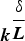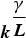Abstract
There is a growing drive within the steel industry to increase scrap usage in the Blast Furnace (BF)-Basic Oxygen Furnace (BOF) integrated route and extend the scrap-based Electric Arc Furnace (EAF) route for steel production due to the resultant reduced energy costs and CO2 emissions and the abundant steel scrap supply in the UK. In general, steel scrap may contain high levels of undesirable residual elements, which may have pronounced effects on the casting process of steel production. This article critically reviews current metallurgical understanding about the behaviour of various residual elements, individually and synergistically at high (>1200°C) and low (<1200°C) temperatures during the casting process, with a focus on local enrichment and cracking caused by the residual elements. This review article aims to help the steel community to increase the utilisation of steel scrap for steel production by identifying the current constraints and opportunities.
Introduction
Steel scrap is used as a raw material in the Blast Furnace (BF)-Basic Oxygen Furnace (BOF) and Electric Arc Furnace (EAF) processes during steel production. Steel production by the EAF route can process 100% of steel scrap as the iron-bearing charge, while BF-BOF can take up to 20% of steel scrap [1–3]. Figure 1 shows the proportions of crude steel production (%) by the two different routes (BF-BOF and EAF) for some of the main steel producing regions of the world in 2017. Most of the countries rely on the BF-BOF route, while India, Turkey and USA have a large share (India 54.5%, Turkey 69.2% and USA 68.4%) [4] of steel production via the EAF route [5]. Among some of the key European Union (EU) steel countries, Italy (80.3%) and Spain (66.6%) have high steel production amounts via the EAF route [4]. Crude steel production (%) by the BF-BOF and EAF routes in 2017 for different steel countries and union (adapted from Steel Statistical Yearbook 2018, World Steel Association [4]).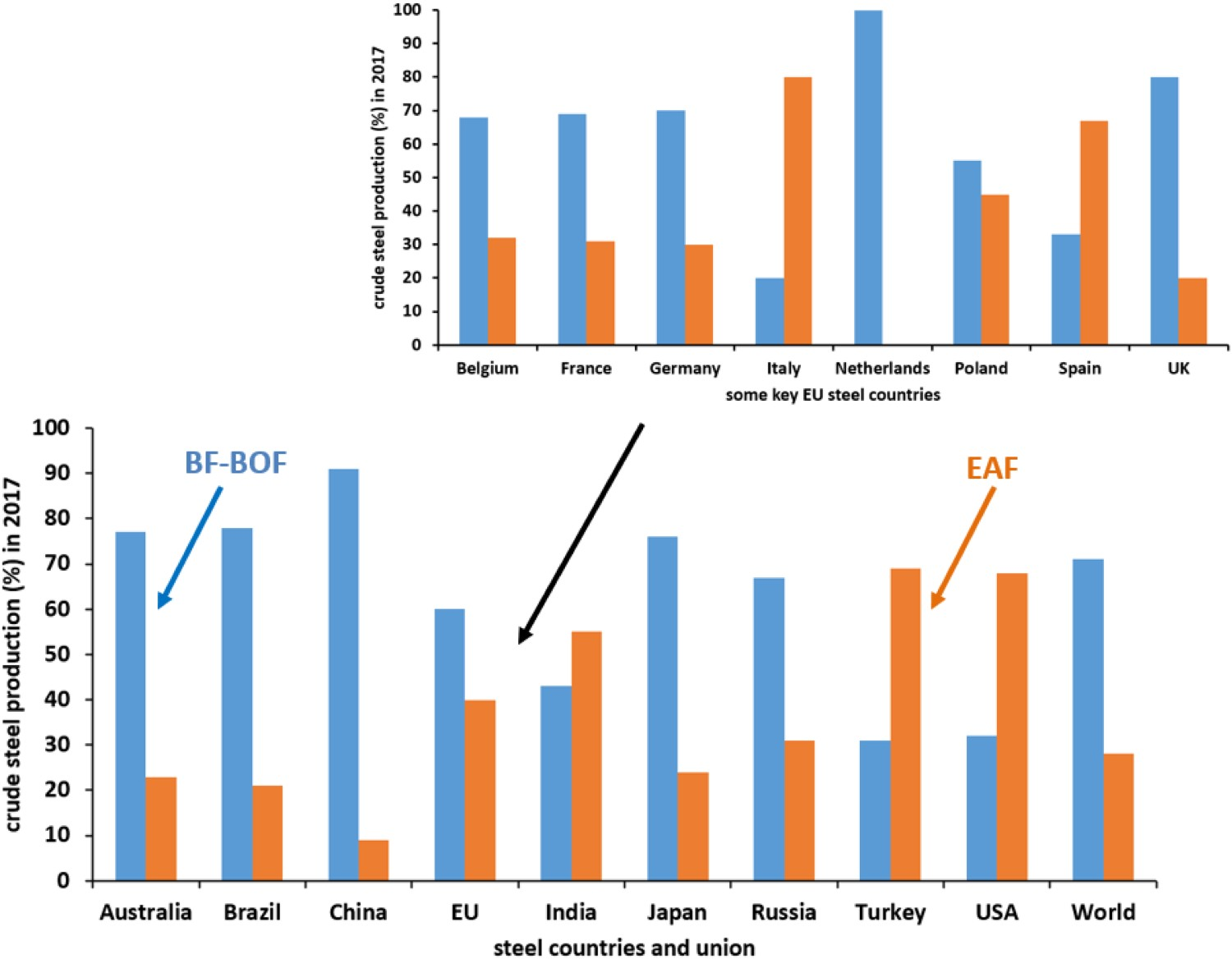
There is a growing interest among steel making industries to shift towards EAF from the BF-BOF route to reduce energy costs and CO2 emissions. It is estimated that steel production by the EAF route will surpass the BF-BOF route globally by 2060 [6]. The energy required to produce steel from steel scrap in the EAF is approximately half of that from the reduction of iron ore in the BF-BOF [3]. The amount of CO2 emissions per tonne of crude steel from the scrap-based EAF route (∼500 kg of CO2 emissions per tonne of crude steel) is less than a third that from BF-BOF route (∼1800kg of CO2 emissions per tonne of crude steel) during steel production [7,8].
