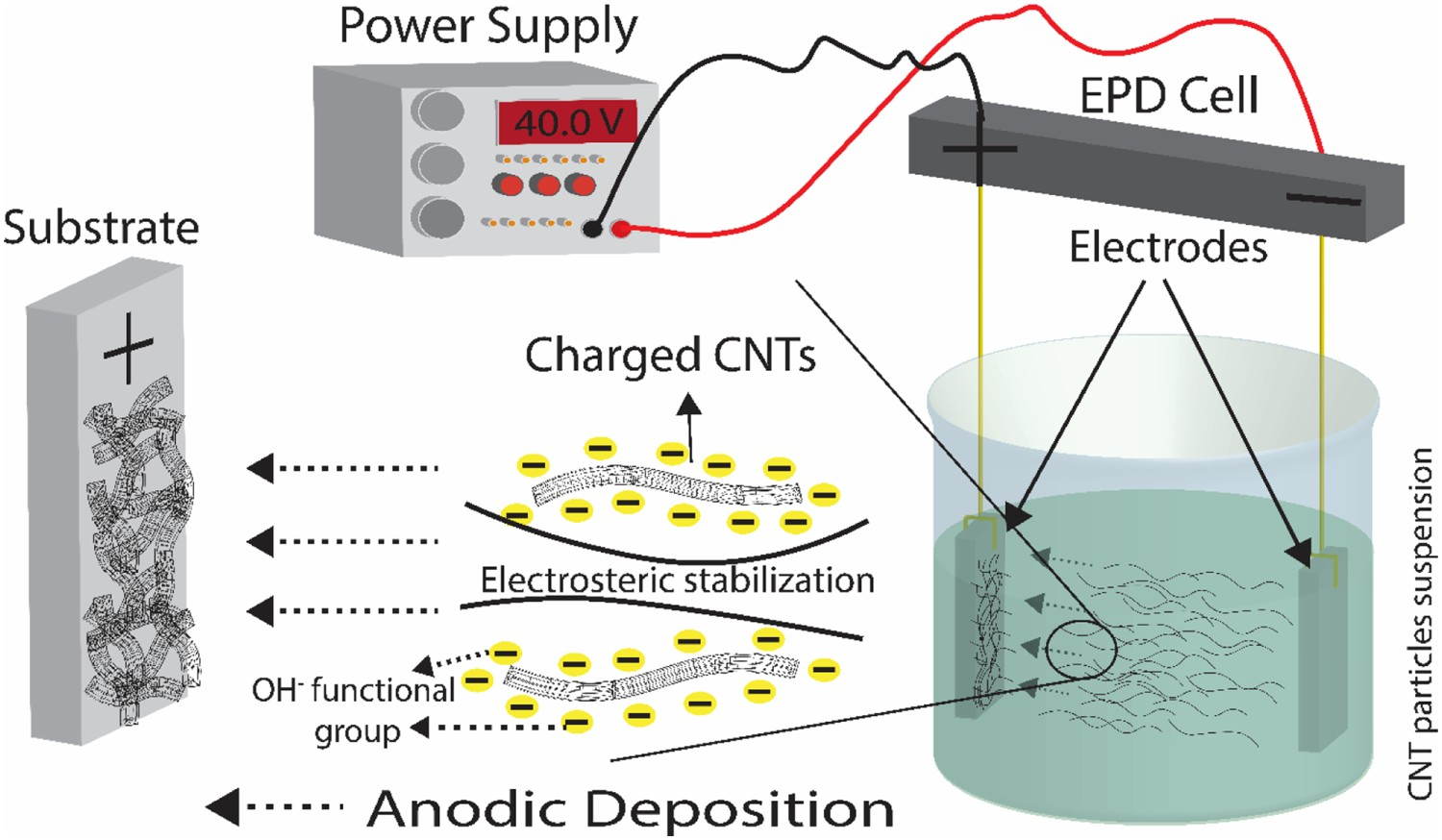
A stable suspension of charged particles is a pre-requisite for successful EPD. In principle, EPD can utilise any particulate solid (metals, ceramics, polymers as well as their composites) or biological entity (cells, proteins, enzymes, etc.), as long as the particles can be stably suspended and develop an appreciable surface charge in contact with the solvent. The charge helps to prevent sedimentation due to agglomeration and to facilitate a high electrophoretic mobility, µ, under influence of the applied electric field [19,23]. Suspension stability is often described by the classical Derjaguin–Landau–Verwey–Overbeek (DLVO) theory [25], explaining that the electrical double layer, the ionic cloud which forms around colloidal particles as a result of the surface charge, leads to electrostatic repulsive forces counteracting the attractive interparticle forces (typically van der Waals forces in the case of CNTs). In addition, besides this electrostatic stabilisation, electrosteric stabilisation can be obtained by grafting or adsorbing larger chemical molecules (bulky polymers, surfactants, etc.) on the particle surface, introducing steric repulsion between the approaching particles [26].
The stability of an electrostatically stabilised colloidal suspension is indicated by the zeta potential (ζ) as a measure of the extent of the repulsive surface charge. A high (absolute value of the) zeta potential is required to avoid coagulation (greater than ±30 mV as a rough guideline). The zeta potential also determines the direction (anodic vs. cathodic EPD) and velocity of particle migration during EPD; a high zeta potential also promotes electrophoretic mobility [19,27–29]. Appropriate EPD solvent media can be organic, aqueous or a combination depending on the targeted particles and the intended application, but should enable a high (absolute value of the) zeta potential, which can be enhanced by varying the ionic strength of the solution, pH, concentration of particles in suspension as well as particle size, shape and chemistry [20,21,23]. However, the ionic conductivity of the suspension should be kept low, as high values reduce the electrophoretic mobility [28], and may lead to undesirable side reactions (bubble formation and oxidation of the anode due to water electrolysis).
In order to improve deposition process control, many attempts have been made to describe the underlying mechanisms of EPD [19]. While the electrophoresis stage is well understood, the exact mechanism of particle aggregation at the electrode surface is still under debate; currently, the double-layer distortion model is most accepted [18,20]. Regardless of the detailed mechanism, the deposition yield can be correlated with different deposition parameters, to develop useful mathematical expressions for EPD kinetics [18,30,31]. These approaches are largely based on Hamaker’s law relating the deposit yield (w) to the electrophoretic mobility (μ) and mass concentration (C) of the particles in suspension, as well as the electric field strength (E), electrode surface area (A) and deposition time (t) [19,22,32]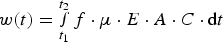
While deposition rate is important, most applications require a ‘high quality’ deposit, usually indicated by a homogeneous microstructure and high packing density. Here, again, the zeta potential plays a crucial role, as particle agglomeration in unstable suspensions will lead to a low deposit density [18,19]. Nanometric particles tend to easily re-agglomerate because of their large specific area, which may be detrimental for the quality of the coatings [29]. Excessive particle accumulation rate at the deposition electrode, for example, at high electric field strength, may also limit deposit density, since fast-moving particles do not have sufficient opportunity to rearrange to more an optimal packing [19,29]. Moreover, at high voltages, especially in aqueous solvents, electrolytic reactions can lead to gas bubble formation, disrupting the deposited film [32].
Overview of key EPD parameters used for CNTs.
SWCNT, single-walled CNT; MWCNT, multi-walled CNT; HA, hydroxyapatite; BG, bioactive glass; PAA, polyacrylic acid; EtOH, ethanol; IPA, isopropanol; DMF, dimethylformamide; ITO, indium tin oxide; FTO, fluorine-doped tin oxide.
aA detailed list is given in the supporting information (Tables S1–S4).
Preparation of carbon nanotube suspensions
CNTs can vary significantly in diameter (typically ∼0.5 to several nanometres for SWCNTs and ∼10–30 and even 100 nm for MWCNTs), length (∼0.02–20 µm or even longer) and atomic structure (helicity) [23,34–38]. Depending on the synthesis route, intrinsic heterogeneity in length, diameter and helical distribution can be observed [39]. In addition, common synthesis methods (e.g. chemical vapour deposition (CVD) or arc discharge techniques) introduce impurities such as residual metal catalyst particles and carbonaceous non-nanotube particles which affect the mechanical, physical and chemical characteristics of the deposited materials [38,40–44]. In addition, strong attractive van der Waals interactions lead to the formation of networked bundles or ropes of (typically up to hundreds of) aligned SWCNTs; while MWCNTs tend not to bundle above two or three walls, they instead often form entangled agglomerates in the micrometre size range. Both effects hinder the formation of ordered, dense packing at longer scales [39,45]. On the other hand, many high-end applications would benefit from, or require, high-purity individualised CNTs with specific and homogeneous characteristics. In structural applications, high aspect ratio, disentangled CNTs are much more efficient reinforcing phases in polymer matrix composites, while SWCNTs with a specific diameter and helicity allow fine-tuning the metallic or semiconductor nature for electronic applications, as transparent conductors, thin-film transistors, thermoelectrics, etc. [5,35]. As such, prior purification and proper dispersion of CNTs is often essential, in order to optimally exploit their superior characteristics [5,39].
CNT purification processes have been discussed elsewhere [38,42,43,46,47] but, briefly, purification can be broadly classified as either chemical or physical, or a combination. While chemical purification processes (e.g. gas or liquid phase oxidation, electrochemical oxidation) rely on a faster oxidation of non-nanotube carbons when compared with CNTs or the selective dissolution of metallic impurities in acids, physical purification (e.g. filtration, centrifugation, chromatography, electrophoresis, high-temperature annealing) exploits differences in physical properties between CNTs and impurities, such as size, aspect ratio, gravity, magnetism, etc. Chemical methods are generally more invasive, typically involving oxidation which inherently introduces defects that are detrimental to the functional properties. Physical methods are relatively mild, therefore largely preserving the CNT structure, but can be time-consuming, and may still involve damaging dispersion steps [42,46]. A systematic comparison of SWCNT purification methods indicates the preferred method will depend on both the starting material and the characteristics required [48]. Often a combination of purification techniques is applied in order to obtain CNTs of desired purity.
On the other hand, structural defects introduced during CNT purification can enable subsequent chemical transformations, which can be of use for further processing and application. As indicated above, strong attractive van der Waals interactions between pristine CNTs favour agglomeration. As a result, pristine CNTs generally form unstable dispersions in organic and aqueous liquids (although some exceptions in ionic and electronic liquids exist) [49]. The extent of dispersion prepared by shear methods (such as sonication, high-pressure micro-fluidic flow, etc.) depends on the nature of the starting material, including the aspect ratio, purity, presence of entanglements, as well as the surface chemistry, and its compatibility with the solvent. Oxidative purification processes have been shown to introduce oxygenated (hydroxyl, carboxyl, carbonyl, etc.) functional groups at the surface of MWCNTs, which can assist in electrostatic dispersion [40,46,50,51]; the associated CNT cutting, reduces aspect ratio and removes entanglements, which simplifies processing but may harm final properties. Functional groups introduced during purification can also drive interaction with larger molecules involved in electrosteric stabilisation [52,53]. Polymers and dendrimers with terminal amino or hydroxyl groups can be covalently coupled to the activated CNTs to give amides or esters [54]. As an alternative, thermochemical grafting can clean and functionalise CNTs, with minimum damage, by utilising pre-existing defects to graft a range of monomers [53]. Surface modification of CNTs is also possible by weak non-covalent interactions, such as π–π stacking, charge transfer and hydrophobic interactions, which can improve CNT dispersion without affecting the CNT framework bonding [55]. Polymers and co-polymers can bind effectively to CNTs through multiple interactions and have been found to disperse both SWCNTs and MWCNTs in various solvents depending on the amphiphilic character of the polymer [56].
