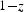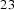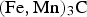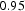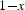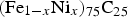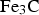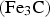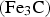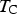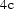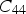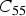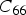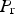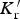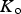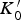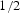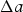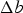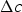Abstract
Cementite occurs in steels, in meteorites, possibly at the core of the Earth and has uses in its pure form. It's composition can deviate from 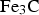

Introduction
In its crystalline, liquid and glassy states, iron has an affinity for carbon in its many forms, whether to form a solution over a wide range of compositions, or in the form of compounds with narrowly defined compositions, such as cementite. It is possible, therefore, to find equilibria between iron and graphite, iron and diamond and iron and cementite, represented conventionally by the respective binary, two-phase diagrams. Such diagrams identify domains, for example, in temperature and composition space, where either a single phase or a combination of phases is stable. However, the term stable is a tenuous concept because there might be something else also consisting of Fe and C, which may be more stable. Instead of considering just two phases together, if we now put iron, graphite and cementite in mutual contact at ambient pressure then the cementite eventually must give way to the more stable equilibrium between graphite and iron. All equilibria in this sense are metastable; even the constituents of atoms will all decay eventually if the Universe keeps on expanding.
Nevertheless, some 50 million tonnes of cementite is produced annually within about 1.6 billion tonnes of steel, adding enormously to the quality of life. This is because it is hard at ambient temperature, as we shall see, due to its crystal structure that has a much lower symmetry than all the forms in which the iron occurs. Its metastability mostly does not matter over the time scale and conditions of normal life. The longest single-span suspension bridge in the world, the Akashi-Kaikyo Bridge, utilises exceptionally strong ropes to suspend the deck. The bridge connects Kobe with Awaji Island and has a span of 1.9 km between the towers. There is enough steel wire used in the bridge to circle the earth seven times, with the bridge being designed to withstand an earthquake of Richter 8.5 magnitude. The bridge represents a magnificient triumph of engineering and steel containing substantial quantities of cementite, without which the ropes would be nothing short of feeble (Figure 1). The Akashi-Kaikyo Bridge in Japan, the longest single-span suspension bridge, which relies on huge cables made from pearlitic steel. Photograph courtesy of Professor Nobutaka Yurioka.
In spite of its metastability, we shall see that cementite is found in meteorites that have cooled at a few degrees per million years, and within diamonds found deep in the bowels of the Earth. It perhaps has played a seminal role in the genesis of carbon nanotubes from gaseous reactions. There is fledgling work to indicate that nanoparticles of cementite may have a useful purpose in biomedicine for the site-specific delivery of healing drugs.
This is a review about cementite as a phase in its own right. How was its chemical composition established given that the nature of carbon inside steel could not have been understood in the very early days of metallography? In 1878, Müller [1] dissolved some steel in dilute sulphuric acid to leave behind a black residue which when analysed contained 6.01–7.38 wt-% carbon. Müller referred to this as amorphous iron. Comprehensive experiments done independently by Abel around 1883 were published in 1885 in a report, on the state of carbon within steel [2]. This confirmed ‘the correctness of the conclusions based on earlier experiments, that the carbon in cold-rollled steel exists in the form of a definite iron carbide, approximating the formula Fe
The name has its origins in the theory of Osmond and Werth, in which the structure of solidified steel consists of a kind of cellular tissue, the iron constituting the nucleus and the carbide the envelope of the cells 1 [3,4]. The carbide was therefore envisioned to cement the iron.
In mineralogy, the carbide is known as cohenite (Fe,Ni,Co)
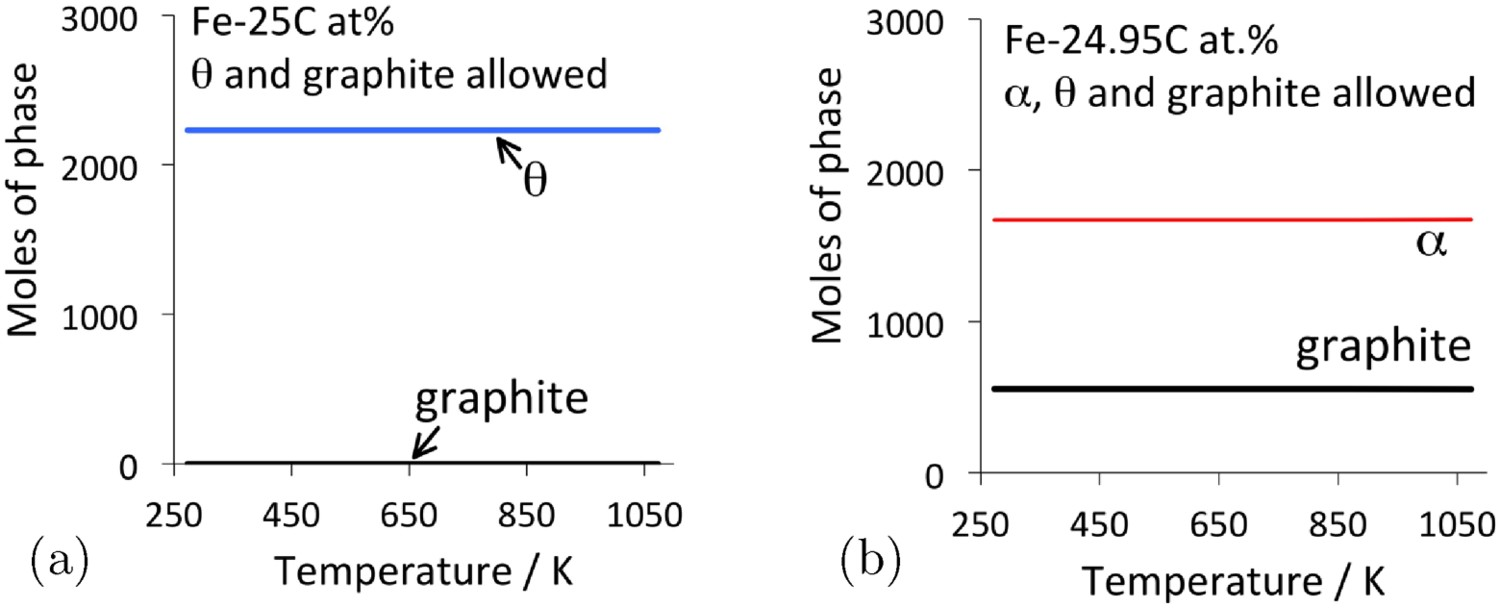
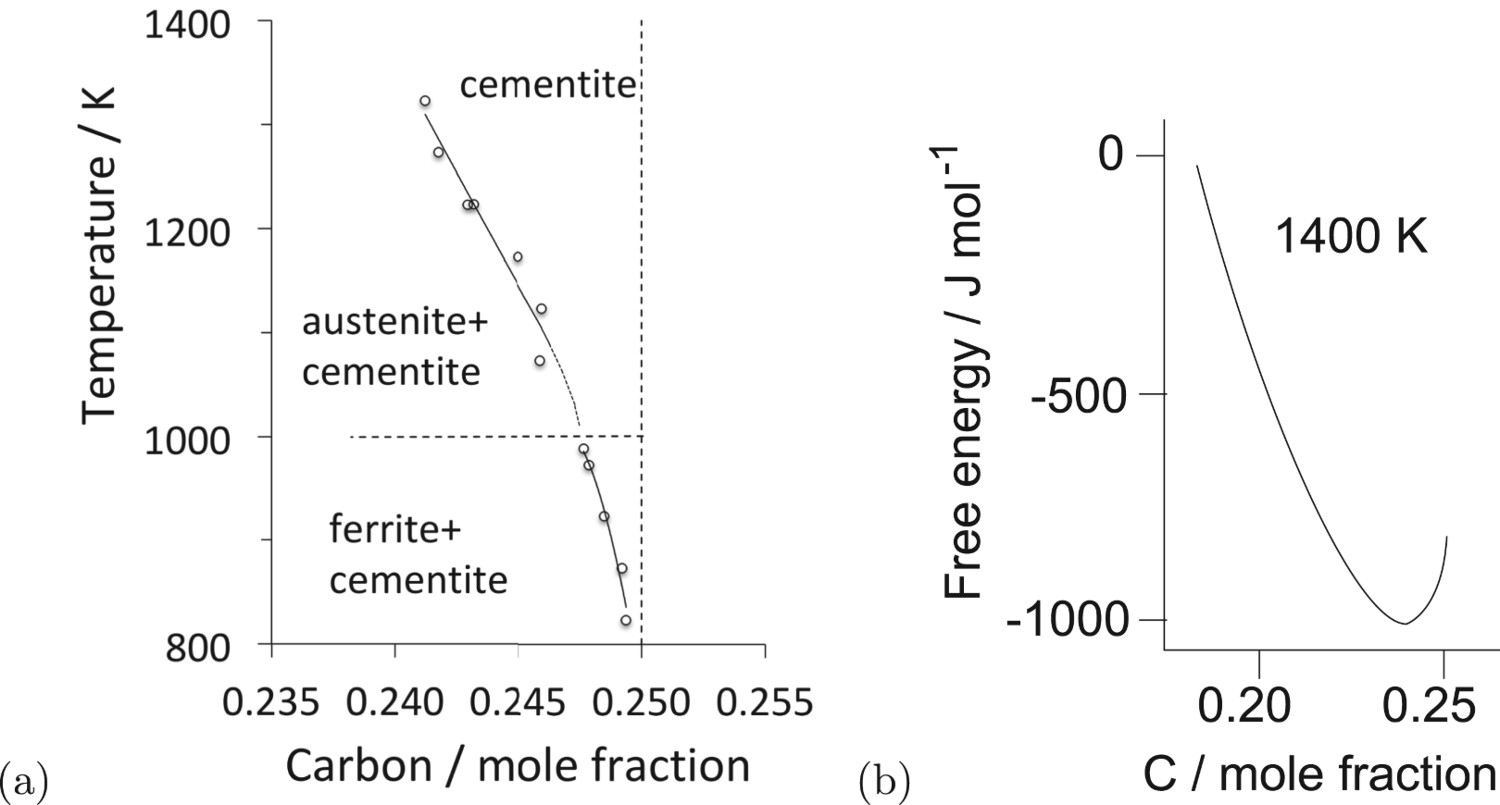
Cementite often is said to be metastable with respect to graphite. However, as shown in Figure 2, pure cementite when allowed to coexist only with graphite, is stable in the presence of graphite, presumably because the iron does not dissolve in graphite. When ferrite on the other hand is allowed to coexist with cementite and graphite, the stable mixture at equilibrium becomes ferrite and graphite. These calculations are consistent with observations on carburised iron, where the cementite in contact with ferrite decomposes more rapidly to graphite during heat treatment, than cementite that is lodged within coke [6]. Nanoparticles of cementite that are surrounded by a thin shell of carbon remain stable as cementite during heat treatment at 700 Phase diagram calculations or 100 kg total weight, using MTDATA [11] and the SGTE thermodynamic database. (a) Fe–25C at.-%, permitting only cementite and graphite to coexist. (b) The average carbon concentration is reduced slightly to allow ferrite to appear, in which case the most stable mixture becomes that of ferrite and graphite.
The carbon atoms in cementite are located in interstitial sites [12,13]; any deficit from the 3:1 Fe:C atom ratio is attributed to interstitial vacancies that normally are occupied by carbon atoms, as inferred from lattice parameter changes [14]. The specific volume of cementite that is in equilibrium with ferrite at ambient temperature is found to be greater than that calculated using its measured lattice parameters, indicating vacant carbon sites, i.e. a deviation from the stoichiometric composition [15]. Similar conclusions have been reached by measuring phase fractions and lattice parameters in rapidly cooled Fe–C alloys containing large carbon concentrations [16]. Indeed, the detailed changes in three lattice parameters of cementite quenched from different temperatures have been shown to be consistent qualitatively with corresponding parameters calculated using ab initio methods where carbon-specific sites are left unoccupied [17].
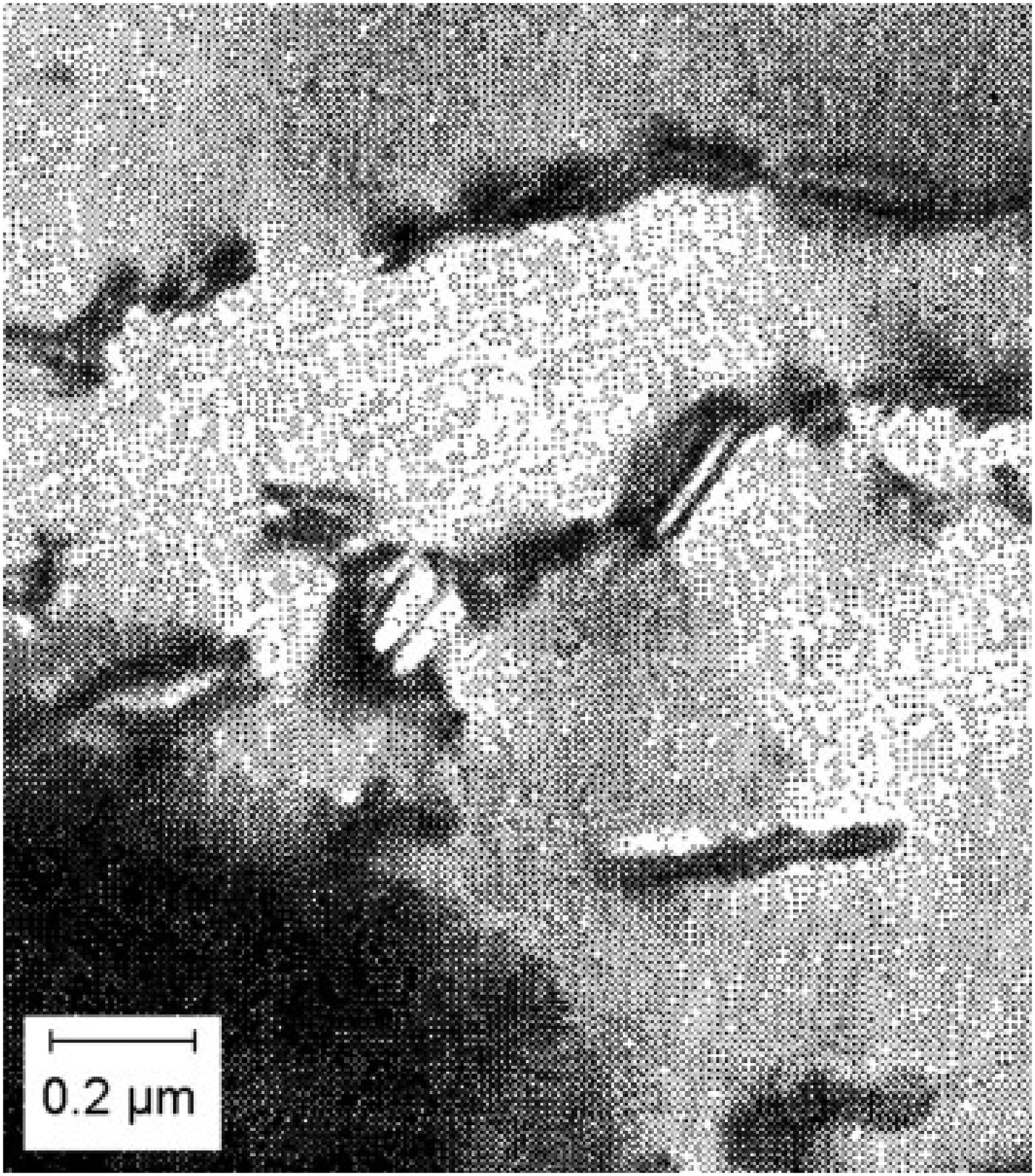
Figure 3(a) shows the thermodynamically assessed phase boundaries between cementite (a) The composition of cementite that is in equilibrium with austenite or with ferrite in an Fe–C alloy. The data are due to Leineweber et al. [17], determined by measuring the lattice parameters of cementite following quenching from the appropriate temperature. (b) Free energy curve of cementite as a function of chemical composition (referred to γ-Fe and graphite). After Gohring et al. [18]. Precipitation of fine platelets of ferrite from cementite. Reproduced with permission of Taylor and Francis from [19].
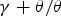
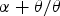
Any deviations from stoichiometry must be small because as demonstrated by Cottrell [20], the bond energy between a carbon atom and iron is greater than that between two iron atoms. Therefore, any deficit of carbon would lead to a reduction in cohesion. Any extra carbon beyond the 3:1 Fe:C ratio would need to be accommodated in less-favoured interstices within the cementite lattice. The nature and energetics of the different kinds of interstitial sites within the cementite structure are discussed later (Section 3.1).
Circumstances can be engineered to make the cementite deviate from the stoichiometric carbon concentration; the decarburisation of pure cementite [21], which leads to changes in the volume of the unit cell and in the Curie temperature of cementite, is an example. The deviation tends to be small, typically Fe
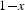
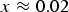