The steel scrap available in the market is either the byproduct of steelmaking and steel product manufacturing or from outdated products/machinery. Pflaum [9] reported the breakdown of scrap purchased in steel mills and foundries in the USA as shown in Table 1. Figure 2 provides the proportions of UK scrap production by different manufacturing sectors [10,11]. Prompt scrap generally arises from manufactured steel products during boring, cutting, punching, turning, etc. operations. Scraps purchased from these various sources (Table 1) are processed by scrap dealers (sorting, shredding, etc.) before shipping to steelmaking facilities [9]. Generally, steel scrap after the refining stage contains higher amounts of elements (known as residual elements) which are not required at the levels that arise and are generally harmful to steel manufacturing processes and final steel product properties. Residual elements commonly present in steel scrap are Cu, Ni, As, Sn, Mo, Cr, Zn, Pb, V, Nb, etc. [3,12–15]. Elements such as Nb, Ti and V are commonly regarded as microalloying elements, for example, being present in High-Strength Low-Alloy (HSLA) steels [16–18] but when present in higher levels these elements could be regarded as harmful and undesirable. Residual elements (such as Cu, Ni, Mo, Sn) are not easily removed from the liquid metal during steelmaking in the EAF and may build up to a higher level with continued steel scrap recycling [12]. Scrap purchased in steel mills and foundries in United States [9].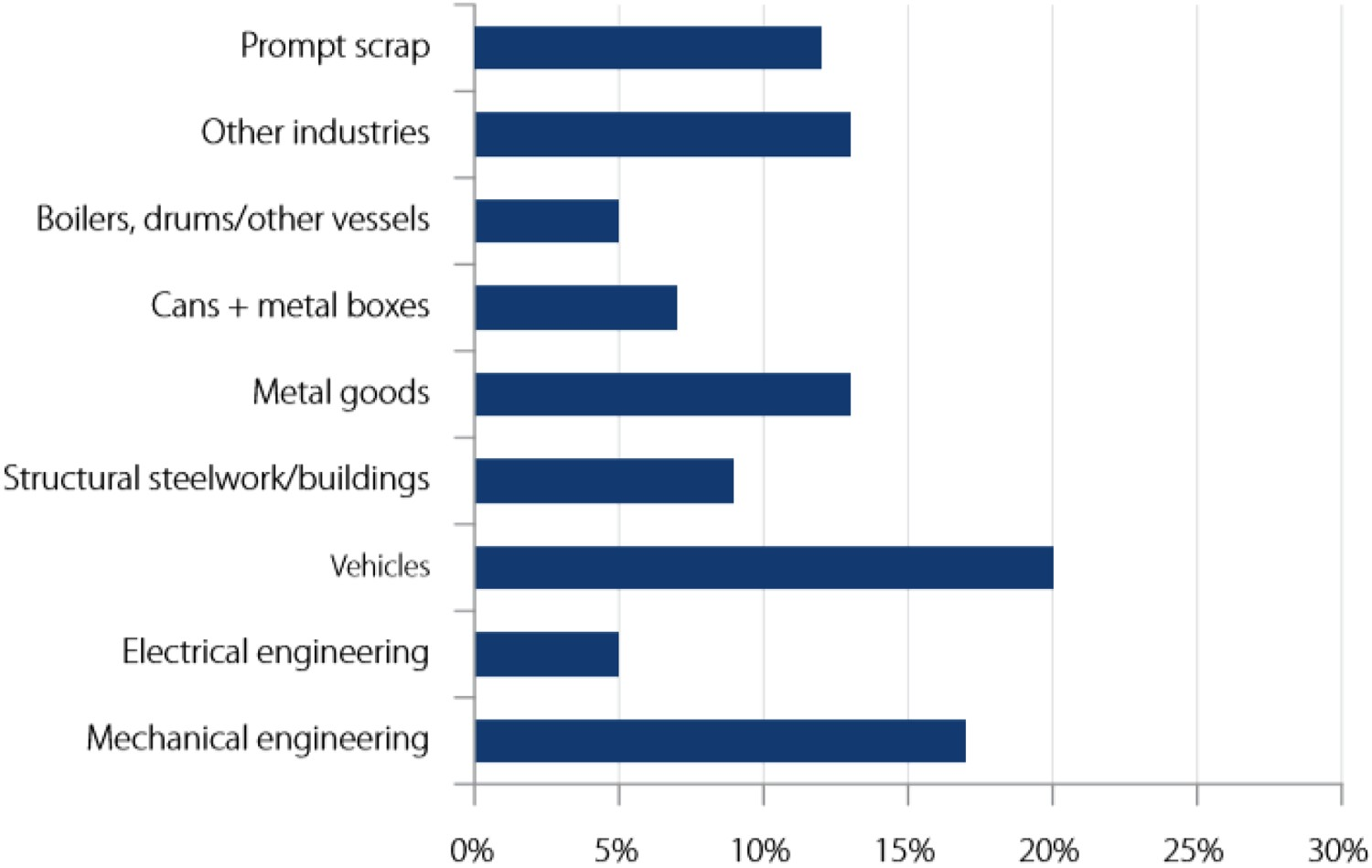
Residuals are introduced into the steel scrap from various sources during recycling of steel products [3,19]: Cu – From bearings [20], electric wires [21], structural steel [22]. Sn – From bearings [23], coated plates such as cans [24] and solders [25]. Zn – From coated plates [24]. Pb – From bearings [26], solders [27], free machining steels [28] and paint colouring agents [29]. Ni – From coated plates [24] and present as alloying elements in steel as Fe–Ni alloy [30]. Cr, Mo, V, Nb, Ti, B – Mostly present as alloying elements in steel [31].
Chemical compositions of residual elements of typical market scrap in Japan (taken from Noro et al. [32]).
*before melting.
In general, higher amounts of residuals in steel scrap have a deleterious effect on downstream processes (hot-working, cold-working, etc.) during steel production such as surface cracking (hot-shortness), grain boundary segregation, and grain boundary embrittlement and consequently lead to poor processability and/or end user properties [35,36]. The presence of residual elements in steel scrap can, however, have a positive impact on final properties. For example, Cu combines with S and forms copper sulphide particles during strip casting and can increase the strength of steel by precipitation strengthening [37,38]. Moreover, fine Cu (∼4 nm) precipitates increase strength and toughness of aged HSLA-80 and HSLA-100 naval steel grades [39,40].
There have been extensive studies on the effect of residual elements on hot-working, cold-working and annealing processes, and end user properties but much less focus on the casting process of steel production [3,36,41]. The casting process is a critical interfacial stage between liquid steel processing and downstream solid processing. The presence of higher levels of residual elements from steel scrap may cause micro- and macroscale phenomena (such as segregation) or problems (such as cracks) during the casting process. These defects may remain present as they are, or in modified forms, in the different stages of downstream processing (hot-working, cold-working, etc.) and could possibly deteriorate the microstructural and mechanical properties of the final steel products. Therefore, it is important to understand the role of residual elements on the casting process and guide the steel community to increase the utilisation of steel scrap in steel production. The casting processes which are considered in this paper are ingot casting (IC) and conventional continuous casting (CCC).
Casting
Ingot casting (IC), where liquid metal is poured into a mould and left to cool for a certain period (from seconds to a day) [42], and continuous casting (CC), where a continuous piece of steel is cast of an un-interrupted length [43] are used for steel production. Steels produced from CC are semi-finished products such as blooms, billets, slabs [44] and sheets [45].
