Abstract
The objective of this study was to develop guidelines for the prescription of ankle-foot, knee, wrist-hand and elbow orthoses for patients with neurological disorders. The study is part of a more comprehensive study focusing on the development of clinical guidelines for the prescription of these orthoses in a wider patient population. Evidence from literature (reviews, randomized controlled trials with good quality), information from literature with a lower degree of evidence and information from structured interviews with experts in the field of orthotics, form the basis of statements presented in a Delphi procedure. The Delphi technique was used to gain consensus on statements published on the Internet. A group of experts in the field of orthotics and neurological disorders was asked to give their opinion on 41 statements. Statements with a general agreement of at least 75% were accepted. Statements with less general agreement were rewritten and the experts could react in a second and possibly in a third round. Afterwards, an opinion-based workshop was organized in which participants could react on preliminary guidelines and on Delphi-statements with between 65% and 75% agreement.
Consensus on 32 statements was gained during the three rounds and the opinion-based workshop. These statements form the basis of the consensus based clinical guidelines for the prescription of orthoses in patients with neurological disorders.
Introduction
Improvement of the quality of healthcare is an important issue in The Netherlands. A shift from “daily clinical practice”-based medicine to evidence-based medicine is considered as a method to improve the quality of healthcare. Another method to improve quality is the development of guidelines. A combination of the previously mentioned methods, namely the development of evidence-based guidelines would probably provide an important tool for healthcare improvement.
Guidelines are a set of statements, directions, or principles presenting current or future rules or policy. In healthcare many guidelines are evidence based. These guidelines will be based on published systematic reviews or on reviews that are executed during the development process of the guidelines.
In The Netherlands some guidelines in the field of prosthetics and orthotics have been developed recently. In the Proguide project, guidelines were developed for the prescription of lower limb prostheses (Van der Linde et al. 2003; Van der Linde et al. 2004). Guidelines for the prescription of orthopaedic shoes were also recently developed (Dutch Healthcare Insurance Board [CVZ] 2004). Guidelines for the treatment of specific disorders in which the orthotic component of the treatment is mentioned, such as in the guidelines for the treatment of acute ankle injuries have been developed as well (The Dutch Institute for Healthcare Improvement [CBO] 1998). These guidelines are partly based on evidence and partly on expert opinion.
The authors were asked by the CVZ, an independent national institute with an advisory role towards the government and the healthcare insurance companies, to develop guidelines for the prescription of ankle-foot, knee, wrist-hand and elbow orthoses. In the field of orthotics for the upper and lower limbs, only a few reviews of literature are published (Hijmans et al. 2004; Morris 2002; Struijs et al. 2001) and they state that no definite conclusions can be drawn from the existing literature. Therefore, the published reviews on orthotics do not provide much information on which the guidelines can be based.
Guidelines for the prescription of orthoses should be based on evidence for as far as evidence is available. When too little evidence is available for a complete guideline, information for the development of the guidelines should be gathered in another way. In the first place, information from literature with a lower level of evidence can provide this information. Another common way to gather information for guidelines in the absence of evidence is to make an inventory of expert opinions.
Several ways to make an inventory of expert opinions are available, such as (structured) interviews with experts and questionnaires. The problem with the use of expert opinions in the development of guidelines is disagreement between the experts. Only when consensus exists on a topic on which no evidence is available, can this topic be used in guidelines. The Delphi technique is an important and commonly used method to gain consensus concerning these kinds of topics (Murphy et al. 1998; Powell 2003).
In the current study, a modified Delphi technique is used in the process of gaining consensus. Besides implicit and explicit information from literature, implicit knowledge of experts in the field of orthotics forms the basis of the statements in the Delphi procedure as presented in this study. The knowledge of the experts was inventoried by means of structured interviews.
The method for the development of guidelines is in line with the Proguide project in which guidelines for the prescription of lower limb prostheses were developed (Van der Linde et al. 2003). In contrast with the field of prosthetics, orthoses are prescribed to a much wider group of patients because a much wider range of disorders can be treated with orthoses. A second problem is identification of the experts. In The Netherlands all lower limb prostheses are prescribed by physiatrists and an identified group of physiatrists exists with a special interest in prosthetics. However, orthoses are prescribed by different medical specialists. Besides, orthoses prescribers are not gathered in any kind of group or organization.