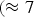
Ambient pressure measurements of the Curie temperature of cementite as a function of its carbon concentration. Data from Walker et al. [22], determined by making cementite as a part of a transformer. Choe [23] reports a somewhat lower
of 167.6
C determined using a superconducting quantum interference magnetometer for ambient pressure.
An interpretation [24] of the change in magnetic properties as a function of pressure attributes the phenomenon to the volume-dependent two-state theory for the high magnetic moment to small-volume low moment transition. Using an X-ray technique and diamond anvil equipment, it has been determined experimentally that the loss of ferromagnetism occurs at about 10 GPa. The change in volume required to induce the magnetic transition is about 5% [24,25].
The atom probe permits the composition of cementite to be measured directly using time-of-flight mass spectroscopy. There are, nevertheless, difficulties in measuring the carbon concentration of cementite [26]. It has not yet been possible to demonstrate small deviations from stoichiometry using such high-resolution methods. However, using conventional atom probe field ion microscopy, extremely small (4 nm) cementite particles in severely deformed mixtures of ferrite and cementite have been shown to contain only 16 at.-% of carbon, a concentration that recovers to the 25 at.-% when the mixture is annealed to reduce the defect density and coarsen the cementite [27]. It is argued that the deformation introduces defects such as vacancies into the cementite, leading to the reduction in carbon concentration. However, it is important to note that the particles containing such a large deviation from stoichiometry were not proven to retain the orthorhombic crystal structure.
One study, based on neutron diffraction intensities measured over at temperatures ranging from ambient to 800
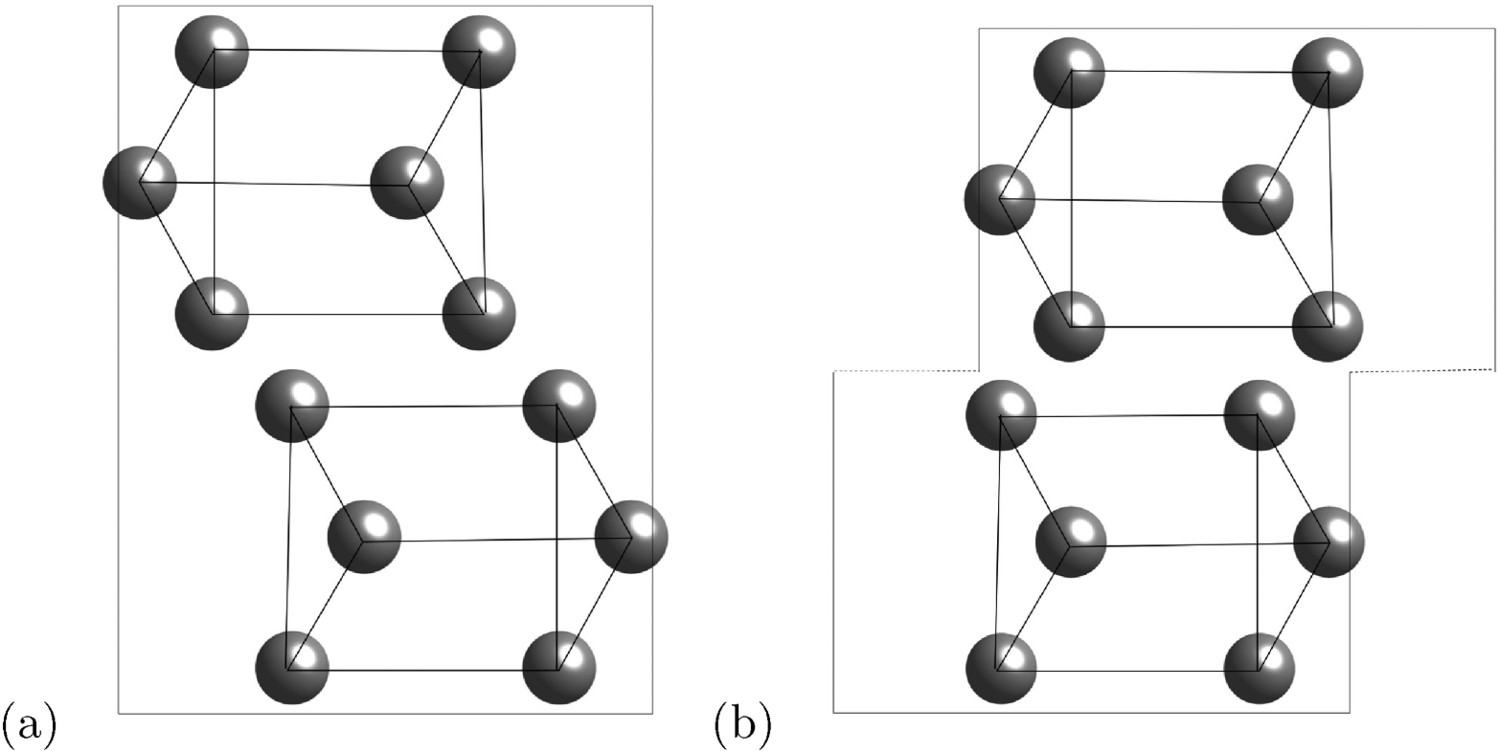
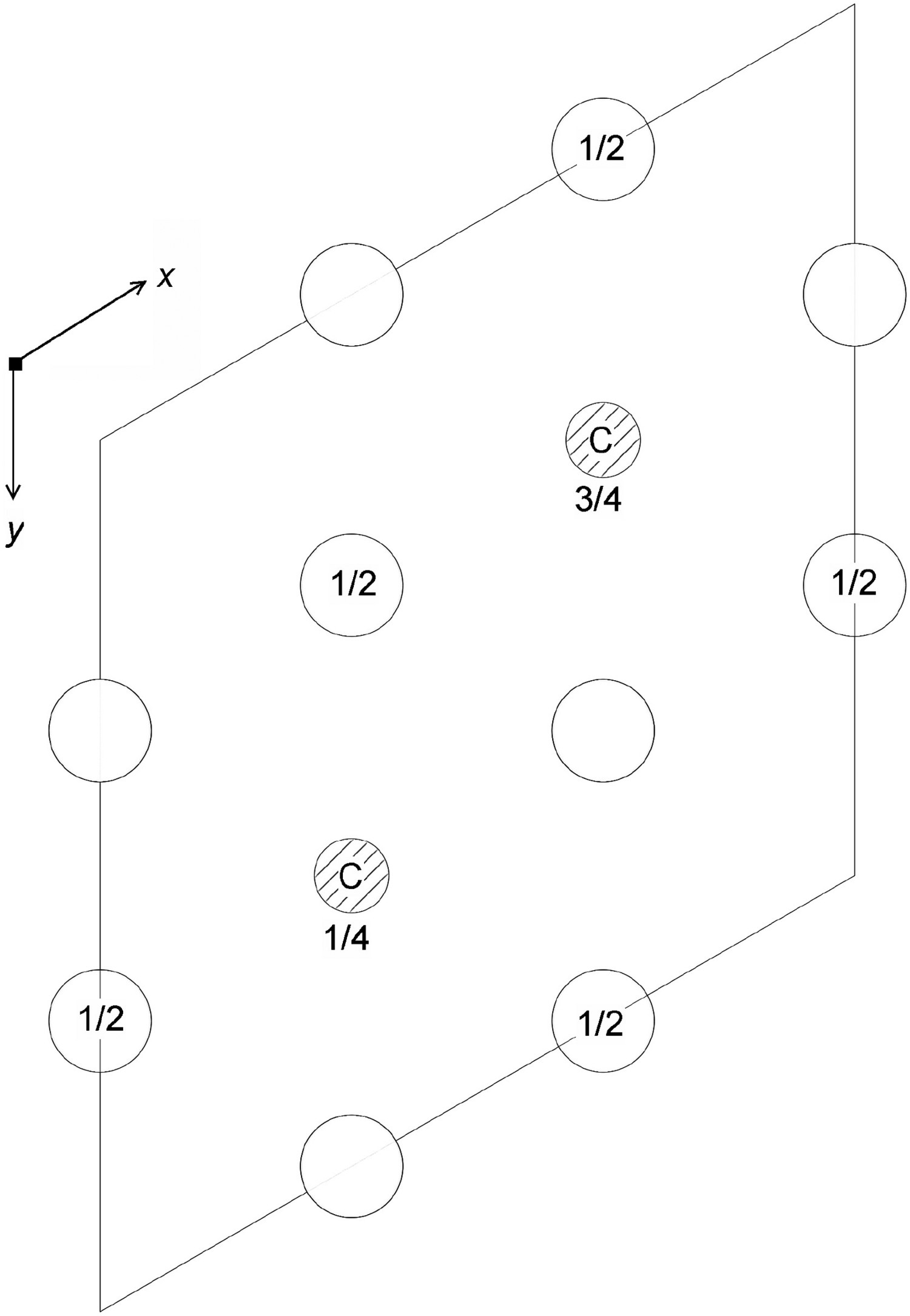
Cementite has an orthorhombic unit cell and the common convention is to set the order of the lattice parameters as a = 0.50837 nm, b = 0.67475 nm and c = 0.45165 nm. There are 12 atoms of iron in the unit cell and 4 of carbon, as illustrated in Figure 5. Four of the iron atoms are located on mirror planes, whereas the other eight are at general positions (point symmetry 1). The crystal structure of cementite, consisting of 12 iron atoms (large) and 4 carbon atoms (small, hatched pattern). The fractional z coordinates of the atoms are marked. Notice that four of the iron atoms are located on mirror planes, whereas the others are at general locations where the only point symmetry is a monad. The pleated layers parallel to (100) are in 

