Abstract
Different alloying concepts to trace deoxidation products, mainly aluminium oxides, using rare earth elements (REEs), were tested on the laboratory scale by melting trials with a high-frequency remelting furnace. Lanthanum and Cerium, which belong to the group of light REEs, were used for these experiments. The formed multiphase inclusions were characterized by scanning electron microscopy with energy dispersive spectroscopy. Concerning the higher atomic numbers of REEs, traced non-metallic inclusions (NMIs) seem brighter than the steel matrix compared to deoxidation products. REE-traced aluminium oxides showed a primarily heterogeneous and almost globular morphology. The mean equivalent circle diameter of REE-containing NMIs is for all trials similar and is about 2 µm. The experimental results pointed out that the recovery rates of the various alloying concepts differ only slightly. In contrast, the values mainly depend on the surface-to-volume ratio and the amount of oxygen in the melt.
Introduction
The formation and modification of non-metallic inclusions (NMIs) have become an important research focus today due to the increased requirements for steel cleanness in the recent years. The type, size, shape and distribution of these particles can significantly impact the mechanical properties, such as toughness, fatigue properties or fracture toughness, and further the corrosion resistance of steels [1–8]. NMIs can also cause problems during production [9,10], in addition to the effect on the final product quality. For instance, clogging during continuous casting is a possible consequence of the deposition of alumina inclusions, which have a higher liquidus temperature than steel [10–13]. The melting point of the deoxidation products (Al2O3) can be lowered through a calcium treatment. Furthermore, the tendency for deposition in the flow control system is significantly decreased due to changes in interfacial conditions. However, mistakes during Ca-treatment or even the reactions between metal and slag can lead to solid Ca-aluminates, further inducing severe clogging problems [14,15].
Tracing NMIs is one option to increase the knowledge regarding inclusion evolution over different process steps since the clogging mechanism is not entirely clarified. Therefore, rare earth elements (REEs) can be applied for tracking deoxidation products, such as Al2O3. Especially, Lanthanum and Cerium are proven as tracers during steel production [16–18]. In addition, REEs start interacting with aluminium oxides on the interface, whereby REE-containing complex inclusions occur [19,20]. As mentioned by Šuler et al. [21], REEs are not best suited for following the modification steps of inclusions because they also interact and influence the morphology, size, number and distribution of NMIs. The addition of rare earths lowers the melting point of inclusions, resulting in modified liquid-type inclusions at production temperatures, similar to the classical Ca-treatment [21–24].
Furthermore, rare earth elements, mainly Ce, are used in the industry for grain refinement. The study of Baumgartner et al. [25] shows the significant influence of Ce on the grain size using the example of super austenitic stainless steel. The refinement is caused by the formation of small NMIs, primarily AlCeO3, which initiate heterogeneous nucleation. More profound knowledge of the inclusion formation and the interaction with Al is essential since REE-containing inclusions are promising for the grain refining of steels.
Many studies on IF(Interstitial Free)- or ULC(Ultra-Low Carbon)- [20,26–28] and high alloyed steel grades [29,30] show that the interactions between deoxidation products and REEs result in modified NMIs, concerning their size, shape and distribution. Referring to Wang et al. [26], the addition of rare earths increases the number density of inclusions, and, aside from that, the NMIs get smaller and spherical. Similar tendencies are observed by Li et al. [31] and Wang et al. [32]. In both studies, the irregularly shaped inclusions become more globular, and at low Cerium addition, their sizes decrease. Furthermore, they found that inclusions start to grow again at a critical amount of Ce, dependent on the chemical composition of the steel.
The present study investigates the tracing of inclusions by adding the rare earths Lanthanum and Cerium at a laboratory scale. The expected inclusion scenario for different additions is determined based on thermodynamic calculations. Small-scale laboratory experiments are performed to test and compare various alloying concepts for rare earth additions. Subsequent inclusion analyses are done using manual and automated scanning electron microscopy with energy dispersive spectroscopy analysis (SEM/EDS). Furthermore, the formed NMIs are characterized by their type, size and distribution. The amount of traced particles is also determined. Finally, predicted and observed inclusion populations are compared.
Methods and materials
Electrolytic iron
Chemical composition of used electrolytic iron.
The preexisting inclusion population primarily consisted of (Si,Mn)-oxides with a mean equivalent circle diameter (ECD) of 2.1 µm. No pure Al2O3 inclusions were observed in the raw material.
Alloying concepts
Three different alloying methods were tested and compared in this work. La and Ce were protected from fast oxidation in the atmosphere by two various concepts. First, the rare earths were put inside steel tubes with closed ends. Second, La and Ce were wrapped with Al-foil.
Besides the addition of REEs as pure substances, a ferroalloy with Cerium was also produced and tested. The mass of rare earth elements is also related to the melting weight. 300 ppm of pure La or Ce was added for every alloying concept. Therefore, only around 19.5 mg of pure REEs are needed, depending on the small melt weight of about 65 g.
Addition via pure substances
Maximal amounts of the chemical composition of E235 steel tubes in wt-% [33].
Another option to add La or Ce was to wrap the pure elements inside a foil. For this study, an Al-foil was used to minimize the oxidation of La and Ce. The amount of pure aluminium for deoxidation must be adapted due to the additional insert of Al as a foil.