The goal of this study is to develop clinical guidelines for the prescription of ankle-foot, knee, wrist-hand and elbow orthoses for patients with neurological disorders that can be used by the wide spectrum of orthoses prescribers in order to improve the quality of healthcare and to make a shift from prescription based on individual opinion to consensus based prescription. The present study is a part of a more comprehensive study focusing of the development of clinical guidelines for orthoses in a wider patient population such as in patients with trauma, rheumatoid arthritis and osteoarthritis.
Methods
A Delphi study, used to gain consensus on statements concerning orthotics, that provides the basis of the guidelines, is presented. The statements in the study were based on literature and interviews with experts.
When no information was available in literature, these gaps had to be filled in. In this study the authors tried to bridge these gaps by gathering information using structured interviews with experts in the field of orthotics. A multidisciplinary group of 15 experts was interviewed in a structured way. The interviews were recorded and typed out afterwards. The experts were selected based on their expertise in this field. Experts were selected from different parts of The Netherlands, in order to avoid the problem that the guidelines would rely on local preferences in prescribing orthoses.
To objectify the expertise of the participants, the interview started with some general questions concerning their experience, education, current activities, and the amount of orthoses prescribed weekly. After the general part of the interview, the interview focused more on the disorders in which the participant was specialized. The complaints of the patients that are eligible for an orthosis, in terms of body functions, body structures, activities and participation (International Classification of Functioning Disability and Health, ICF) and the specifications of the orthosis that should be prescribed in order to reduce these complaints were discussed. In addition the experts were asked what contribution the patient (consumer) had in formulating the goals of the treatment and the selection of the orthosis. The results from the interviews combined with information from literature formed the basis of the statements in the Delphi procedure.
In this study a modified Delphi procedure was chosen, which has been developed by the RAND corporation (Bernstein et al. 1997). Delphi is a method for structured group communication with the goal to treat complex problems. This method offers all participants the opportunity to give their individual opinion. The participants were able to react on the statements anonymously, which guaranteed open reactions from the participants. In this study the procedure consisted of three Internet rounds. The Internet rounds were followed by an opinion-based workshop in which all experts that contributed to the study (participants in structured interviews or Delphi) were invited to react on preliminary guidelines based on approved statements from the Delphi procedure.
A multidisciplinary group of experts in the field of orthoses and neurological disorders was selected and asked to participate in this study. Selection of the experts was based on the following criteria:
No contribution to the orthotic guidelines project so far (e.g. no contribution in the structured interviews).
Expert in the field of orthotics and affinity with neurological disorders, vice versa or expert in both orthotics and neurological disorders.
Ten specialists in physical and rehabilitation medicine (physiatrists), two physical therapists, two neurosurgeons, two certified orthotists, one orthopaedic surgeon, and one neurologist participated in this study. The participants were asked to read the statements presented on the Internet and to react with “I agree” or “I do not agree”. Besides, they could comment on the statements or justify their answer. Each round lasted for 14 days and it was possible to quit at any time and to continue later that round.
Based on the literature and the interviews and reckoning with a viable amount of statements, 41 statements were formulated and presented in the first Internet round. When necessary, an explanation of the terminology used was added to the statements. When 75% general agreement was reached, the statement was accepted. The percentage of agreement was calculated against the total of agreement plus disagreement; participants who did not react on the statements were left out of consideration in this percentage. This way of calculation was chosen because the participants were asked explicitly not to respond on a statement when they had too little knowledge about the specific topic in that statement.
Statements on which less than 75% general agreement was reached in the first round were rewritten based on the comments of the participants and returned to in the second Internet round. In addition, in the second round, the level of evidence (Appendix A) and, if necessary, the references to literature on which the statements were based, were added to the statements.
In the third Internet round, statements were returned to on which still less than 75% general agreement was reached. In this final round, the level of evidence and references were presented as well. In addition, the participants were given an insight to the results and the comments of the participants from the second round, so they were provided with an explanation on what grounds the statements were changed. Moreover, in this round, the participants were able to react on comments and answers given by the other participants, and could change their opinion, based on comments of other participants during the round.