Organic suspensions
Attempts have been made to prepare CNT suspensions for EPD in a wide range of organic solvents, including ethanol [57–59], isopropyl alcohol (IPA) [10,51,60–62], butyl alcohol [63,64], acetone [65,66], methanol [67], acetonitrile [15,68], dimethylformamide (DMF) [69–74], cyclohexanone [75] as well as their mixtures [76–79] or mixtures with water [80–82]; a detailed summary of the suspension parameters used for the EPD of CNTs is listed in Tables S1 and S3 (supporting information). Purified and functionalised CNTs can be (partially) dispersed in a great variety of solvents by simply sonicating the suspension, yet the degree of dispersion quality varies and often sediments of entangled/agglomerated CNTs are removed by an additional centrifugation step [25]. Typically, dispersions are metastable and continue to precipitate over time, depending on the concentration or the presence of stabilising agents. Successful co-deposition of CNTs with other components depends on the preparation of a stable biphasic suspension in a suitable solvent. For example, in comparison to ethanol, DMF was found to produce more stable, homogeneous dispersions, not only of pure CNTs but also of CNT/alumina hybrids, resulting in agglomerate-free deposits [83]. In general, the success of DMF (and other amides) has been widely attributed to their solvent character, specifically, their high Lewis basicity and low hydrogen-bond donation. In a quantitative analysis, solvents with Hildebrand solubility parameter (δ
t) around 21 MPa1/2 were found to be effective, with a particularly strong sensitivity to the dispersive contribution (δ
d in the Hansen formalism). As such, DMF (δ
d = 17.4 MPa1/2) is preferred over ethanol (δ
d = 15.8 MPa1/2), water (δ
d = 15.6 MPa1/2), acetone (δ
d = 15.5 MPa1/2) and methanol (δ
d = 15.1 MPa1/2) [83]. However, other effects, such as sonochemical degradation of the solvent, may also play a role [84]. In addition to the dispersion quality, the interaction of solvent molecules with the CNTs can affect the electrophoretic processes, either through charging effects or by modifying the effective solvent dipole. For example, differences in the dielectrophoretic behaviour of MWCNTs in water and IPA when compared with cyclohexanone (Figure 2) were attributed to reduced mobility, and hence reduced dielectric permittivity, of the cyclohexane molecules within the CNT solvation shell [75]. SEM images (top) of electrodes connected via CNTs deposited following electrophoresis. Statistical analysis (bottom) of the successful connection yield, using CNTs dispersed in different solvents, under otherwise equivalent conditions. (Reproduced from Ref. [75] with permission of the American Chemical Society)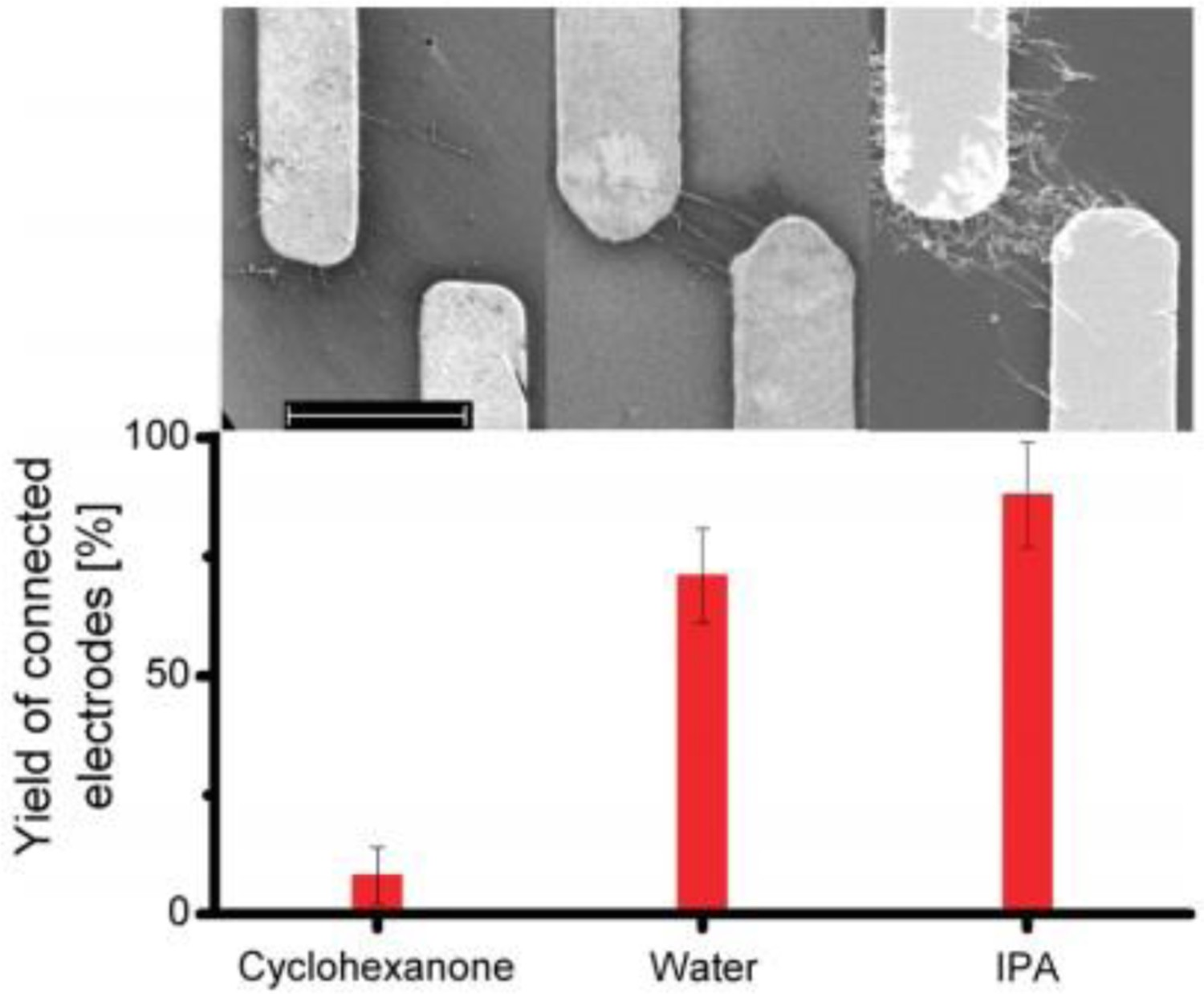
Aqueous suspensions
Aqueous solvents for dispersing CNTs offer several advantages over organic solvents, such as a better environmental compatibility, a lower cost and lower electric potential requirements [32,33]. The suspension parameters for the EPD of CNTs from aqueous solvents are summarised in Tables S2 and S3 (supporting information). The most common approach for dispersing CNTs in water relies on the electrostatic stabilisation by the oxygenated groups introduced during chemical oxidation (see above) [53]. In many cases, this approach leads to good aqueous dispersions in the absence of charging salts or surfactants [33,53,85,86], as indicated in Table S2. However, the use of aqueous solvents introduces a potential problem of gas evolution at the electrodes due to water electrolysis at higher voltages. This hydrogen gas evolution at the cathode and oxygen gas evolution at the anode can damage the deposits due to bubble formation, as well as reducing Coulombic efficiency [33,87–91]. To suppress water electrolysis during EPD, modulated electric field-related approaches are emerging, as discussed in the Alternating current EPD section.
Alternative aqueous dispersion strategies
Due to the structural damage caused by acid oxidation, as well as the introduction of possibly cytotoxic oxidation reaction debris (carboxylated carbonaceous fragments), alternative dispersion strategies are being explored to enhance the aqueous suspension stability [53]. The addition of small amounts of charging salts is a common auxiliary method to disperse CNTs and modulate their zeta potential. Mg(NO3)2 has been frequently used for charging CNTs through adsorption of Mg2+ ions, encouraging the formation of an electric double layer [10,62]. More recent approaches to enhance the surface charge include the addition of Al(NO3)3 [12,78,92–95], NiCl2 [59], I2 [51,60,96], Mn(NO3)2 [97], Co(NO3)2 [98], NiSO4 [99] and organic dyes such as crystal violet (CV) [100], methyl violet (MV) [101] or safranin [102] (Tables S1–S3 in supporting information). The addition of Al(NO3)3·9H2O into CNT-based EPD suspensions increases the deposition rate and also improves the adhesion of CNTs to the substrates [78,92,94]. A similar effect was observed with the addition of NiCl2 for the deposition of CNTs on ITO [59]. In other cases, adsorption of metal ions on the CNTs enables co-deposition of other nanoparticles adding specific functionality to the final coatings [97–99], for example, the in situ hydrothermal synthesis of hydroxyapatite (HA) particles on the surface of CNTs [103,104]. The decoration of CNT surfaces with various metallic or ceramic nanoparticles or polymer macromolecules in order to prepare composites is discussed further below (EPD of CNT-based nanocomposites).
Although the addition of charging salts is a valuable approach, high concentrations of salts can also increase the ionic strength, destabilising the suspensions and introducing impurities into the deposits [32,105]. In comparison to the direct adsorption, doping of CNTs by some salt solutions such as AuCl3, Na2PtCl4 and NOBF4, can follow a different reaction mechanism. In this case, a direct redox reaction occurs between the CNTs and the salt solution, depending on their relative positions in the electrochemical series; typically, the cations in solution have a tendency to receive electrons from the CNTs. Due to this charge transfer, positive ions such as Au3+, Pt2+, and NO+ cations are reduced to metal clusters or gas, while the CNTs accumulate positive charges, aiding solvation, for example, in ionic liquids [53].
Another approach to optimise the stability of CNT suspensions for EPD is by varying the pH (e.g. by the addition of HCl [106–109], sulphuric acid [110] or NaOH [106]) to adjust surface charge, particularly through the ionisation state of the oxygenated functional groups on CNTs. At lower pH (pH < 4), carboxylic (and other acidic) groups tend to remain protonated, with lowering surface charge, leading to agglomeration. At higher pH, increased ionisation produces more negative surface charges on the CNTs for improved stability [106]. As an illustration that the dispersion state controls the microstructure of the deposit, EPD coating were found to contain predominantly agglomerated CNTs at pH 4 but individual CNTs at pH 10 (Figure 3) [106]. Optimised pH values not only facilitate dispersion of oxidised CNTs, but also enhance their electrophoretic mobility as a result of the higher surface charge, leading to higher deposition rates [106]. On the other hand, excessive pH adjustment can reduce stability by increasing ionic strength, screening repulsion between the particles; strongly basic conditions may also separately solvate oxidation debris [40]. FE-SEM micrographs of SWCNT electrodes processed from suspensions with different pH values. (Reproduced from Ref. [106] with permission of Elsevier)
Finally, various types of surfactants can be added to enhance the stabilisation of CNT dispersions, such as sodium dodecyl sulphate (SDS) [63,111–115], hexadecyl trimethyl ammonium bromide (CTAB) [63,116], trioctylphosphine oxide (TOPO) [63], triethanolamine (TEA) [67], poly(vinyl butyral) (PVB) [59], ethyl cellulose [117], polyethyleneimine (PEI) [74], ribonucleic acid (RNA) [115], Triton X [118–125], stearyl ether [126], TNWDIS [9,127], Darvan C [128,129], Nanosperse-AQ™ [130], tetramethylammonium (TMAH) [124,125]. A detailed overview can be found in Tables S1–S3 (supporting information). Many of these surfactants are amphiphiles of which the hydrophobic part, typically an alkyl chain, has a high affinity for CNTs, while the hydrophilic part, which can be an ionised functional group, ethylene oxide group or a combination, associates with the water phase preventing aggregation [131,132]. Polymer molecules are of particular interested where they not only act as surfactants improving the dispersion, but serve as a functional component of the final product after EPD. Several examples of diphasic or triphasic colloidal suspensions for the fabrication of CNT/polymer composite films, such as CNT/chitosan or CNT/polyacrylic acid (PAA) composites, have been investigated [26,52,133].
UV–Vis absorbance spectroscopy can be used to evaluate the stability of CNT suspensions, by measuring concentration as a function of time. In the example shown in Figure 4, SDS was found to be a more effective stabiliser than CTAB, showing well-individualised SWCNT, with no significant change after 2 weeks sedimentation time [63]. The packing of different surfactants, amphiphilic polymers and their mixture around CNTs is an extensive topic in its own right, particularly in the content of separating single-wall nanotubes by their helicity [134]. (a) UV–vis absorption spectra of as-sonicated suspensions of SWCNTs in deionised (DI) water with SDS (top) or CTAB (bottom) as surfactant; (b) high-resolution transmission electron microscopy (HSTEM) image of an SWCNT sonicated in DI with SDS for 3 h. (Reproduced from Ref. [63] with permission of Elsevier)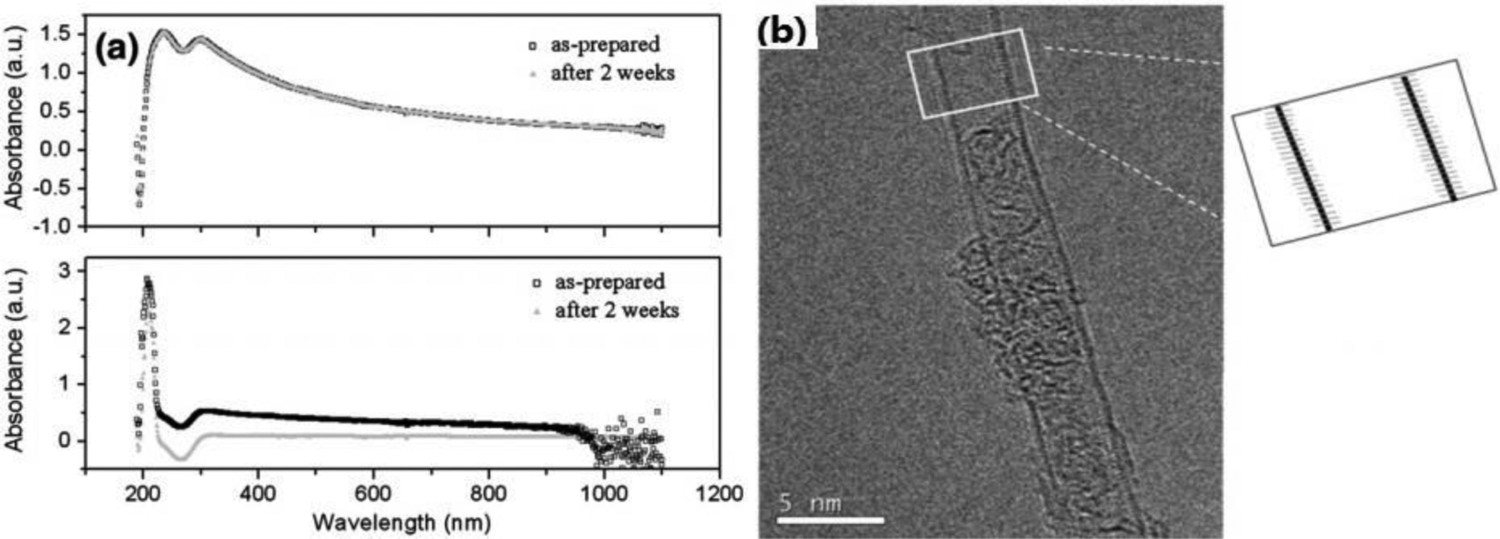
Generally, SWCNTs are much more difficult to disperse than MWCNTs due to stronger intertube attractions [132,135,136]. However, both ionic and non-ionic surfactants can successfully disperse CNTs in water; for ionic surfactants, the dispersion mechanism mainly relies on electrostatic repulsion, whereas non-ionic surfactants depend on steric repulsion [132]. There is no clear conclusion that either cationic or anionic surfactants are better for dispersing CNTs; the adsorption of ionic surfactants may be complicated by the SWCNT purification process and associated inadvertent side wall functionalisation of the tubes [135].