Addition via ferroalloys
For the FeCe production under inert conditions, a plasma reactor, layout seen in Figure 1, located at the Chair of Ferrous Metallurgy at the Montanuniversitaet Leoben, was used as a pre-melting unit. For this purpose, Cerium metal and electrolytic iron were melted with an Argon plasma to prevent Cerium oxidation. Layout of the plasma reactor, used as pre-melting unit [34].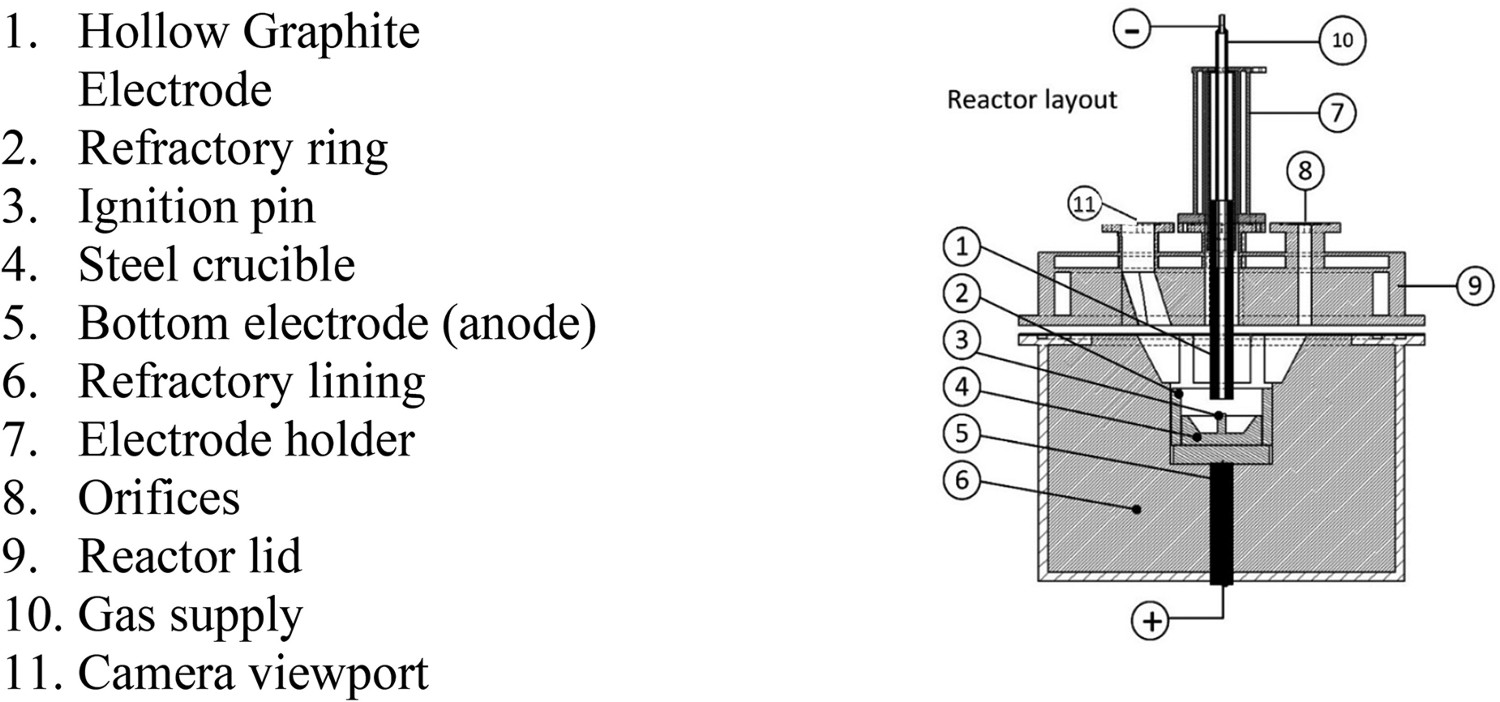
The water-cooled plasma reactor consists of a DC power supply with a maximum output of 16 kW, a visual observation via a camera, an off-gas analysis with a mass spectrometer and a current/voltage measurement by a four-channel analogue logger. A DC transferred arc was ignited between a hollow graphite electrode (cathode) and a steel crucible (anode) with a welded ignition pin to produce the FeCe alloy. A mixture of 85 g Cerium and 20 g electrolytic iron was batch-wise charged into the crucible. After the reactor was evacuated entirely with Argon, an arc was ignited by contact of the graphite electrode with the ignition pin and an electrode gap of 20 mm was set. About 5 l min–1 of Argon 5.0 was supplied through the hollow electrode to stabilize the arc during the process.
Experimental procedure
Laboratory experiments were performed in a Lifumat-Met-3,3-vac high-frequency remelting (HFR) furnace (Linn High Term GmbH, Eschenfeld, Germany) with a spin caster unit. This furnace was chosen since a homogenized melt can be produced without oxygen absorption during the process. Furthermore, the whole procedure is under an inert Ar atmosphere. Continuous bath movement is ensured due to the inductive heating and stirring.
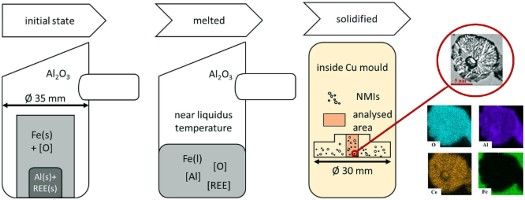
The steel sample (18 × 18 × 22 mm, appr. 65 g) was heated and melted close above the liquidus temperature (TLiquidus + 30−50 K). Afterward, the steel was cast through copper by centrifugal casting. The high mean cooling speed is an additional benefit, leading to quick solidification. Therefore, inclusion interaction is limited. A new, highly purified corundum crucible was used for every trial. The contact time between melt and crucible is about 1 min due to the fast melting and cooling. Hence, the influence on the inclusions is negligible.
The deoxidation agent (Al) and alloying elements (La/Ce) were placed in a blind hole (Ø 6mm) drilled inside the steel samples to prevent too early melting or evaporation losses. Subsequently, the samples were turned upside down. The addition of deoxidation and tracing agents was simultaneous compared to the industry, where the deoxidation takes place several minutes before REEs are alloyed. Hence, the deoxidation products were almost concurrent formed and traced due to the fast melting at the HFR process. Figure 2 shows a schematic overview of the three main states during the sample production in HFR. In addition, in the right picture in Figure 2, the area subsequently analysed with automated SEM/EDS is illustrated. States during sample production in HFR, adapted from Mayerhofer et al. [35].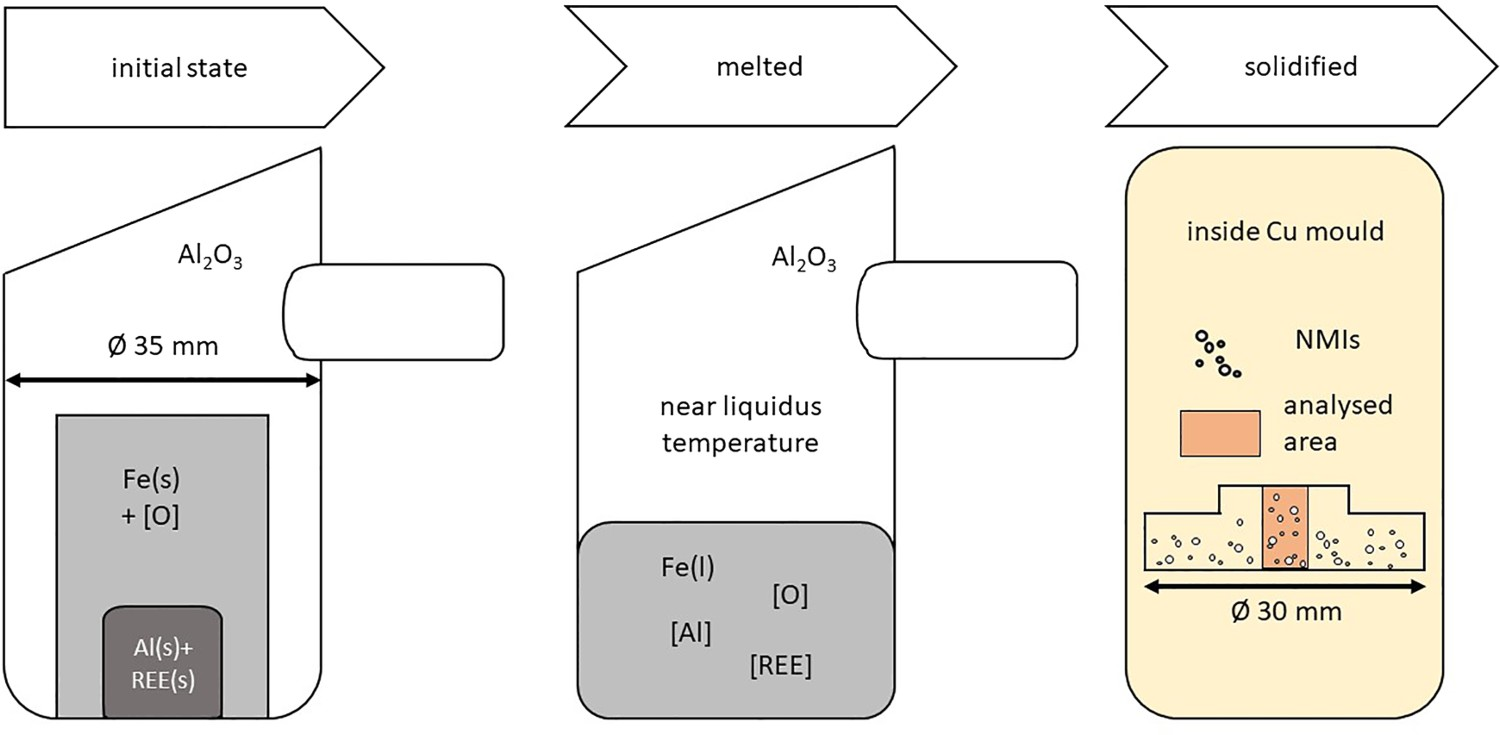