Segregation during the solidification stage of casting process
Solidification of steel during the casting process comprises of (i) dendrite formation during solidification, (ii) dendrite coarsening and impingement during solidification and (iii) equiaxed grain formation at the end of full solidification and bridging [46,47]. There is also a chill zone at the mould surface where relatively rapid cooling produces fine equiaxed grains [48]. Inter-dendritic segregation is a common problem in as-cast steel. During solidification the solid phase rejects solutes into the liquid phase because the solubility of rejected solute in the solid phase is lower than in the liquid state. Solutes tend to release from the solid phase ahead of the solidification front and get trapped in between dendrite arms, commonly known as microsegregation [49–51] (length range from 0.01 to 0.1 mm) [46] or forms a highly enriched core of solute of length range from 10 to 1000 mm within the sample, commonly known as macrosegregation [42,52,53]. Microsegregation can be minimised by a homogenisation heat treatment allowing re-distribution of solute atoms by diffusion. For most continuous cast steels the reheat time/temperature do not allow full homogenisation of substitutional alloying elements so microsegregation remains. While macrosegregation [54] cannot be removed by diffusion as the length scale of segregation is in the order of ∼104 µm [42]. In this review, the inter-dendritic segregation (microsegregation) of residual elements is considered. For detailed information on macrosegregation, the reader is referred to the review work, ‘Macrosegregation in Steel Ingots: The Applicability of Modelling and Characterisation Techniques’ by Pickering [42].
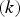
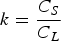
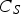
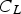
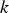
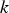
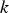
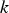
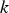
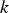
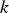
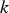
Slater et al. [74] predicted increased inter-dendritic segregation of Nb solute (increase in ratio of Nb content in solute maximum to solute minimum region from 2.9 to 4) with an increase in cooling rate from 1°C s−1 to 100°C s−1 (in the study by Slater et al. [74], cooling rate is defined for the stage of cooling from liquidus temperature to 1000°C). The higher cooling rate promotes higher segregation and minimises the time for back-diffusion to occur [74,75]. Dobrovska et al. [76] estimated a back-diffusion parameter for different solute elements (C, Si, Mn, P and S) in steel based on different microsegregation models. They predicted that the back-diffusion parameter of Mn (∼1.4×10−3 at a cooling rate ∼1.1°C s−1 and ∼8.3×10−4 at a cooling rate ∼6.4°C s−1, using the Brody-Flemings model [77]) is the lowest as compared to other elements. With an increase in cooling rate, the back-diffusion parameter decreases and therefore, the rate of diffusion balancing of concentration decreases.
The partition coefficient of Sn is low (Table 3) which increases the chances of microsegregation of Sn during the solidification process [78]. Zhang et al. [79] calculated the extent of Sn microsegregation in boiler and pressure vessel steels, it was found that the initial contents of Si, Mn, P and S have an insignificant effect on the degree of microsegregation of Sn. The extent of inter-dendritic segregation of Cu during solidification has not been reported in detail in the literatures. Since the partition coefficient reported for Cu is higher than Sn (Table 3) and therefore, Cu may not show strong tendency of inter-dendritic