Abstract
A dual-phase steel chemistry has been processed with varying intercritical annealing temperatures and the properties investigated. It was found that increasing the temperature in the intercritical region led to an increase in the volume fraction (52%–65%) and grain size (1.8–3.3 µm) of martensite whilst the volume fraction and grain size of ferrite decreased. The highest tensile strengths were found at low intercritical temperatures, achieving 935 MPa whilst the highest elongation values of 21% were achieved at the highest intercritical temperatures.
Introduction
Automotive manufacturers are increasingly turning from traditional high strength steels to advanced or ultra-high strength steels (AHSS/UHSS) in a bid to save weight and therefore reduce automobile exhaust emissions, in particular CO2 [1,2]. One of the most extensively used grades of AHSS is dual-phase steels (DP). These DP steels consist of a ductile ferritic matrix, interspersed with a harder martensitic second phase, though other phases such as bainite and retained austenite may be present depending on the chemical composition and thermal treatment [2,3].
Dual-phase steels combine high strength, good formability and continuous yielding [4,5]. The duplex structure is achieved via intercritical annealing, in which the steel is heated and held within the ferrite (α) + austenite (γ) region [6], allowing for austenite to form in the pre-existing ferrite and pearlite microstructure. Rapid cooling is then essential, as it promotes a diffusionless transformation from austenite to martensite [7–9].
An increasing body of research has looked at improvements of DP steels over the last few years, particularly around intercritical annealing. Movahed et al. investigated the effect of intercritical annealing on the tensile properties and work hardening behaviour of DP steel, suggesting equal amounts of ferrite and martensite exhibit optimum mechanical properties [10]. Further developments were made by producing intercritically annealing a cold-rolled DP chemistry with a starting martensitic microstructure, the work done has shown to improve the strength-ductility balance through the refined final microstructure [11–13].
Other bodies have work have focused on the effect that the holding time has on the microstructure and the mechanical properties of DP steels during the intercritical annealing stage, some suggest that the effect of increased holding times does not greatly affect the microstructure [14,15], whilst others showed an improvement in tensile strength compared to other commercially available DP steels [16].
Increasing the intercritical temperature increases austenite formation, which can lead to an increase in the final martensite start temperature [17,18]. This start temperature is important for austenite transformation, and whether it transforms to martensite or bainite on rapid cooling [19]. Additional challenges exist for DP production, particularly on an industrial annealing line which would incorporate an overage section which aids the production of different steel grades.