Preliminary guidelines were written based on the approved statements. These guidelines were presented in an opinion-based workshop. All contributors to the project (e.g. experts that contributed to the interviews or Delphi) were asked to participate in the workshop. In a structured discussion, the participants could react to both the content and the form of the guidelines. Besides, statements on which between 65% and 75% general agreement was reached were presented again and discussed. After the discussion, those present could give their opinion on the statement. When at least 75% agreed with the statement, it was finally accepted.
Results
In the first Internet round 11 of the 41 statements were approved (Figure 1). These statements did not re-enter the second round. Besides, based on the comments of the participants and contradictory results from the approved statements, three statements were eliminated and did not enter the second round. The resulting 27 statements were rewritten based on the comments of the participants and presented in the second round. In addition, two extra statements were added. The addition of the extra statements was based on comments of the participants. The response in the first Internet round was 67%. One participant decided not to participate in the future rounds due to only having knowledge of orthotics in peripheral nerve disorders.
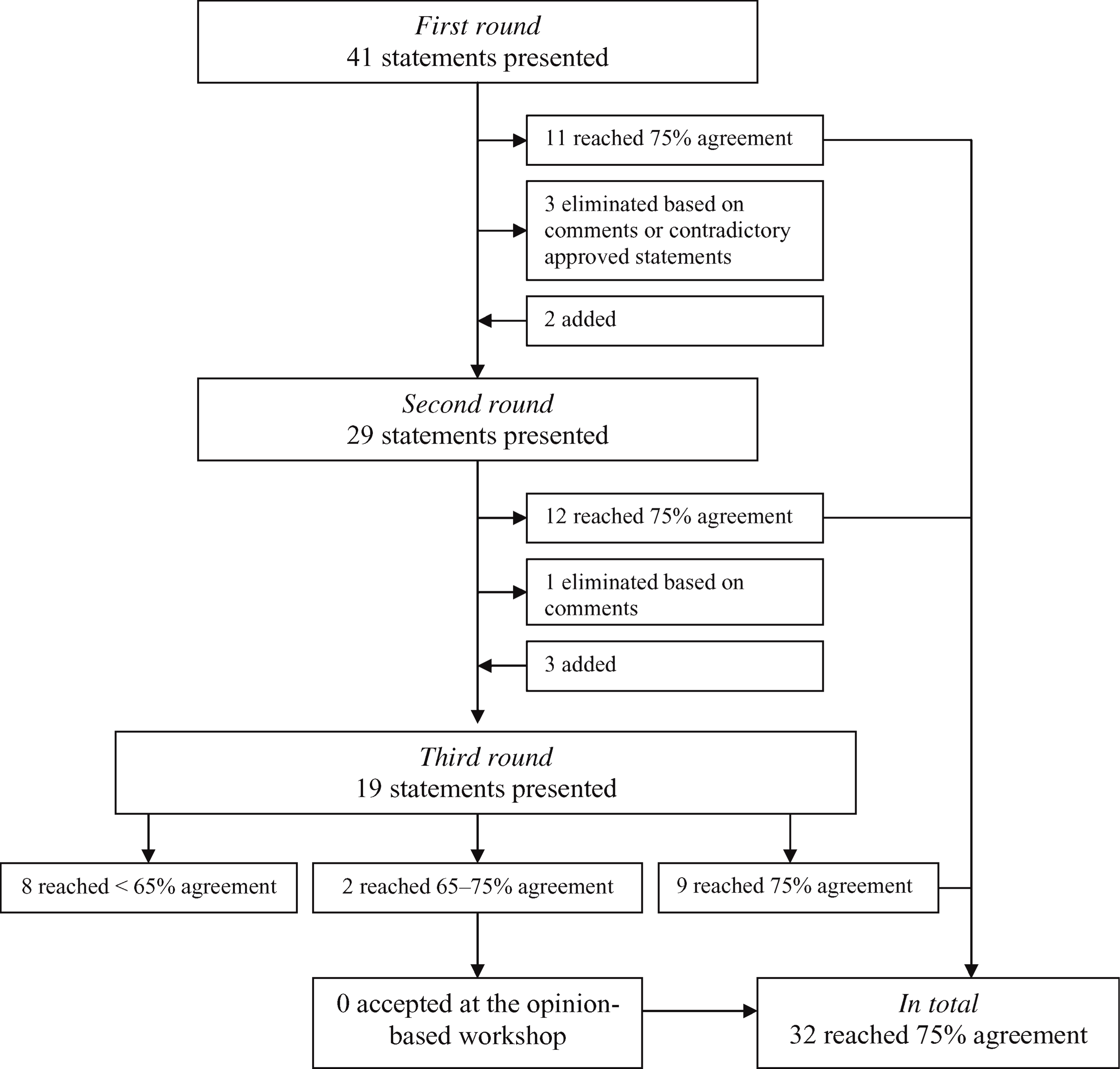
Flowchart of the Delphi procedure.
