Abstract
A dynamic one-dimensional mathematical model was developed for predicting the thermal state of a steelmaking ladle. The model is intended to be used in process control applications, in which fast computational times are desirable alongside model accuracy. The calculation domain was discretized using the finite difference method, and time integration was performed using both the implicit Euler and Crank–Nicolson methods, the performances of which were compared. The model was implemented in Python programming language and validated using data from our own measurements and other studies available in the literature. The results indicate that the model can reproduce the measured temperature evolution of the ladles within 5°C at best. The worst performance was observed during cooling, where the model underestimates the temperature at the innermost measurement point by up to 200°C. With computation times of around 16–23 s for one hour of simulation, the model is computationally sufficiently fast for online applications.
Introduction
A comparison of mathematical models for the thermal state of a ladle.
The thermal models focus on specific parts of the ladle [2] or a specific process such as preheating [4,20] or teeming [17,19]. More complex models may consider the complete process cycle and more complex geometry for the ladle. Ladle thermal models have been successfully used in parametric studies, [4,6,12,17,21] ladle design work [1,2,5,11,23], and online applications [7,14–16]. As computing power increases, two-dimensional and three-dimensional models have become more feasible model geometries. The majority of the multidimensional thermal models have been based on computational fluid dynamics (CFD) software, which offers accurate and valuable information about fluid flow and the thermal behaviour of the molten steel and ladle. However, CFD-based models are computationally too expensive for online process modelling, and instead, their results have been used to build simpler models [12,17,22] which can then be used in online applications.
The majority of the non-CFD models assume a uniform bath temperature, although there are also CFD models that assume a completely mixed melt at some point in the model cycle [21,22]. The results of Jormalainen and Louhenkilpi [17] indicate that the temperature in the ladle wall is quite uniform in the axial direction. The thermally more conductive refractory on the slag line is obviously hotter than the lower portions, but it does not seem to significantly affect the lower lining. The simplification of a uniform melt temperature may be less significant for the model results when calculating either the ladle thermal state or the average melt temperature. However, when modelling the tapping or casting temperature, the thermal stratification of the melt may significantly affect the temperature of the molten steel flow.
As computing power increases, two-dimensional models may become more feasible for online calculations. Compared to a one-dimensional model, two-dimensional ones may better account for different materials in different zones, the effects of corners, the changing view factors in different positions, and the effects of the melt height. However, with such a uniform refractory temperature along the contact surfaces, [14,17,24] their benefits may be marginal compared to a well-defined one-dimensional model.
This work aimed to develop a fast one-dimensional model for the thermal state of a steelmaking ladle. The work is based partly on the master’s thesis by Mäkelä [25], which presented the first version of the model as well as its preliminary qualitative validation. It presented and applied the main heat transfer mechanisms on the ladle wall. The calculation domain was discretized using the finite difference method. For time integration, two methods (implicit Euler and Crank–Nicolson) were implemented and compared. The model was implemented in Python programming language for fast model development. In the present study, the model is expanded to consider the top slag and the heat losses from it, and the model is validated more thoroughly using data recreated from a previous study by Fredman and Saxén [10].