It was essential to keep the time between cutting off a small piece of the REE metal, weighing in, and heating as short as possible for alloying rare earth elements. Otherwise, REEs oxidize and are lost as tracers for deoxidation products.
Methods of analysis
Determination of rare earths content
The amounts of Lanthanum and Cerium were determined with an inductively coupled plasma-quadrupole mass spectrometer (Agilent 7500ce ICP-QMS; Agilent Technologies, Tokyo, Japan). Small pieces of each sample with a total mass of approximately 500 mg were digested and measured. Bandoniene et al. [36] described the ICP-QMS measuring procedure in detail. The sample digestion differs for solid steel samples. Complete dissolution was achieved by additionally adding drops of nitric acid and longer dwell times on the heating plate.
Analysis of REE-traced NMIs
The steel samples were cut in half, conductive embedded, ground with SiC paper and polished with diamond suspension for 9, 3 and 1 µm. A field-emitting SEM (JEOL 7200F; JEOL Germany GmbH, Freising, Germany) equipped with a 100 mm2 SDD-EDS detector (Oxford Instruments Ultim Max 100; Oxford Instruments GmbH NanoAnalysis, Wiesbaden, Germany) was used for the measurements. The chemical composition was automatically analysed with AZtec Feature (AZtec 5.0, Oxford Instruments GmbH Nano Analysis, Wiesbaden, Germany) and manually with AZtec Mapping. First, the automated detection and measurement of the particles were performed. The size distribution of non-metallic inclusions and the amount of traced and non-traced NMIs were determined by this method. Afterward, the representative NMIs were targeted and manually measured. Microscopic NMIs were the focus of this paper. Therefore, inclusions larger than 15 µm were deleted at the characterization step.
A threshold for the greyscale needs to be specified before an automated measurement. Therefore, all the particles defined within this threshold were analysed with the AZtec Feature tool. Since rare earth elements have higher atomic numbers than typical inclusion forming elements, such as Al or Si, their associated greyscales are significantly brighter. Hence, two greyscale sectors were necessary to detect the traced and non-traced particles within the same measurement. The measurement duration is extended within this method, and multiphase NMIs with high contrast are divided into two separate particles.
The double-threshold scan splits heterogeneous multiphase inclusions into several particles. This split results in an inaccurate description of the distribution and the number of NMIs in the investigated area. This error can be solved using a custom-built Matlab tool wherein the related parts of a divided inclusion were recombined. Finally, the correct size, shape and chemical composition of NMIs obtained.