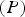
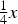
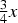
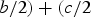
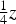
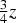
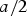
Wyckoff positions for space group Pnma.
Note: Table of space group symbols, July 2018, http://www.cryst.ehu.es/cgi-bin/cryst/programs/nph-wp-list.
There are prismatic, octahedral and three kinds of tetrahedral interstices in the cementite unit cell; if the space within each is defined from the centre of the interstice to the boundary of the nearest iron atom, then the sizes are 0.71, 0.53, 0.34, 0.26 and 0.28 Å [34]. The centres of the prismatic interstices lie on mirror planes so there are four per cell (4c, Table 2) and they all are filled with carbon atoms in the stoichiometric form of cementite [13]. The smaller octahedral interstices, of which there are four per cell (4a, Table 2), are empty in pure cementite unless the carbon concentration exceeds 25 at.-%, and the tetrahedral interstices are too small to be occupied by carbon. When hydrogen enters the cementite lattice, it locates in the octahedral [35] interstices because the prismatic ones are occupied by carbon (Figure 6). Two kinds of interstices in the cementite unit cell. (a) Prismatic. (b) Octahedral.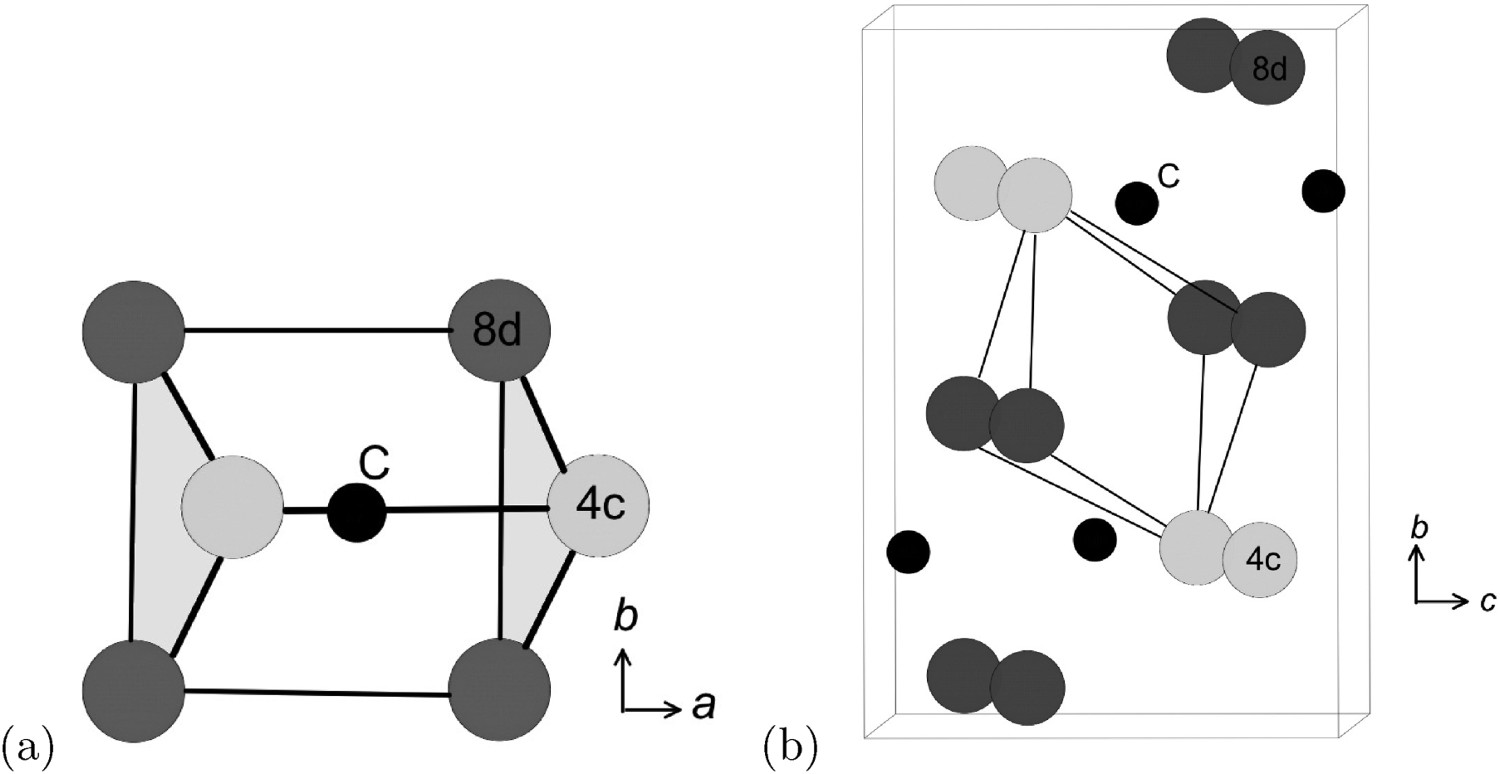
Given the orthorhombic structure, the elastic moduli of cementite vary with the direction within the crystal [36]. The shear modulus 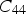
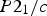
The experimentally observed slip systems in cementite include 
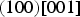
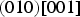
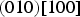
The common slip system appears to be 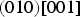
The stress relaxation of cementite at 1250
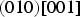
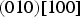
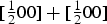
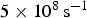
Planar striations can sometimes be observed in cementite, particularly when it precipitates in the solid-state. Nishiyama et al. [48] identified stacking faults on (010)θ using transmission electron microscopy, involving translations by vectors parallel to [100]θ that are not lattice vectors (Figure 7). It is known that dislocations with the Burgers vector equal to the lattice vector [001]θ, which lie in (010) slip plane, are not in general dissociated except when they lie in the (130)θ plane [49]. More complex faults occur on other planes. Cementite that grows at low temperatures can contain planar defects that are identified as two-layer thick regions of transition carbide χ-Fe Creation of a stacking fault on (010)θ by a partial displacement parallel to [100]θ that does not recreate the lattice. Carbon atoms have been omitted for clarity. (a) Unfaulted structure. (b) Faulted structure. Adapted from [48].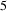

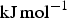
Calculated formation energies for point defects in cementite, referred to ferromagnetic bcc iron and diamond as the reference states. Data from the 128 atom simulations by Jiang et al. [53]. The mole fraction of carbon in cementite is denoted x. For example, placing an iron atom in a prismatic interstice leads to a carbon concentration that is less than 25 at.-%.
Simulations show that cascades produced by irradiation can lead to vacancies in the iron sites, and anti-site defects where iron atoms lodge in interstitial positions [54]. These point defect concentrations tend to be greater than those that occur in ferrite under the same conditions [55]. Similar simulations suggest that the moduli of cementite are affected by irradiation, but the lattice parameters used do not seem to be correct [56].
A hexagonal form of cementite (Fe Projection of the possible crystal structure of the hexagonal form of cementite, using the parameters and symmetry proposed by Nagakura [57]. The fractional z-coordinates of atoms not located at z = 0,1 are marked. The carbon atoms (hatched) are located in a third of the octahedral interstices formed by the iron atoms.
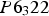