This study focuses on the influence of intercritical temperature, also known as soak temperature, and the effect this has on the final mechanical properties of a DP steel processed using a similar annealing cycle that would be used on an industrial continuous annealing line.
Methods
VIM cast chemistry of DP steel.
Values shown in weight %.
The bars were then cut to 70 mm × 70 mm blocks using a band saw before being reheated to 1250°C and hot rolled to a target thickness of 3.5 mm. The hot rolling process was controlled to achieve a ferrite/pearlite microstructure. The material was subsequently pickled in HCl to remove the mill scale before being cold rolled to achieve a 55% reduction.
The annealing process was simulated on a continuous annealing simulator (CASIM), which uses resistive heating to heat the material to the required temperatures in air. Rapid air quenching to the required cooling rate is achieved using blow boxes incorporated in the machine. The temperature is controlled via a control thermocouple at the centre of the strip providing continuous feedback, whilst five additional thermocouples which are spot welded to the strip to allow for live temperature readings across the length and width of the sample. The annealing cycles used for this investigation are shown diagrammatically in Figure 1 below and replicates what is seen on a modern continuous annealing line. Continuous annealing cycle showing variations in intercritical temperature, also known as soak temperature.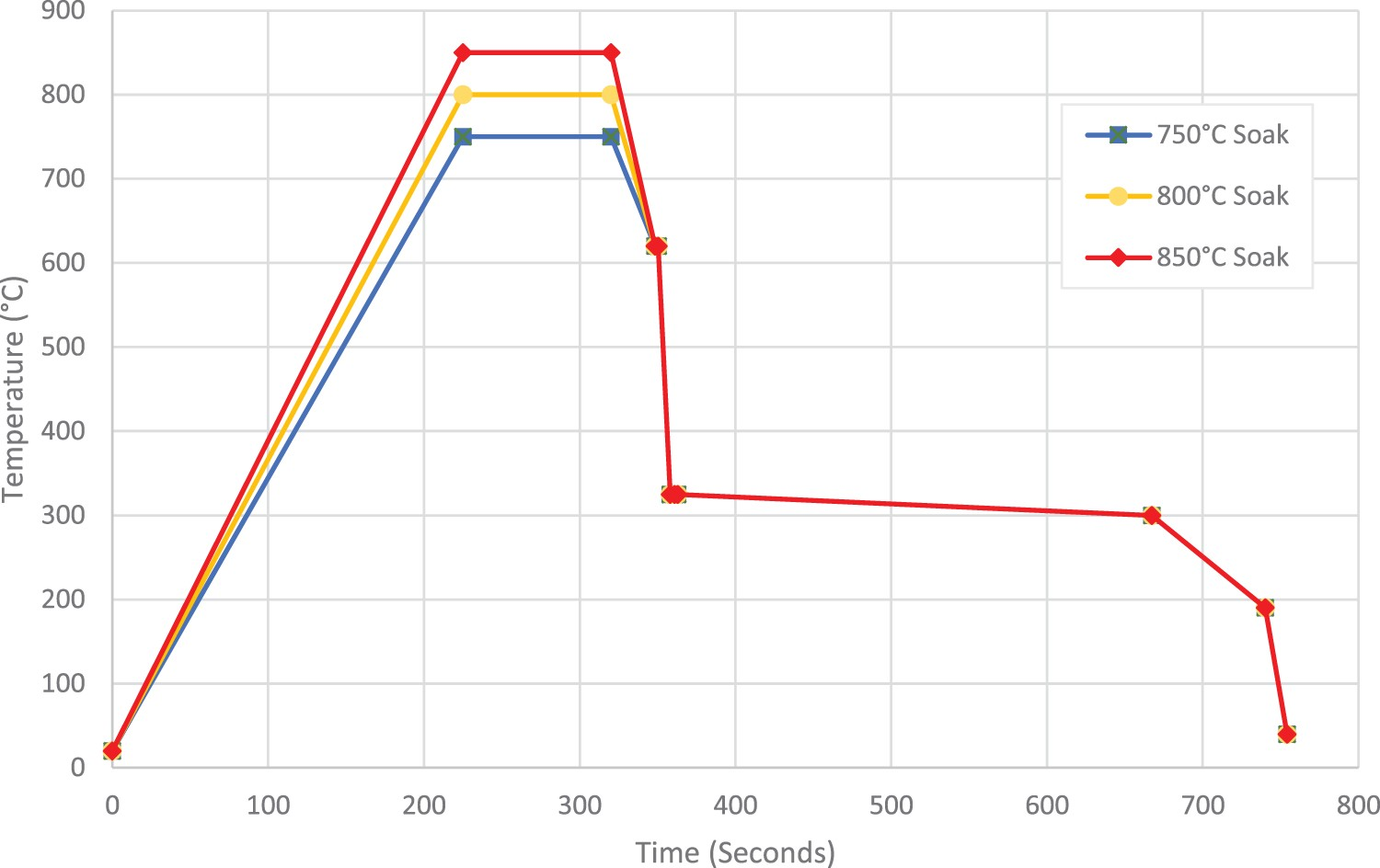
Once annealed, the material was cut to tensile sample sizes with a gauge length of 50 mm, gauge width of 12.5 mm and total length of 180 mm, and tensile tested on a Zwick 1474 tensile testing machine. Testing was performed in a longitudinal direction in accordance with ISO 6982 with an initial displacement rate of 0.00025 mm/s applied, followed by an increase to 0.0067 mm/s once 2% strain was surpassed until fracture. For bake hardening, the samples were subjected to a 2% pre-strain then baked at 170°C for 20 min in accordance with standard EN10325. The samples were then tensile tested in accordance with the ISO 6892 standard to measure the bake hardening (BH) effect.
Metallographic analysis was achieved by setting the samples in Bakelite and grinding through successively finer grit papers to a 1-micron finish and then etched in 2% Nital for 10 s. Samples were then imaged under a Zeiss Observer light microscope and a Zeiss Evo LS25 scanning electron microscope (SEM). Volume fractions of constituent phases and grain size analysis were obtained using ImageJ imaging software.
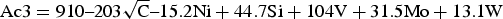
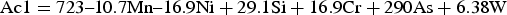
Annealing processing parameters investigated.