segregation as that of Sn during solidification stage of casting process. Partition coefficient of Cu is similar to Mn (Table 3) but due to higher diffusion coefficient of Cu (∼2.24×10−10 cm2 s−1 at 1050°C [80]) than Mn (∼7.84×10−12 cm2 s−1 at 1050°C [64]), the tendency of Cu back-diffusion could be more than Mn and therefore, chances of inter-dendritic segregation of Cu could be less than Mn during solidification stage of casting process. Later in the review, solid state segregation (controlled by diffusion of vacancy-impurity complexes [81]) is also discussed in relation to Cu and Sn and their effect on straightening operations during the continuous casting process.
Cracking problems in cast steel in temperature zone higher than 1200°C
Cracks can form in continuous cast steel due to internal strain generated during surface expansion (thermal strain) or during mechanical bending and straightening between rollers (mechanical strain) [82]. Thermal stresses originate due to temperature gradients through the cast [56]. The mould associated with the continuous casting process is constantly oscillating to avoid melt from sticking to the walls. These oscillations give rise to transverse ripples (or oscillation marks) on the surface of the continuous cast strand. Owing to the oscillations, cracks initiate inside the mould and align in the direction of the oscillation marks on the surface and/or at the edges of the continuous cast strand. During the straightening operation (Figure 3), the top surface and edges are in tension and promote crack propagation. These cracks are observed up to 5–8 mm depth below the surface and propagate along austenite grain boundaries [83]. Schematic diagram of continuous casting machine (taken from Mintz et al. [83]).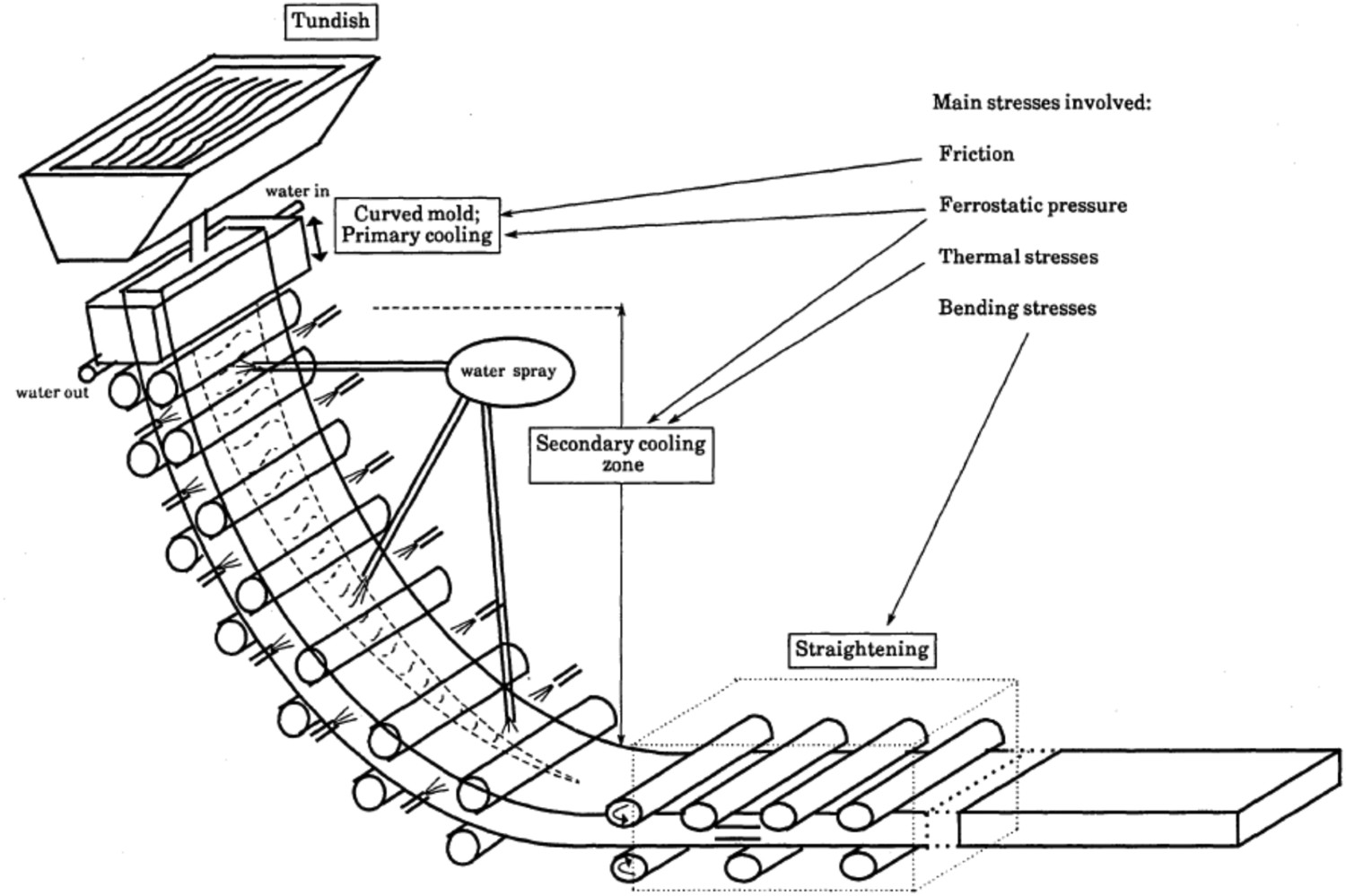
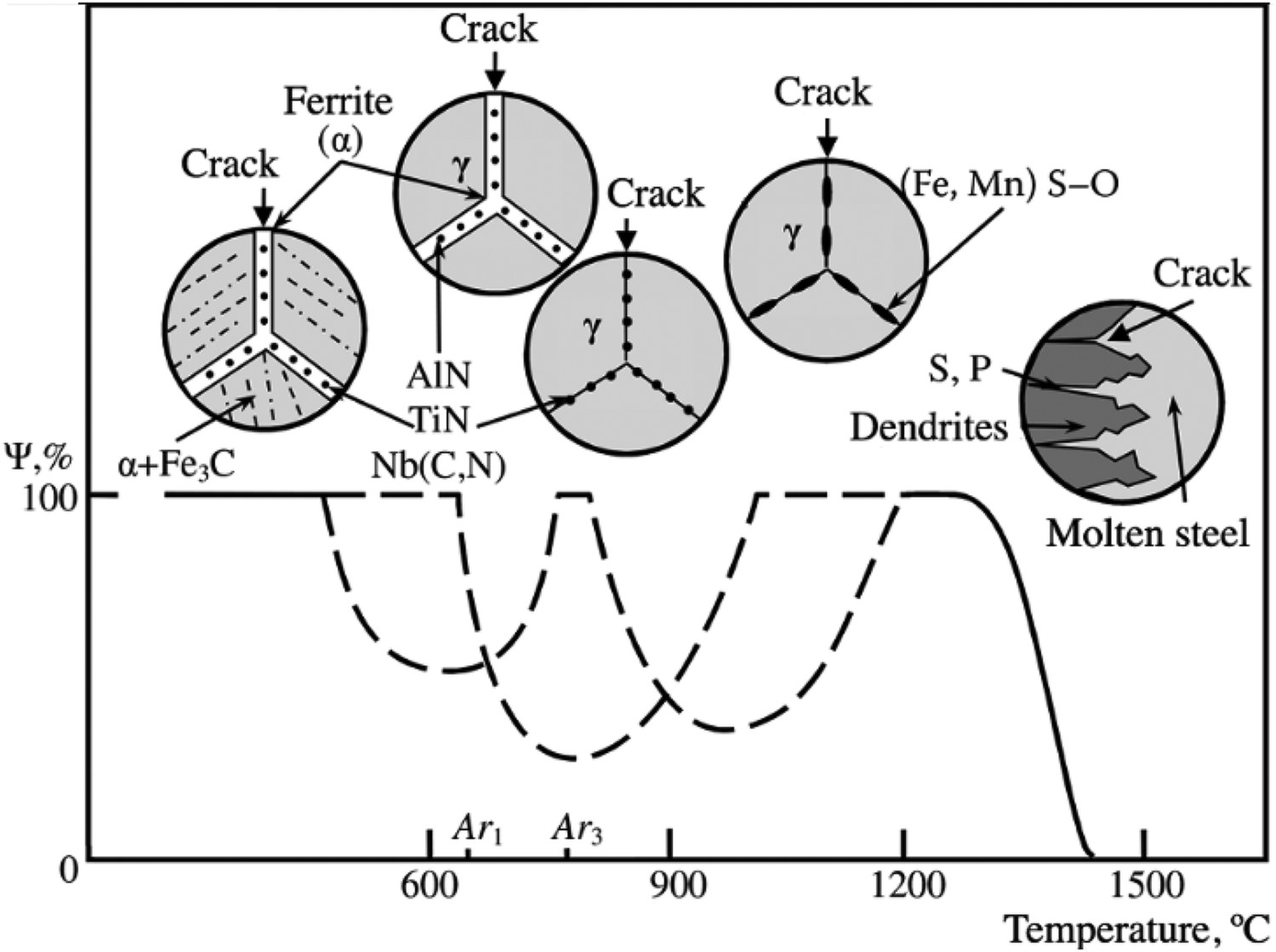
Previous works [56,84,85] have used high temperature tensile tests to simulate and understand the formation of solidification cracks during the casting process. Scheller et al. [86] schematically (Figure 4) presented the temperature range where the material will show a propensity for cracking due to low-ductility during the casting process. For the temperature range from temperature of zero ductility (TNZ) to temperature of zero force (TNF), the sample is brittle due to there being a partially solidified microstructure. Strength and ductility increase on full solidification of the steel because of a gain in plasticity [87]. TNZ is the temperature at which the microstructure starts transforming from low ductility (reduction in area, RA % ∼ 0%) to high ductility in nature (fraction of solid is in between 0.98 and 1) [46]. The parameter, ΔTO (= TNF – TNZ) [86,87] is known as the brittle temperature range [46] and is used as a physical quantity to describe the tendency of hot-crack formation during the continuous casting process [84,86]. Moreover, the temperature range of ΔTO is equivalent to the temperature range of the low-ductility zone (T > 1200°C) as shown in Figure 5. Schematic representation of reduction of area (RA) and maximum force as function of temperature (taken from Scheller et al. [86]). Schematic representation of different temperature zones of reduced hot-ductility of steel for different embrittling mechanisms (taken from Fedosov et al. [85]). Ψ % in y-axis is reduction of area (RA (%)).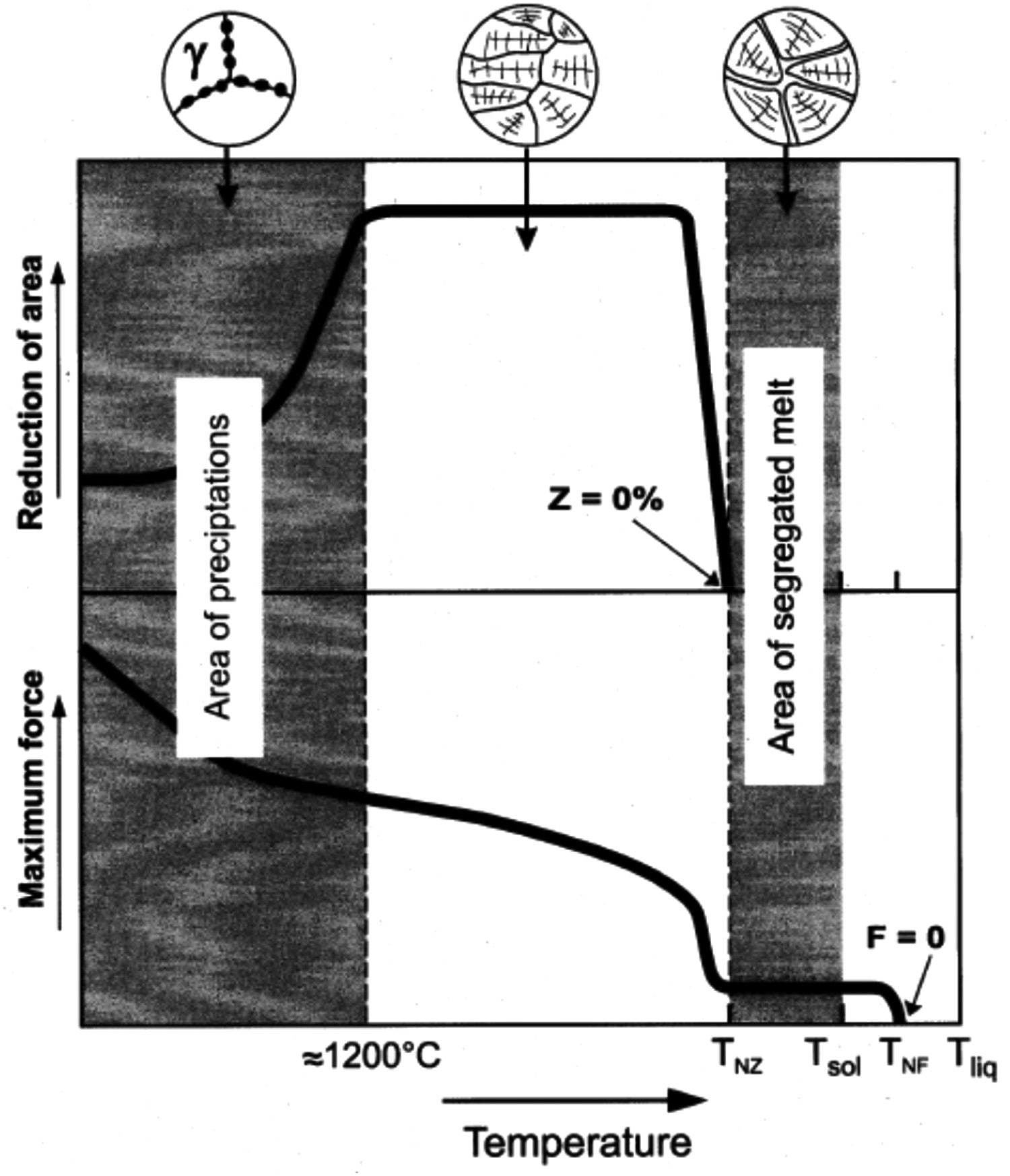
Role of residual elements in casting process in temperature zone higher than 1200°C