The original structure proposed by Jack [58,59] was rather different than that of Nagakura, with the iron atoms in a hexagonal close-packed arrangement and carbon atoms in octahedral interstitial sites, with chemical composition Fe
It is not clear whether a hexagonal form of cementite with a composition Fe
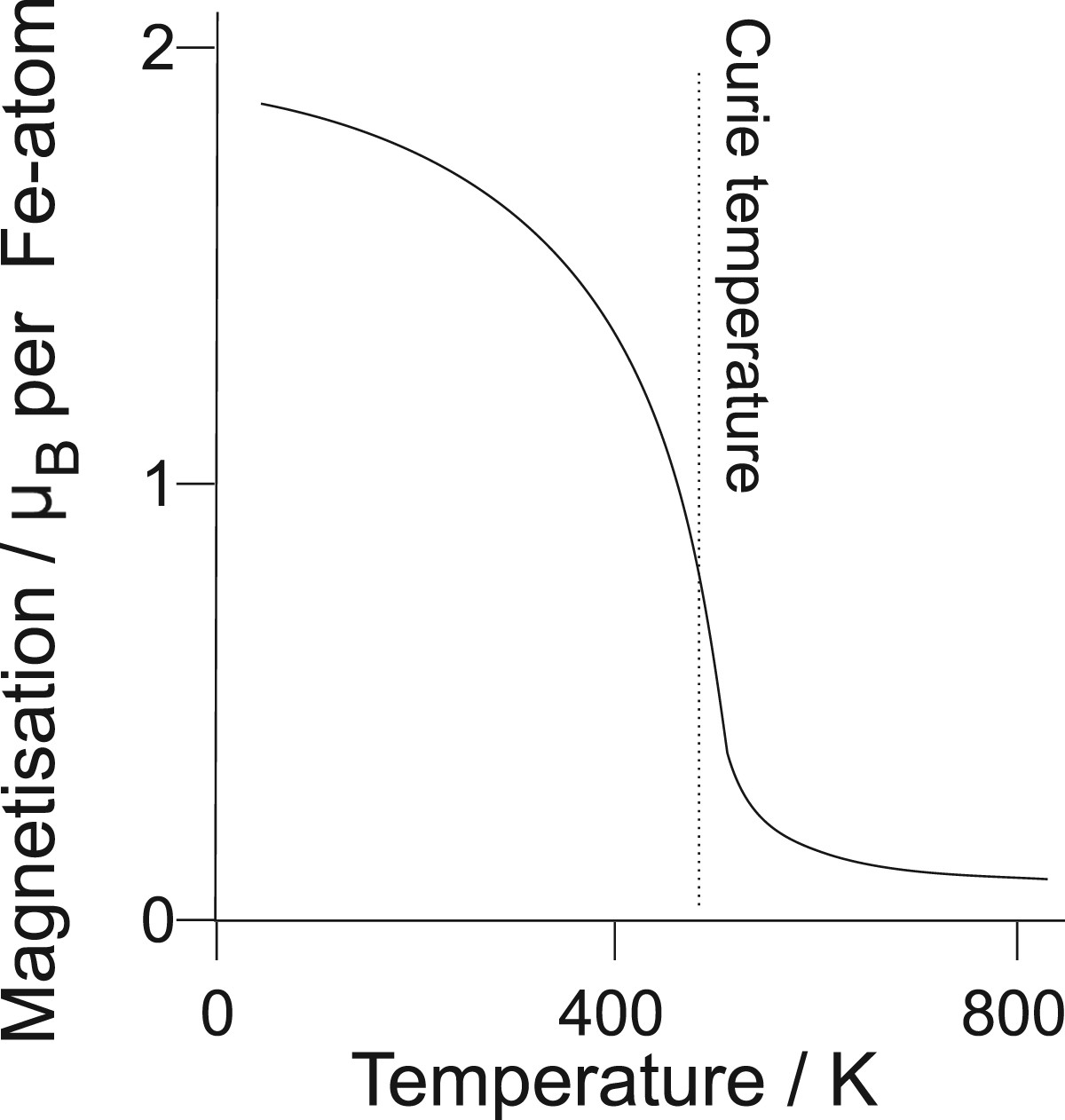
Cementite at ambient pressure and room temperature is a metallic ferromagnet that becomes paramagnetic beyond the Curie temperature 





The calculated magnetisation of cementite as a function of temperature is illustrated in Figure 9 [68], where the average magnetisation at 0 K is about 1.86 Calculated magnetisation of cementite as a function of temperature, after Dick et al. [68].
There is a transition from ferromagnetic to nonmagnetic states at 25 GPa pressure and 300 K [73]. The term nonmagnetic is used here because it is not clear whether the magnetic collapse corresponds to a loss of spin correlation or to a transition from a high-spin to a low-spin state. There is a volume contraction of 2–3% following the transition from the ferromagnetic state. The structure, with its orthorhombic symmetry, is magnetically anisotropic, with [001]θ, [010]θ being the easiest and second easiest, and [100]θ the hardest magnetisation directions [23,74,75]. The magnetocrystalline anisotropy energy is 334 Magnetic domain structure of cementite. Reprinted from [76], with the permission of AIP Publishing.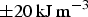

The effect of manganese goes beyond the dilution of the magnetic moment per atom when manganese substitutes for iron [81]. Calculations for The orthorhombic unit cell with eight metal atoms in the 8d positions (circles), four in the 4c locations and four carbon atoms (small circles). The magnetic structures are from calculations representative of 0 K. (a) Ferromagnetic cementite of composition Fe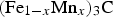




Cementite exhibits a magnetocaloric effect [82]. During adiabatic demagnetisation, the alignment of magnetic spins decreases. Since the total entropy remains constant during the adiabatic conditions, the increase in magnetic entropy on the removal of the applied field is compensated for by a decrease in temperature. If demagnetisation occurs isothermally, then the change in magnetic entropy leads to a corresponding change in total entropy. Measurements indicate an adiabatic change in temperature of 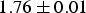
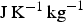
There is a single report [83] of two modifications of cementite, one ferromagnetic and the other paramagnetic, coexisting at ambient temperature. This conclusion was based on the interpretation of Mössbauer spectra. There has been no follow-up on this observation or any theoretical interpretation since the original publication.
The average thermal expansion coefficient of polycrystalline cementite changes from The linear thermal expansion coefficient of polycrystalline cementite as a function of temperature and magnetic state. Adapted using data from Umemoto et al. [64].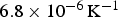
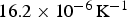
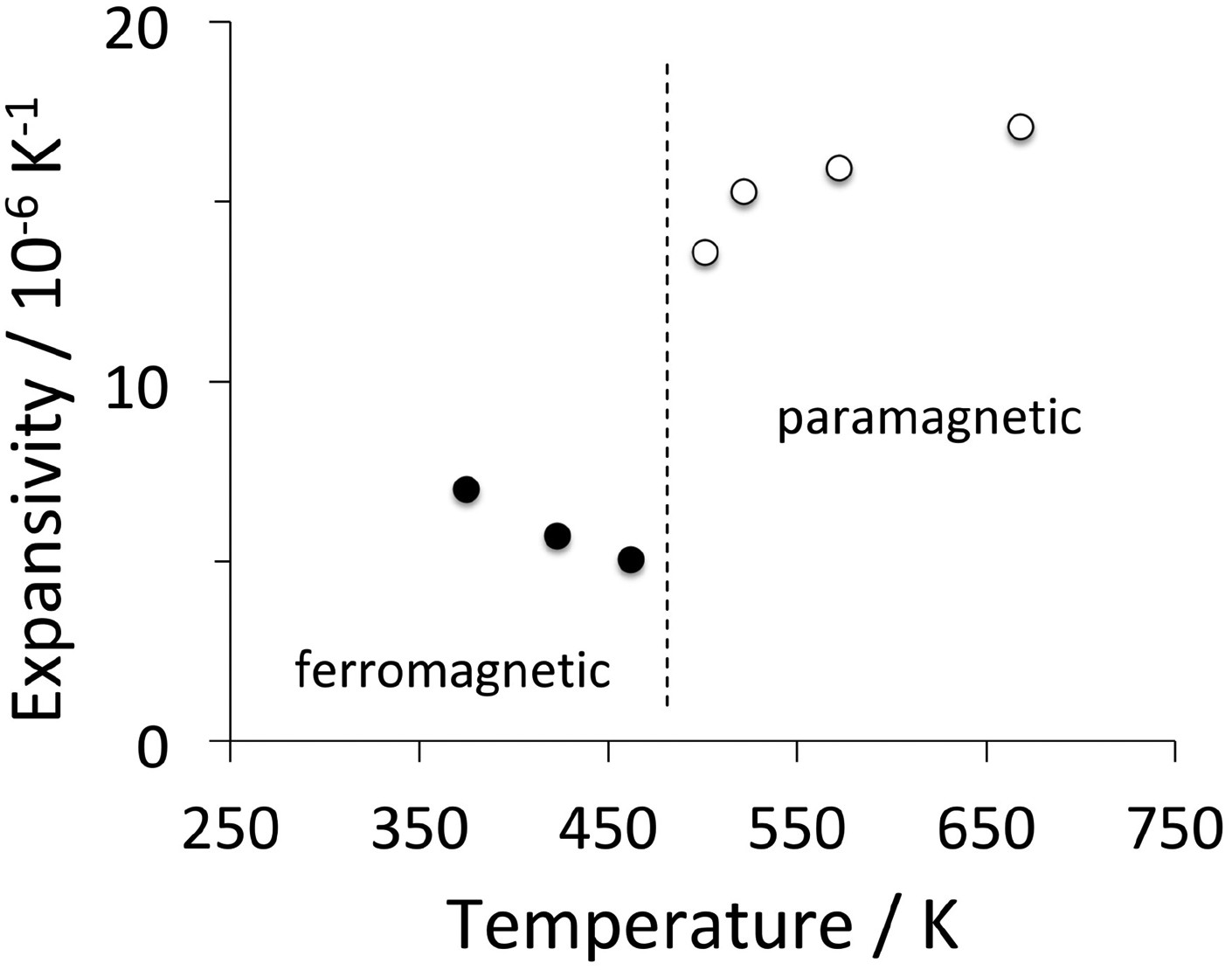
Figure 13 shows diffraction data [84–86] for each of the lattice parameters of cementite as a function of temperature. The parameter a is most sensitive to the change from the ferromagnetic to paramagnetic state, with a contraction evident as the temperature is raised within the ferromagnetic range. An increase in the amplitude of thermal vibrations in an anharmonic interatomic potential causes expansion, but the spontaneous magnetisation leads to a contraction, and this latter effect dominates the a parameter below Neutron and X-ray diffraction data on the three lattice parameters a, b and c of cementite as a function of temperature. Data from [85] (small circles with error bars), [84] (filled circles) and [86] (crosses). The dashed line in each case identifies the Curie temperature. The calculated pressure dependencies of the lattice parameters are as follows [87]: 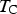
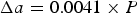
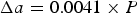
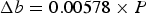
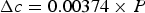
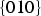
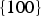
Calculated surface energies of cementite in a vacuum. A further broadly similar set of data calculated using interatomic potentials is available in Reference [89] but the authors concerned did not express confidence in the relative values of the moduli.
Calculated surface energies of cementite in a vacuum. A further broadly similar set of data calculated using interatomic potentials is available in Reference [89] but the authors concerned did not express confidence in the relative values of the moduli.
First-principles calculations of the elastic moduli are presented in Table 6; the anisotropy is illustrated on stereographic plots for cementite and for comparison, Mn Stereographic projections showing the variation of calculated single-crystal elastic moduli as a function of orientation, for the setting Pnma. (a) Fe

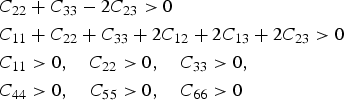
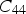
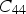
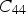
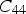