In the second Internet round 29 statements were presented and, as previously mentioned, the level of evidence was added, and when evidence was available references were presented in the addition. Twelve statements reached at least 75% agreement, one statement was eliminated based on the comments and did not enter the third round. So, 16 statements returned in an adjusted way in the third round. Besides, three extra statements were added. The adjustments and additions were based on comments of the participants. The response in this Internet round was 76%. One participant decided not to participate in the final round due to lack of time.
In the final round 19 statements were presented. As mentioned previously, the results from the second round were presented in the addition. An agreement of 75% was reached in nine of the 19 statements. The response in this final Internet round was 69%.
In total, 32 statements were accepted. These statements formed the basis of the preliminary guidelines presented in the opinion-based workshop. Based on this workshop, the guidelines were given the finishing touch to, but none of the statements with between 65% and 75% general agreement were accepted by the participants. So the final guidelines were based on 32 accepted statements. A translation of these statements is presented in Appendix B, together with their level of evidence.
Discussion
In this study, guidelines were developed using various information gathering methods. In the first place, information was gathered from literature and structured interviews. Based on this information, 41 statements were formulated and presented to a group of experts using the Delphi technique. The Delphi procedure was the key process in this guidelines developing project. In the three Internet rounds, agreement on 32 statements was reached. The accepted statements were used to develop preliminary guidelines that were presented to a group of experts. During an opinion-based workshop the experts were able to react on both the form and contents of the guidelines, in order to give the finishing touch to the guidelines.
The field of orthotics is complex and because of the wide range of disorders found a wide range of experts exists. Most of the experts are expert in the field of a specific disorder, but not specifically an expert in orthotics as part of the treatment of that disorder. There are no experts who have knowledge of the whole field. Even the neurological subdivision seems to be too wide to cope with, for the majority of the experts. It may have been better if it had been possible to split up the expert group into a group of experts in orthotics in adults and a group with expertise in orthotics in children. The lack of expertise can easily result in lack of commitment to the field of orthotics, which can be an explanation for the low response (average 71%) compared to the response (average 98% in two internet rounds) of the Proguide project (Van der Linde 2004). Another explanation for this lower response can be the lack of knowledge resulting in not answering at all. However, it was possible to develop guidelines which were based on consensus.
As stated by the International Society for Prosthetics and Orthotics (ISPO), it is important that patients or consumers participate in the prescription of orthoses (Geertzen and Craig 2003). In the development of guidelines, consumer participation is important for the acceptance of the guidelines when they are brought into use. In this study a member of the national board for people with chronic diseases or disabilities was part of the group of supervisors of the project. His task was to ensure that the wishes of the consumer were taken into consideration during de development of the guidelines. In addition in the study a questionnaire was sent to consumers of orthoses, assessing their opinion concerning the important issues when orthoses are prescribed.
Guidelines based on consensus are generally well accepted by the prescribers. In future these guidelines should be used by all medical specialists that prescribe elbow, wrist, knee, or ankle-foot orthoses. The method of implementation will be crucial to the acceptance of the guideline. The National Scientific Boards for Physical and Rehabilitation Medicine, Orthopaedics and Rheumatology and the Dutch National Member Society of ISPO will play an important part in the acceptance of the guidelines by the prescribers. In this study representatives of these boards supervised the process of guideline development. Positive effects of the co-operation with these boards are to be expected.
Footnotes
Acknowledgements
This work is supported by a grant from the Dutch Health Care Insurance Board (CVZ).