Results
Soak temperature effect of microstructure
Heating to the intercritical temperature range between Ac1 and Ac3 will promote the nucleation of recrystallised ferrite (α) and austenite (γ). Austenite has been shown to predominately form during this range at the interphase boundaries of pearlite colonies and ferrite grains [21]. In DP steels, the typical industrial cooling section rapidly reduces the temperature of the steel strip by 30–40°C/s which is sufficient to allow the austenite to transform to martensite (α′), producing the dual-phase structure.
A number of variables from chemistry to processing times and temperatures can affect the microstructure and the final mechanical properties of the material [15,22–25]. The stability of austenite is key to whether martensite, tempered martensite or bainite is produced in the final microstructure. The carbon content in austenite plays a key role in austenite stability, dependant on the volume fraction of austenite in the microstructure and the total carbon content of the alloy. For a given steel composition, the lower the heating temperature in the intercritical region, the higher the carbon content in austenite which therefore leads to lower cooling rate requirements for the martensitic transformation of the austenite phase [26].
As a DP steel is heating up to its soak temperature, the relative proportion of ferrite to austenite will change. At the lower soak temperature, it would be expected that a lower proportion of austenite would be present leading to a lower volume fraction of martensite in the final microstructure, whilst higher soak temperatures will increase the austenite formation and subsequent martensite volume fraction in the final microstructure [18]. The light microscope images below show the initial cold-rolled microstructure in Figure 2, whilst Figure 3 shows the effect of changing soak temperature on the microstructure. Initial cold-rolled microstructure before annealing. White regions are ferrite with dark pearlite islands interspersed. Optical images of 750°C (A), 800°C (B) and 850°C (C) soak temperatures and the effect on the microstructure. The white background represents ferrite (F) with the brown areas being martensite (M).

The 750°C soak temperature shows a heavily banded microstructure, with large veins of martensite and ferrite running parallel with the cold-rolling direction. The low soak temperature has resulted in recovery and recrystallisation of the parent ferrite. The martensite present in the microstructure is also less equiaxed due to the low temperature and short soak time limiting austenite grain growth.
Increasing the soak temperature from 750°C to 800°C increased the volume fraction of martensite in the final microstructure from 52% to 55%, respectively. Large grains of ferrite are clearly present, so grain growth has occurred as well as more refined ferrite formation. An increase from 750°C to 800°C resulted in martensite grains increasing from 1.8 µm to 2.7 µm, whilst the ferrite grain size decreased from 4.2 µm to 3.1 µm. The increased volume fraction of martensite has also been accompanied with an increased martensite grain size which is more equiaxed.
At 850°C soak temperature, there is less banding present and the grains are more equiaxed. The ferrite and martensite phases have become more uniformly distributed and the grain size of the martensite is much larger compared to the lower soak temperatures. The larger ferrite grains present in the lower soak temperatures are also not present, therefore it is likely that the austenite growth is competing with the ferrite, this leads to the average ferrite grain size decreasing from 3.1 µm to 2.6 µm. The volume fraction of martensite and grain size of martensite is highest at 850°C soak temperature at 65% and 3.3 µm, respectively.
Figure 4 shows the SEM micrographs of the three different heat treatments. Scanning electron microscope images (SEM) of 750°C (A), 800°C (B) and 850°C (C) soak temperatures. Ferrite (F) is shown as the dark background whilst martensite (M) is the lighter phase. Also note the presence of tempered martensite (TM) at higher soak temperature.
From these SEM images shown in Figure 4, the banding of martensite and ferrite seen in the light microscope images are present at lower magnification. At the higher temperature, the final martensite grains become larger and more equiaxed.
Phase fraction analysis and grain size analysis on each of the soak cycles.
Effect on mechanical properties
Changes seen in the microstructure under the microscope influences material behaviour during mechanical testing. The values below are shown as averages, with the error bars dictating the minimum and maximum values achieved from the tensile tests.