Modulus data for cementite (space group Pnma) at 0 K and zero pressure unless otherwise indicated: the nine independent, calculated elastic stiffness constants (GPa) using the stress-strain method, for 0 K.
Many of the first-principles calculations assume that the composition of cementite is exactly Fe Experimentally determined plot of pressure versus density for polycrystalline cementite. The gradient increases with density, indicating an increase in the bulk modulus with pressure. Data from Fiquet et al. [101].
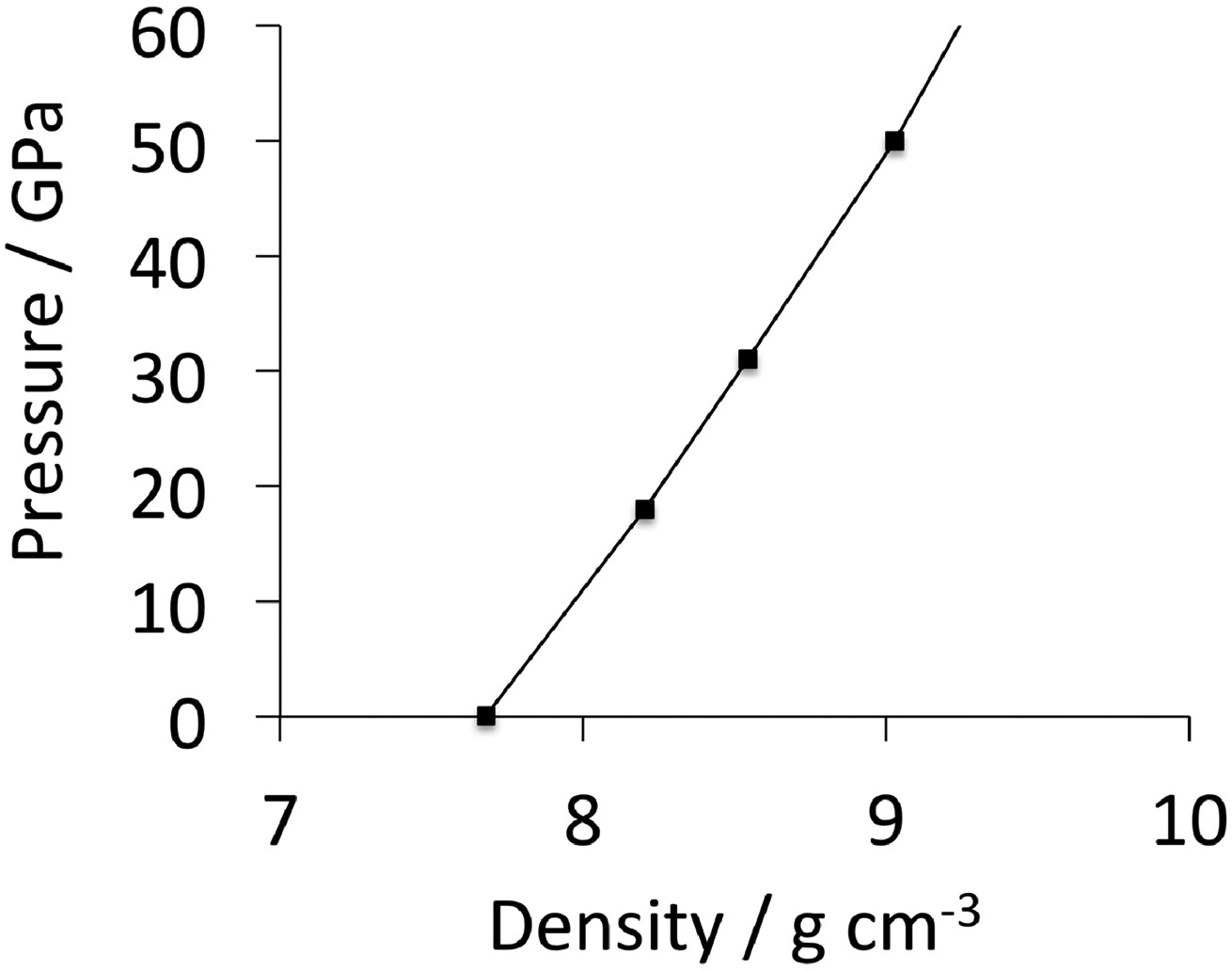
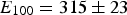
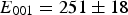
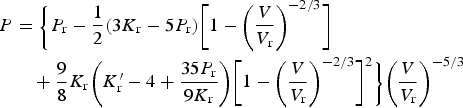
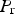
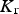
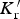
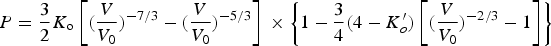
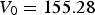
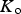
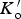
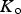
The data from first-principles calculations of single-crystal elasticity can be used to estimate the elastic properties of polycrystalline cementite by assuming uniform stress (Reuss) or uniform strain (Voigt) throughout the cementite [109]: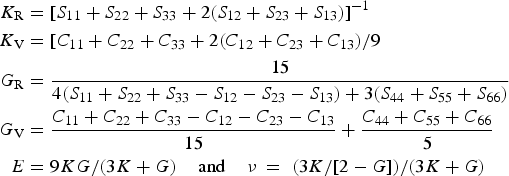
Samples of bulk, pure cementite are difficult to prepare given that cementite in contact with iron is less stable than the corresponding equilibrium between graphite and ferrite. The largest samples have been manufactured by mechanical alloying in experiments by Umemoto et al. [113]. Powders of iron and graphite in the correct stoichiometric ratio are milled together, resulting in a solid solution, as indicated by very broad (a) A sample of cementite, courtesy of Professor Minoru Umemoto of Toyohashi University. (b) Reaction of 80 wt-% Fe and 20 wt-% graphite for 10 min at the temperatures and pressures indicated. Selected data from [115].
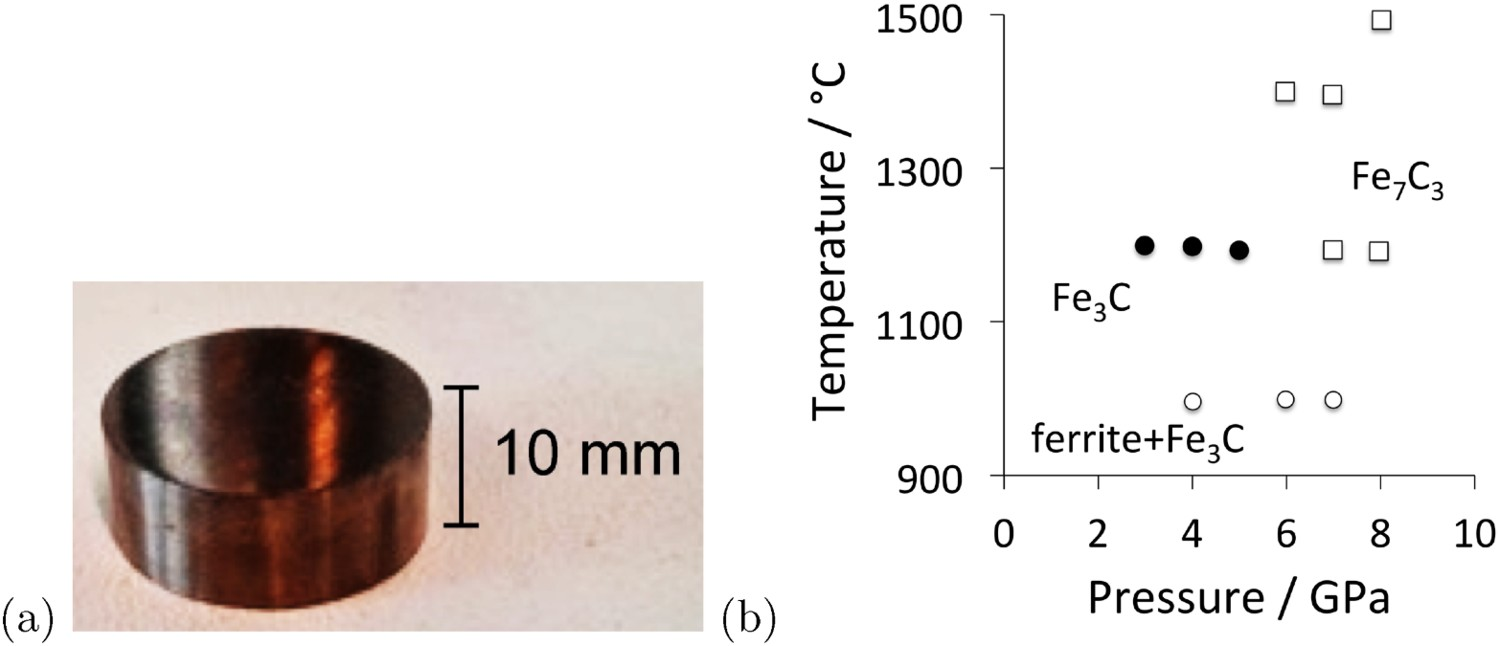
The sintering step has been unnecessary in other work where cementite was obtained directly during the milling process [116–118]. This might be explained by the fact that Umemoto et al. [113] milled their powders for a much longer time. A comparison of the 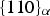
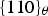
It has been proposed, based on evidence from Mössbauer spectroscopy, that there are intermediate stages between the formation of the solid solution during milling, and that of cementite. The process may first involve transition carbides such as Hägg (Fe


A clever method [75] for fabricating a ‘single crystal’ of cementite is to incorporate electrolytically extracted cementite particles into a resin which then is subjected to a 10 Tesla magnetic field for some 24 h with the composite periodically rotated in the field to magnetically align the particles as the resin solidifies. This enabled the magnetocrystalline anisotropy of the cementite to be determined experimentally.
Iron can be converted into cementite by exposing it to a carburising gas mixture, if the activity of carbon relative to graphite is maintained at greater than one. Graphite is deposited preferentially unless the surface of the iron is contaminated with blocking atoms such as sulphur, in which case cementite is precipitated [124]. It has been demonstrated that cementite can be made by carburising magnetite (Fe