Model description
Simplifications and assumptions
We made the following assumptions and simplifications: The steel bath is completely mixed and of uniform temperature, and the refractory surface in contact with the melt is at the same temperature as the melt Heat is conducted only in the radial direction in the ladle wall, and in the axial direction in the slag There is no contact resistance between lining layers Thermal conductivities of materials are constant with respect to temperature There is no heat flux through the ladle bottom Heat losses from the inside surfaces are caused only by radiative heat transfer When used, the lid is closed perfectly, and there is no heat loss During preheating, the lining surface is at the same temperature as the gas phase inside the ladle Tapping and teeming take place instantaneously.
Nodal structure of the model
Figure 1 has a schematic example of the different nodal configurations in the model. All the reference points in the network are Δr apart from each other, and the temperature and position of the nodes are represented by Tn and rn. The slag has a similar structure, with the exception that the relative positions of the nodes are not needed, only their distance from one another. Schematic example illustration of the nodal structure of the model.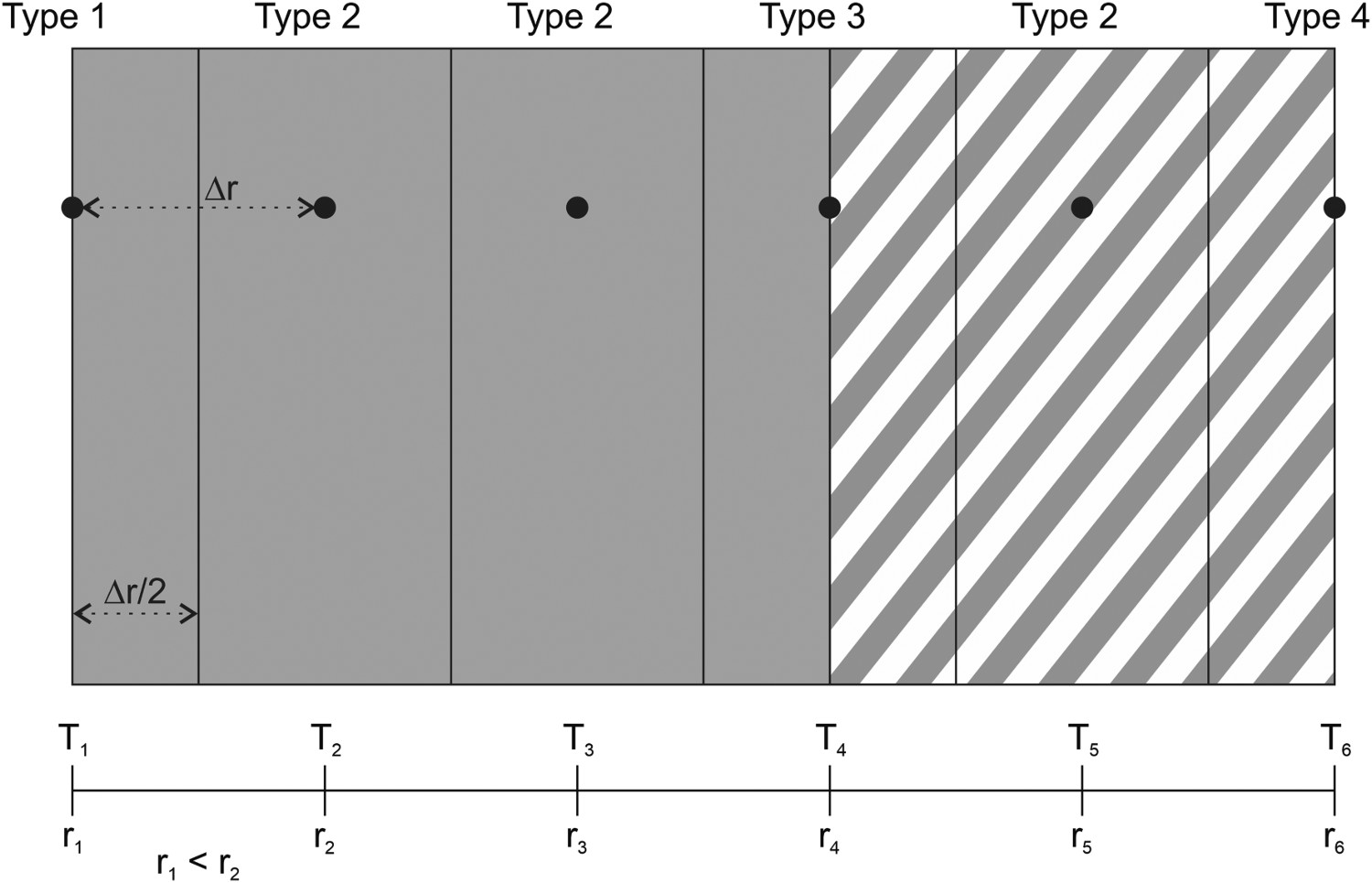
Four types of nodes were implemented for the model’s nodal network (see Figure 1): The Type 1 node, i.e. the inner surface node in contact with the melt. The Type 2 nodes, i.e. the bulk nodes are surrounded by a singular material and represent the bulk of the refractory or slag. The Type 3 nodes, i.e. the boundary nodes are at the interface of two different refractory materials. The Type 4 node, i.e. the outer surface node represents the outer surface of the ladle shell or slag.
Heat transfer mechanisms
Heat losses through the ladle wall
The ladle is modelled as a multilayer cylindrical object, for which the radial thermal conduction can be described by Equation (1),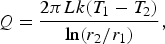
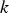
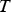

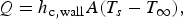
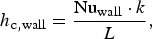
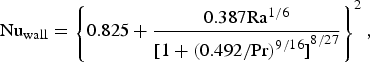
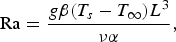
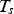
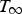
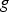
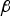
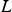


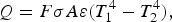
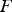
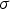
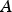

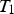
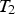
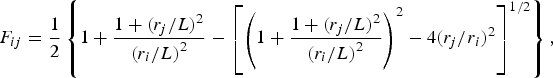
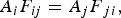
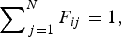
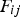

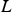
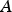
Heat losses through the slag
The slag is modelled as a flat surface, through which the axial thermal conduction is described by Equation (10),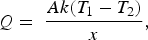
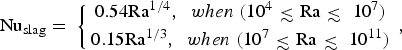
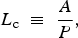
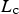
Material properties
Thermal properties of common ladle materials.
Emissivity of surface materials.