Deposition of carbon nanotubes
Direct current EPD
Since the publication of our previous review paper in 2006 [9], a large number of investigations have been carried out on the EPD of CNT films on a wide variety of electrode surfaces, both anode and cathode, depending on the charge of CNTs. Besides the required stability of CNT suspensions as discussed above, the influence of various electric field parameters on the characteristics of CNT deposits has been assessed. Table S4 (supporting information) summarises the electric field parameters used for DC-EPD of CNTs, including electrode materials, electric field strength, deposition time and interelectrode distance.
At low applied voltages (<2 V), an extremely low deposition rate is generally observed, which greatly improves above a threshold voltage. For intermediate voltages, the kinetics of CNT deposition as predicted by Hamaker’s law were found to be in good agreement with the experimental results, linearly correlating the deposition yield, and thus the thickness of the deposited films, with the applied electric field strength and deposition time [33,137]. Higher voltages (>5 V) produce much thicker coatings but with strong structural irregularities [33,121]. CNT layers of desired thickness and microstructure can, therefore, be prepared at a suitable voltage, simply by adjusting deposition time, to suit the final structural, electrochemical or photocatalytic application [91,138,139]. For example, at longer deposition times, a thicker network of SWCNTs provides a higher areal electrical conductivity [113].
Film quality and thickness are also affected by the conductivity of the electrode substrate used for EPD [19,79]. For identical suspension and electric field parameters, CNT films fabricated on a Cu sheet had a higher thickness (∼70–80 μm) than on Cr-coated Si wafers (∼50–60 μm), whereas CNT coatings on Au substrates were thicker than on stainless steel substrates, suggesting that EPD is facilitated on substrates with higher electrical conductivity [87,140]. At the same time, more conductive substrates produced denser CNT deposits with reduced surface roughness and pore volume, leading to more conductive coatings with potential for use in electronic devices such as sensors or electrodes [138–141].
Conventionally, CNTs are deposited on conductive electrodes of simple planar geometry. However, for a wide variety of applications, there is a growing interest in producing CNT films in a patterned fashion, on substrates of complex geometry and even on non-conductive materials. For example, patterned CNT films have been prepared on indium tin oxide (ITO) by pre-depositing a thin photoresist on the electrodes [59]. After EPD over the whole substrate, from an Ni-ion containing ethanol solvent, the CNTs deposited on the photoresist lines were selectively lifted-off by ultrasonication, while the CNTs on the ITO areas remained immobilised in Ni metal layer co-deposited on the ITO (Figure 5). A similar process using a more sophisticated patterning scheme has been applied to selectively deposit CNT field emitters [117]. CNTs selectively deposited on a silver mesh served as a protective layer against oxidation increasing the stability of the electrode [142]. Other organised porous substrates can be used to create regular arrays; for example, CNTs have been deposited on insulating metal oxide structures such as anodised aluminium oxide (AAO), filling the nanochannels with vertically aligned CNTs [65,143,144]. EPD is usually considered for large area/bulk applications, but EPD has also been used to deposit individual SWCNTs into electronic devices [145], for example, using pre-patterned electrodes. (a) Optical microscope image and (b) cross-sectional SEM image of CNTs deposited on patterned ITO substrates after ultrasonication. (Reproduced from Ref. [59] with permission of Elsevier)
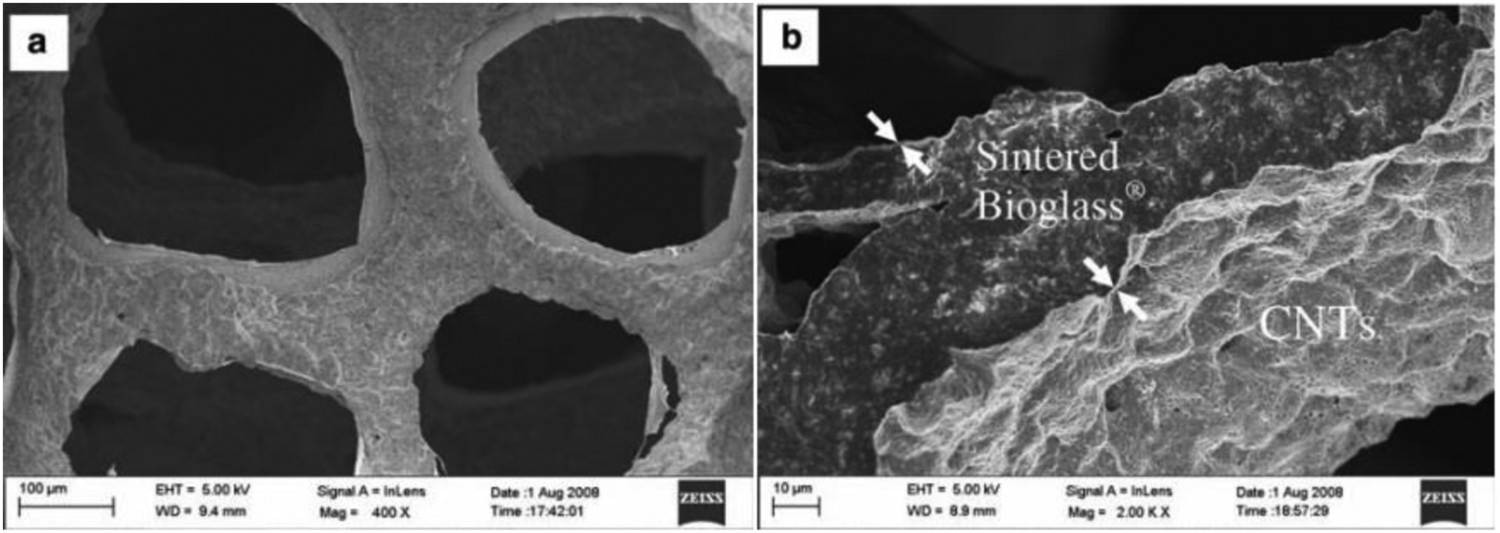
EPD can also be utilised to produce uniform coatings on 3D curved substrates by designing EPD cells that provide a uniform electric field through the electrolyte [87,146]. EPD can even be used to infiltrate porous non-conductive substrates, such as polymeric or ceramic scaffolds [147]. Polymeric substrates in different hierarchical structures have been coated with CNTs for a wide range of applications [52,79,119,133,148]. A typical example (shown in Figure 6) involves infiltration of CNTs into a polyurethane (PUR) foam [119], situated in the middle of the EPD cell between two stainless steel electrodes; the CNTs were driven through the pores under the application of a DC field. Under optimised EPD conditions, homogeneous CNT coatings formed on both the outside and the interior surface of the foams while retaining a highly open porous interconnected scaffold structure as required for bone ingrowth [119]. A similar approach was used for the production of CNT coatings on non-conductive poly- Schematic diagram of the EPD cell used to infiltrate non-conductive PUR foams with MWCNTs. (Reproduced from Ref. [119] with the permission of IOP science) (a) Low and (b) high magnification SEM images showing the typical microstructure of a CNT-coated Bioglass® scaffold obtained by EPD from an aqueous suspension, the CNT coating is indicated by the arrows in (b). (Reproduced from Ref. [146] with permission of Springer)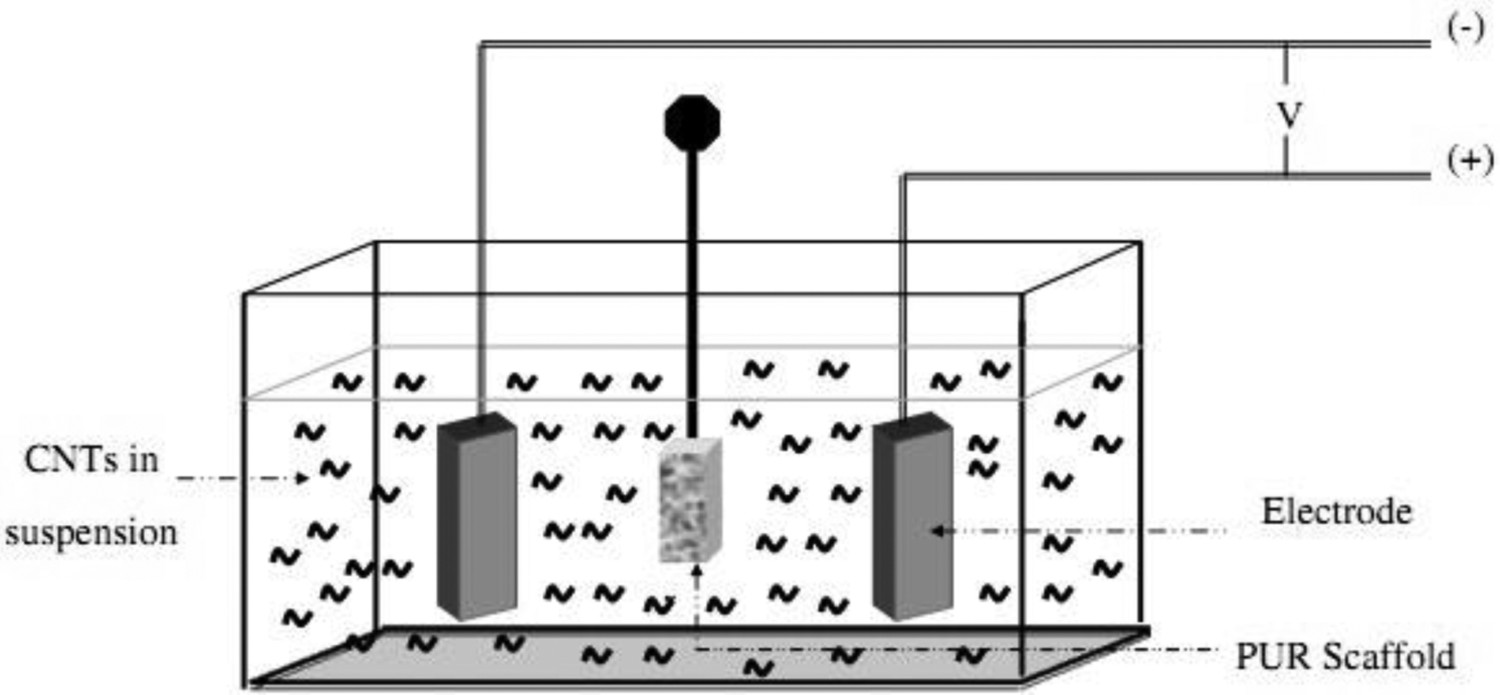
Alternating current EPD
As indicated earlier, the use of aqueous CNT suspensions for EPD is attractive; however, water electrolysis and concomitant bubble formation at the electrodes can be problematic in the case of DC-EPD. This effect can be limited by restricting the deposition time and by applying lower electric fields; however, shorter deposition times and lower applied electric fields also lead to thinner coatings [91,113,126,151,152]. In contrast to DC-EPD, AC-EPD is known to decrease the electrolytic decomposition of water, enabling the deposition of smooth and thick films with high packing density [24,153]. Deposition is a complex function of AC-EPD parameters such as frequency (0.01–1000 Hz) and waveform (sinusoidal, rectangular and triangular); for example, deposition yield was found to decrease for increasing frequencies when applying a triangular waveform, but increase for sinusoidal waveforms. Moreover, the spatial arrangement of CNTs correlates with the applied frequency, an effect ascribed to the distribution of the electric field lines [153,154]. The use of AC-EPD has been reported to produce more uniform coatings of CNTs on carbon fibres for improving load transfer [155], and enhance field emission properties in comparison to DC-EPD-coated substrates [76].
Dielectrophoresis
Dielectrophoresis (DEP)-assisted deposition is formally a process based on the motion of polarised particles rather than charged particles (electrophoresis) [75]. CNTs have a high polarisability, due to the delocalised electrons on the backbone, generating a dipole in response to an applied field. The polarisability is a strong function of size, crystallinity and electronic character; dielectrophoretic mobility in gels has been used to separate SWCNTs according to their different electronic character [134]. Since CNTs also develop surface charge in contact with solvents, motion in an electric field may be a combination of electrophoresis and dielectrophoresis. Where surface charge is low, dielectrophoresis may dominate, leading to symmetric deposition at anode and cathode. DEP has been used to deposit single SWCNTs onto device electrodes and to create aligned arrays [145]. MWCNTs can also be applied to increase the polarisability of other particles (Figure 8(a)). For example, polystyrene (PS) microparticles were successfully manipulated by DEP after coating with CNTs, to increase their conductivity and permittivity [156]. Figure 8(b) shows that the CNTs served as a nanoelectrode extension, protruding from the edge of lithographically patterned microelectrodes. Under optimised conditions, particles with different dimensions were deposited in selected regions, depending on the local field [157]. (a) Layout of a dielectrophoresis system equipped with curved microelectrodes; (b) patterning of Pt/N-CNTs between the microelectrodes of the dielectrophoresis system. (Reproduced from Ref. [156] with permission of Wiley)
Post-treatments of carbon nanotube deposits
Increasing the adhesion of the CNT layer to the substrate material can significantly improve the stability and functional properties of EPD coatings. Therefore, post-EPD treatments are increasingly applied to introduce chemical bonds at the coating/substrate interface [10,63]. For example, pre-coating an Si wafer with a conductive Ti film before the EPD of CNTs allows the subsequent formation of a TiC interfacial layer during vacuum annealing of the dried CNT films at high temperature, improving field emission performance [11,63].