During the casting process, higher concentrations of P and S, due to segregation [84,88], occur at the solidifying dendrite interfaces as shown in the low-ductility zone (T > 1200°C) of Figure 5. Micro-segregation of P and S residual elements decrease the solidus temperature locally in the inter-dendritic regions [89,90] and widen the ΔTO [46]. Application of stress (originating from bending or sticking of the cast in the mould [56] or thermal contraction or volumetric change during delta to gamma phase transformation [87] or other external source) leads to inter-dendritic cracking (commonly known as hot-tearing) in the low-ductility zone (T > 1200°C). During grain formation at the end of full solidification and bridging there might be a thin liquid film with high concentrations of solute elements (S and P) present along the grain boundary which could lead to inter-granular cracking on application of external stress [46].
Segregation of P and S has a more significant effect on the critical strain, εc (function of strain rate and brittle temperature range [87]) required for inter-dendritic failure than Si and Mn. There is a significant decrease in critical strain required for inter-dendritic failure with an increase in concentrations of P (0.01 wt-% to 0.08 wt-%) and S (0.01 wt-% to 0.08 wt-%). While, there is no significant change in critical strain (for example, Δεc ∼ 0% for 0.4 wt-% of C) with an increase in Si (0.015 wt-% to 0.12 wt-%) and Mn (0.2 wt-% to 1.6 wt-%) levels as shown in Figure 6 [87]. Lower equilibrium partition coefficients (Table 3) and diffusion coefficients (Table 4) between the solid and liquid phases for P and S as compared to Si and Mn promote severe inter-dendritic segregation of P and S and lead to hot-tearing. Effect of silicon (a), manganese (b), phosphorus (c) and sulphur (d) concentration on critical strain (εc) for internal crack formation at a strain rate ∼10−4 s−1 (taken from Won et al. [87]). Equilibrium diffusion coefficients of elements (data taken from Ueshima et al. [64]). Note: R is 8.314 J mol−1 K−1; T is temperature in Kelvin, 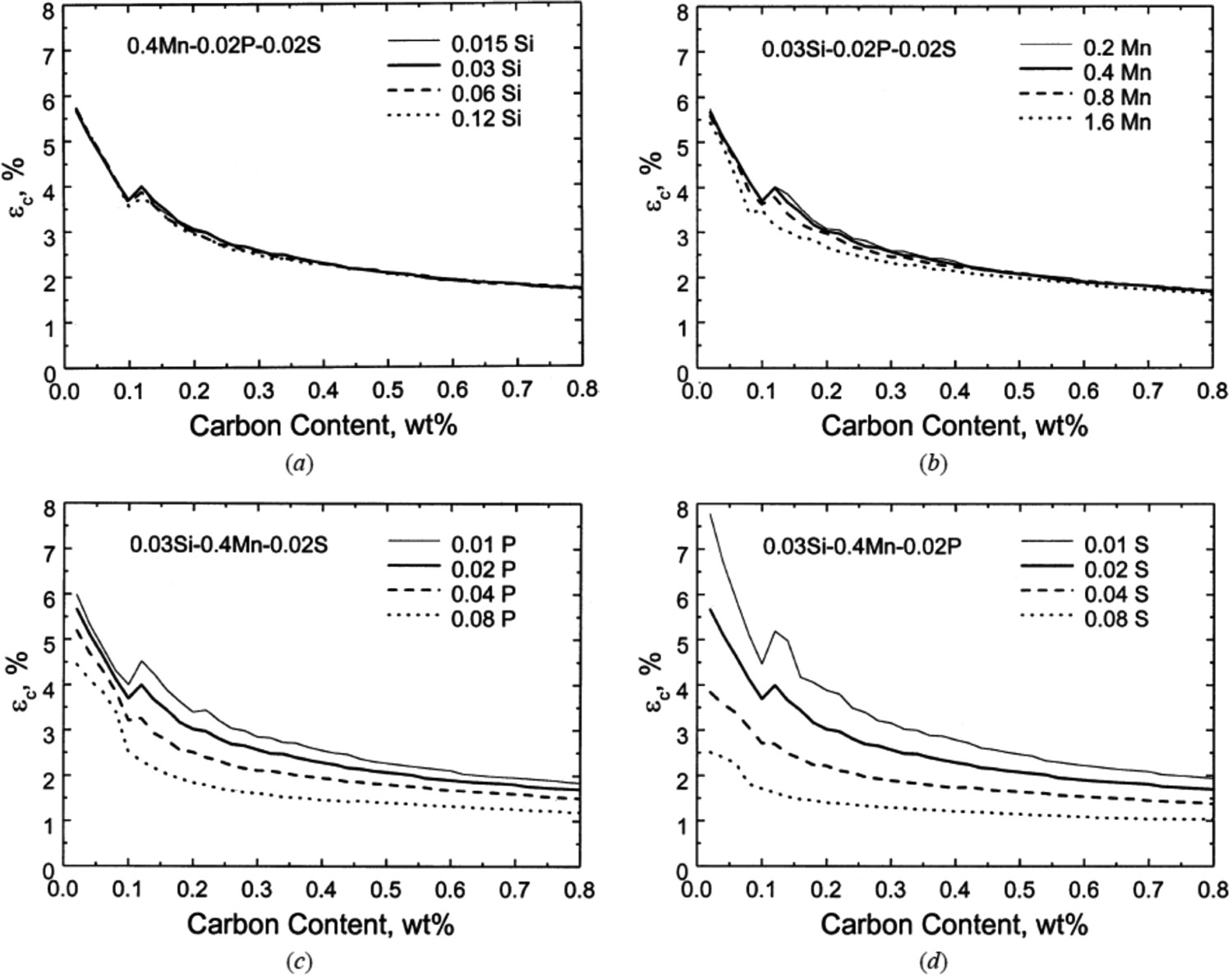
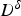
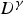
Suzuki et al. [91] have observed high ductility (reduction of area, RA (%) >60%) of 0.7 wt-% C – 0.04 wt-% P steel grade tensile deformed at temperatures (700°C–1200°C) after cooling at different cooling rates, 0.05°C s−1, 0.1°C s−1 and 1°C s−1 (in the study by Suzuki et al. [91], cooling rate is defined for the stage of cooling from the melting temperature of ∼1500°C for 60 s to the tensile deformation temperatures to examine the hot-ductility trough (700°C–1200°C)). Steel fractured at a deformation temperature of 900°C showed that the grain boundaries were enriched with metastable P enriched plate shaped phosphide, (Fe,Mn)3P. These are formed along austenite grain boundaries at T ∼ 950°C by a eutectic reaction which involves the transformation of liquid to austenite phase and (FeMn)3P.
Won et al. [92] have numerically shown the effect of increased cooling rate (in the study by Won et al. [92], cooling rate is defined for the stage of cooling from liquidus temperature to TNZ) on TNZ and TNF for different carbon steels. With an increase in cooling rate (0.1°C s−1 to 100°C s−1), TNZ and TNF decrease due to severe inter-dendritic segregation of solute elements (S, P and Si). As mentioned previously, the higher cooling rate promotes higher segregation and minimises the time for back-diffusion to occur [74,75]. Therefore, lower cooling rate (for the solidification stage of continuous casting process of steel) is effective to reduce inter-dendritic segregation of residual elements and minimise the chances of hot-tearing [91–94].