Thermodynamic considerations
One major characteristic of REEs is their high oxygen affinity. Rare earths, such as Lanthanum and Cerium, have a higher negative free Gibbs energy than manganese, silicon or aluminium oxides. Consequently, the formed oxides were more stable than the common deoxidation products, e.g. Al2O3 or SiO2 [16]. The Richardson-Ellingham diagram (Figure 3) illustrates the stability of the most frequent oxides in secondary metallurgy. The calculation of the free Gibbs energies for deoxidation products and other typical alloying elements in steel was done by the thermodynamic software HSC Chemistry 6.1. Richardson-Ellingham diagram including Lanthanum and Cerium.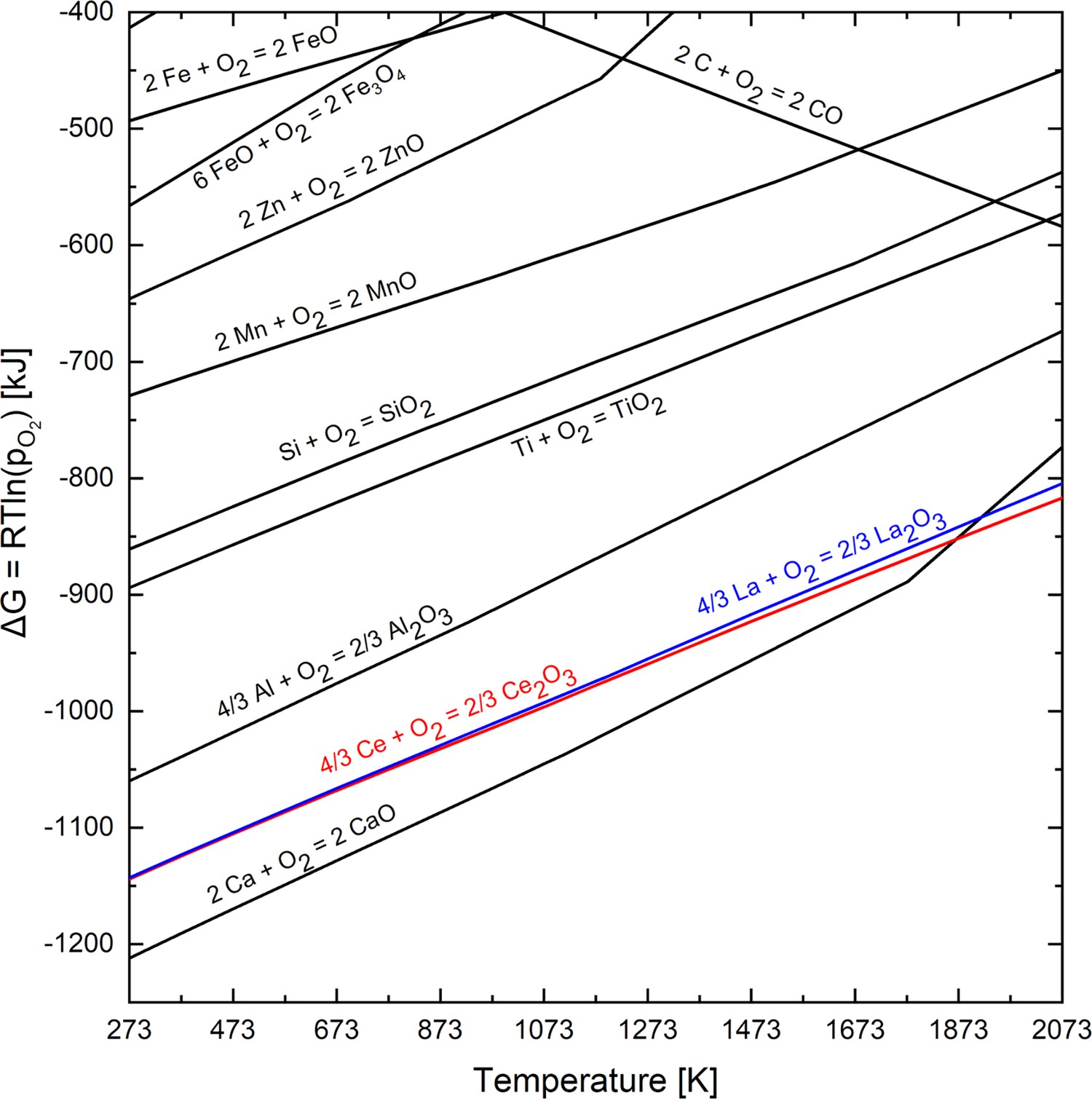
The thermodynamic calculations to analyse the needed REEs amount for tracing alumina inclusions are performed using the software FactSage 8.2 with FSstel, FToxid and FactPS 2022 databases. The influence of the rare earths, La and Ce, on the inclusion formation is also pointed out within these evaluations.
Results
The recovery rates of rare earth elements for different alloying concepts
Measured chemical composition of steel samples in wt-%.
Recovery rates [%] of rare earth elements in different alloying concepts.
Characteristics of REE-traced inclusions
It was observed that most of the inclusions were heterogeneous multiphase types with isolated parts representing the REEs oxides being brighter than the steel matrix. Almost all inclusions in the samples were Al2O3(>99%). Hence, only this inclusion type was considered for the evaluation.
Amount of traced Al2O3–NMIs [%] and corresponding sample number.
Mean ECD and deviation for traced Al2O3–NMIs [µm].
Cross-sections of the samples were analysed by manual and automated SEM/EDS measurements. Figure 4 shows BSE images of representative NMIs traced by Lanthanum in samples 1 and 2. Furthermore, the distribution of the chemical composition is seen in Figure 4. In both cases, the traced NMIs have a similar size. Sample 1 has a distinct border between the lanthanum oxide in the core and the surrounding alumina inclusion. In comparison, no segregation could be observed in sample 2. BSE images and mappings of La-traced NMIs: (a) sample 1, La inside steel tube, (b) sample 2, La wrapped in Al-foil.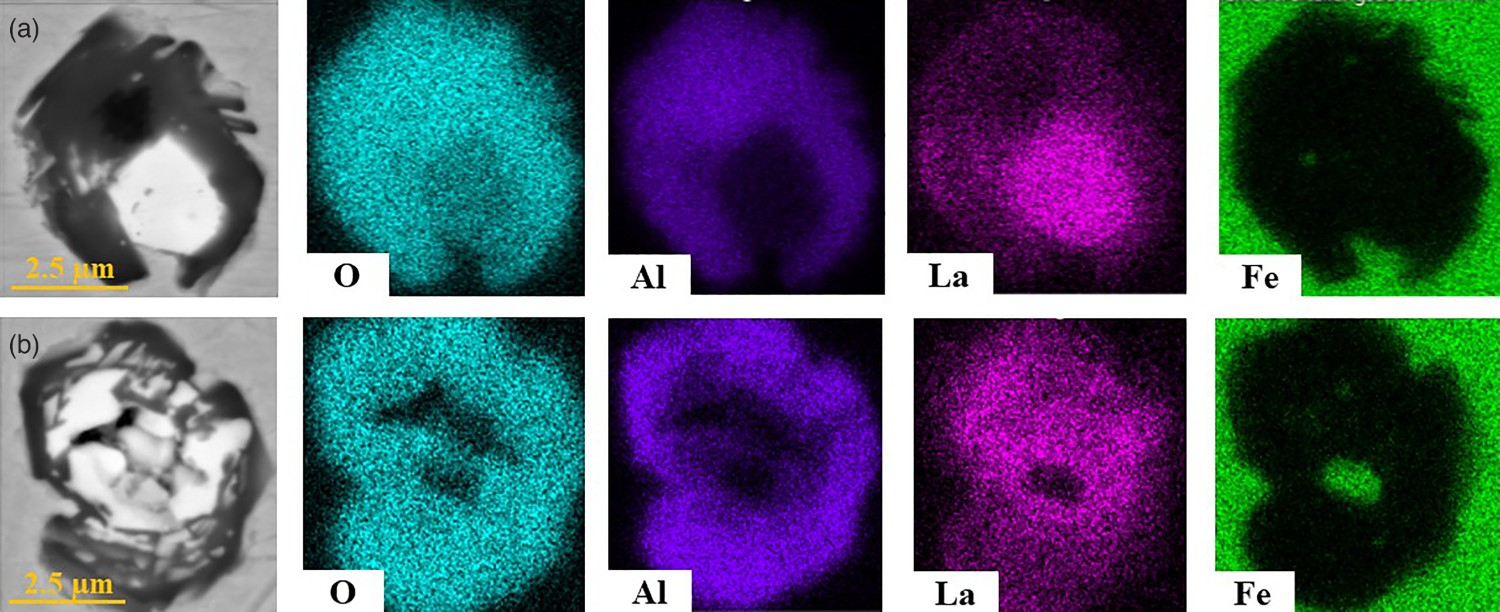
BSE images of typical NMIs traced with Ce and their chemical compositions for the three tested alloying concepts can be seen in Figure 5. The Ce-enriched parts were finely distributed in the Al2O3 inclusions in all cases. More agglomerated minor inclusions can be observed in sample 5 (Figure 5(c)). BSE images and mappings of Ce-traced NMIs: (a) sample 3, Ce inside steel tube, (b) sample 4, Ce wrapped in Al-foil, (c) sample 5, Ce as FeCe (56 wt-% Ce).