EPD of CNT-based nanocomposites
Overview
In addition to the deposition of pure CNT coatings discussed above, EPD is increasingly used to prepare CNT-based composite coatings incorporating other nanoparticles, using a variety of strategies, such as sequential deposition from separate dispersions, one-pot co-deposition from multicomponent dispersions, or deposition of preassembled CNT-based composite particles, as schematically summarised in Figure 9(a) [23]. An overview of the wide variety of nanoparticles that have been successfully combined with CNTs by means of EPD is given in Figure 9(b), a more detailed overview of the specific suspension parameters used in different studies is presented in Table S3 (supporting information). (a) Schematic illustration of common EPD strategies applied for the production of CNT-based composite coatings. (b) Summary of typical compositions, reported in recent literature.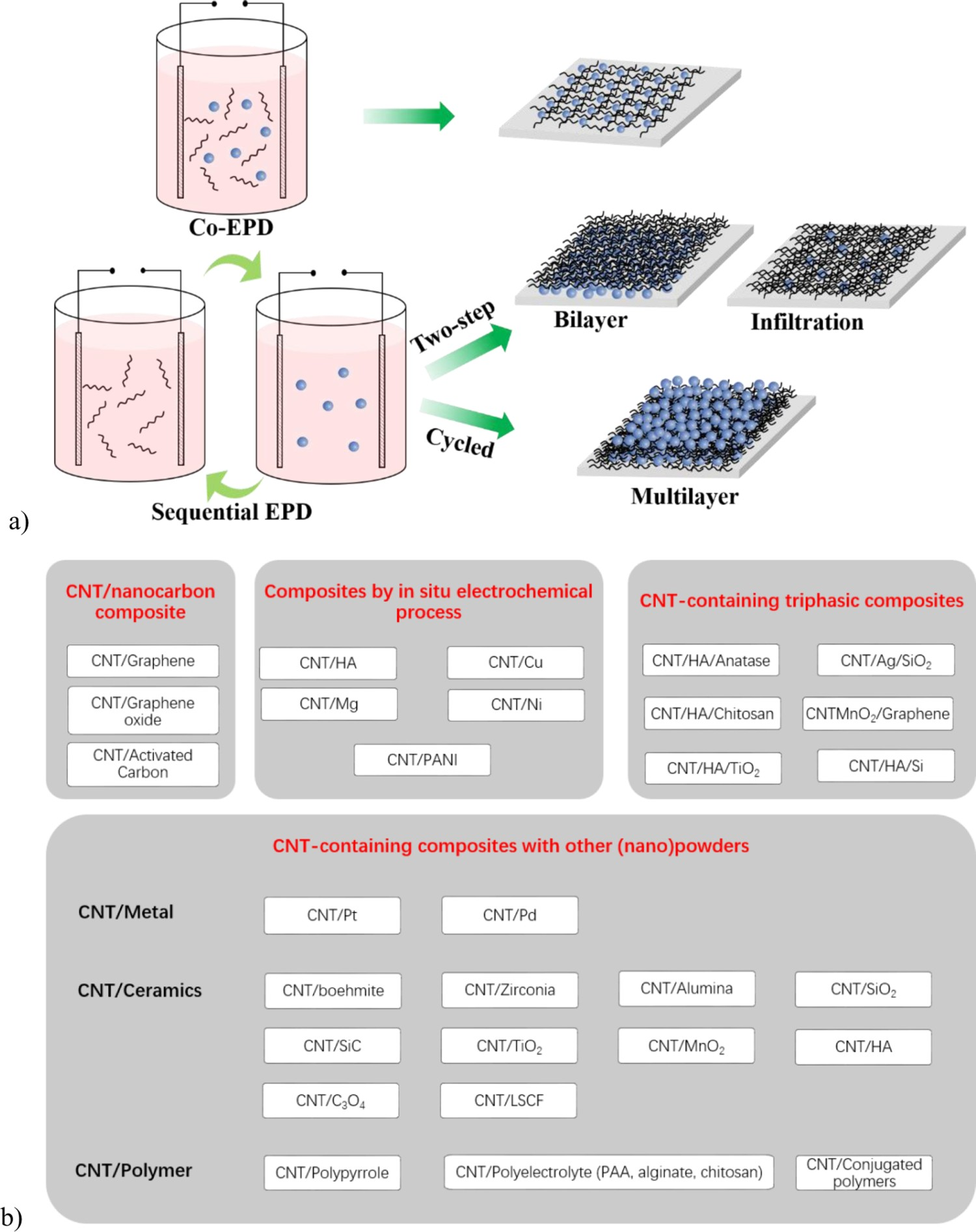
Sequential deposition from single-phase dispersions
Layered composite materials (laminates) can easily be obtained by simple alternating EPD from two or more single-phase dispersions [158]. As such, CNT/TiO2 laminate coatings of up to four layers (CNT/TiO2/CNT/TiO2) have been produced demonstrating that the CNT layer can provide a crack deflection and delamination path effectively reinforcing the ceramic coating [123]. Sequential deposition has also been used to deposit nanostructured fibrous meshes of CNTs covering both Bioglass (BG) [121] and hydroxyapatite (HA) [145] layers, with the potential to promote cell attachment and proliferation as desired in tissue engineering applications.
Co-deposition from multiphasic dispersions
A wide variety of materials, especially ceramics and polymers, have been combined with CNTs by co-deposition in a single EPD step. As highlighted in the Electrophoretic deposition of carbon nanotubes section, the stability of the multiphasic dispersions is the key factor for producing homogeneous composite coatings. If both types of particle are to remain individualised, and deposit at the same electrode, they must possess the same charge. A previous comprehensive review on EPD of CNT/ceramic nanoparticles discussed the combination of CNT with nano-sized ceramics for functional, structural and biomedical applications [36]. By way of examples, CNTs and nanocrystalline MnO2 have been co-deposited, after charging with sulphuric acid [159] and various phenolic molecules [160], to create porous, well-connected networks, useful for improving the cycling efficiency of hybrid electrodes in supercapacitors. In a mechanical context, micro-cracking was reduced in co-deposited TiO2–CNT [161] and triphasic HA/anatase/CNT [162] composites films. Larger (micrometre-sized) BG particles were co-deposited with CNTs on stainless steel substrates, using Triton X100 as a surfactant [121].
CNTs can also be co-deposited with other nanostructured carbon materials, such as graphene. Well-dispersed CNTs can prevent agglomeration of graphene sheets, preserving their large surface area and serving as conductive bridges that decrease the resistance of hybrid CNT/graphene transparent conductive films [92,163–165]. In order to achieve homogeneous co-deposition of CNT/graphene composites, the usual approach is to create stable mixed dispersions using the strategies discussed in the Preparation of carbon nanotube suspensions section. Several dispersion systems were successfully used including those stabilised by simple pre-oxidation of CNTs in air or in acids, by Mg(NO3)2·6H2O in IPA [165], by Al(NO3)3·9H2O in a mixture of acetone and ethanol [92], or by using methyl violet or safranin in water [101,102]. However, insulating additives (including long-chain surfactants or polyelectrolytes) can limit the conductive connection between CNTs and graphene flakes, while pre-oxidation treatments introduce structural defects limiting the intrinsic conductivity of the nanocarbon. A balance of different dispersants, or post-treatments, may be needed to further optimise the performance. CNTs can also be co-deposited with more conventional nanocarbons, such as carbon black, using similar methods [49].
CNTs can be deliberately co-deposited with polymeric components (e.g. PAA, alginate, chitosan) in order to create composite structures. PAA chains adsorbed on CNTs can act as dispersant and charging agent due to deprotonation of the carboxylic groups, as well as the eventual matrix, allowing anodic EPD of uniform PAA/CNT films [133,166,167].
Sequential co-deposition from different multiphasic dispersions can be used to produce laminates with multicomponent layers. Chitosan/MWCNT/HA laminate nanocomposites containing chitosan–MWCNT layers separated by chitosan–HA layers were successfully prepared by EPD [145,164]. Co-deposited monolayer chitosan–MWCNT–HA composite films were also produced from suspensions consisting of homogeneously dispersed HA particles and CNTs in chitosan aqueous solution (Figure 10(a)); sequential deposition produced laminates (Figure 10(b)) with controlled layer thicknesses in the range of 0.2–5 μm by varying the deposition time. The layers with different compositions were strongly adhered, indicating the convenient use of chitosan as a matrix for all layers, which enables the effective integration of the individual layers into a robust multilayer structure [166]. SEM images of (a) a chitosan/MWCNT/HA composite film obtained by co-deposition from triphasic dispersion (arrows indicate MWCNTs) and (b) a laminate consisting of alternating chitosan/HA and chitosan/MWCNT layers obtained by sequential co-deposition from the corresponding biphasic dispersion (arrows indicate chitosan/MWCNT layers). (Reproduced from Ref. [166] with permission of Elsevier)
Deposition of preassembled composite particles
Rather than depositing individual CNTs with other (nano)particles from well-dispersed suspensions, it is also possible to deposit homogeneous composite films by assembling hybrid particles in advance. CNTs can be bound to other components, either covalently or electrostatically, to form hybrids which behave as single entities during subsequent EPD. Simple heterocoagulation uses complementary electrostatic interactions between CNTs (typically negative after oxidation) and nanoparticles (typically positively charged), with careful selection of concentrations, relative proportions and sizes, as applied, for example, to CNT/HA [166] and CNT/TiO2 [67] hybrid particles. In the case of TiO2, the nanoparticles were positively charged in a range of alcohols and hence heterocoagulated on the surface of the negatively charged CNTs to form TiO2/CNT composite particles with a net positive charge, which could be further enhanced using TEA as a charging agent [67].
Alternatively, CNTs can be introduced during the syntheses of ceramic nanoparticles, acting as nucleants for the inorganic phase precipitation. CNT/HA composite powders were produced by an in situ chemical method and deposited on Ti substrates (shown schematically in Figure 11(a)) [104,128,129,149]. Acid-treated, carboxylated MWCNTs have been used as nucleants for the precipitation of both nano-sized HA [104], and MnO2 [161], from aqueous salts, before EPD of crack-free coatings. Similarly, CNT/boehmite hybrid powders were prepared using hydrothermal processing to both functionalise the MWCNTs and deposit the inorganic phase (Figure 11(b)) [168].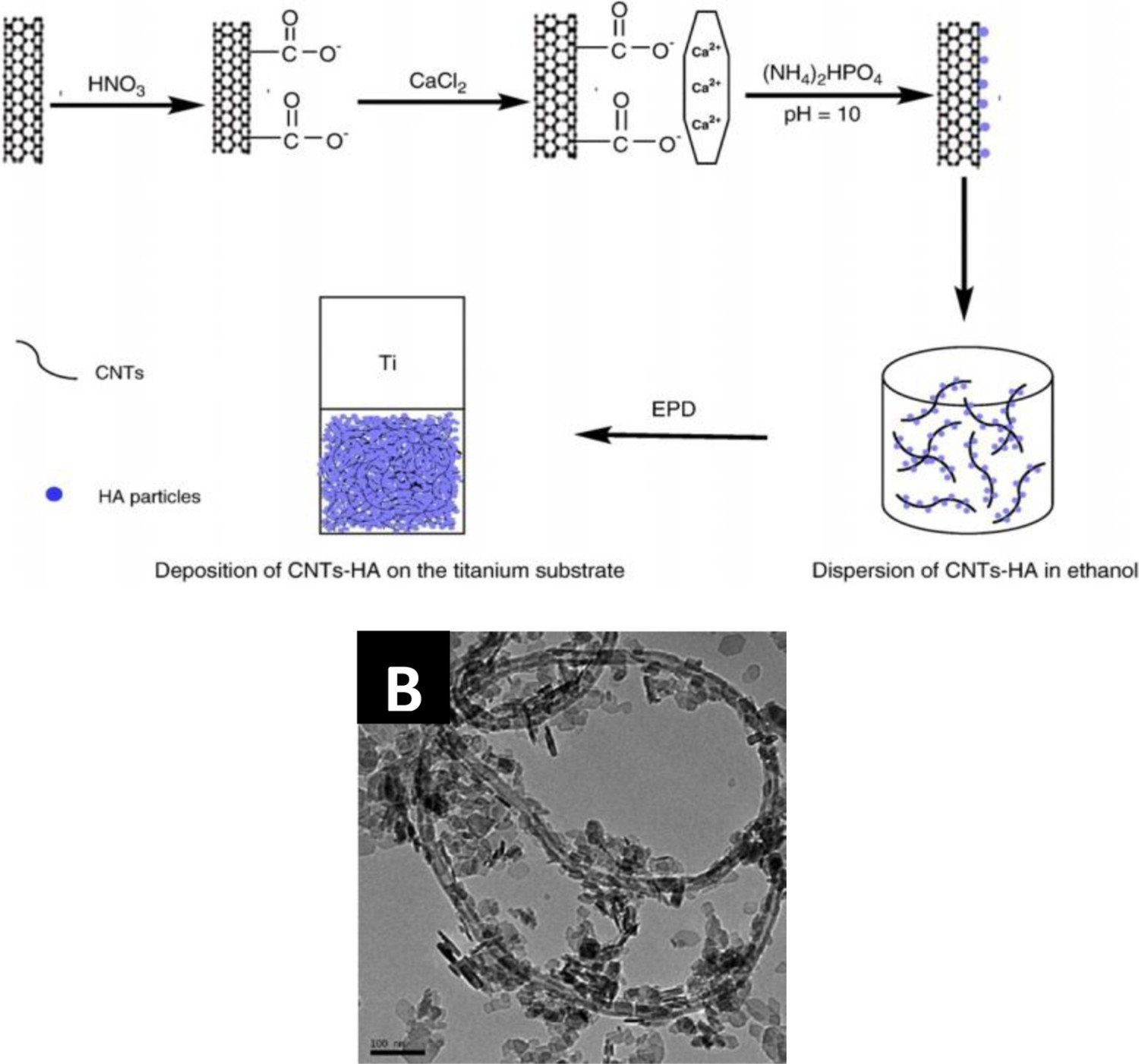
As noted above, pre-adsorption of polymers both enhances dispersion stability, providing (non-covalent) functionalisation, and allows the fabrication of CNT/polymer composite films [133,148,169–172]. Individual CNTs can be wrapped by a variety of amphiphilic polyelectrolytes including PAA [173] and a variety of DNA sequences [174]; the binding can be strengthened electrostatically, for example, by linking carboxylated MWNTs to DNA though Mg2+ ions, leading to the formation of core–shell CNT/DNA coatings after EPD [175]. Alternatively, conjugated polymers, such as polythiophene [171] and fluorene-phenylene [170], adsorb strongly to the pristine CNT surface through π–π interactions, forming stable suspensions in a variety of typically organic solvents, depending on the polymer side chains. Since the interaction is non-covalent, and does not require functionalisation, the intrinsic properties of the CNTs are maintained, though modulated by the presence of the polymer. Side chains that carry charges, for example, by protonation of amines, can facilitate EPD through additional particle charge, and charge neutralisation (for example, deprotonation) at the working electrode [170]. Other charge bearing, adsorbed composite components may act similarly; for example, robust and homogeneous MWCNT/hexamethylene diisocyanate (HDI) composite coatings were deposited on Cu substrates by optimising the concentrations of HDI and the dispersant Gum Arabic (GA) from an aqueous suspension [172]. An alternative to simple adsorption is to polymerise suitable monomers, in situ, around the CNTs. For example, aniline introduced during the purification of CNTs can yield homogeneous CNT/polyaniline (PANI) hybrid precipitates for EPD [169].
