Abstract
In this study, the reaction kinetics of carbon monoxide reduction of magnetite ore concentrate in the temperature range of 973–1173 K (700–900°C) has been investigated using the thermal-gravimetric analysis. Various methods were used to obtain the governing mechanism and reaction rate constant of this reaction. It was observed that the sintering phenomenon affects the concentrate particles’ reduction at temperatures higher than 1173 K (900°C). CO reduction of magnetite concentrate to pure iron or iron carbide occurred as a single-step reaction. The first-order reaction model and diffusion models are two important mechanisms for this reaction in the operating temperature range. The following equation is the result of our research work relevant to carbon monoxide reduction of the magnetite concentrate particles in order to predict the progress of the reduction reaction (with R2 about 0.98):
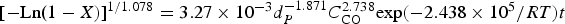
Get full access to this article
View all access options for this article.