Discussion
The output of FeCe production
An option to trace inclusions was to produce a ferroalloy of Ce wherein it was added directly to the melt as in industry. The amount of Cerium in FeCe used in the industry is usually much lower than the alloying material in laboratory trials. Ren et al. [39] added FeCe to trials on the laboratory scale. This study used a significantly higher Ce-concentrated ferroalloy with 30 wt-% for experiments with an ULC-steel in a Si–Mo high-temperature tube furnace.
The lab-produced FeCe was analysed, and the real composition was compared with the FactSage predicted value. A deviation in the chemical composition can be observed, as seen in Figure 6. The FeCe system was calculated with the software FactSage 8.0 and the databases FactPS and FSstel 2020. At 93 wt-% Ce, a low melting eutectic is formed between Ce and Fe. The planned Ce amount was 81 wt-% concerning the added amount of Fe and Ce as described in the materials section. The produced alloy had finally only 56 wt-% Ce according to the analysis. On the one hand, this difference can be attributed to a slight pre-oxidation of the pure Cerium during the comminution of large pieces into chargeable grain sizes. On the other hand, a deviation can occur due to its reaction to the residual oxygen present in the electrolytic iron during the melting process. Equilibrium diagram of the FeCe system (including planned and real composition).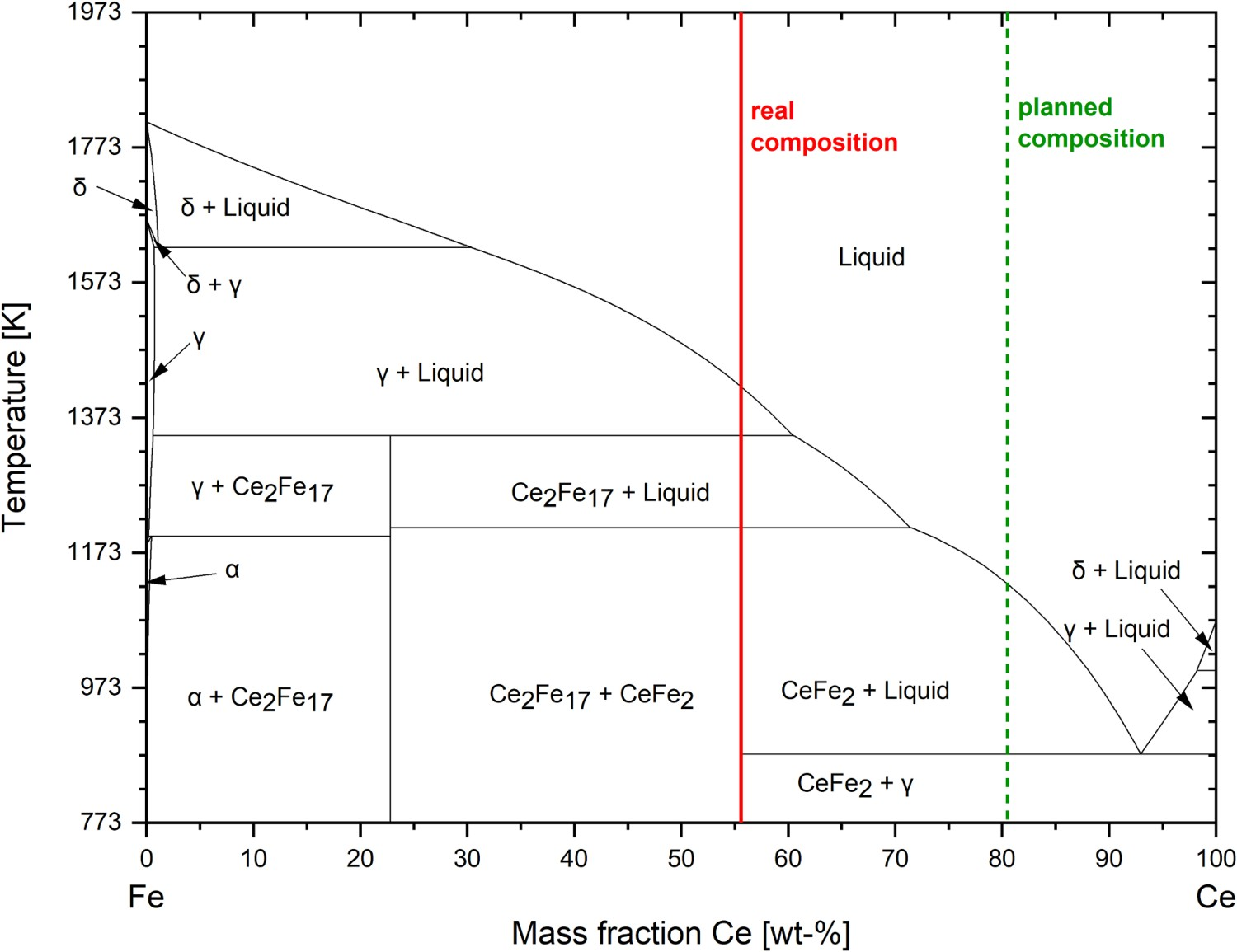
Evaluation of the alloying concepts
Examining different alloying concepts for trials in the laboratory is essential because smaller amounts of tracers were added compared to the industry. Former studies have already dealt with the problem of a smaller output for rare earths, which were significantly lower in the case of laboratory experiments [19,29] compared to industrial trials [26,37]. The main reason behind this is its high oxygen affinity. The associated oxidation rate is faster due to a large surface-to-volume ratio in the small dimensions of the added tracers. Therefore, only a little part of the original content can act as a tracer and interact with deoxidation products. The vapour pressure of light REEs, such as La and Ce, is relatively low at process temperatures compared to heavy REEs, as mentioned by Okabe et al. [38] Hence, the losses for heavy REEs would be significantly higher due to evaporation.