Previous researchers [95–97] observed negligible direct effect of C on the hot-tearing mechanism. Microsegregation of C is negligible due to rapid back-diffusion during the solidification of steel [77,95–97]. Tendency of P microsegregation is higher in austenite phase than delta ferrite phase because of the lower partition coefficient and back-diffusivity in austenite phase than in delta ferrite phase [95] (Tables 3 and 4). Higher carbon concentration favours the formation of austenite phase [98] and therefore promotes P microsegregation during solidification [95]. Moreover, S activity in liquid steel increases with an increase in C concentration [89]. Increase in S activity favours microsegregation of S and increases the chances of solidification cracking during casting [89]. Therefore, C could play an indirect role on solidification cracking during steel casting. Hot-tearing problems in steels due to microsegregation of S could be minimised by addition of Mn (Mn/S ratio > 20), as Mn combines with S to form MnS particles [84].
Zhang et al. [79] observed an insignificant effect of Sn content (0 wt-% to 0.20 wt-%) on the temperature of zero ductility (TNZ ∼ 1300°C) and temperature of zero force (TNF ∼ 1420°C) in boiler and pressure vessel steels. Li et al. [99] also observed similar insignificant effect of Sn content on TNZ and TNF in Sn bearing ferritic stainless steel. Scheller et al. [86] studied separately the effect of Cu, Sn and Pb on the low-ductility zone, T > 1200°C (Figure 5) in AISI 317 and AISI 304 steel grades. Contrary to previous work by Zhang et al. [79] and Li et al. [99], the addition of Sn lowers the characteristic (liquids, solidus, TNZ and TNF) temperatures of AISI 317 and AISI 304 steel grades. With an increase in Sn content from 0.006 wt-% to 0.15 wt-%, ΔTO increases from 34°C to 67°C and then decreases to ∼58°C with a further increase in Sn content to 0.3 wt-% in AISI 317 steel grades. In the case of AISI 304 steel grades, with an increase in Sn content from 0.009 wt-% to 0.22 wt-%, ΔTO increases from ∼8°C to 16°C and then remains constant with a further increase in Sn content to 0.41 wt-% [86]. The increase in Cu content from 0.07 wt-% to 2.34 wt-% leads to a small increase in ΔTO from 34°C to 48°C in AISI 317 steel grades. In the case of AISI 304 steel grades, with an increase in Cu content from ∼0.2 wt-% to ∼1 wt-%, ΔTO increases from ∼8°C to ∼15°C and then remains constant with a further increase in Cu content to ∼2.4 wt-% [86]. With the increase in Pb content from ∼0 ppm to ∼10 ppm, ΔTO increases from ∼5°C to ∼35°C and then decreases to ∼20°C with a further increase in Pb content to ∼14 ppm in AISI 304 steel grades [86]. A small increase in the width of ΔTO occurs with the increase in Sn, Cu and Pb contents, which increases the susceptibility to hot-tearing during solidification and decreases the hot-ductility of the solidified microstructure under the application of external stress. The change in chemical compositions of Cu, Sn and Pb have resulted in the different variations in ΔTO but there has been no discussion in the literature [86] for these differences in observations based on different chemical compositions of Cu, Sn and Pb. There has been no discussion in the literature [86] of the synergetic effect (Cu, Sn and Pb) on ΔTO.
Cracking problems in cast steel in temperature zone lower than 1200°C
The second zone of low-ductility extends from the TAr3 temperature to 1200°C (Figure 5). The failure mechanisms reported for this low ductility zone includes precipitation pinning along the austenite grain boundaries [100,101] or grain boundary sliding or delayed dynamic recrystallization (DRX) [102,103]. The third zone of low-ductility occurs below TAr3 temperature. The failure mechanisms reported for this low ductility region include local strain concentration along the interface of the ferrite and austenite matrix or pearlite colonies [101]. The third zone of low-ductility could further extend below TAr1 (finish temperature of austenite to ferrite phase transformation). Local strain concentration along the interface of pearlite colonies and recrystallized ferrite matrix [104] or partially recrystallized ferrite matrix [16,17] or precipitation pinning along ferrite grain boundary [17] have been suggested to lead to failure during elongation for T < TAr1.