Nanoparticles of cementite can be prepared by the thermal decomposition of Fe(CO) Cementite nanoparticles produced using laser pyrolysis of a gaseous mixture. Reproduced from Morjan et al. [129] with permission from Elsevier.
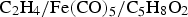
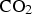
Cementite powder containing pores about 20 nm in size from an aqueous mixture of iron chloride, colloidal silica and 4,5-dicyanoimidalzole. The dicyanoimidalzole is the source of carbon when the mixture is heated to 700
Electrical conductivity data from first-principles calculations, measurements made on cementite-containing microstructures and on pure cementite are compiled in Figure 19. The large difference between the calculated value and measurements is attributed to the fact that a real material is likely to contain defects that reduce electrical conductivity. The Umemoto data on pure, bulk cementite are from its polycrystalline state [113]; it is not clear why those due to Lee and Simkovich [132] correspond to a much lower conductivity, although it is noted that the sample preparation methods for the two studies are different. The fact that the electrical resistance (i.e. reciprocal of conductivity) increases with temperature confirms that cementite is metallic rather than a semiconductor [132].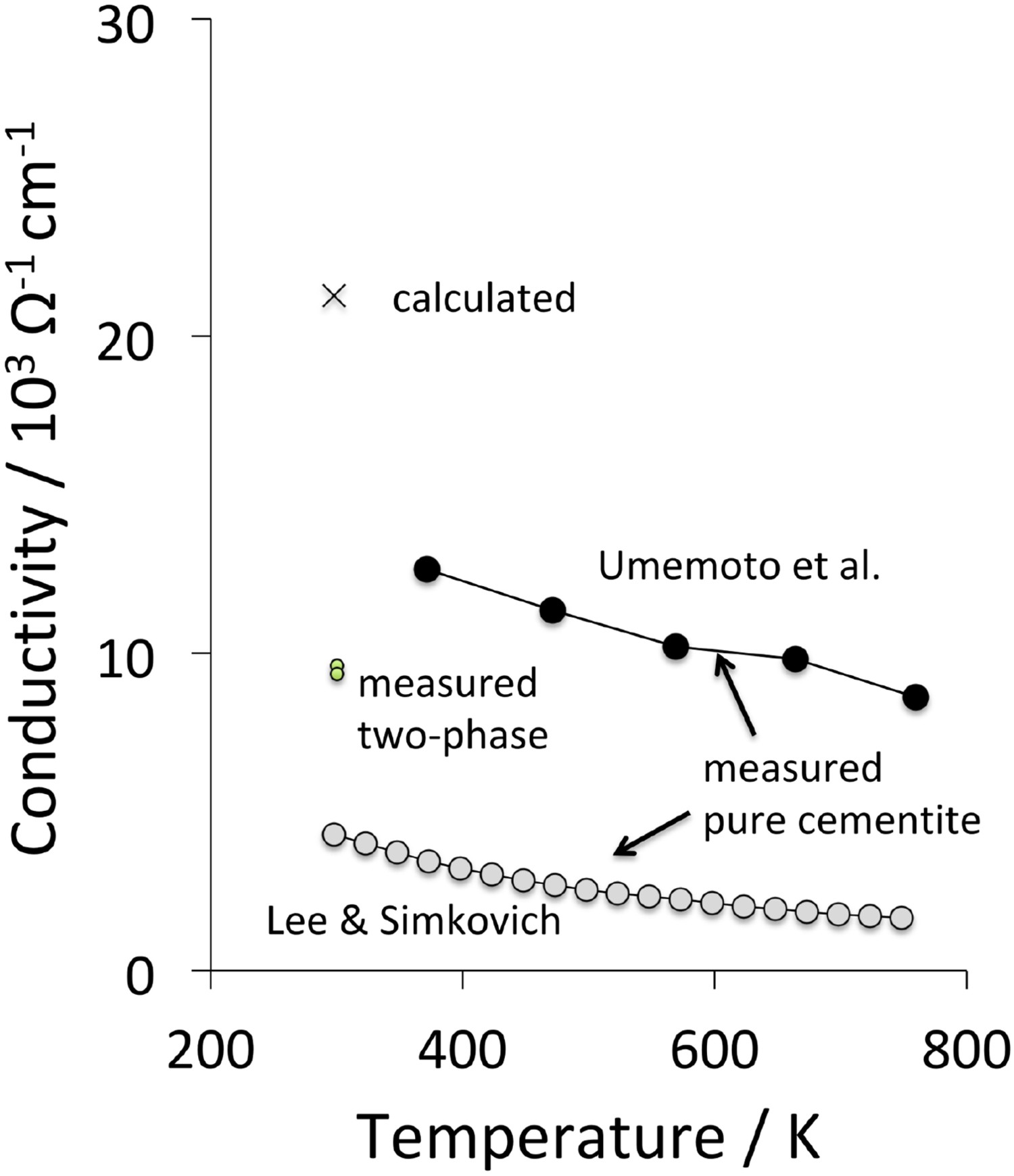
The ideal strength of cementite, i.e. in the absence of dislocations, can be estimated using first principles methods. An increment of strain is applied to a unit cell which then is allowed to relax both in shape and atomic positions, so that only the stress along the applied direction is non-zero. The stress corresponds to the derivative of the free energy with respect to strain. The maximum in strength is given by an instability when an inflexion occurs in the free energy versus strain curve. Calculated data using this method are illustrated in Figure 20; as expected, there is considerable anisotropy in properties. Calculated ideal values of ultimate strength at corresponding engineering strains for perfect crystals of cementite. (a) Tensile deformation along crystallographic axes parallel to the cell axes. (b) Shears on the planes and directions indicated. Uniform colour indicates data from Garvik et al. [51] whereas the cross-hatched bars are from Jiang and Srinivasan [37].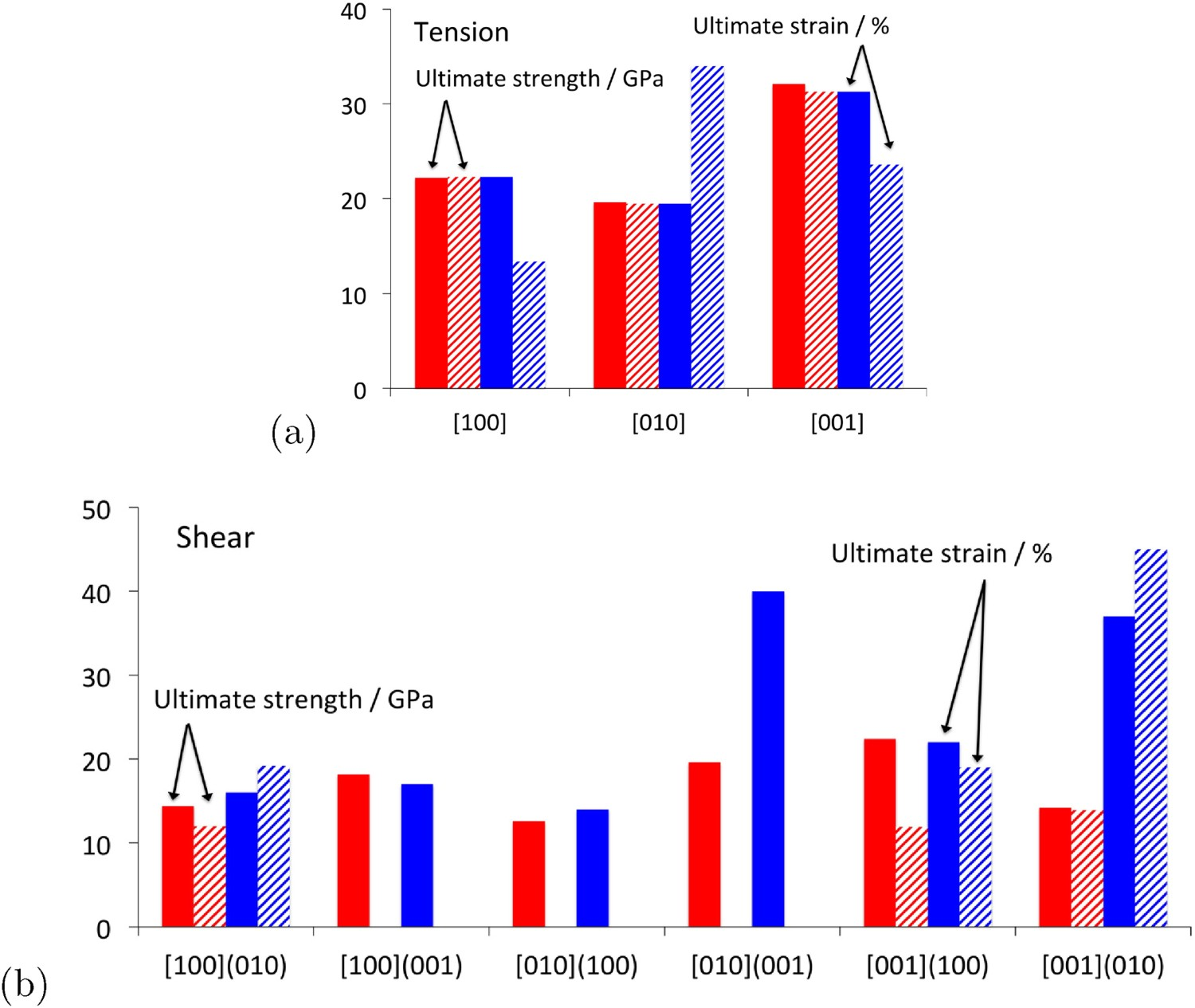
Early experiments designed to measure the strength of cementite were confined to small samples extracted from high carbon steel. Ribbon-like samples of cementite 1–2 μm thick and 1 mm long when tested by bending gave strength estimates in the range 4.6–8 GPa [134]. Experiments on 2.5μm thick films of cementite with a grain size of about 50 nm, on samples prepared using plasma vapour deposition, revealed a microhardness of about 1230 HV at ambient temperature, decreasing to about 650 HV at 673 K [110]. Young's moduli measured on polycrystalline thin films range from 160–180 GPa. More comprehensive hardness data are shown in Figure 21; it is clear that hardness measurements from bulk cementite samples prepared by mechanical alloying and spark plasma sintering, are systematically lower than some measurements made on eutectic cementite within cast iron, or prepared by plasma synthesis. In the latter case the grain size can be as small as 50 nm which may add some strengthening, but the intrinsic resistance to plastic deformation due to the Peierls barrier is likely to be a much greater contribution. The sintered samples all contain some porosity which can reduce the strength [135], however, the data from Kagawa and Okamoto [136] from cementite in cast iron, are remarkably consistent with those on bulk cementite (Figure 21). There is an intriguing study by Drapkin et al. [137] where primary cementite was found to be much harder (≈ 1070–1350 HV) than eutectic cementite (≈ 740–960 HV) in cast iron; these data are unexplained. (a) The Vickers hardness of cementite. The data for pure cementite, filled circles and crosses, are from Umemoto et al. [113] and Kagawa & Okamoto [136]. Umemoto's data are from bulk cementite, Kagawa's from cementite within cast iron. Those for cementite containing chromium at the concentrations are from Zheng et al. [138]. The curves represent measurements on cementite in cast iron, alloyed with an unspecified amount of chromium or boron [136]. The hardness values may be underestimates due to some porosity in the samples. (b) Ambient temperature microhardness data for cementite within a cast iron microstructure [139,140] and from plasma deposited Fe