The tensile test results for UTS in Figure 5 show the highest strength was measured on the sample exposed to the 750°C soak temperature, achieving an average of 935 MPa. The second highest strength values were found at 850°C soak temperatures at 893 MPa, whilst the medium value of 800°C soak had the lowest strength on average at 860 MPa. The highest soak also provided the most variability in results, though all UTS results were shown to be within 20 MPa of each other. Effect of soak temperature on the ultimate tensile strength (UTS). Effect of soak temperature on 0.2% proof strength.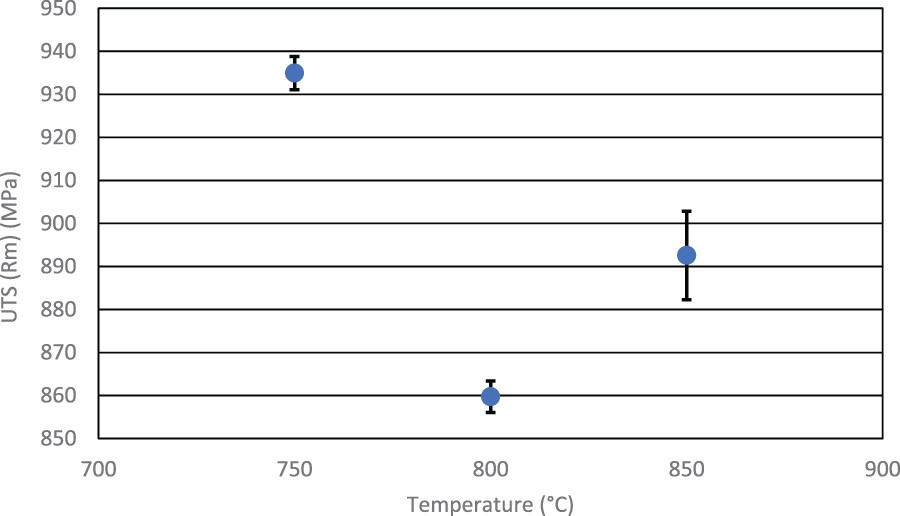
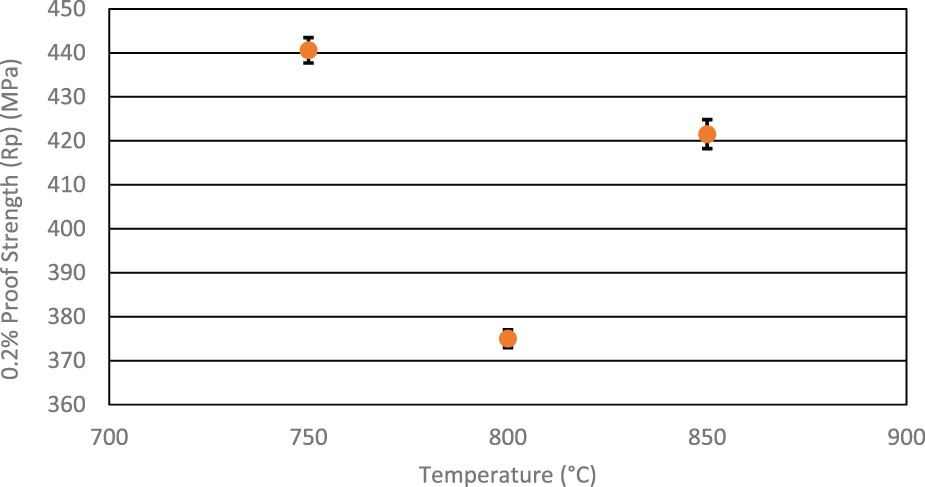
The results from the proof strength shown in Figure 6 follow the same trend as the UTS results. The highest proof strengths were seen on the 750°C soak, at 441 MPa, and 850°C soak, at 422 MPa soak temperature. The lowest values were seen at 800°C soak temperature with results of 375 MPa.
Figure 7 shows that total elongation values increase with soak temperature. The 750°C soak temperature achieved 14%, whilst at 800°C the value increased to an average of nearly 18%. The 850°C soak temperature achieved an average of 21% but showed the highest scatter. Effect of soak temperature on total elongation properties.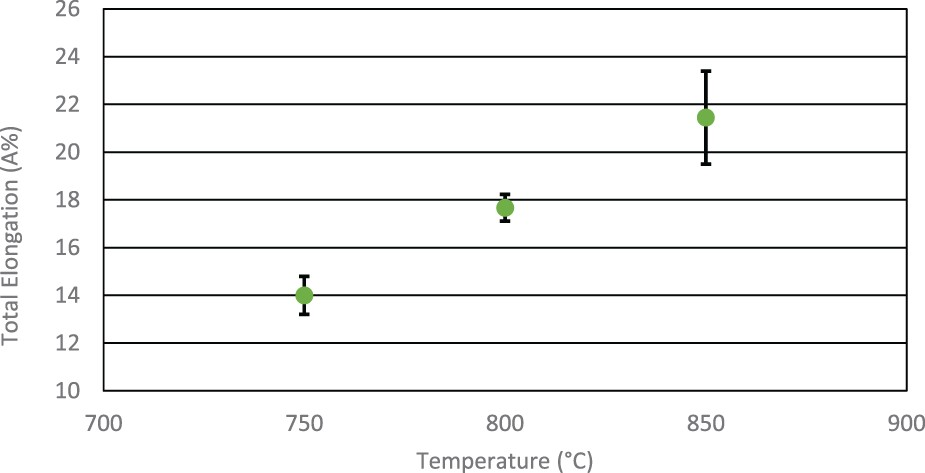
Bake hardening occurs when an automotive panel is heated to around 150–200°C to cure the paint layer. At this temperature free dislocations become pinned by carbon in Cottrell atmospheres, resulting in an increase in yield strength and is defined as the bake hardening response [27].