In addition to these diphasic hybrids, a variety of ternary systems have been investigated, including the addition of graphene to CNT/MnO2 films [108], as well as MnO2/alginate/CNT [176], HA/chitosan/CNTs [80,166,177] and calcium phosphate (CaP)/chitosan/CNT [81] composites. The adsorption of the charged polymer on both the CNTs and the other particulates not only increased the suspension stability, but also provided a binder to reduce cracking and to strengthen the adhesion of the deposited film to the substrate. The composition, microstructure and thickness of such triphasic composites can be controlled by varying the concentration of raw materials in suspension and the EPD parameters [177].
Due to the relative high density of metallic materials when compared with other coating components including ceramics and polymers, obtaining stable multiphasic colloidal suspensions containing well-dispersed metal particles can be challenging. Therefore, CNT/metal composites are commonly produced by the in situ reduction of a metal nanoparticle precursor solution either on the surface of CNTs to form hybrid particles or on directly on the deposition electrodes. As discussed previously (Preparation of carbon nanotube suspensions), the addition of metal charging salts can effectively facilitate the EPD of CNTs, but may also contaminate pure CNT deposits due to the deposition of metal components. However, where the metal component is desired for certain applications, the approach is particularly attractive, exploiting the function of metal ions not only for the stabilisation of CNTs in suspension but also as precursor for the metal phase in CNT/metal composites. Following this approach, various CNT/metal composites including CNT/Mg [178], CNT/Ni [60] and CNT/Cu [179,180] composites have been developed for applications in capacitors, catalyst supports, etc. Taking CNT/Cu composites as an example, Cu2+-complexed CNTs move towards the cathode. Upon deposition, electrons are conducted from the cathode, though the porous conducting deposit of CNTs, reducing the Cu2+ ions to Cu nanoparticles on the surface of individual CNTs. The film density and dimensions of the metal particles can be controlled by the applied potential, deposition time and concentration of Cu2+ ions in the suspension [179]. However, preferential reduction of excess metal ions in solution can lead to greater metal deposition on the outer CNT layers, producing a rougher surface morphology compared to bare CNT deposits (Figure 12). On a more local scale, the metal nanoparticles may also grow heterogeneously, depositing preferentially at junctions in the CNT network. However, this morphology may be advantageous, as shown, for example, in the deposition of a CNT/Cu hybrid network with enhanced electrical conductivity and mechanical properties on carbon fibres [180]. CNT/Ni composite films were fabricated by a modified two-step process involving first the EPD of pristine CNTs followed by electrodeposition of the metal [65,88]. This approach yielded a uniform composite coating with a CNT concentration as high as 12.5 wt-%, much higher than that obtained by conventional co-electrodeposition [88]. Alternatively, a thin Ni film can also be fabricated on the pre-deposited CNTs by electroless deposition. Ni clusters formed on the surface of the CNTs by adding NiSO4·6H2O to the EPD bath can serve as nuclei inducing the growth of a homogeneous metal layer covering the overall CNT network [99]. This secondary metal deposition is an example of composite preparation by post-EPD treatment. Similar methods can be applied to polymer composites, by resin impregnation [179], and ceramic-matrices by post-EPD mineralisation. For example, CNT/nacre particles, co-deposited to form crack-free coatings on a Ti substrate, were mineralised in phosphate buffer solution [106]: a dissolution-precipitation process released Ca2+ ions from the nacre powders which reacted with PO4
3- ions to form carbonated HA nanoparticles. SEM showing the surface morphology of CNT films deposited by EPD onto an ITO electrode: (a) Cu/CNT assemblies; (b) bare CNTs. (Reproduced from Ref. [179], Open Access article by SpringerOpen distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0))
Applications of electrophoretically deposited CNT films
As noted above, the widespread use of CNTs is often limited by difficulties preparing microstructures that fully manifest their intrinsic properties. The versatility of EPD to form CNT-based films, coatings or free-standing layers with controlled architecture has attracted enormous interest as a means to control mechanical, electrical or optical properties [23]. EPD-derived CNT systems have been considered for structural reinforcements in composites [91,155,181–187], field emission display devices [69], electrochemical electrodes [188,189], transparent conductive films, dye-sensitised solar cell electrodes [178], catalyst supports, solid-phase microextraction fibres, sensors and biomedical devices [120,190]. This section will briefly summarise the advantages of EPD for these applications, with focus on more recent developments.
Structural films and composites
Pure CNT films, or Buckypapers, are often studied for their pure mechanical properties and used as precursors for composites. Although Buckypapers are most often prepared by filtration, EPD of CNTs on stainless steel sheets has been developed as a cost-effective, rapid and reproducible alternative method [191]. The introduction of a reversed electric field (post-EPD) effectively detaches the Buckypaper from the metallic substrate without appreciable degradation of surface morphology or mechanical properties [192]. Composite coatings may be formed directly on substrates by either co-deposition or post-EPD infusion of a variety of matrices (Deposition of preassembled composite particles); the presence of CNTs is reported to significantly increase the modulus and hardness of polymer matrix composites [52,177], increase the hardness of metal matrices, as well as to inhibit cracking of ceramic coatings. Masked co-deposition of CNTs and boehmite nanoparticles was used to produce composite microgears [168]. The presence of chemical interactions appears to improve performance, for example increasing bonding between oxidised CNT and chitosan [52] and HA [21,64,104,193] matrices, providing particle bridging and pull-out mechanisms. Even small additions of CNTs (1–5 wt-%) were found to significantly reduce the cracking of TiO2 films produced by co-deposition [161]. In an HA/TiO2/CNT triphasic composite, the CNTs enhanced the surface hardness and adhesion strength to a Ti alloy substrate [162]. In sequentially deposited CNT/TiO2 laminates, CNTs substantially raised the interfacial strength between the different layers acting as a crack inhibiting barrier [123,161]. In Ni/CNT composite films, MWCNT concentrations as high as 12.5 wt-% provided a significant enhancement in hardness (870 VPN) due to the high loading and uniformity of the microstructure [88].
As well as the above mentioned reinforcement application of pure CNTs, there is interest in combining CNTs with conventional, micrometre-scale, structural reinforcements, in order to form hierarchical composites [2]. In fibre-reinforced composites, the interfacial region between the fibres and the matrix determines the extent of stress transfer, and plays a critical role in the overall composite mechanical properties [182]. Due to their size, stiffness and strength, CNTs have the potential to strengthen the fibre/matrix interface when used as an auxiliary reinforcement at the fibre surface, leading to increased load-bearing capacity and fatigue resistance [181,182]. There are a variety of routes to grow or graft CNTs onto the fibre surface, but EPD is particularly straightforward, providing uniform deposits with a homogeneous microstructure, in a scalable, potentially continuous fashion. Notably, the CNT coating process can be carried out at ambient conditions, preserving the fibre strength and modulus, in contrast to most CVD or other high-temperature processes. EPD has been applied to graft functionalised CNTs onto conductive carbon fibres (CF) as well as non-conductive glass fibres (GF) and the resulting epoxy matrix composites have shown improved interfacial shear strength, interlaminar shear strength, flexural strength and flexural modulus, when compared with composites made using commercial fibres [91,155,181–187]. CNTs can be deposited on continuous fibre tows [194] or infused into woven fibre preforms (Figure 13) [185]. The approach is analogous to the extensive use of EPD to infiltrate porous preforms with nanoparticles to fabricate ceramic matrix composites [22–24,144]. Increasing CNT film thickness by either ultrasonication during EPD or longer deposition times can improve the efficiency of stress transfer between the CNT-coated CF and the resin matrix [116,127,195]. Experimental set-up for the deposition of CNTs into glass fibre preforms, suitable for the manufacture of glass fibre-reinforced polymers (GFRP). Glass fibre textiles were vertically attached to the electrodes with a non-conductive spacer to enable penetration of CNTs through the fibre mats during EPD. (Reproduced from Ref. [185], licensee MDPI, Basel, Switzerland, open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/))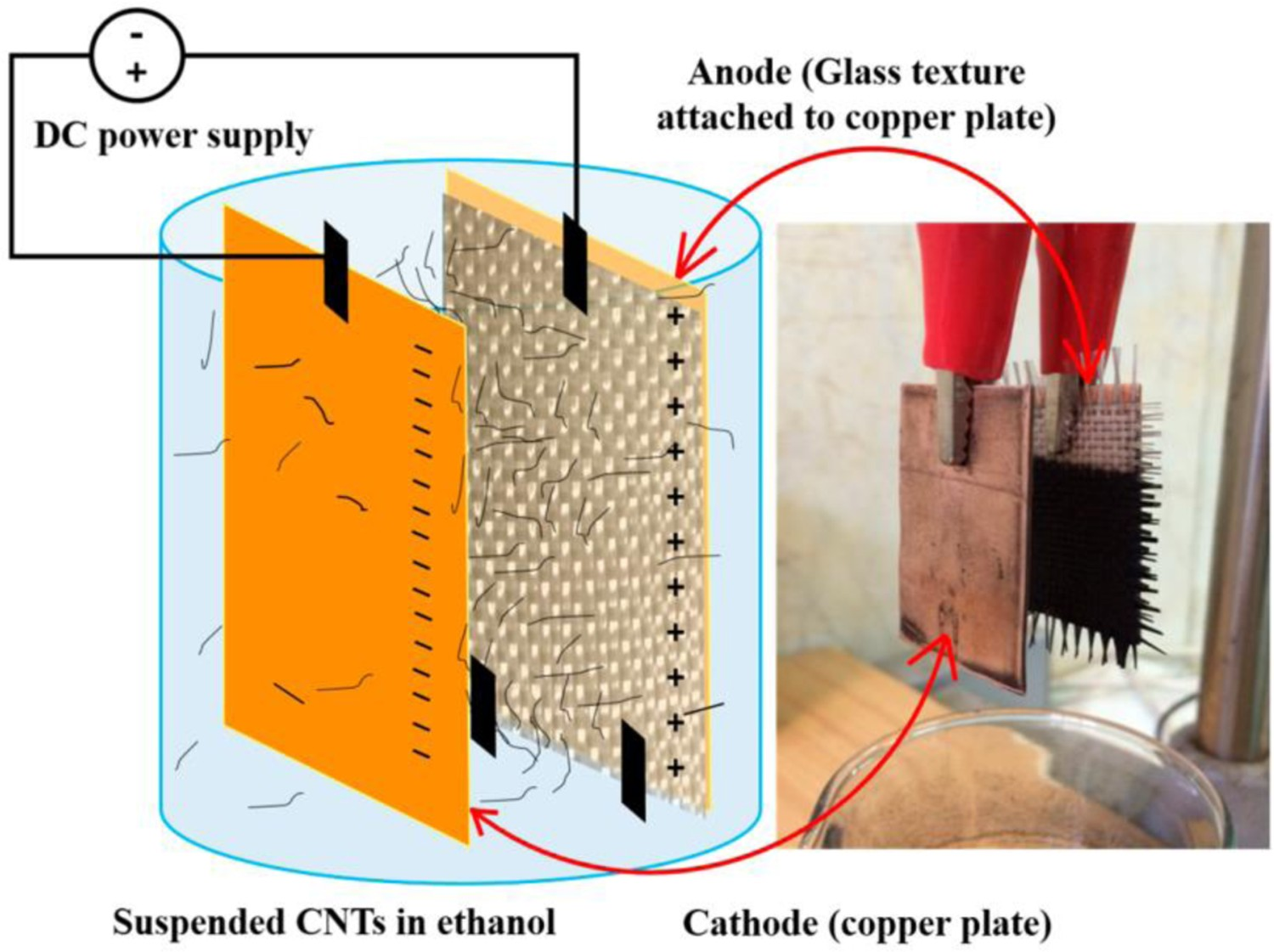
While the primary reinforcement mechanisms are physical, relating to mechanical interlocking, increased fibre/resin contact area, and direct local load bearing, surface chemistry can be used to enhance the adhesion between fibres and resin. CNT/graphene oxide (GO) hybrid particles introduce reactive oxygen-containing groups that can couple to the epoxy resin matrix during the curing process, improving mechanical properties [196]. Similarly, deposition of amine functionalised MWCNTs onto CFs, by AC-EPD, enhanced composite fatigue life [197]. The use of a chemically reactive dispersant, such as PEI, incorporating amine groups, not only reacts with epoxy, but also provides a locally thermoplastic interphase. Compared to the brittle fibre/epoxy interface, a PEI-CNT-rich interphase toughens the transition layer, facilitating a homogeneous load transfer, more ductile fracture and increased fracture (flexural) strength by shifting the fracture surface away from the fibre surface [184]. CNT/metal hybrids deposited onto CFs can improve both mechanical and electrical properties of the resulting CF/epoxy composites, as exemplified by anodic EPD of CNT/Cu nanoplatelets [198].