Boundary conditions, initial conditions, and time integration
The model has been divided into three operating modes based on the different working periods of the ladle. The working periods along with the used boundary conditions are illustrated in Figure 2. Working periods of the ladle.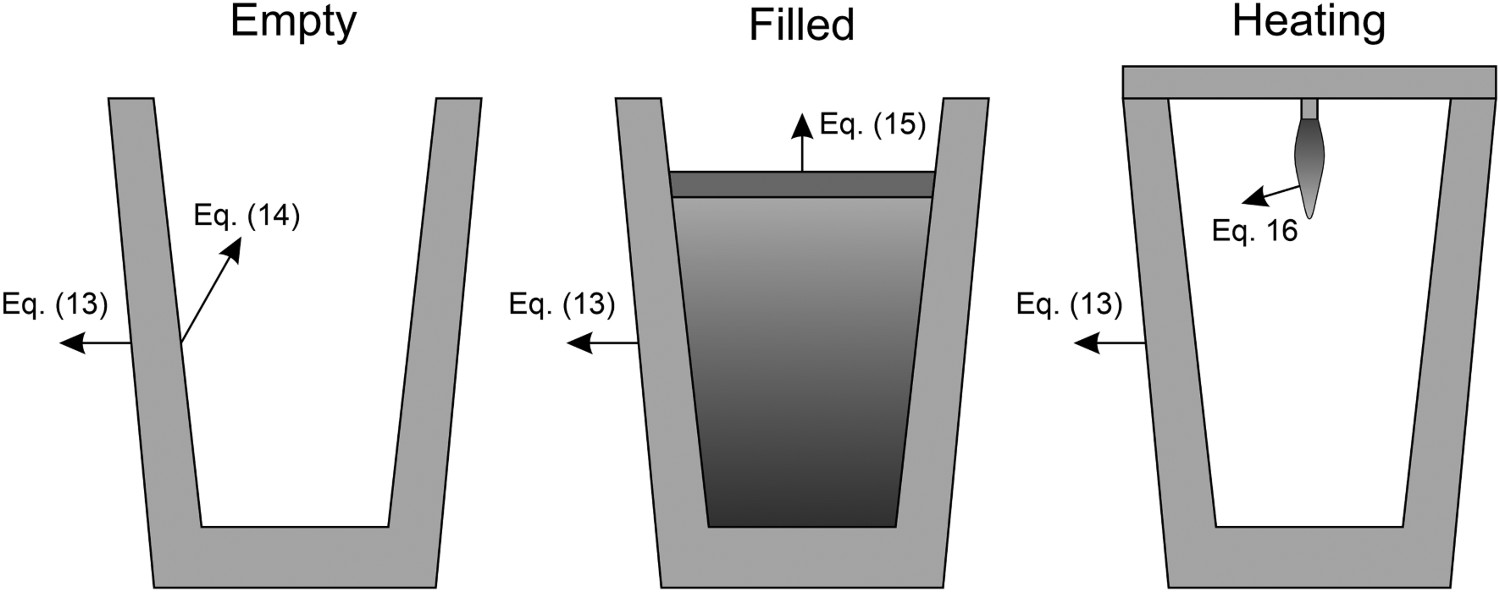
During all operating modes, the boundary condition on the ladle shell stays the same and is described by Equation (13), Empty period. During the empty period, the inner surface node is cooling according to grey body radiation, and transfers energy to the adjacent node through conduction. The boundary condition applied to this surface is described by Equation (14). Charged period. During the charged period, the ladle is filled with molten steel, and the inner surface node of the wall and slag are at the same temperature as the melt. The inner surface nodes transfer energy through conduction to the adjacent nodes. The boundary condition is applied to the slag top surface and is described by Equation (15). Preheating. During the preheating period, the ladle is filled with air and the inner surface node is at the same temperature as the air. The air and the inner surface node are heated according to the burner power until they reach a predetermined maximum heating temperature where it is then kept. The inner surface node transfers energy through conduction to the adjacent node. The heating is described by Equation (16),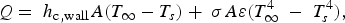
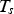
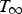
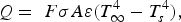
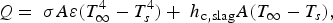
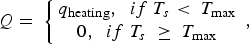
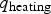
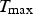
Time integration was performed using the implicit Euler and Crank–Nicolson methods. The performance of these two methods is compared in the results section. The initial conditions were chosen based on the performed validation. During the original validation, the refractory temperature profile was set to room temperature of 25°C and then heated with the burner for 36 h. Validation using data from Fredman and Saxén [10] used initial conditions, where the inner surface was at 400°C and linearly decreased to room temperature of 20°C on the outer surface. In all validations, the initial temperature of the slag was set to that of the ambient temperature.
Numerical solution
The one-dimensional temperature profile at the new time is solved using the Newton–Raphson method, by iterating Equation (17),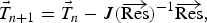
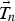
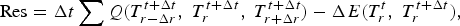
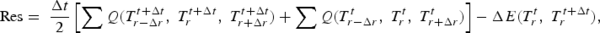
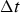
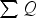
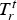
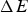
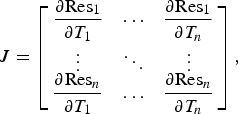
Validation data
Measurement campaign
During the model development, a measurement campaign was conducted at the Outokumpu Stainless Oy Tornio Works, melt shop line 2. Further details regarding this production line are provided by Spiess et. al. [30]. In this campaign, the ladle mantle temperatures of 150-tonne ladles were measured with a thermal camera pointed at the casting turret.
The thermal camera had a resolution of 0.11 megapixels, a field of view of 18° by 14°, and the temperatures were given to the hundredth of a degree. The camera could not be safely calibrated at the steel plant and was therefore calibrated in laboratory conditions. To do this, a piece of generic carbon steel was fitted with a thermocouple, heated in a laboratory furnace to ∼400°C and used for calibrating the camera. The offsite calibration of the camera may have been a source of an issue, where the measurements significantly differed from the model predictions. This may have partly been due to the emissivity of the steel piece used in calibration differing from the emissivity of the steel used in the ladle shell.