Role of residual elements in casting process in temperature zone lower than 1200°C
Continuously cast slabs undergo a complex combination of thermal and mechanical (bending and/or straightening) stresses which result in strain and cause surface cracking during the casting process [84,105,106]. The amount and distribution of residual elements such as S, Mn, Nb, Ti, V, Cu, Sn [3] have a significant role on the high temperature (600°C to 1200°C) ductility properties of slabs during continuous casting process [3,56,84].
Researchers have performed high temperature tensile [107,108], bending [109] and compression [57] tests to study surface cracking mechanisms during the bending and straightening operations in the continuous casting process. High temperature (∼1000°C) tensile deformation (>0.1 s−1) of a re-melted and cast steel grade containing a high amount of S (0.012 wt-%) exhibits significant austenite grain boundary embrittlement [101]. With an increase in S content (0.004 wt-% to 0.016 wt-%) in low-carbon steel, the ductility trough widens [101,110,111] and deepens [56,101,111,112] as shown in Figure 7 [113]. Steel grades [113] exhibiting high ductility loss (RA % <20%) have their prior austenite grain boundaries decorated with fine (<50 nm) sulphide particles (Fe-rich (Fe,Mn)S particles) with an average inter-particle spacing of ∼200 nm. Along with grain boundary particles, fine (average size ∼30 nm) sulphide particles are also present inside the prior austenite grains outside the precipitate-free-zone (PFZ) [114] of 1–1.5 µm wide along the grain boundary [113]. Variation of reduction of area (RA %) to failure at different tensile deformation temperatures of various low-carbon steel grades (A, B, C, D, E and F) solution treated at 1623 K and strain rate 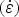
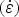
On application of external stress, submicrometre voids are formed because of decohesion of particle-matrix interfaces on the grain boundary (Figure 8). Submicrometre voids further coalesce with each other to form microvoids (Figure 8(b)). Final inter-granular ductile fracture of the sample occurs by coalescence of microvoids (Figure 8(c)) [107,113]. During high temperature deformation, distribution of fine (average size ∼30 nm) sulphide particles within grains lead to further enhancement of strain localisation within PFZs formed along grain boundaries (Figure 8(b)) [113]. In addition, Fe-rich (Fe,Mn)S particles could also deteriorate ductility at high temperatures (T > 900°C) due to their lower melting point (T ∼ 980°C) as compared to stable MnS particles (melting point T > 1300°C) [56,112,115]. Schematic demonstration of inter-granular microvoid coalescence of Nb-bearing steel during deformation at low temperature (< ∼1200°C) within the austenite phase regions (a–c) and ferrite-austenite duplex region(d–f) (taken from Maehara and Ohmori [107]).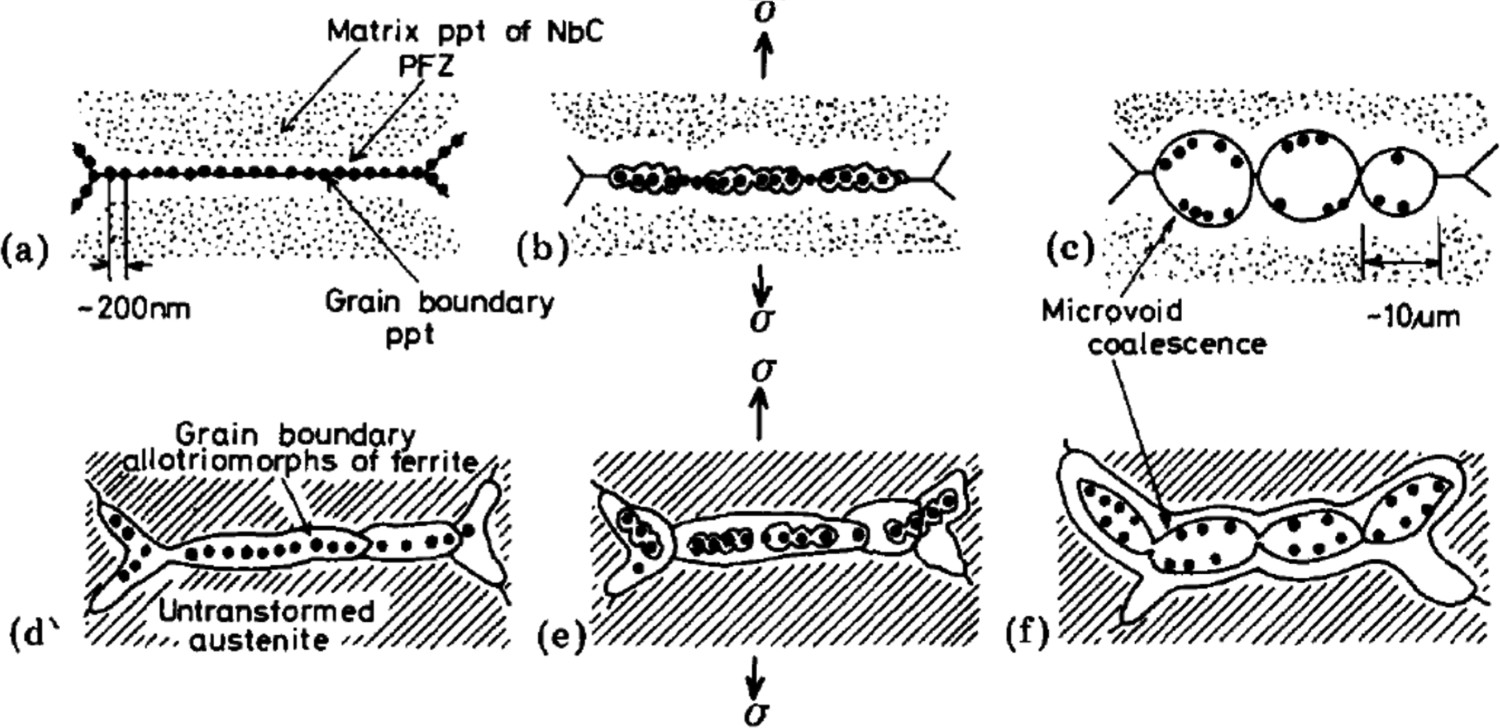
The inter-granular ductile fracture mechanism observed for sulphide particles is almost the same mechanism explained by Maehara and Ohmori [107] (Figure 8) for carbide and/or nitride particles of Nb and Al, although the strain rate required for high temperature embrittlement because of sulphide particles is larger (∼104 times) than in the case of carbide and nitride particles (Figure 9). The role of Nb carbide and nitride particles on high temperature inter-granular fracture [107] will be discussed later in this paper. S atoms also have a strong tendency to segregate along the grain boundary, which attracts an electronic charge from