The amount of La or Ce depends on the oxygen content in the steel and the number of deoxidation products intended to be traced.
Various methods have already been used in the literature for laboratory and industrial trials. Lanthanum [19] and Cerium [29] were wrapped in pure iron foils for alloying in the laboratory trials of Li et al. [19] and Luo et al. [29] with Si–Mo high-temperature tube furnaces. Based on the REE content in these two studies, the recovery rate for Lanthanum was about 40% and for Cerium 20%. These results must be carefully evaluated since many parameters influence the outcome of REEs, such as the oxygen content in the melt, the flow rate of the protective gas and the duration of stirring, which were different for these two studies. The results in the present study were about 30% for La and about 36% for Ce concerning the tested alloying concept, REEs inside an Al-foil. The findings are comparable to the prior research mentioned where a pure iron foil was used.
In comparison, when Al-foil wrapped La and Ce were added as described in this paper, the losses for Lanthanum were slightly higher, within a difference of approximately 6%. This recovery rate difference between La and Ce could be possible as the added REEs wrapped inside the Al-foil were not completely air-tight. Further deoxidation will occur within this alloying method, and the amount of Al in the melt will subsequently increase. The reason, therefore, is the additional Al-source, which leads to the formation of extra Al2O3 inclusions. The rare earths reduce these NMIs, so the Al content in the melt continues to rise.
In contrast, losses of REEs due to oxidation can appear, especially by adding La or Ce using steel tubes. The reason was the relatively high amount of oxygen and the resulting unavoidable oxidation inside the tubes. As a result, the recovery rates were comparatively lower than for the other methods described in this work. Furthermore, using a steel tube for insertion can contaminate the melt due to the introduction of new alloying elements and preexisting NMIs which might be present in the steel tube.
Considerably lower losses were expected when Ce was added as a ferroalloy because the interaction between REEs and oxygen is inhibited. As anticipated, the highest recovery rate at about 38% was achieved in this case.
The obtained recovery rates for each alloying concept in this study can only serve as guide values for the industry since parameters, such as treatment time or bath movement, differ. The tendency between the tested concepts would be similar on the industrial scale, but to evaluate the total achieved recovery rates of REE addition in the industry further experiments are needed. Further studies are necessary to determine the output of the different alloying methods at various furnaces to obtain accurate recovery rates for rare earths on the laboratory scale.
Assessment of the inclusion characterization
Mainly alumina inclusions (>99%) were found in the investigated samples. The Al2O3 inclusions would have originated from the Al-deoxidation of the samples since the raw material consists of only (Si,Mn)-oxides and no Al2O3.
No pure REE-containing inclusions were found in the manual and automated measurements in the samples. There are two possibilities for the occurrence of REE-containing inclusions. On the one hand, rare earth oxides can nucleate on preexisting alumina inclusions, and on the other hand, new oxides can form, which contain REE and Al.
Owing to the fast cooling, the difficulties of a precise temperature setting and the centrifugal cast, NMIs were not homogeneously distributed in all samples. Thus, directly comparing the five samples regarding their number of inclusions and distribution was impossible. However, the samples can be compared concerning their amount of traced Al2O3-NMIs and their inclusion size. No significant differences occurred with respect to the mean ECD of the inclusions. The values for all samples were in the range of 1.92–2.56 µm. The ECD deviation was relatively similar for all samples and between 0.92 and 1.48 µm.
As mentioned, the alloying concepts were not only compared by their resulting recovery rates but additionally by their achieved tracing rate. The alloyed amount of REEs was 300 ppm, about 1.75 times higher than the required addition for complete tracing. Therefore, all five samples reached a high tracing rate. Sample 3 achieved the lowest tracing rate compared to the others, with around 96%.
The morphologies of traced inclusions were analysed and compared for all five samples. First, a comparison of the La-traced samples was made. It was found that the La-containing areas of sample 2 were more irregularly distributed than in sample 1 (Figure 4). Furthermore, there was no fixed border between pure Al2O3 and the La-enriched parts in sample 2, where La was added wrapped in an Al-foil. The fineness of the distribution for marked areas inside inclusions mainly depends on the reaction time concerning segregation tendency. Larger and coarser distributed areas were formed at longer reaction times.
Moreover, complex multiphase inclusions would show stronger distinct boundaries between traced and non-traced regions if there was enough time for segregation. It can be concluded that sample 1 had a longer reaction time than sample 2 based on the clearly formed border between the different areas inside the inclusion. La-traced NMIs were irregularly shaped and were more globular than elongated.
Compared to the La-marked NMIs in Figure 4, no significant differences were found in manual SEM/EDS measurements for the Ce-containing inclusions in Figure 5. The segregation process is identical to the inclusion in sample 2 (Figure 4(b)). Even size, shape and elemental distribution are similar. Furthermore, this analysing method can identify no variations between the alloying concepts. The fine distributed areas of REE oxides are primarily owed to the used melting aggregate and the fast cooling within the HFR process. The REE-traced inclusions formed within this aggregate differ from REE-containing aluminium oxides of industrial experiments. The reasons are the tendency of REEs to segregate and the longer reaction and dwell times in the industry. Hence, the REE-containing areas of NMIs formed at industrial trials are more clearly separated and larger compared to the inclusions in this study. An attempt will be made in the next series of experiments to map the industrial process sequence more precisely, i.e. REEs will be added a certain time after deoxidation.