Images were recorded every 4 s, whenever an object hot enough entered the field of view. The camera measured the temperatures of each pixel, allowing us to port the data into Excel, where they could then be formatted into a recreation of the thermal image. This allowed us to then process the data using commands readily available in Excel. In Figure 3, the camera positioning is shown along with a recreated thermal image, and an outlined area, from which an average temperature was taken. The area was chosen by hand from the lower portion of the ladle and covered an area of approximately 1.5–2 m2. From each cast, two thermal images were chosen, one before casting and another after. The images were chosen based on quality and ladle positioning, with shaky images being ignored and importance placed on having the ladle in the same position before and after casting, allowing us to choose the same area of the ladle more consistently. From the collected data, 71 consecutive melts were chosen, which included 8 different ladles, at different stages of life. Thermal image of the ladle, and a schematic of the imaging position.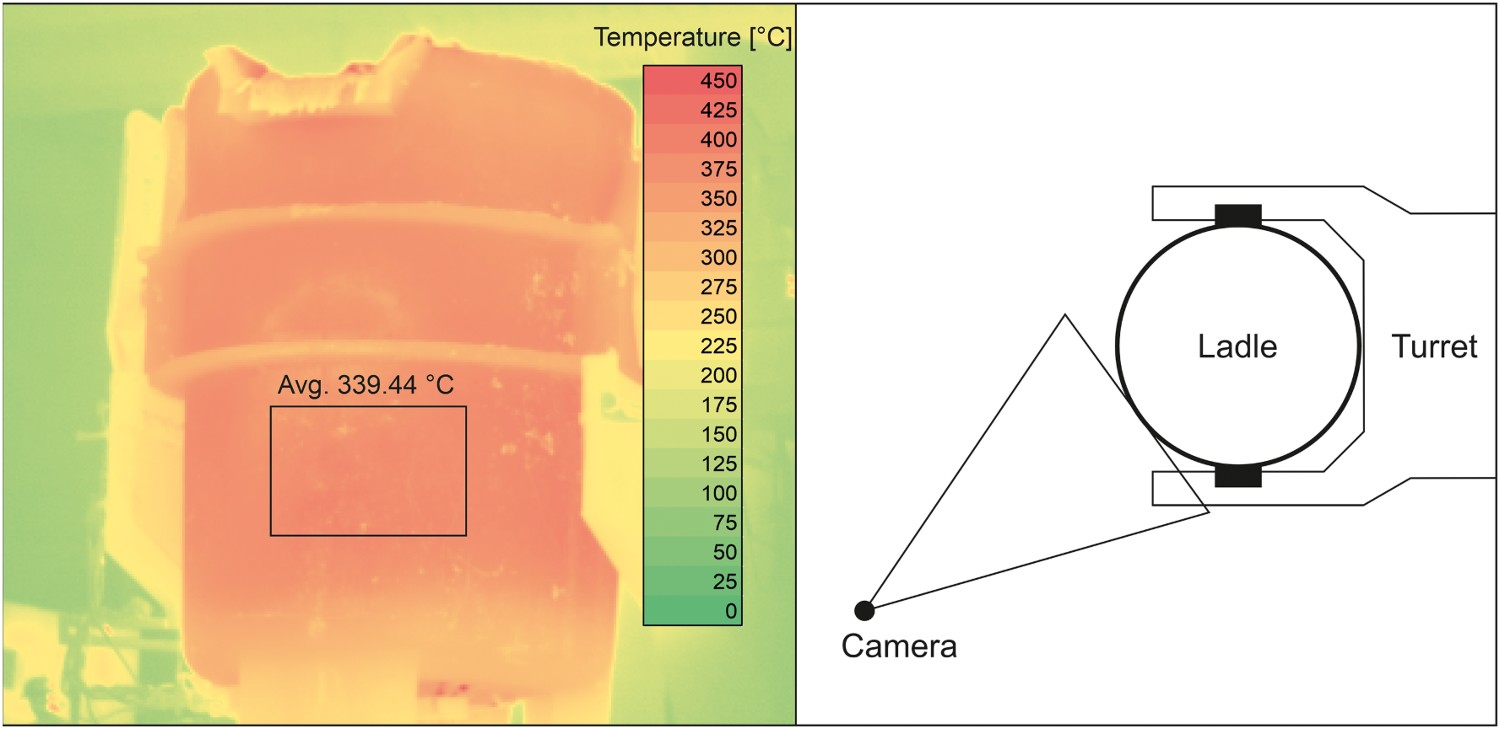
Data from the literature
Material properties from Fredman and Saxén [10].
Results and discussion
Sensitivity analysis
The effects of the Δr and Δt sizes on the model performance were studied by changing these parameters and then simulating three hours of heating, melt holding, and cooling. The temperatures of the wall inner surface and the wall outer surface nodes were logged and compared.