CNT/CF hybrids have generally been studied in polymer matrix composites; however, they are applicable in other matrices. Carbon/carbon (C/C) hybrid composites reinforced by CFs with radially grafted CNTs exhibited an improved out-of-plane and in-plane compressive strength, as well as interlaminar shear strength [199]. Similarly, in CF-reinforced SiC (C/SiC) ceramic matrix composites, the deposition of CNTs increased the work of fracture through a ‘double-scale pull-out’ energy dissipation mechanism at both the micro- and nanoscale. Interestingly, this composite had two different fibre/matrix interfaces due to the application of a pyrolytic carbon coating over the primary fibres. The CNTs were only found to be effective when introduced at the weaker CF/pyrocarbon interface, not at the pyrocarbon/SiC interface [200]. The addition of a CNT/SiC hybrid coating at the interface of conventional C/SiC composites, via CNT EPD followed by SiC CVD, increased the ultimate flexural strength by 40% compared to a pure SiC coating [201]. Such SiC-based coatings also protect C/C composites from oxidation at high temperatures. SiC/CNT–SiC double-layer coatings obtained by EPD showed an improved thermal shock resistance, which further increased with EPD time [9].
Although CNT-coated CFs are effective, the high production costs and low yields have limited commercial uptake. Continuous electrophoretic processes offer one approach that is applicable on an industrial scale, as schematically shown in Figure 14 [202,203]. However, EPD is less effective at creating a strong bond between the CNTs and the CF, compared to some alternatives such as continuous CVD [204]. Adhesion can be enhanced by chemical functionalisation, or alternatively by applying a pyrolysis treatment after EPD [205]. Schematic diagram showing the continuous EPD process for manufacturing multiscale CNT-coated carbon fibre reinforcement. (Reproduced from Ref. [194] with permission of Elsevier)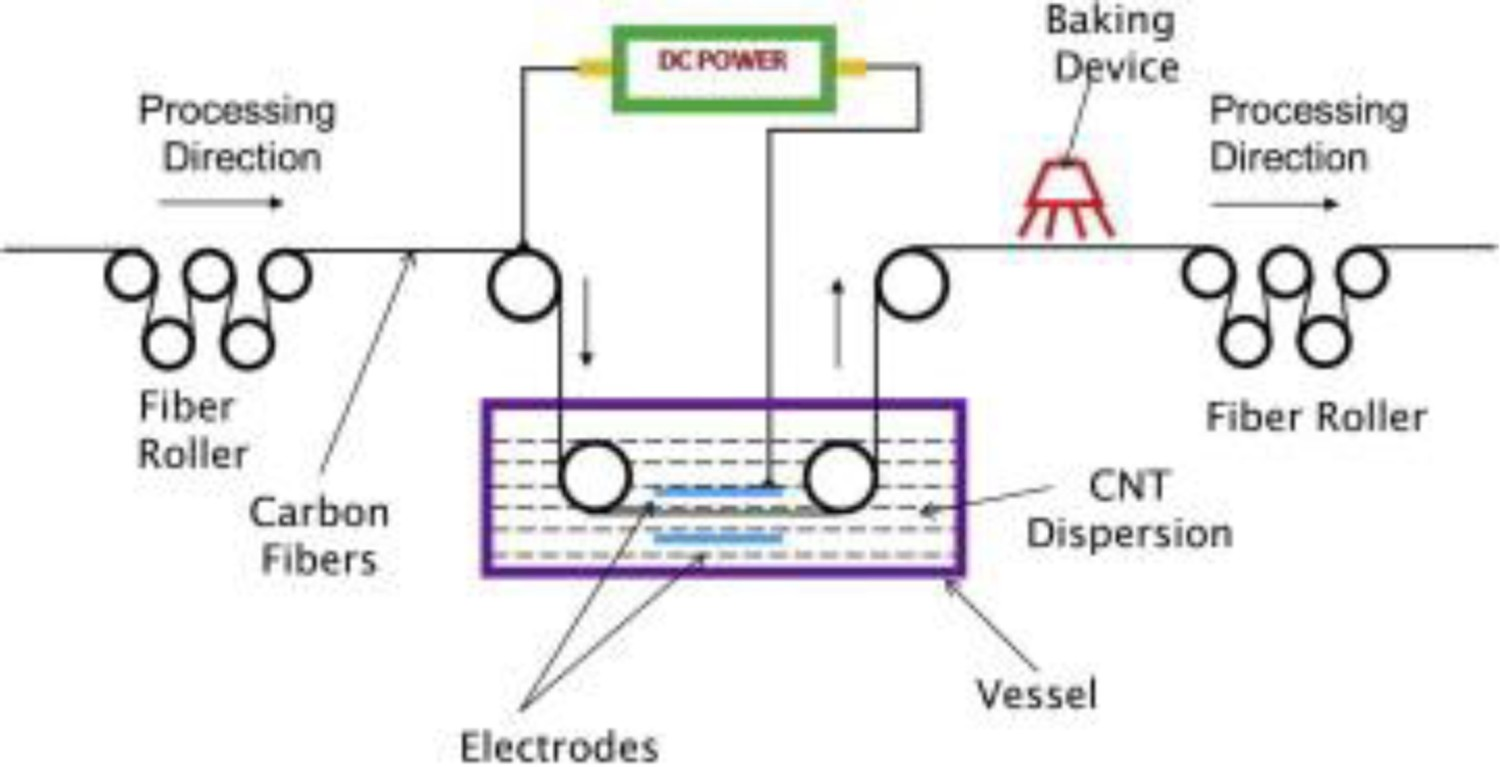
Field emission devices
The high aspect ratio, small radius of curvature, high electrical conductivity and excellent chemical stability of CNTs contribute to promising field emission characteristics, including low turn-on voltage, large emission current density and long-term stability. These advantages have triggered the use of CNTs as ‘cold-cathodes’ for a wide range of applications, such as field emission displays (FED), back light units for liquid crystal displays, microwave amplifiers, X-ray sources, etc. [69,179,206,207]. Usually, longer (high aspect ratio) CNTs offer a lower turn-on field; however, the degree of field-enhancement improves with increasing separation between the individual structures [10]; on the other hand, the net total current density depends on the density of active tips packed together within the coating. The competition between these effects means the spacing, orientation and density of the CNTs must be carefully controlled. EPD can selectively deposit vertically aligned CNTs onto large area cathodes with a simpler process and at a lower cost than conventional CVD methods [10]. Planar-gate triode devices, exploiting EPD-based CNT field emitters exhibited a lower turn-on voltage and better emission uniformity than screen-printed CNT cathodes [62,117]. In addition, it was found that controlled nanotube orientation and density could significantly improve the field emission current density and long-term stability under high operating voltages [69]. However, the adhesive strength between EPD-derived CNT films and the substrate is usually relatively weak, which may lead to a deteriorating uniformity and stability of emission at high electric fields. Post-EPD treatments, such as vacuum annealing at high temperatures, can introduce a TiC interfacial layer between the CNTs and the substrate, significantly improving the contact properties at the interface, including both adhesion strength and electrical conductivity [11,200]. Post-deposition annealing of both MWCNT [63,110] and SWCNT [115] coatings in air on various substrates also improved emission stability at high emission current density and emission uniformity, by removing contaminating dispersants (Figure 15). Field emission images of (a, c) low-purity SWCNTs and (b, d) high-purity SWCNTs without (a, b) and with (c, d) heat treatment to remove the RNA dispersant, on applying a field of 4.1
In recent years, the composition of CNT FED devices has been shifted from pure CNTs towards CNT-based composites such as CNT/graphene, CNT/carbon nanoparticles and CNT/CdSe hybrid coatings [165]. The sheets of graphene can connect more widely separated CNTs, considerably decreasing the contact resistance and sheet resistance of the film [165]. Similarly, carbon nanoparticles, applied by electrochemical process on conventional EPD CNT coatings, were found to improve the conductivity as well as the adhesion of the MWCNT films to the substrate [208]. In a related approach, Cu nanoparticles were deposited on the outer wall of CNTs increasing their surface roughness for improved field emission and electrical contact [179,209].
Energy storage and conversion devices
Due to their high electrical conductivity and accessible specific surface area, CNTs are also being considered as electrode materials for energy storage or conversion devices, such as supercapacitors, fuel cells and solar cells [110,210].
Supercapacitors
Compared to rechargeable batteries, supercapacitors offer the advantages of a more rapid charge/discharge characteristic (i.e. higher power density) and a longer cycle life, although with lower energy densities [189,210–212]. As energy density is given by ½CV 2, where C is the capacitance and V the cell voltage, it is helpful to increase capacitance and voltage windows [189]. CNTs intrinsically offer a high electrochemical surface area, especially at small diameters, when exfoliated or debundled, and deposited as thin-film electrodes [188,189]. Thinner films manifest higher specific performance relative to the mass of active material, as losses associated with electron and ion transport are minimised; however, while these values indicate intrinsic performance, they are not directly relevant to practical devices. In complete supercapacitors, the weight of the current collectors, separator and electrolyte must also be included, and thicker films give a better specific performance normalised to the mass of the whole device. EPD conveniently allows the thickness of the electrode film to be adjusted, on a metal current collector, in the relevant (tens of micrometre) range. Ragone plots of energy density versus power density summarise performance, but some recent examples combine specific data normalised in different ways, leading to confusion. EPD not only provides a versatile tool to coat complex electrodes homogenously with CNT composite films of controlled thickness, but it is also a potentially binder-free method, avoiding the use of adhesives which can occlude or electrically isolate a fraction of the active electrode [210,213].