Bake hardening occurs with ageing of ferrite and the tempering of martensite in the following stages: the formation of Cottrell atmospheres, carbon clustering and growth and tempering of martensite [28].
The values in Figure 8 show the highest response occurs at the middle range of the soak at 800°C, achieving an average of 50 MPa. The worst BH response occurs at 850°C where only a 24 MPa increase is obtained. The results show the highest soak temperature also exhibits the highest variability in BH response. Effect of soak temperature on bake hardening properties.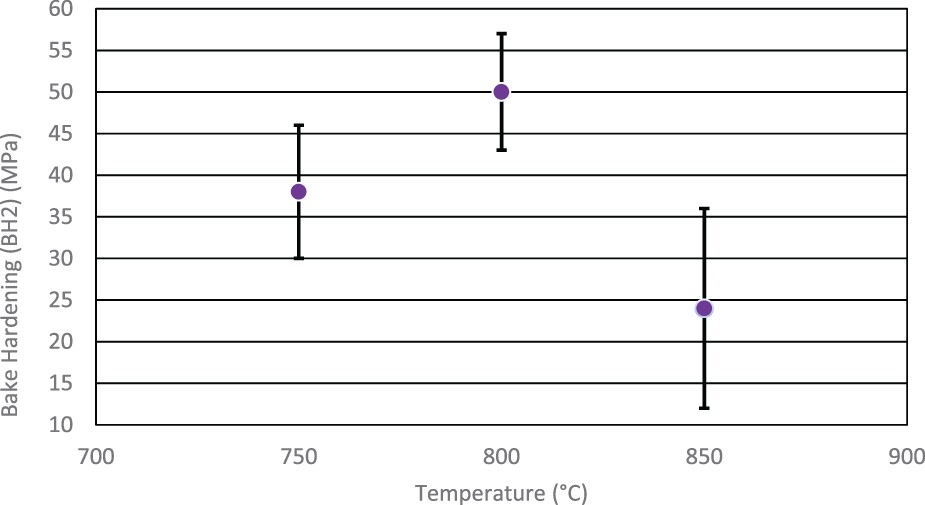
Discussions
The banded microstructure at low soak temperatures is prominent. During the cold-rolling process, the grains are deformed in the direction of rolling leading to thin elongated grains that provide high hardness but also poor ductility. The cold-rolling process generates a large volume of dislocations within the grain structure which act as sites for nucleation during the annealing process.
During the soak stage of the continuous annealing cycle, the time at elevated temperature promotes nucleation of austenite, which occurs primarily at the interphase boundaries of the existing pearlite and ferrite [29]. These grain boundaries tend to be more energetically favourable as sites for nucleation than within grains themselves.
The time in which ferrite takes to recrystallise can be restricted by the presence of manganese, as well as the additions of chromium and niobium. Work done by Garcia and DeArdo [30] shows that at low soak temperatures, combined with short soak holding times, allow only for ferrite recovery, rather than recrystallisation. Even at low temperatures, there looks to have been sufficient time to allow for recrystallisation to occur from the annealing cycles used in this investigation.
The growth of austenite along the rolling direction is related to the banding present in the existing cold-rolled structure. The potential for manganese to segregate within the pearlite bands can cause regions of high manganese-containing pearlite which has been reported to accelerate the transformation of pearlite to austenite [21].
One of the major determining factors for the final transformation to austenite to either martensite or bainite is related to its carbon content [31]. Typically, a low volume fraction of austenite in the microstructure would lead to austenite that has high levels of carbon present with high hardenability [32]. At lower soak temperature, the stability of the austenite would increase due to the lower volume fraction of austenite becoming saturated with carbon. This shifts the bainite transformation to lower cooling rates, increasing the likelihood of martensite in the final microstructure [33].