Tables 5 and 6 provide the tabulated temperatures of the inner surface node after three hours of simulated heating. These results are visualized more intuitively in Figure 4. The results of the implicit method significantly depend on both the Δr and Δt sizes. The Crank–Nicolson method, on the other hand, seems to depend mainly on the Δr size, with changing Δt resulting in negligible changes in results for the most part. However, the Crank–Nicolson method was prone to instability in the form of slowly dampening oscillations in the temperature profile. This was a problem mainly when using small Δr with large Δt. The temperature of the wall inner surface node after three hours of simulated heating using different time integration methods: (a) implicit Euler method, (b) Crank–Nicolson method. The black dots represent the simulated points. Implicit model inner surface node temperatures after three hours of simulated heating. Crank–Nicolson model inner surface node temperatures after three hours of simulated heating.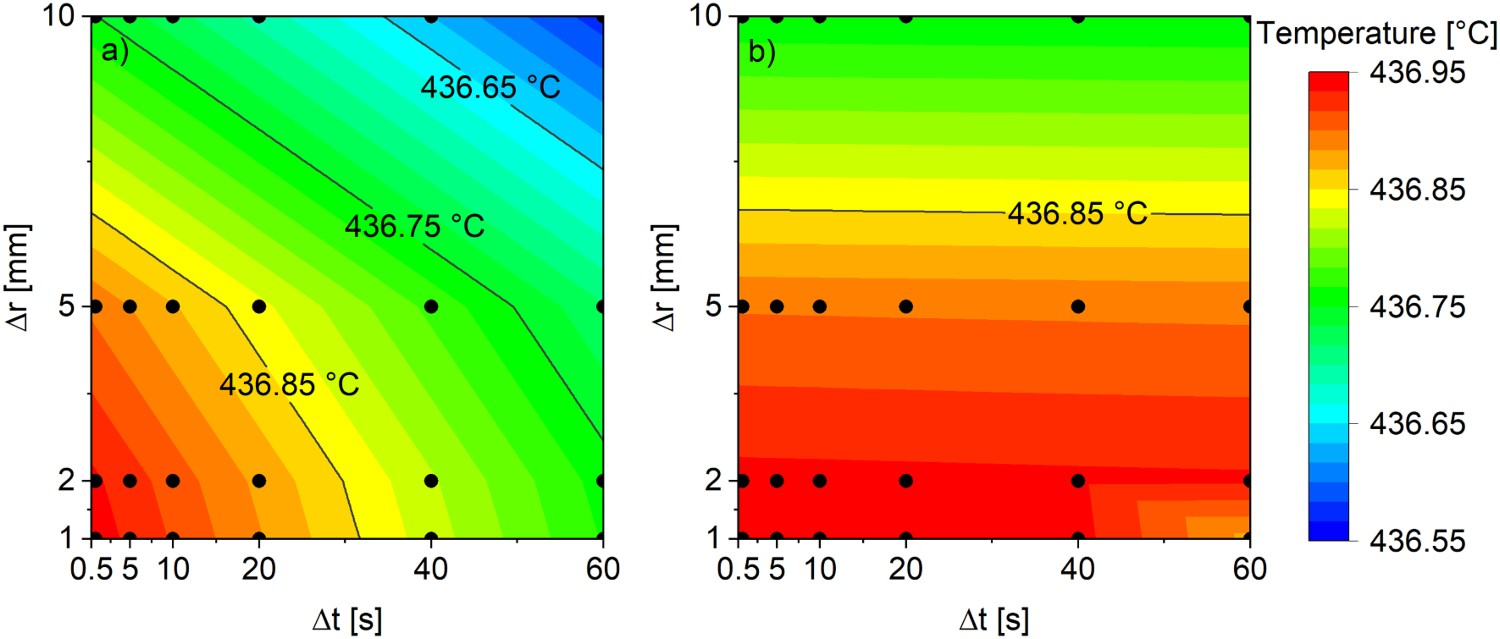
It should be noted that the Crank–Nicolson method was around 5–20% slower than its fully implicit counterpart when using the same model parameters. However, since the result of the Crank–Nicolson method was less dependent on the used Δt, it could be faster by using larger Δt, with the risk of some instability. The instability could be resolved by applying adaptive timesteps. However, this could not be implemented in this study’s timeframe.
The implicit Euler method took around 16–23 s of real-time to simulate 1 h when using Δt = 5 s and Δr = 1 mm, on a laptop with an 11th Gen Intel Core i5-1135G7 2.40 GHz processor. This is sufficiently fast for online applications. Since the implicit method is sufficiently fast, simpler and produces almost identical results with the Crank–Nicolson method at small timestep sizes, it was deemed the more suitable option.