Indentation fracture toughness of cementite present in cast irons at ambient temperature [141]. The hardness data measurements include elastic strains and hence will be underestimated. The chemical compositions represent energy-dispersive X-ray analysis data of the cementite phase alone. The indentation toughness of cementite of unspecified composition in a nickel-containing cast iron has been reported to be 4.09
MPa m
although the reported hardness of 1340 HV is quite large [140].
Cementite in steels is often touted as helping resist wear in a variety of applications such as bearings and rails [143–145] where the carbide is present as a minor phase embedded within a matrix of ferrite or martensite. Figure 22 shows the reciprocal of the wear rate for a variety of samples of ferrite containing different amounts of cementite [146], including data for pure cementite made by mechanical alloying followed by plasma sintering. The reciprocal wear rate is given by PL/V, where P is the applied pressure, and V is the volume of material lost as the pin of the sample material rubs against a rotating alumina disc over a distance L. Data from the abrasion of a pin made out of the material of interest, against an alumina disc. After Sasaki et al. [146].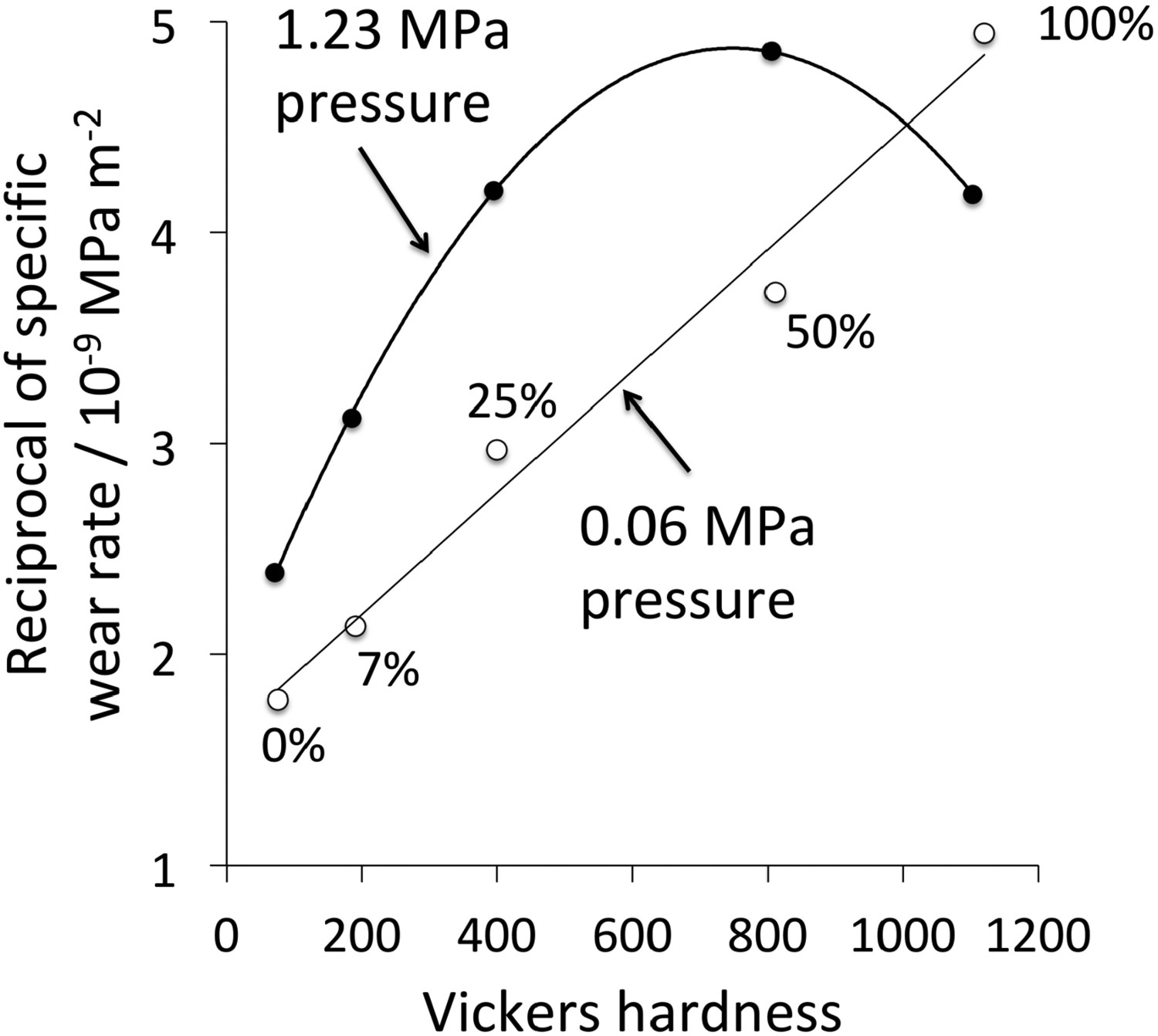
As concluded by Sasaki et al. [146], the data show that at low pressures, the wear rate decreases as the amount of cementite in the sample increases, as long as the abrasion process does not lead to the formation of large brittle chips of cementite. At the high pressure, the brittle chip formation makes the pure cementite wear more rapidly than at low pressures.
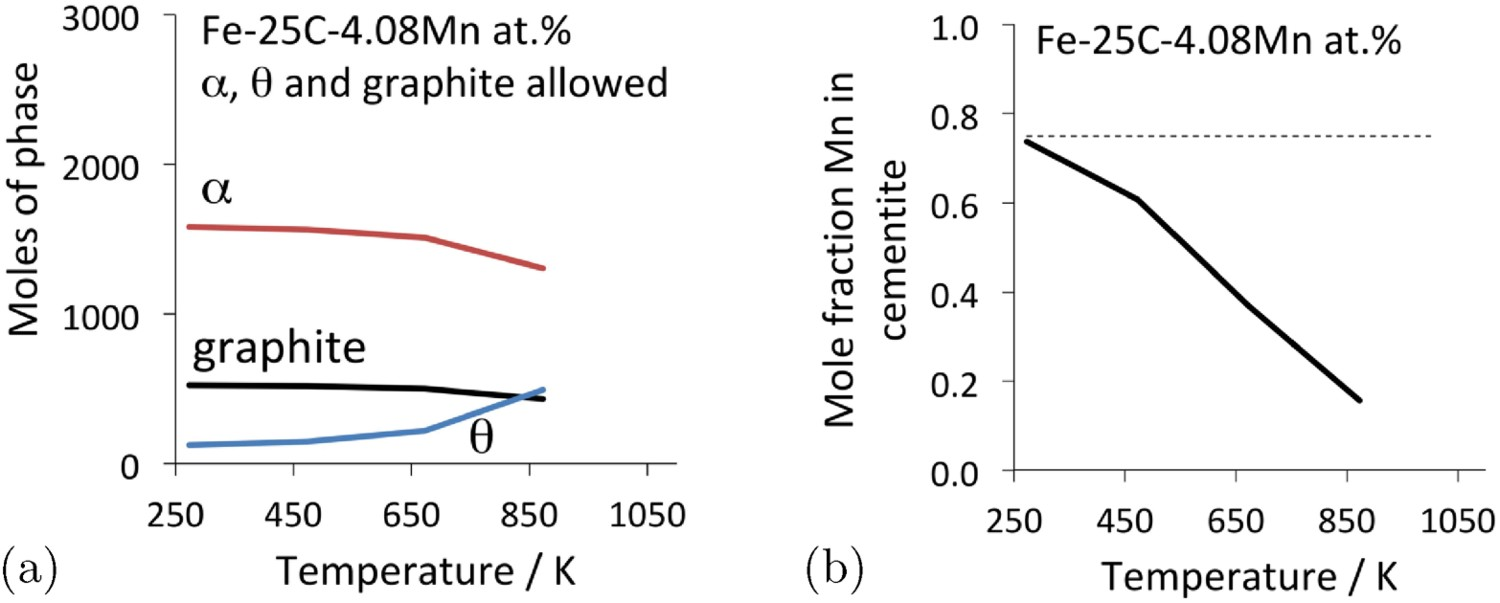
Alloying cementite with manganese reduces the rate at which it might decompose into graphite [147]; it has been known for some time that cementite becomes more stable when it ‘unites with manganese’, sometimes resulting in the growth of robust single-crystals known as Speigeleisenkristall [148]. Figure 23(a) shows that the addition of manganese permits cementite to co-exist with graphite and ferrite, whereas in the same circumstances, a Fe–25C at.-% steel would, at equilibrium, consist only of a mixture of ferrite and graphite. The cementite in the Fe–C–Mn alloy contains manganese, the equilibrium composition of which at low temperatures is more akin to Mn Phase diagram calculations for 100 kg total weight, using MTDATA [11] and the SGTE thermodynamic database. Fe–25C–4.08Mn at.-%, permitting cementite, graphite and ferrite to co-exist. (a) Equilibrium phase mixture as a function of temperature. (b) The equilibrium manganese concentration in cementite for the calculations presented in (a).

Chromium hardens cementite, presumably by solid solution strengthening; Figure 21 shows that the effect is not large. Therefore, the effect of chromium additions on the wear resistance of cementite is also minimal, when the comparison is limited to data for single-phase cementite (i.e. neglecting samples with Cr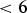
The magnetic properties are affected in line with expectations, i.e. substituting an atom with a lower magnetic moment reduces the saturation magnetisation [78,151]. Iron atoms in cementite have local magnetic moments of 1.97 
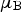
There are circumstances where nickel-containing cementite has a favourable free energy of formation [156]. (Fe,Ni) The free energy of formation associated with the reaction 

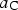

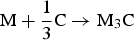
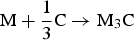

Chromium has a strong affinity for carbon and hence when alloyed in cementite, makes the latter more stable with the free energy of formation decreasing systematically with concentration [80,157]. Zhou et al. [158] have published data for chromium in cementite showing similar trends although the absolute values of formation energy are much greater, even for pure cementite. Manganese too is a carbide former, and once some complex magnetic effects (Section 4) are accounted for, leads to a thermodynamically more stable cementite. A compilation of data on a variety of solutes affecting the formation energy of cementite at 0 K are presented in Figure 25. There is a significant variation in The calculated formation energy 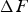
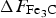
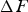
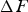
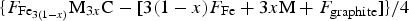

Figure 25 shows that silicon reduces the stability of cementite, a fact that is of considerable technological significance. It often is added to steel to suppress cementite while ensuring that the concentration is small enough that graphite does not form during the fabrication or use of the alloy [160]. If cementite is suppressed, then any carbon partitioned due to ferrite formation ends up in the austenite, thus permitting it to be retained and enhance ductility and toughness. The influence of silicon on the precipitation of cementite is substantially greater when the matrix phase is supersaturated austenite, because the driving force for precipitation from supersaturated ferrite is much greater [161]. The effect of silicon on cementite has been exploited for many decades in the design of steel microstructures [162–170]. A detailed review, covering both steels and cast irons, can be found in [171].
Boron, which is a larger atom than carbon but much smaller than iron, lodges within the prismatic interstices when it substitutes for carbon, thereby causing the volume of the unit cell to increase (Figure 26); this location is consistent with the lack of change in the relative intensities of the X-ray diffraction peaks when pure cementite and that containing boron are compared [173]. Figure 26 shows that large concentrations of boron can be introduced into cementite without changing its orthorhombic symmetry. The saturation magnetisation increases with the boron concentration, as does the Curie temperature which reaches 578 The lattice parameters of Fe



Helium atoms have closed 1s electron shells and hence interact repulsively in compression. When in cementite, the He will therefore tend to locate in the largest available spaces. Calculations indicate that it prefers to substitute for the iron rather than carbon atoms in cementite; the energy needed to substitute C, Fe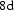
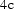
Change in the lattice parameters of cementite (Pnma) as a function of the concentration in wt-%. The coefficients are derived from the work of Kagawa and Okamoto [177].
The are no data on the solubility of copper in cementite; the usual phase diagram calculation packages indicate zero solubility, but this may simply be a reflection of the absence of appropriate thermodynamic data as inputs. There are now observations showing the precipitation of copper or copper-rich precipitates within cementite [178], as opposed to precipitation at the 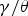
Thermodynamic properties
There are significant calculations of the heat capacity of cementite, using a combination of density functional theory and quantum Monte Carlo methods [68]. These permit the individual contributions of phonon, electronic and magnetic components, with the total heat capacity in good agreement with thermodynamic assessments based on the CALPHAD method [172]. The calculated values of the different components are illustrated in Figure 27. Polynomial functions describing the free energy of cementite as a function of temperature have been derived by Hallstedt et al. [172]. CALPHAD type data on cementite are widely available in the literature so are not reproduced here, other than in context where they add to insight or interpretation. The calculated components of the heat capacity of cementite as a function of temperature at zero pressure; adapted from Dick et al. [68].
It has been known for some time [182–184] that cementite is metastable with respect to the equilibrium between graphite and α-iron for all temperatures below the eutectoid in the iron-carbon binary phase diagram. Graphite and γ-iron saturated with graphite form a more stable mixture than cementite and γ-iron for higher temperatures. On the other hand, if cementite and α-iron can somehow coexist at temperatures above the Fe–C eutectoid, then free energy of formation data indicate that the mixture would be stable relative to α-iron+graphite, Figure 28. The formation energy 
The data presented in Figure 28 are essentially consistent with first principles calculations as far as the temperature dependence of 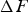
Cementite presumably is easier to nucleate in the solid-state than graphite, hence its ubiquitous in its metastable form. One consequence is the phenomenon of metal dusting, associated with the formation of cementite due to the desorption of gases such as CO on the steel surface at an elevated temperature (400–800
The formation of graphite leads to a large expansion in volume:
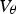
Samples of cementite have been prepared by the reaction of Fe