Thermodynamic calculation of inclusion formation after the addition of REEs
The modification of Al2O3 inclusions due to the REE addition on electrolytic iron is shown for Lanthanum (a) and Cerium (b) in Figure 7. Inclusion evolution process of electrolytic iron at 1873 K (a) for La and (b) Ce addition.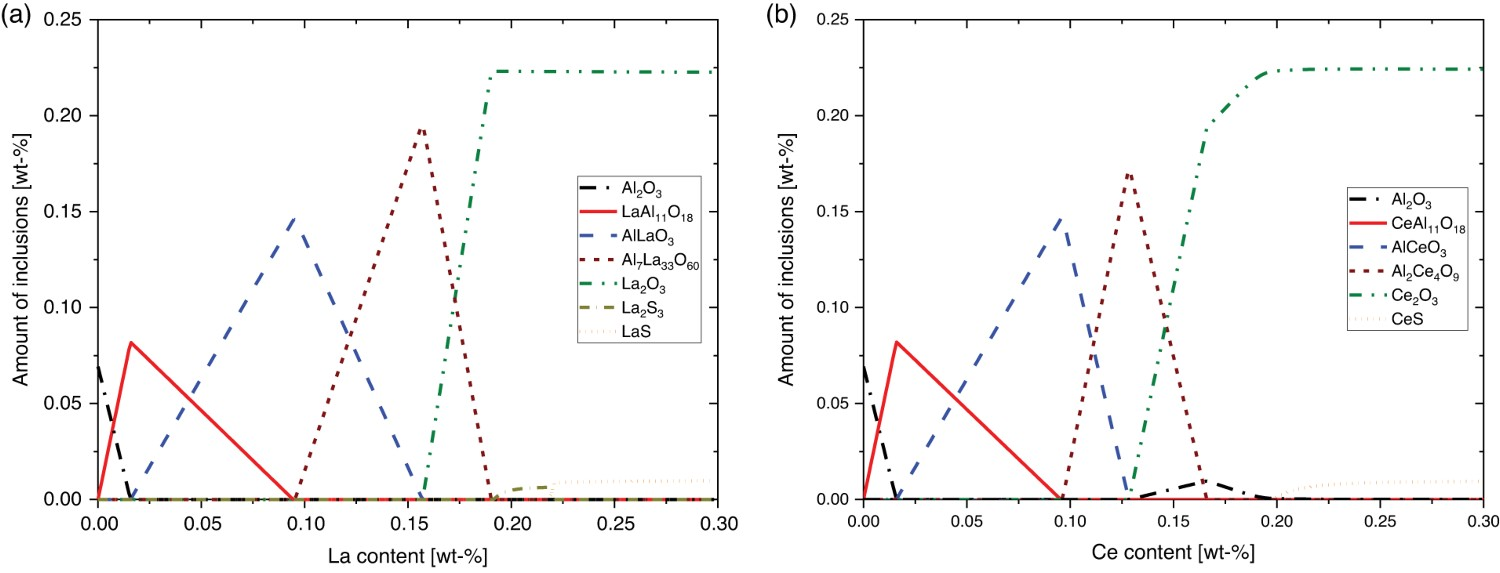
The inclusion evolution was comparable between La and Ce at REE concentrations below 0.13 wt-%. The LaAl11O18- or CeAl11O18-NMIs originate after aluminium oxides, Al2O3, were formed. Higher La-concentrated, complex Al7La33O60 inclusions were generated through the Lanthanum addition above 0.1 wt-%. Compared to La addition, a different complex inclusion type, Al2Ce4O9, occurred for Ce. Parallel to the appearance of Ce2O3 inclusions, Al2O3 has formed again. REE-containing sulphides were formed in both cases at around 0.2 wt-%. The prediction for La contains two different sulphide types, La2S3 and LaS; for Ce, only CeS is predicted. From these thermodynamic calculations, the predicted inclusion path for Ce is Al2O3 → CeAl11O18 → AlCeO3 → Al2Ce4O9 → Ce2O3 + Al2O3 → Ce2O3 + CeS, and for La, it is Al2O3 → LaAl11O18 → AlLaO3 → Al7Ce33O60 → La2O3 + La2S3 → La2O3 + LaS. Luo et al. [29] and Pan et al. [30] determined similar modification processes for Ce additions at high alloyed steels. The changes for ULC steels were illustrated by Wang et al. [27], where the inclusion type CeAl11O18 is defined as a complex of Ce2O3·11Al2O3.
The oxygen content for the electrolytic iron was much higher compared to the steel grades used in these studies. Although the required amount of REE is not comparable to the mentioned studies, the modification steps of inclusions are equal. The inclusion evolution of Lanthanum up to the formation of Al7La33O60 has not yet been described in the literature.
Since REEs bind with oxygen in the melt, the already existing aluminium oxides get reduced and thus modified. Furthermore, an excessive amount of rare earths can also lead to the formation of homogeneous pure REE oxides. Wang et al. [40] have cited the free Gibbs energies for forming oxide, oxide-sulphide and pure sulphide REE bonds. The reactions and the associated free Gibbs energies are shown below [40]: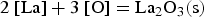
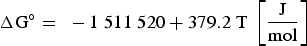
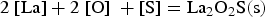
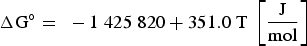
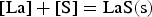
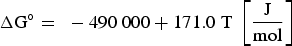
It can be concluded from these reactions that REEs have a greater affinity for oxygen than sulphur. Therefore, mainly modified aluminium oxides and pure REE oxides are formed. It is also possible that chemical oxide-sulphide bonds occur in the presence of sufficient sulphur. Furthermore, it must be noted that dissolved REEs are essential for interactions with aluminium oxides. Concerning thermodynamics, small amounts of dissolved REEs partly reduce the deoxidation product Al2O3 and lead to the modification of these NMIs, as the following equation shows for dissolved [Ce] [19,29]: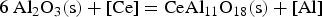
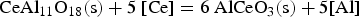
REEs reduce aluminium oxides in both reactions and form new complex inclusions enriched with REEs. Moreover, the amount of dissolved aluminium in the melt increases. The presence of dissolved rare earths also leads to interactions with refractory wear. The melt would be even more enriched with dissolved Al if corundum crucibles were used. [19] No further reactions are predicted in the present study since the reaction time between the crucible and the melt is low during the HFR-melting process.
Comparison between predicted and effectively traced NMIs
Concerning the thermodynamic calculations in Section ‘4.4. Thermodynamic calculation of inclusion formation after the addition of REEs’, the predicted inclusion types depend on the two parameters, REE content and process temperature. Owing to melting in the HFR furnace and accompanying fast cooling, the state of the melt was as-frozen, and so were the therein-contained NMIs. Furthermore, no modification and segregation of other inclusion types took place.
Hence, the addition of 300 ppm of REEs predicts mainly complex Al–REE–oxides. REEAl11O18 and AlREEO3 were formed as per thermodynamic calculations (shown in Figure 7). The amount of La or Ce effectively achieved in the melt was only between 72 and 114 ppm for all samples. A high number of Al2O3 inclusions was also predicted to be present in addition to REEAl11O18 for these REE contents. The theoretically predicted inclusion population was compared to the measured NMIs of the samples using the automated SEM/EDS analysis. The measurement results showed mainly REE–Al–oxides and nearly no pure Al2O3 inclusions. Further, no pure REE oxides were found in any of the investigated samples.