Pure CNTs offer only electrochemical double-layer capacitance (EDLC), through the association of counterions at the surface. Energy density can be increased by incorporating nanoparticles that introduce a pseudo-capacitive response, through redox activity across a range of potential that resembles a capacitive response [110,159]. Examples of such pseudo-capacitive components include noble- and transition metal oxides such as MnO2, RuO2 and their mixtures [110,159,196,211,213,214], rare-earth oxides such as Eu2O3 [215]. Post-deposition annealing can be used to transform (co-deposited) metals into active particles, for example, by oxidising nickel particles into active oxides/hydroxides [60,189]. CNT/Ni hybrids have been produced using NiSO4 as charging salt during the EPD of pure CNTs to form Ni clusters that served as nuclei for subsequent Ni electroless deposition (without using a Pd sensitiser) [99]. In general, the conductivity of CNTs assists current collection from relatively low conductivity pseudo-capacitive materials, such as MnO2 and Ni(OH)2, and provides a porous nanostructured support framework. Such combination allows both electron and ion access to the nanostructured pseudocapacitor component, minimising transport losses [109], and providing a high electrode surface area for charge and discharge reactions [138,141,216]. The porous structure of CNT films obtained by EPD allows the electrolyte to penetrate deeply into the active electrode material, resulting in an improved specific capacitance of CNT/MnO2 composites [159,176,213,214]. Low losses are evidenced by the nearly symmetrical rectangular shape of the cyclic voltammetry (CV) curves [92,110,139,210]. In addition, the presence of a robust CNT network inhibits the structural breakdown of MnO2 particles contributing to a high stability under repeated charge/discharge cycling [159,160,176,213,214,217]. Finally, the specific capacitance can also be influenced by the use of charging additives during EPD, such as the cationic tyramine hydrochloride which enables cathodic deposition of CNTs or the anionic gallic acid for anodic deposition. As anodic deposition leads to oxidation of the usually metallic electrode and the formation of an interfacial layer with low conductivity, cathodic deposition is preferred, in many cases, for capacitor applications [160]. Effective hybrid pseudo-capacitors can also be formed by EPD of CNTs into active 3D substrates, such as anodised Al2O3, nickel cobaltite nanograss and coaxial Ni nanocables, leading to higher specific areal capacitance and improved cycling stability [218]. In general, small pores (<5 nm) contribute to an increased electrochemically active surface area, while larger pore channels (>20 nm) offer a fast transport path for the electrolyte through these hybrid architectures [97,98,144]. Figure 16 summarises the specific capacitance of different CNT-based composites developed by EPD.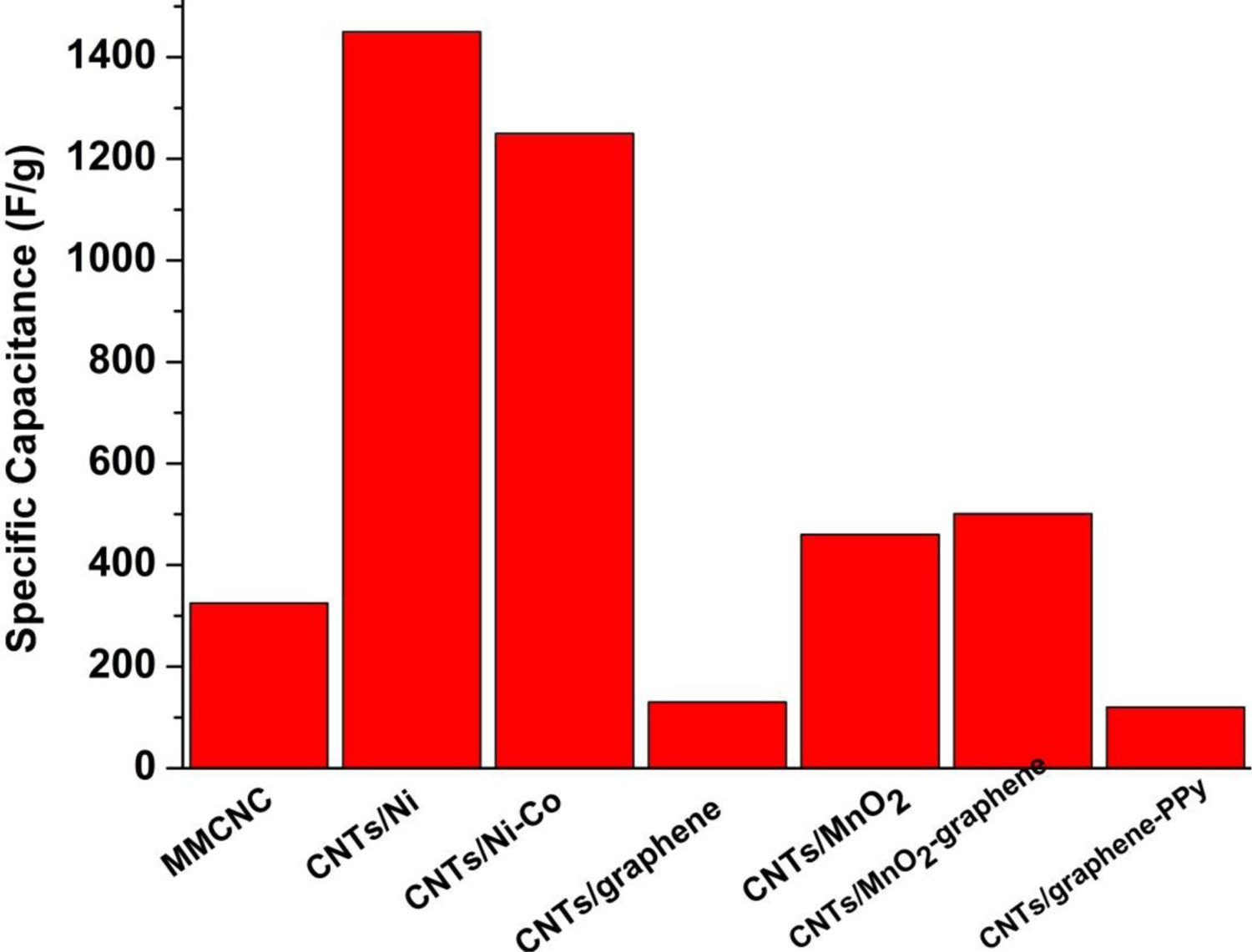
The EDLC performance of CNT-based electrodes can be improved by combining CNTs with graphene, reduced graphene oxide (RGO) [101] or activated carbon [49]. Graphene has a high intrinsic surface area but tends to re-stack, dramatically reducing access. MWCNTs can serve as spacers maintaining the interlayer distance between adjacent graphene layers to prevent their agglomeration and restacking, effectively enlarging the contact area between electrolyte and electrode. In addition, CNTs are efficient network formers and help to maintain a high electrical conductivity in the porous composite. These CNT/graphene hybrids are also efficient supports for pseudo-capacitive components, forming triphasic composite systems, for example, incorporating MnO2 [108]. CNTs can control the stacking of other electroactive 2D materials; for example, co-deposition of Ti3C2 MXene with CNTs spacers significantly enhanced the electrochemical performance [82]. It may also be possible to use CNTs to create structured electrodes for conventional dielectric materials, to create so-called 3D capacitors; BaTiO3 nanoparticles have been deposited on aligned CNTs by EPD for this purpose [188].
Fuel cells
Fuel cell research is currently focused on the development of new electrode materials with lower cost (i.e. by reducing the amount of precious metal catalyst used) while retaining electrocatalytic activity [167]. CNT films are an attractive electrocatalyst support due to their high conductivity, porosity, and high surface area. By facilitating electron, ion and mass transfer kinetics to/at the electrocatalyst interface, catalyst utilisation can be increased compared to the conventional carbon black supports [137,219]. It can be, however, challenging to obtain consistent, compact layers with a high catalyst content by means of conventional coating techniques (such as painting, spraying, printing, brushing). EPD is a fast and economical process capable of producing uniform and compact catalyst layers with controlled metal catalyst content and morphology, potentially from environmentally friendly, aqueous suspensions [137]. As such, EPD has been employed to fabricate direct methanol fuel cell electrodes from a semi-aqueous dispersion of preassembled CNT/PtRu hybrid particles stabilised by an ionomer dispersant (Nafion®) using HClO4 as the supporting electrolyte. Thin and smooth composite layers were obtained at low DC voltages, but preferential deposition of ionomer limited the fuel cell performance [137]. The ionomer binder can be avoided by performing EPD of pure CNTs followed by electrodeposition of noble metal catalysts [167] or Cu/Pd bimetallic particles [220]. Alternatively, CNTs have also been applied as a sacrificial spacer to produce ordered porous cathodes for an application in intermediate temperature solid oxide fuel cells [13]. Co-deposition of CNTs and La0.6Sr0.4Co0.8Fe0.2O3−δ (LSCF) created a uniform connected dispersion of CNTs in the ceramic matrix, which resulted in a highly porous LSCF film after removal of the CNTs by thermal oxidation.
Dye-sensitised solar cells
Dye-sensitised solar cells (DSSCs) are attractive for their potentially low-cost fabrication and relatively high photovoltaic conversion efficiency (PCE). CNTs are potential counter electrodes in such cells, as a replacement for platinum metal, offering lower cost, superior chemical stability in corrosive electrolytes, good catalytic activity and high conductivity, and significant flexibility. When produced by EPD, CNT films have an excellent uniformity and controlled thickness [12,178], and have been demonstrated within a DSSC operating at a PCE of 7.0%, comparable to the performance obtained with conventional Pt counter electrodes [12]. Mg(NO3)2 is commonly used to stabilise CNT suspensions for EPD; however, the inorganic contamination can block the catalytic sites and limit electron transfer to tri-iodide (I3 −) ions [178]. Alternatively, the combination of CNTs with RGO can produce composite coatings with a high conductivity and catalytic activity [163], for similar reasons as the hybrid EDLC electrodes discussed above. Further improvements can be obtained by adding a metal catalyst; for example, doping with Au nanoparticles increased the PCE from 6.2% to 8.8% for CNT/Au hybrid films [163]. Overall, CNT-based counter electrodes obtained from an environmentally friendly solution, at low temperature, by means of EPD can be a viable alternative over conventional DSSC counter electrodes [12]. CNTs can also be incorporated in the active photoanode to improve conductivity and reduce recombination. For example, EPD films combining CNTs with TiO2 on FTO or ITO showed improved transport of photo-generated electrons, and consequently an enhanced PCE [221].
Biomedical applications
CNTs have attracted a wide interest for use in biomedical applications, including nanostructured coatings for implants or scaffolds for tissue engineering, drug delivery systems, biosensors and electrodes for neural system stimulation [149,222,223].
Biocompatible coating systems
CNTs are widely investigated as components for coatings on implants and scaffolds in tissue engineering because nanostructured CNT surfaces show improved bio- and cytocompatibility when compared with traditional materials [190]. The nanodimensionality of CNTs corresponds to the size of proteins which promotes protein adhesion and subsequent cellular adhesion and tissue growth [120,190]. Additionally, the introduction of electrical conductivity can increase cell viability and differentiation under electrical stimulation [16,119]. EPD produces coatings of high purity to a degree which cannot be achieved easily by other processing techniques [21], on a particularly wide variety of substrate materials and complex geometries, including non-conductive and porous scaffolds, as discussed in the Deposition of carbon nanotubes section.
EPD of CNT coatings on Ti modified with TiO2 nanotubes improved the bioactivity of Ti dental implants. In combination with the TiO2 nanotubes, the uniform CNT coating not only enhanced HA formation when soaking in simulated body fluid (SBF), but also promoted osteoblast attachment and proliferation [57,224]. Similarly, co-deposition of CNTs with HA and chitosan improved the apatite formation on Ti implant surfaces [80,177]. Moreover, these and similar biopolymer/HA/CNT hybrid coatings offer effective corrosion protection for metal implant surfaces in various physiological solutions [80,166,177,225].
Optimised EPD parameters allow adequate infiltration and homogeneous coating of foam-like scaffolds (such as polyurethane) with CNT films, enhancing the bioactivity owing to the introduction of a nanostructure on the pore walls, while preserving a highly open pore network with potential for applications in bone tissue engineering [119]. CNT coatings on porous bioactive glass (BG) scaffolds did not impede the subsequent mineralisation process upon immersion in SBF, or produce any measured cytotoxic effects [16,122]. Scaffolds combining CNTs with PLLA nanofibres have been developed with a functional gradient structure mimicking the complexity of natural tissue. In principle, structures simulating different length scales in tissue engineering can greatly improve the scaffold performance [120]. On the other hand, in temporary biosensor applications, tissue integration should be limited in order to allow facile removal of the sensor after use and therefore, surfaces that inhibit connective tissue cell growth and proliferation are of interest. CNT films deposited on Ti by EPD showed good conductivity as required for sensor applications or for electrical stimulation of bone growth and were non-cytotoxic [141].
The incorporation of CNTs into various bioceramic coatings, including those based on HA [64] and BG [121], is often motivated by the need to overcome the low mechanical reliability of these brittle coating materials. MWCNTs deposited along with HA on Ti or with HA and Si on NiTi substrates significantly enhanced the adhesion strength without inducing any cytotoxic effects and were shown to even enhance osteoblast cell attachment [64,226]. Similarly, co-deposition of CNTs with HA and chitosan also improved the adhesion strength of the resulting coatings [80,177]. The addition of CNTs to an ultra-thin HA layer led to substantially improved mechanical properties, in particular in interlaminar shear strength between coating layer and implant surface [178]. Despite promising results, applications of CNTs in tissue engineering are being restricted due to the uncertain cytotoxicity in long term in vivo applications. More extended investigations are required in order to establish the cytotoxicity of different types and concentrations of CNTs [156].
Biosensors
CNTs are being considered for electrochemical biosensors due to their excellent electrochemical properties, high electrical conductivity and low overpotential, biocompatibility, high surface-to-volume ratio and high resistance to surface fouling [15,68]. Enzyme-based electrochemical biosensors, which rely on an efficient electron transfer from immobilised enzymes to the electrode, can benefit from the use of CNTs as supports and current collectors [68,227]. The large loading capacity for biomolecules on both the inner and outer surfaces of CNTs can increase the redox current [15]. EPD CNT films obtained, with a high degree of alignment and ordering, can significantly improve the electrode sensitivity as a consequence of a facilitated electron transfer process [175]. In principle, ballistic conduction could improve performance, although most commercial CNTs are too defective to take advantage of this phenomenon. Nevertheless, CNT-modified graphite electrodes had an improved stability, electron transfer rate and reproducibility for heparin detection when compared with bare graphite [228]. EPD was also shown to be a reproducible technique to prepare SWCNTs on CF electrodes with a more consistent response than other methods like drop-casting. The resulting electrodes showed a high selectivity and stability during in vivo measurements of ascorbate in rats [229].
Carboxylated CNTs (c-CNTs) offer simple surface functionalisation for direct covalent attachment of biomolecules in order to reduce the leaching that may occur with adsorption. Various organic molecules have been immobilised on EPD CNT films for high-efficiency biosensors. c-CNTs were deposited on ITO and functionalised with urease and glutamate dehydrogenase for urea detection, which led to a biosensor with a high sensitivity and wider detection range [68]. Similarly, c-CNTs homogeneously coated on electrospun Au fibres by means of EPD provided an anchor for the covalent immobilisation of glucose oxidase in a highly sensitive glucose biosensor [227]. ITO/c-CNT electrodes functionalised with monoclonal aflatoxin B1 antibodies revealed an improved detection limit for aflatoxin immunosensors [15]. c-CNT/DNA nanobiowires deposited by EPD on ITO allowed selective detection of complementary DNA with high sensitivity [175]. Polymer/CNT hybrids, such as PANI/SWCNT composite films, significantly reduced the response time and sensitivity of a tributyrin bioelectrode while ensuring an improved storage stability of 13 weeks [169]. CNTs were coated on Ti substrates and functionalised with glutamate oxidase (GOx) and lactate to produce a biosensor with a site-specific enzyme response [230]. Similar sensors have been produced using AC-EPD to deposit the CNT enzyme-support on Pt electrodes [153].