The data from Fredman and Saxén [10] was used to further inspect the sensitivity of the implicit model to the employed Δt and Δr, by comparing the change in the absolute error. The absolute errors from two working periods, (a) the third melt holding period and (b) the third empty period, and from three refractory positions are presented in Figure 5. The change in absolute error in all cases is quite small and should not be a cause for concern. The absolute error of the predicted temperature at three refractory positions (1.250, 1.355 and 1.440 m) at two different times: (a) third melt holding period and (b) third empty period.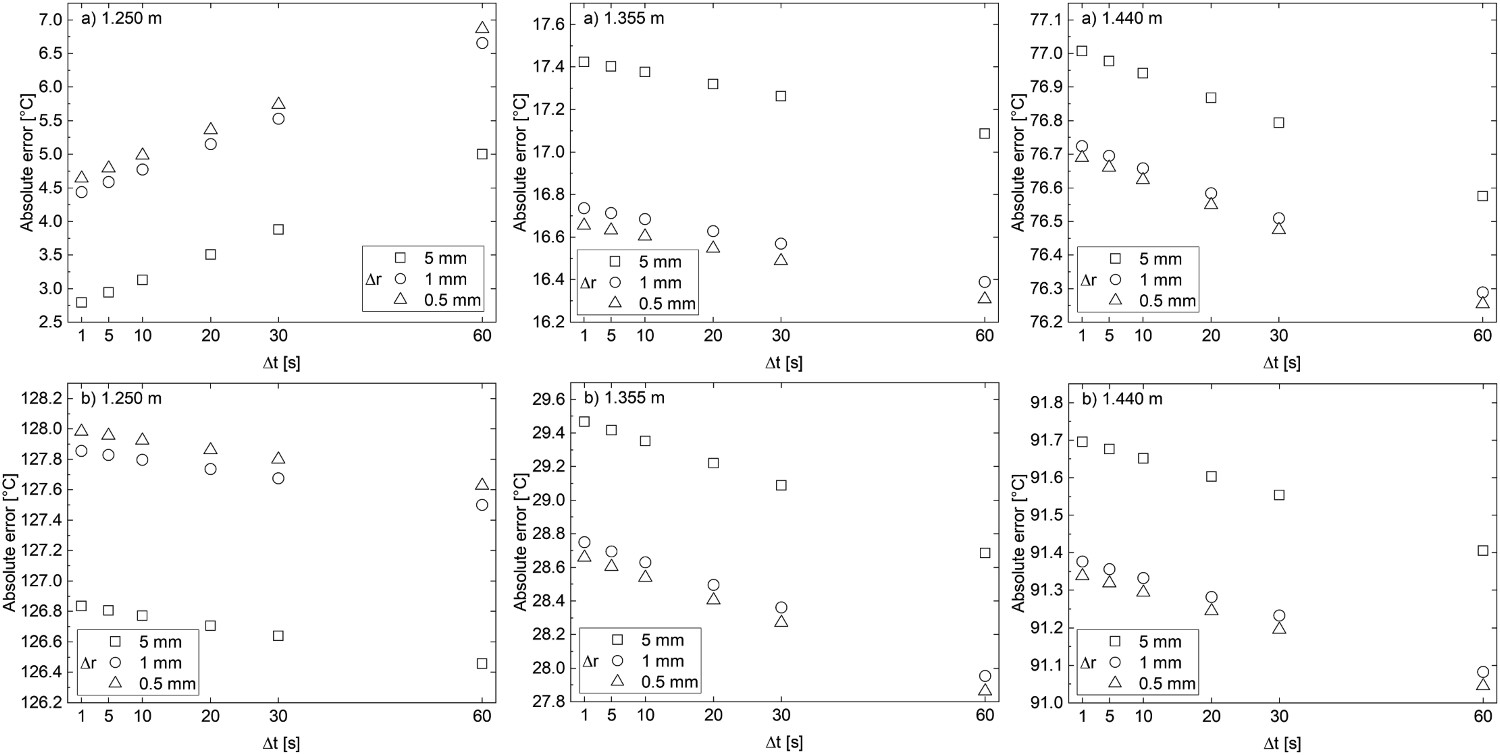
Model validation
The studied ladle has three refractory layers and a steel mantle. Starting from the hot face, the used materials were dolomite, 90% high-alumina brick, insulating brick and steel. The model was validated using the collected ladle process data to run the model and log the temperature of the steel shell at each timestep. These model results were then combined with the collected thermal imaging data, from which Figure 6 was compiled. The square marks are the measured mantle temperatures taken from the thermal images, and the black lines are the model results. The model results had to be raised by ∼100°C to obtain comparable results with the thermal images. It is suspected that this is largely caused by the inadequate calibration of the thermal camera. The temperature adjustment for each ladle is noted in Figure 6. Model validation results for the qualitative validation.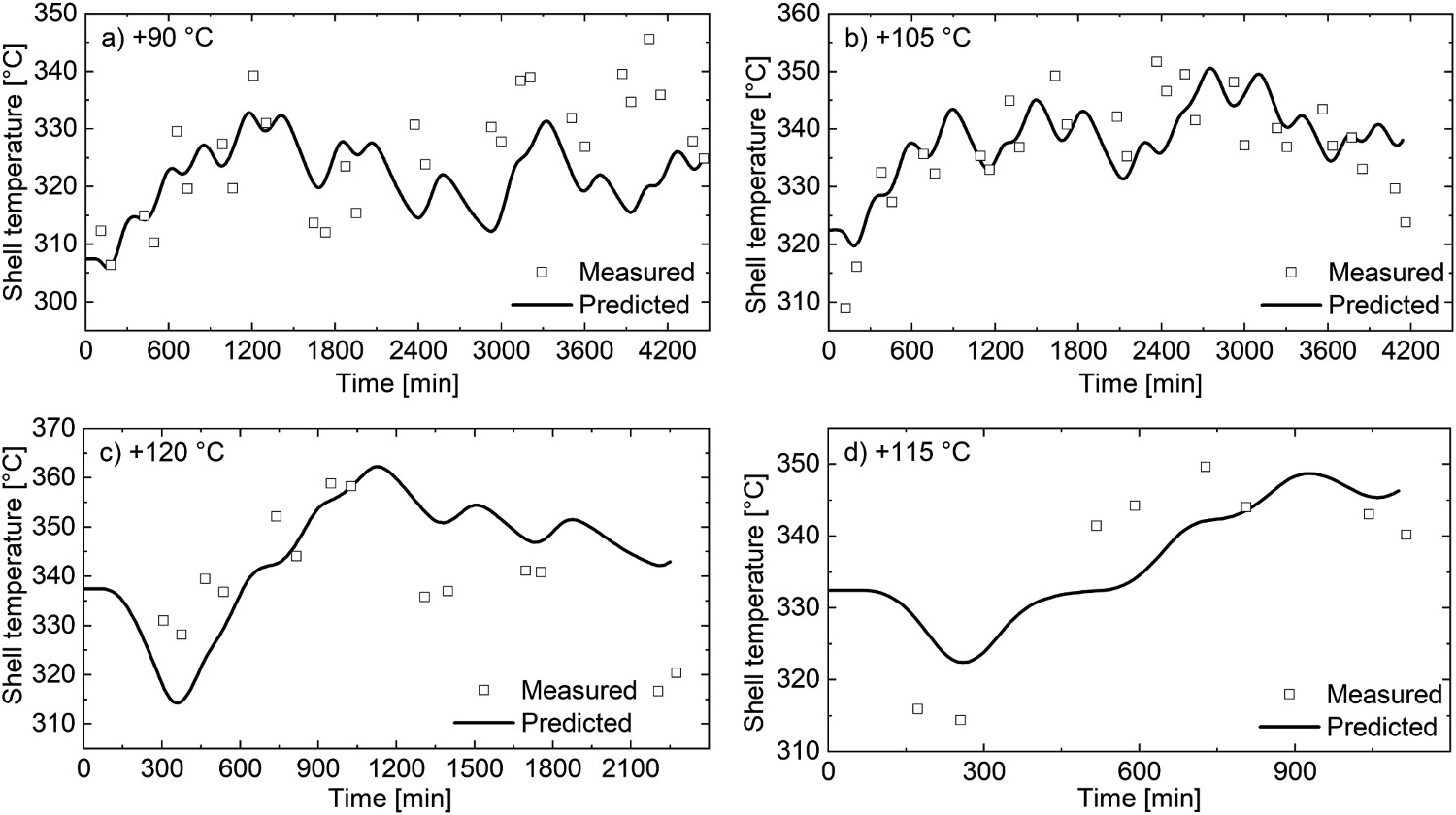
While the absolute deviations are in some cases quite large, the predicted trends are in qualitative agreement with the measured values. The inadequate validation results could be caused by improperly defined material properties, improper process data, or the overly simplistic nature of the model and its heat transfer phenomena.