Cementite can be synthesised by gas ( Time, temperature and 50% transformation diagrams for the decomposition of cementite into elemental iron and carbon. In one case, the cementite is made by carburising haematite ore (Fe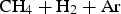






Some calculations indicate that the orthorhombic η–Fe


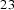

Amorphous alloys of iron precipitate cementite when their carbon concentration is sufficiently large; it is difficult to be specific because there is no phase diagram relating to the equilibrium between cementite and the glassy alloy or even whether an equilibrium mixture of glass and cementite is possible. Figure 30 shows cementite and ferrite obtained by the devitrification of a binary glassy-steel 500 nm thick film during heat treatment at just 300 Cementite (majority phase, containing planar faults) and equiaxed ferrite, crystallised from metallic glass films of composition Fe–13.6C at.-% by heat treatment at 300


Metallic glasses have a larger free volume than supercooled liquid because they essentially become configurationally frozen once below the glass-transition temperature. It is expected therefore that diffusion coefficients will be greater than in the crystalline version of the material. The measurement of diffusion coefficients is complicated by the fact that the glass will undergo structural changes such as relaxation and ultimately, devitrification, when measurements are conducted. Experiments on the diffusion of iron in a variety of metallic glasses in their relaxed condition, show that there is indeed an enhanced diffusivity in the glassy state (Figure 31, [193]).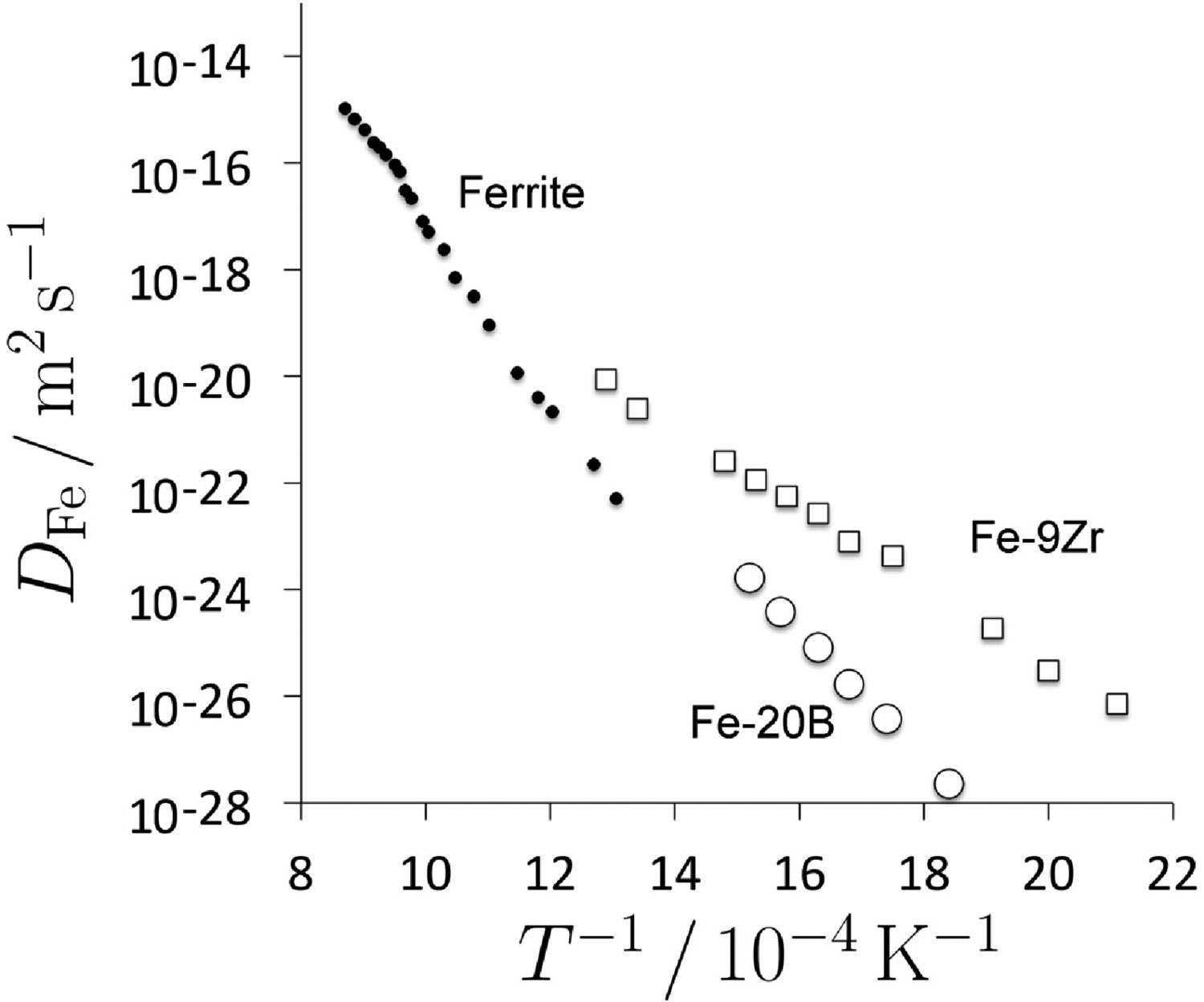
Carbon nanotube formation from gaseous hydrocarbons is catalysed by fine particles of transition metals, particularly iron. Bulky iron or thin films of iron are not as effective as dispersed fine particles [196]; this is because flat surfaces do not form good templates for the growth of tubes, and indeed, the size of catalyst particles correlates with the diameters of the nanotubes generated [e.g. [197]].
Iron does catalyse the breakdown of hydrocarbons in appropriate circumstances, but there has been discussion [reviewed in Section 5.1.3, [198]] about whether it is the iron particles or the cementite particles that form subsequently, that induce the nucleation and growth of multiwalled or single-walled carbon nanotubes. Environmental transmission electron microscopy has provided clear evidence for ‘graphitic networks’ first forming on cementite particles, followed by the genesis of carbon nanotube growth [199,200]. The process of carbon depositing on the cementite particle is not uniform, so carbon diffuses through the cementite from the graphite-rich region to that which is not coated, leading to the expulsion of carbon filaments [196].
Not everyone accepts these conclusions about the role of cementite; Tessonnier et al. [201] comment on electron beam-induced artefacts and the possible role of surface diffusion. Nevertheless, X-ray diffraction experiments involving nanotube formation in a fluidised bed where a mixture of ethylene, hydrogen and nitrogen is catalysed to decompose by iron supported on alumina, indicate that the iron is converted into metastable cementite which then decomposes into a more stable mixture of iron and carbon-nanotubes, rather like the ordinary process of graphitisation [188]. Mössbauer spectroscopy and transmission electron microscopy of nanotubes synthesised by the catalytic decomposition of acetylene on iron particles have shown that while α-iron is the active centre for the breakdown of acetylene, cementite formation induces the growth of the carbon nanotubes [202].
The presence of α-iron or cementite particles within carbon nanotubes can add a magnetic function that has the potential for exploitation in devices. Tubes synthesised the pyrolysis of liquid hydrocarbon in a mixture containing ferrocene [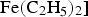
Conclusions
The need to understand cementite comes from its metallurgical importance, it can help interpret data on the Earth's inner core and in order to understand why it occurs in meteorites which will have cooled incredibly slowly. Raw particles of ferromagnetic cementite may in the future play a role in the delivery of drugs to specific locations within the body. And it is clear that cementite, like body-centred cubic iron, has catalytic properties, but is more resistant to oxidation than the iron. Cementite can now be manufactured in sizeable samples, and there are a number of mechanisms by which nanoparticles of cementite can be made. As a result, its intrinsic properties have been characterised in detail over a very large range of temperatures and pressures. The phase has been a fertile ground for first-principles calculations of the effect of alloying elements substituting for iron or carbon, and on its magnetic structure. There are a few puzzling observations that have been published but do not seem to have been followed-up: There are large discrepancies in hardness data which do not seem to be at fault or attributable to whether they represent micro- or macro-hardness measurements. There is a credible observation that cast iron contains two kinds of cementite, one of which is much harder than the other. The mechanism for this is not known. There is a single observation that indicates the existence of both paramagnetic and ferromagnetic forms of cementite at ambient temperature. There are intermittent reports in the literature of a hexagonal version of cementite; it is not clear whether these are correct interpretations of experimental data, but would be worthy of detailed validation. There are conclusions based on atom probe experiments that severely deformed cementite has a hugely depleted carbon concentration. However, evidence that the cementite lattice is maintained in these circumstances is lacking.
It always is difficult to predict the future. But study of cementite spans the disciplines so I can speculate that there my be some unusual outcomes, some of fundamental significance and others in producing immediately useful materials. It would be worth focusing on the production of large quantities of cementite in the form of powder for subsequent consolidation into structural materials. Imaginative ideas, such as the artificial pearlite made using alternating layers of cementite and ferrite [204], could then be implemented without limits on the fraction of cementite that can be incorporated into the structure. Thus, strong steel wire is essentially limited by the amount of cementite that can be introduced into the microstructure by the equilibrium phase diagram.
There is a yearning for economically viable high modulus iron alloys – the concepts and potential are described elsewhere [205]. The modulus along a particular direction of a single crystal of cementite alloyed with manganese can exceed 500 GPa. This can be exploited in a polycrystalline cementite by introducing crystallographic texture – cementite deforms nicely at elevated temperatures. The cementite particles could even be subjected to ‘poling’ to magnetically texture the material prior to consolidation. One example used to discover the hard and soft magnetic directions of cementite [75] has already been described in this review.
From a fundamental point of view, there is amazing microscopy by Ohmori [206] and Nagakura et al. [50] to show the intercalation of really thin layers of cementite with another transition carbide of iron, χ, which has a monoclinic crystal structure and a chemical composition Fe
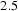
Footnotes
Disclosure statement
No potential conflict of interest was reported by the author.
1
‘une sorte de tissu cellulaire, le fer constituant le noyau et le carbure l'enveloppe des cellules’
2
The space group Pnma has been reported to be inconsistent with cementite that has been annealed at a high temperature, based on a comparison of experimental electron energy-loss fine structure spectra with calculations. A technique such as this gives qualitative information about the local atomic configurations within the structure [32]. However, Mossbäuer and X-ray diffraction experiments show that the correct space group for annealed cementite is in fact Pnma [![]() ].
].
3
The labelling of the orthogonal axes of the unit cell is selected here to be consistent with the space group Pnma i.e. a=0.50837 nm, b=0.67475 nm and c=0.45165 nm. Any crystallographic data referring to the alternative convention Pbnm
References