The ternary systems of La–Al–O and Ce–Al–O, illustrated in Figure 8, show the distribution of REE-containing Al2O3 inclusions for all samples. Furthermore, the areas of the predicted REEAl11O18 and REEAlO3 inclusions are marked in the diagrams. All trials led to a similar distribution of REE–Al–O inclusions in the ternary systems. The most significant number of inclusions lies in the region of 25 wt-% of REE, next to the predicted REEAl11O18–NMIs. Ternary system of (a) La–Al–O with the area of LaAl11O18 and AlLaO3 marked, (b) Ce–Al–O with the area of CeAl11O18 and AlCeO3 marked.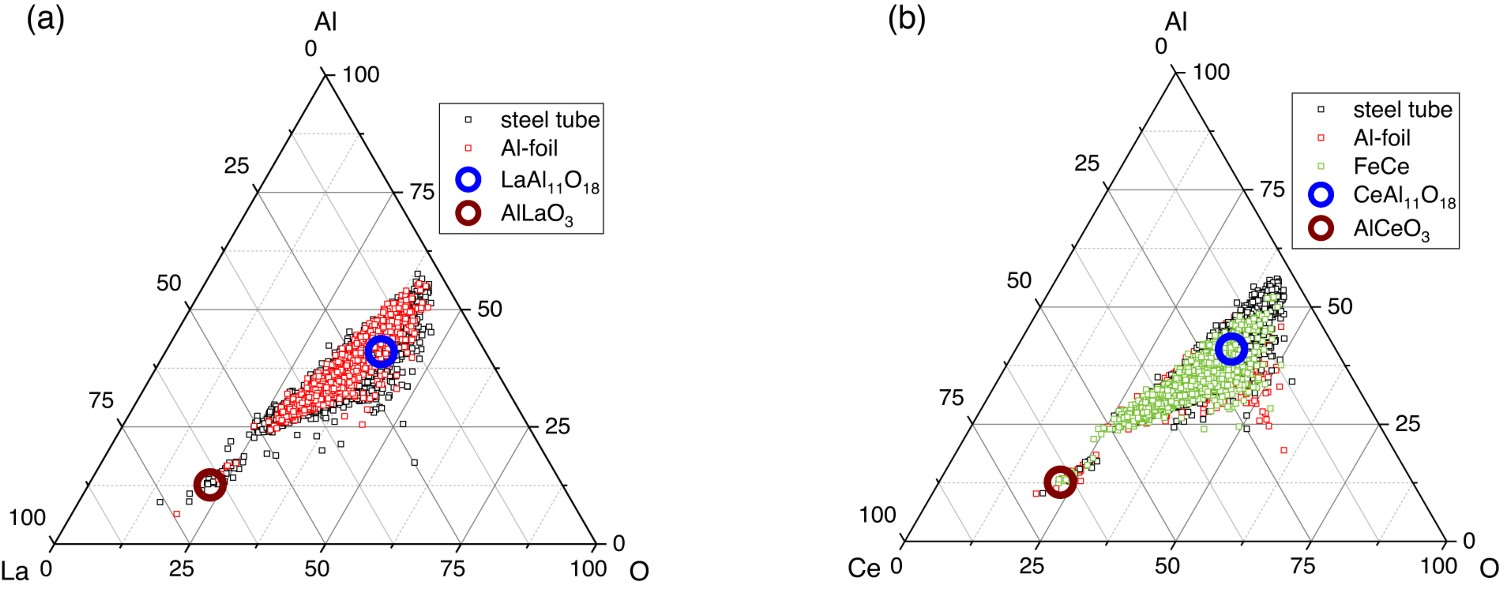
Thermodynamics only depicts the formation of complex REE-containing inclusions and modification of existing alumina inclusions in an equilibrium state. The higher number of traced NMIs can also result from the heterogeneous nucleation of REE oxides to the preexisting Al2O3 inclusions. Comparisons between the predicted and actual inclusion numbers were challenging because kinetics plays a substantial role in forming NMIs within the HFR furnace. However, the influence of kinetics is not considered in thermodynamic calculations. Therefore, thermodynamically expected inclusion types are also valid for industrial trials. Models to contemplate kinetics are necessary for precise prediction of trials on a laboratory scale as well as industrial ones.
Conclusion
In this study, different alloying concepts for the addition of rare earth elements to the steel were investigated on a laboratory scale. The percentage of traced inclusions for three alloying concepts and the associated recovery rates were determined. The formed NMIs were characterized in detail. The main findings relating to the recovery rate of rare earth elements on the laboratory scale can be summarized as follows: On the laboratory scale, significant losses of alloying elements with a high oxygen affinity, such as La and Ce, occur due to the high surface-to-volume ratio and the small added amounts. At the tested alloying concepts for Lanthanum, the addition inside an aluminium foil led to a recovery rate of 30.78%. The recovery rate was higher than the result of the second concept, where La was alloyed inside a steel tube. The difference between them was about 6%. Similar tendencies for the recovery rates of the different alloying concepts occurred for Cerium. The lowest output, 24.33%, was measured by sealing Ce inside a steel tube. The alloying of Ce inside an aluminium foil led to a higher recovery rate of 36.40%. Compared to this concept, the addition of Ce as a ferroalloy with 56 wt-% achieved a slightly higher recovery rate of 38.40%.
Further, the tracing potential and the modification of inclusions due to the rare earth elements La and Ce were investigated, and the following findings can be concluded: (3) The evolved NMIs traced with REEs were similar in size with mean ECDs ranging between 1.9 and 2.6 µm. The inclusion types are mainly REE-marked deoxidation products (Al2O3 + La/Ce) after treatment in the high-frequency remelting furnace. Despite the excessive addition of rare earth elements, no pure REE oxides occurred. (4) As determined by SEM/EDS measurements, complex REE–Al–oxides are the dominating inclusion type in all samples. These NMIs are primarily REEAl11O18, as seen from thermodynamics and the experimental results. Accelerated cooling in the HFR furnace prevents inclusion modification during solidification, and only partial segregations occur. (5) Concerning the effective REE contents in the samples, thermodynamics predicted less traced NMIs than effectively observed in the samples. Based on the thermodynamic calculations, only complex REE-containing inclusions are expected. Considering that heterogeneous nucleation plays an important role, complex multiphase NMIs can also be formed. Since kinetics is not taken into account in thermodynamic calculations, it is primarily a good indicator of which NMIs are produced rather than an accurate prediction. The thermodynamic inclusion predictions can be related to both – industrial scale trials and experiments on the laboratory scale. A detailed analysis of the morphologies via three-dimensional characterization is planned to differ between these inclusion types.