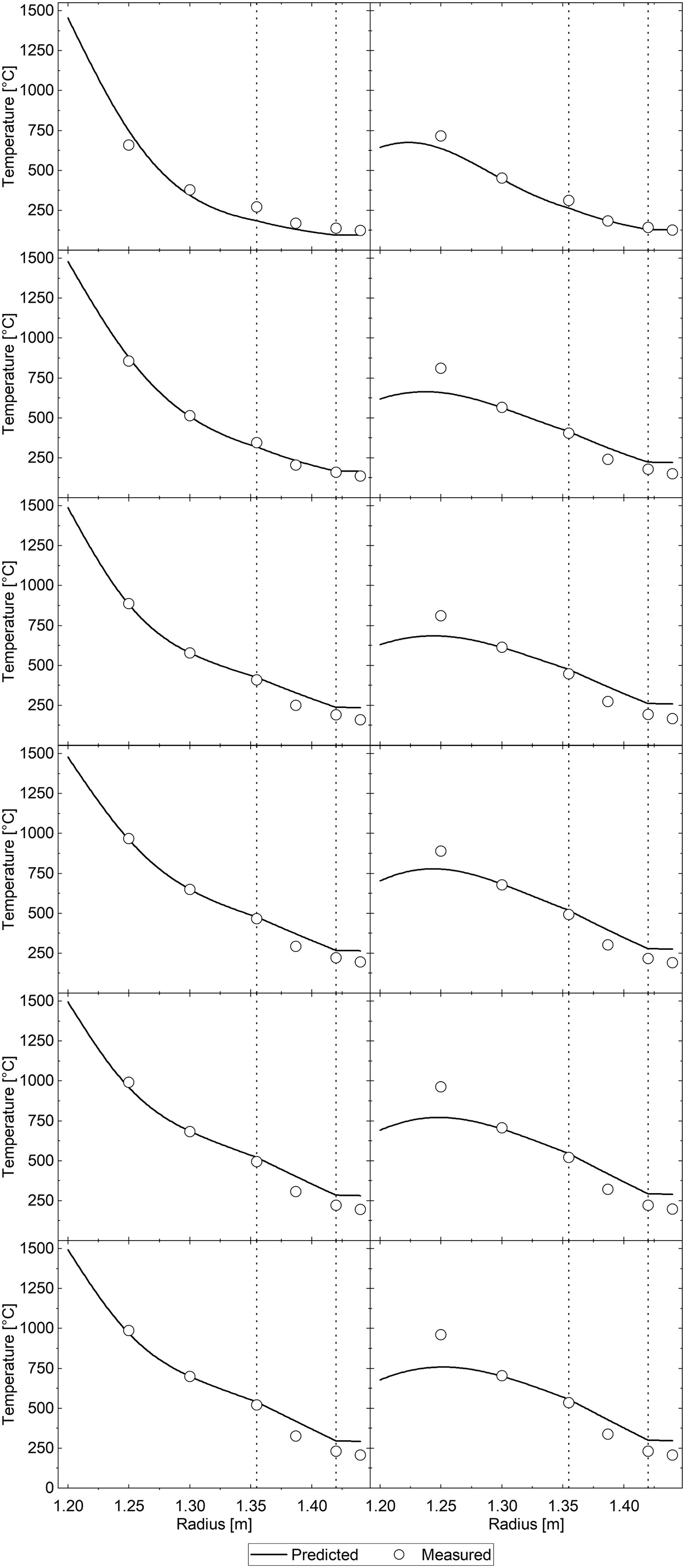
Additionally, the model was validated quantitatively using data recreated from the previous study of Fredman and Saxén. [10] It appears that the present model performs relatively well when examining the situation at the end of holding (see Figure 7). At the end of cooling, the results are noticeably worse. Figure 8 contains the predicted temperature profiles and measurements for the first six ladle cycles. It can be seen that the predicted mantle temperatures at later cycles are significantly higher than the measured values and that at the end of the cooling periods, the innermost measurement point is significantly hotter than predicted. Either our model overestimates the heat loss from the inner refractory, or some unaccounted-for process practice decreases the heat loss. Model validation results with data from Fredman and Saxén [10] at different refractory positions at two times: (a) at the end of holding and (b) at the end of cooling. Thermal profiles generated using data from Fredman and Saxén [10].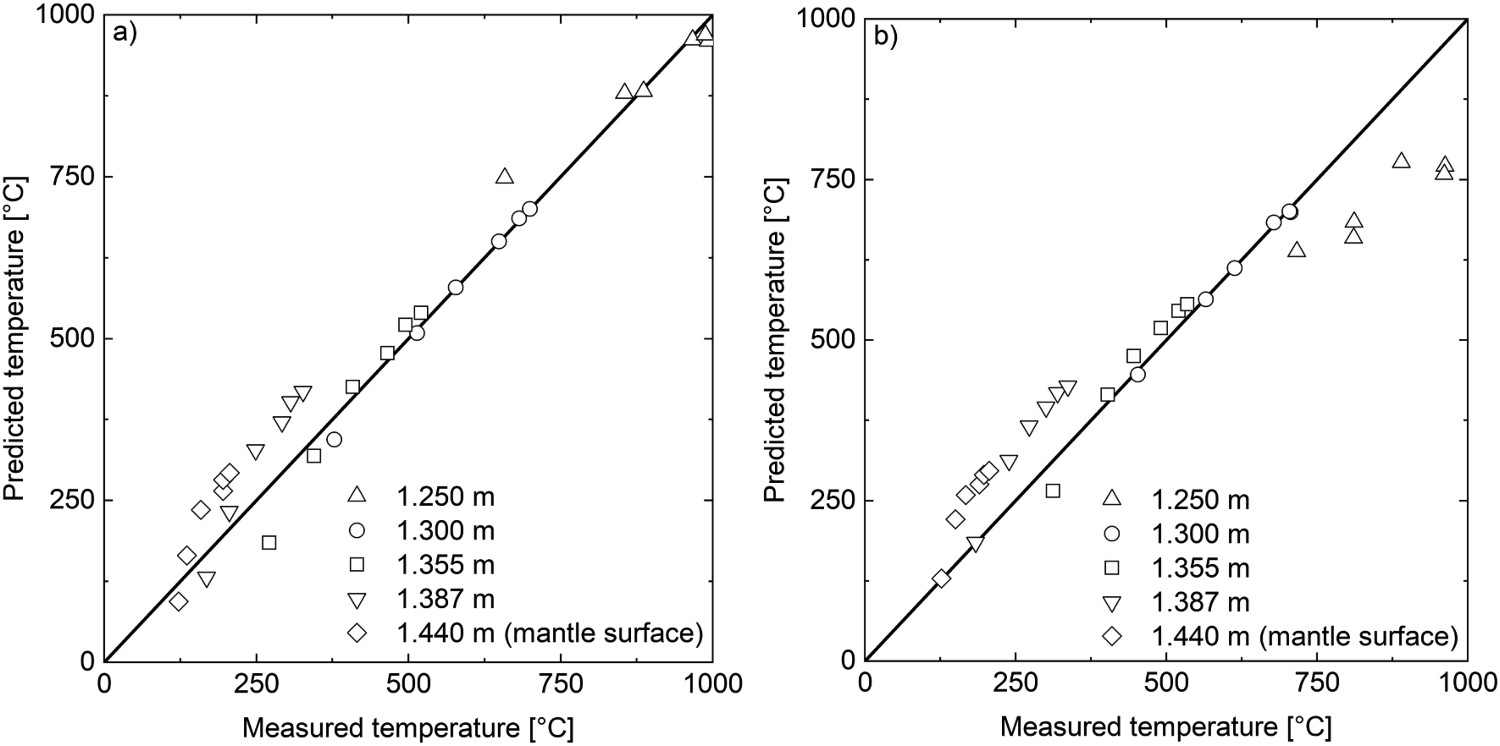
Conclusions
A dynamic one-dimensional model was developed to predict the thermal state of a ladle. The model accounts for the main heat transfer mechanism for heat losses through the ladle walls and bath surface. Two different time integration methods (implicit Euler and Crank–Nicolson) were implemented and compared. The main advantage of the Crank–Nicolson method was its insensitivity to the timestep size, which could allow the use of larger step sizes and thus shorter calculation times, provided the step sizes were not too large to cause instability. However, as the implicit Euler method was not only faster and more stable, but also produced almost identical results at smaller timesteps, it was deemed more appropriate for the intended online applications.