Other applications
Transparent conducting films
CNTs are emerging materials for transparent conducting films (TCFs), potentially offering lower surface resistance for a given transparency, a high degree of flexibility and neutral tint, at lost cost on a wide range of substrates. However, the cylindrical morphology, together with the presence of kinks and agglomerates, can restrict the formation of the smooth film needed for optimum performance and for many opto-electronic applications (to reduce haze) [142,231]. EPD can be an effective method for the fabrication of suitable CNT films with a smooth and homogeneous surface morphology. However, methods are needed to deposit/transfer the films onto the required insulating substrates, or to combine with other TCF strategies. EPD coatings on an, in principle re-usable, stainless steel anode can be transferred to a transparent substrate; for example, EPD CNT films hot-pressed onto to a poly(ethylene terephthalate) (PET) substrate showed strong adhesion, and the conductivity remained stable after >10,000 cycles of repeated bending [113]. Hybridisation, by co-deposition, can combine the areal coverage of graphene, with the network connectivity of CNTs, for improved TCF performance [163,164]. In an n-type Si heterojunction solar cell, PCE was nearly doubled for the hybrid compared to control devices using pure CNT or graphene films as current collectors [163].
CNTs can also be combined with other transparent conductors, such as PEDOT:PSS (poly(3,4-ethylene-dioxythiophene):poly(styrenesulfonate)). Bilayer coatings have been deposited on glass substrates via a two-step process involving CNT spray coating followed by EPD of PEDOT:PSS particles; the polymer filled the voids in the CNT film, forming a conductive bridge for electron transfer and eventually decreasing the sheet resistance of the hybrid electrodes [231]. Rather than acting as a primary conductor, CNTs can also be used to modulate other TCF solutions. For example, for an application in touch screen panels, CNTs were deposited by EPD on Ag and Cu nano-meshes, substantially reducing both reflectance and ageing through oxidation [142,232].
Catalyst supports
CNT networks are often considered as substrates for heterogeneous or hetrogenised catalysts; providing a stable, high surface area material, with excellent reagent accessibility. One example is the use of CNT networks to support hydrogen storage and evolution systems: a four-layer hierarchical catalyst support was produced by EPD of CNTs on a CF gas diffusion layer, followed by pyrolysis of an adsorbed polymer-derived SiCN and electrodeposition of a transition metal film. This system showed an exceptionally high hydrogen generation rate with a high stability over multiple cycles, attributed to covalent bonding between the catalyst and the CNT/SiCN support [233].
Black body absorbers
Despite the transparency in thin-film form, CNTs are extremely effective broadband light absorbers; as thick layers, they can form some of the blackest materials known, particularly where porosity is controlled or graded to minimise surface reflection. Large area thick films can be used as optical baffles for sensitive instruments or, when patterned, as elements of miniaturised integrated photonic devices. EPD has been used to deposit optically absorbing CNT films on tantalum substrates (Ta). To enhance absorption in relatively thin (∼1 µm CNT layers), a resonant cavity structure, was defined by adding a Ti/SiO2 multilayer. The structures may be used for sensing, antenna and thermophotovoltaics (TPV), offering advantages in bandwidth, compactness and cost [234].
Solid-phase microextraction fibres
Solid-phase microextraction (SPME) is a solventless extraction technique used to capture dilute analyte molecules from aqueous or gaseous phases within a coating material. In this concentrated form, the compounds can be easily presented for separation and analysis by chromatographic processes [235]. CNTs deposited onto a fibre or wire can form a porous coating with high surface area for large sample capacities and fast mass transfer characteristics [236]. Besides low cost, easy control over coating thickness and scalability of the process to complex shapes, EPD can avoid binding agents that might reduce the adsorption capacity of the resulting SPME fibres [70,236].
SWCNTs have been applied on a Pt fibre via EPD from a DMF suspension without the use of adhesives [70]. The coatings showed good stability in organic solvents without swelling or stripping from the substrate; the physical robustness was attributed to the strong Van der Waals attractions between the SWCNTs. After optimisation of the desorption conditions, which included pH, ionic strength and extraction time, the conductive SPME fibre enabled the extraction of phenols from seawater and tap water with detection limits and recoveries similar or superior to commercial polyacrylate fibres [70]. An additional heat treatment at 500°C in an H2 atmosphere following EPD significantly enhanced the extraction of benzene, toluene, ethylbenzene and xylene from aqueous samples to efficiencies similar to commercial carboxen polydimethylsiloxane fibres. In addition, a high thermal stability, up to 350°C, allowed direct transfer of the analytes into the injection port of a gas chromatography (GC) instrument for sensitive quantitative analysis. The same fibre was successfully reused up to 120 cycles, confirming excellent durability of the coatings. After thermal treatment, the coatings showed a high degree of reproducibility, offering a reliable alternative to commercial fibres [71]. Similarly, a stainless steel wire coated with MWCNTs by means of EPD from an aqueous suspension stabilised by SDS served as SPME fibre and was used for GC detection of polycyclic aromatic hydrocarbons from aqueous media with improved performance compared to commercial polyacrylate and polydimethylsiloxane fibres [237]. Furthermore, in contrast to commercially available silica-based SPME fibres, the SWCNT-coated Pt fibres showed excellent stability in strong organic desorption solvents used in high-performance liquid chromatography (HPLC), enabling an accurate quantitative analysis of the extracted compounds. As such, EPD-based SPME fibres were successfully coupled with HPLC for the sensitive and reproducible detection of several endocrine-disrupting compounds in water samples [73]. In addition, the conductivity of the SWCNT-coated Pt substrates, which is preserved upon EPD, opens up opportunities for electrosorption-enhanced SPME in order to selectively extract ions and ionised compounds by the application of a potential to the SPME substrates [70,72]. A positive potential applied on SWCNT-coated Pt plates prepared by EPD allowed trace anions to be extracted from water significantly enhanced efficiencies than conventional methods, over 50 cycles [72].
CNT/polymer nanocomposites are also being considered as coatings for SPME fibres. MWCNTs applied on stainless steel fibres by EPD from a DMF dispersion were coated by a sol–gel-derived polymer film and showed high mechanical strength, good stability and long service life for the GC detection of polycyclic aromatic hydrocarbons from aqueous samples [236]. In another study, MWCNT/PANI hybrid precipitates were applied on a stainless steel wire by EPD. The fibre base was first platinised in order to provide a larger and coarse surface for an improved coating adhesion. The resulting SPME fibre in combination with HPLC allowed the recovery and analysis of thymol and carvacrol from aqueous solutions with improved performance compared to commercial fibres [148].
Wastewater treatment
Zero-valent iron (ZVI) can initiate a series of spontaneous reactions in water, leading to the removal/destruction of a variety of contaminants from groundwater. The introduction of a high surface area cathode can improve this ‘internal-electrolysis’ efficiency. CNTs with an excellent electrical conductivity and high specific surface area are a promising alternative to conventional carbons for use with ZVI. EPD can be used to deposit CNTs onto the surface of ZVI, followed by calcination to fix them in place. Depending on the optimal coating thickness, such hybrid ZVI-CNT materials considerably enhanced the reaction rates in water as demonstrated for the degradation of methylene blue as a model compound [143]. The catalytic activity of CNT/Ni hybrids for the decoloration of methyl orange may follow a similar mechanism [99]. These ZVI composites can also serve as a pH adaptable ozone catalyst, showing high catalytic performance either in acidic (pH 3) or basic (pH 9) conditions [238]. A more conventional approach to water purification uses the photocatalytic activity of titania to drive the destruction of organic contaminants through reduction by photoexcited electrons. The creation of CNT/titania hybrid coatings can improve activity by improving electron–hole separation and reagent access. Adjusting the CNT content of co-deposited EPD films through the composition of the mixed suspension, allowed the photocatalytic properties to be optimised [66].
Sensors
CNTs/PDMS bilayers containing localised regions of highly loaded CNT networks have been fabricated by EPD of CNTs into a patterned mould followed by transfer-micromoulding. Such structures exhibit high sensitivity to both transverse compressive and tensile forces, and may be used to monitor local pressure/structural changes in tissues or micro-fluidic systems [151]. Co-deposition enabled the production of nanocomposite networks of MWCNTs/CuO/Cu2O on polyethylene terephthalate sheets, for which the film resistance was linearly correlated with the bending angle. After optimisation of the EPD parameters, a highly sensitive angle sensor with a durability up to 1000 bending cycles was realised [239]. Alkali metal-doped CNTs deposited into the pores of the Ag foam showed a significantly enhanced photoconductivity when compared with pristine CNTs. These results indicated that as-prepared alkali doped CNTs can be used as a light sensor without the necessity of separation between metallic and semiconducting CNTs [240].
Discussion and outlook
This review comprehensively summarises the latest developments related to the use of EPD to process CNTs in a great variety of structures/material systems for numerous applications. The literature highlights the advantages of EPD as a technique to produce CNT structures, but also the challenges that need to be addressed in order to optimise CNT deposition for different applications. The first problem is to identify a source of suitable CNTs, which manifest the required properties, and then to form a suitable suspension without unwanted damage or contamination. The deposition parameters must then be developed to control the physical and chemical characteristics of the film deposited on the substrate. Nevertheless, many promising strategies for CNT EPD have already been demonstrated for specific contexts.
The deposition medium affects the stability of CNT suspensions and the nature of subsequent deposits obtained by EPD. One of the most popular pathways in an aqueous medium relies on strong oxidation of the CNTs in 3:1 HNO3:H2SO4. However, this approach induces structural damage in the tubes, which cannot be entirely recovered, and is only appropriate for certain applications. Organic media such as IPA, ethanol and DMF have been frequently used to produce stable CNT suspensions. Depending on the inherent ability of the organic medium to donate electrons, the zeta potential of CNTs can either be positive or negative. However, many simple CNT suspensions, especially without acid treatment, show poor stability during EPD. In many cases, additives such as metallic ions, metallic salts, organic salts, cationic dyes and covalently linked molecules are introduced, not only to stabilise the suspensions, but to co-deposit functional hybrids.
In some applications, co-deposition of other micro or nano-sized particles and/or polymers alongside CNTs is essential. This hybridisation can be accomplished during EPD, via sequential deposition (layer by layer) or co-deposition of CNTs and other contingent particles. The later process is of interest as it offers flexibility in the composition of the deposited materials; however, the EPD mechanisms are relatively more complex than in the case of single-component deposition, and less well understood. For instance, the additives used for stabilising multicomponent suspensions need to be compatible with all components. On the other hand, co-deposited particles may also act as stabiliser elements for the dispersion of CNTs. Triphasic composites are increasingly popular, for example, combining CNT/polymer/ceramics or different types of nanocarbons. Low voltages are usually recommended, especially when dealing with aqueous suspensions to limit the unwanted hydrolysis of water. Modulated current EPD can be useful to suppress hydrolysis of water at higher voltages; however, this strategy has not been widely used yet in the context of CNTs and may be a useful future direction.
While many aspects of the deposition mechanism are understood, further research is needed to understand the detailed microstructure that forms under different process conditions. Hamaker’s law provides a simple predictive framework for designing the EPD process, but is rarely applied in the literature. Hamaker’s law typically describes a linear relationship between the EPD parameters and the deposited mass. Deviation at long deposition times or at high applied voltages are very common, and can indicate the changes in deposit conductivity, dispersion composition, dispersion aggregation state or the influence of side reactions involving either the deposit or medium. In addition, the high aspect ratio of CNTs and their ability to orient in an applied field complicate the usual simplifying assumptions about electrophoretic mobility. These factors should be explored in more detail to identify the limits to EPD stability and rate, which may largely determine the commercial viability of CNT films. Models predicting the porosity, anisotropy and even properties of CNT networks formed through EPD would be useful for accelerating research and developing robust industrial processes.
EPD of CNTs has been used commercially to fabricate activated carbon (AC)/CNT-based supercapacitor electrodes and CNT-coated CF reinforcements on an industrial scale. Yet, commercial and large-scale development of CNTs applications via EPD remains limited. As the cost of the CNT raw material continues to fall, a wider range of applications are likely to become economically feasible. In particular, relatively thin-film coating applications may be good candidates for developing valuable benefits with relatively small quantities of CNTs. Many applications exploit CNTs as a superior carbon black, with improved accessible surface area, conductivity and robustness. However, other advanced applications, exploiting the fundamental properties of CNTs, especially SWCNTs, continue to emerge. The enormous research effort, over the last decade, has demonstrated the versatility and utility of EPD for CNT and CNT-composite or -hybrid films. The groundwork has been laid, both for developing new systems designed for specific applications, and scaling existing systems to a practical scale.
Footnotes
A.B. acknowledges the receipt of a starting grant from Internal Funds KU Leuven (STG/17/024) and a travel grant from “Fonds Wetenschappelijk Onderzoek - Vlaanderen” (K225418N). Q.C. acknowledges support from the National Natural Science Foundation of China (31800802) and Fundamental Research Funds for the Central Universities (China) (3102017OQD041).
Disclosure statement
No potential conflict of interest was reported by the author(s).