The refinement of the martensite in the final microstructure explains the higher strengths present in mechanical properties at 750°C soak. A lower volume fraction of refined martensite, as described above, has an increased carbon content and increased hardenability that results in higher strength, similar to results seen in work by Girina et al. [31].
At 800°C, there is grain growth occurring in the austenite; however, this is competing with the recrystallisation of ferrite and is therefore still limited in the direction of rolling. The other notable difference between the 750°C and 800°C soak is the presence of large ferrite and martensite grains dispersed throughout the microstructure. These can benefit the ductility of the steel but would also lower the overall strength.
Girina et al. suggest that the low stability of austenite is responsible for lower strengths in mid-range soak temperatures in comparison to lower soak temperatures. In the same study, increasing the temperature close to the fully austenitic intercritical region showed an increase in strength, primarily due to the refinement of ferrite [31], which is also seen in this study with the highest soak temperature leading to the smallest ferrite grain size. One noted improvement at the 800°C soak was the bake hardening response. During the rapid cooling, the carbon concentration can be retained within the ferrite, this can then lead to an increase in the strengthening by free carbon pinning dislocations within the ferrite [21].
As the austenite grains grow, there is a restriction of the ferrite growth as the existing pearlitic structure is consumed by the austenite. As the austenite grain size increases, carbon content and hardenability decreases. During the slow cooling section, the austenite grains that have low carbon content are first to transform to new ferrite. This in turn enriches the remaining austenite as carbon is ejected from the ferrite [34].
Areas of austenite that are supersaturated in carbon sufficiently depress the Ms temperature and transform readily to martensite during the final cooling after the overage section, rather than during the overage section. Conversely, an increased volume fraction of austenite can mean lower carbon enrichment [5]. This means the austenite is not sufficiently enriched, allowing for martensite transformation to occur before or during the overage section. This in turn leads to tempering of the martensite which can be seen in Figure 9. Tempered martensite presence after cooling from high soak temperatures. Tempered (TM) shown middle of the image surrounded by ferrite (F) and martensite (M) islands.
The presence of bainite can also lower the overall strength of a DP steel. Work by Fonstein et al. [35] suggests that bainite provides 30% lower strengthening than martensite. In the same work, it is suggested that the presence of bainite leads to an improved yield strength and elongation. Tempered martensite would also have a similar effect [36].
Along with equiaxed ferrite, the presence of bainite or tempered martensite can account for the higher levels of elongation shown in comparison of the 750°C and 800°C soak. Typically, a larger grain size can also contribute to higher levels of ductility too. As previously mentioned, larger martensite grains contain less carbon, which reduces its hardenability but increases its ductility.
The higher strength observed in the 850°C in comparison to the 800°C soak temperature is likely to due to the 10% increase in the volume fraction of martensite present in the final microstructure. Whilst a small fraction may be tempered, there is sufficient martensite present to increase the overall strength of the steel by 33 MPa, this can also be combined with the fact that the ferrite grain size is more refined than that of both the 800°C and 750°C soak temperatures, due to the increasing martensite volume fraction.
At 850°C, it is likely that the increase in new ferrite in the microstructure occurring during cooling from a higher soak temperatures would reject carbon, therefore leading to significant enrichment of the surrounding portion of austenite with carbon [21]. The enrichment of the surrounding austenite would increase its hardenability and also delay the onset of bainite transformation. Conversely, the interstitial free ferrite would have insufficient free carbon to pin dislocations in the ferrite, this in turn would lower the BH response.
Conclusions
Volume fraction and grain size of martensite increase with increasing soak temperature, higher soak temperatures also led to tempered martensite in the final microstructure. Increasing the soak temperature led to an increase in martensite grain size and a decrease in ferrite grain size. The best combination of strength and ductility was achieved at the highest soak temperatures, but resulted in poor BH performance.
Footnotes
Acknowledgements
The authors would like to acknowledge the M2A funding from the European Social Fund via the Welsh Government, the Engineering and Physical Sciences Research Council and Tata Strip Steel UK that has made this research possible. We would also like to acknowledge the SEM support from the Advanced Imaging of Materials (AIM) centre at Swansea University.
Disclosure statement
No potential conflict of interest was reported by the author(s).