To validate the model, a measurement campaign was conducted in a stainless steelmaking melt shop. Based on qualitative validation using the data obtained, it was found that while the model was unable to accurately predict the short-term temperature changes at the ladle mantle, it could reproduce the main trends of temperature evolution. The next step was to conduct a quantitative validation using data available from a previous modelling study. The results suggest that the model can capture the temperature profile of the ladle wall within 5°C at best. However, the model fails to produce good predictions for the innermost refractory layer during cooling, underestimating the temperature by up to 200°C. This may have been caused by operating practices which we have not accounted for in our validation. Other than that, the next most significant error is seen on the outer shell of the ladle, where the model overestimates the temperature by around 90°C. With computation times of around 16–23 s for one hour of simulation, the employed 1D approach provides a fast basis for online temperature monitoring applications, given that the accuracy is further improved in some areas.
Footnotes
Acknowledgements
The research was carried out within the framework of the TOCANEM research programme, funded by Business Finland. The authors thank Sapotech Oy for conducting the measurement campaign at Outokumpu Stainless Oy. Furthermore, Seppo Ollila from SSAB Europe Oy and Esa Puukko from Outokumpu Stainless Oy are acknowledged for providing the technical details regarding the ladles used in this study.
Disclosure statement
No potential conflict of interest was reported by the author(s).