the iron atoms. This process reduces the amount of electronic charge available to participate in Fe–Fe bonds which holds the grain boundary together and leads to inter-granular decohesion fracture [113,116]. Addition of Mn is beneficial to steel as it combines with S to form MnS particles and prevents the formation of lower melting point (T ∼ 980°C) [56,112,115] Fe-rich (Fe,Mn)S particles and also reduces the chance of S segregation along grain boundaries [84]. Schematic representation of strain rate regions inducing hot-embrittlement in steels due to dynamic precipitation of carbides and/or nitrides (A) and sulphides (B) (taken from Maehara et al. [105]).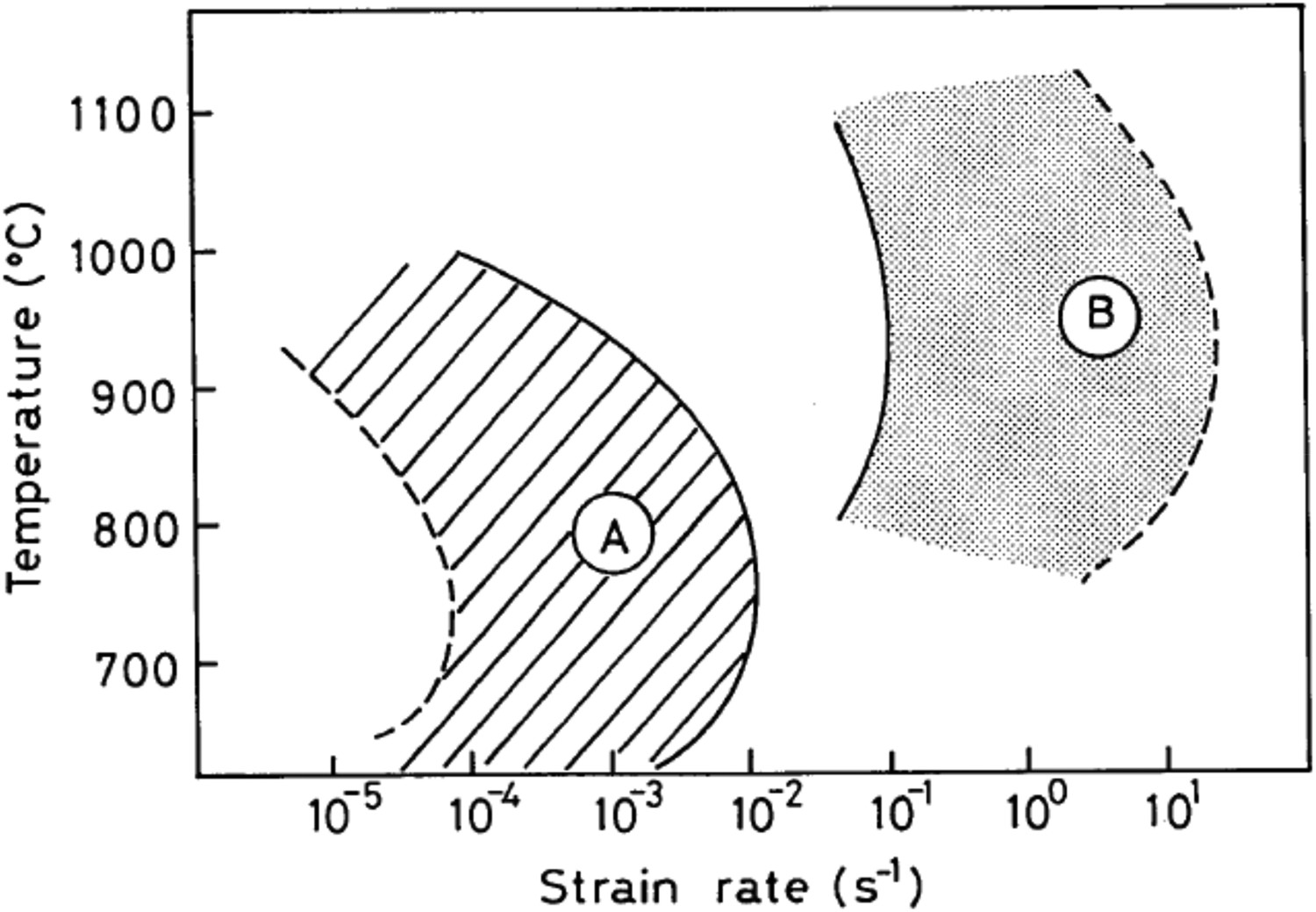
Nb, Ti and V are commonly regarded as microalloying elements in High-Strength Low-Alloy (HSLA) steel [16–18] but when present in higher levels these elements could be regarded as harmful and undesirable. Nb is beneficial to steel as it provides additional strength by precipitation strengthening and is also used for recrystallization control during hot-rolling to achieve a fine grain size microstructure [117]. However, higher levels (0 wt-% to 0.05 wt-% [102]) of Nb in low-carbon steel can have detrimental effects in deepening and widening the hot-ductility trough [83,100,102,118–123]. Combination of low-strain rate (10−2–10−5 s−1 [56,100,124,125]) deformation and presence of fine particles of Nb(C,N) along austenite grain boundaries are responsible for surface crack formation in continuous cast (during straightening operations [102,107]) and ingot cast (due to strain generation from thermal stresses [106]) steel.
Fine (<50 nm) NbC particles pin austenite grain boundaries and retard DRX of austenite grains [107,117]. Prevention of DRX of austenite grains would prevent the formation of finer austenite grains and result in low hot-ductility at high temperatures (1000°C to 1200°C) [105,106,125]. DRX of austenite grains and austenite grain boundary movement prevent the inter-linking of voids and results in high ductility [126]. It is important, however, to note that the majority of the surface cracks in CC slabs occur at fracture strains less than the critical strain required for DRX of austenite grains [105,106].
During low-strain rate (∼4×10−3 s−1) deformation at relatively high temperature (800°C to 900°C, i.e. within austenite phase region) of HSLA steels, initiation of inter-granular fracture occurs by grain boundary sliding mechanism [106]. With an increase in Nb content (0 wt-% to 0.074 wt-% [106]), the number density of fine (20–50 nm) Nb(C/N) particles increases along the austenite grain boundaries. During deformation, decohesion of the precipitate-matrix interface will occur along the grain boundary and form voids. As deformation progresses, voids coalesce with each other and lead to inter-granular fracture (Figure 8(a, b and c)) [107]. Although grain boundary sliding is necessary to initiate inter-granular fracture, the amount of Nb plays a significant role in controlling high temperature ductility [106,127].
With an increase in Nb content (0 wt-% to 0.074 wt-% [106]), the temperature of minimum ductility shifts from the lower temperature range (700°C–750°C, i.e. within the ferrite-austenite phase region) to the higher temperature range (800°C–900°C, i.e. within austenite phase region) [106]. The ductility in the lower temperature range is significantly affected by the thickness of the ferrite film that forms along the austenite grain boundary. The RA (%) increases (from 40% to 70%) with an increase in ferrite thickness (from 10 to 43 µm) [106]. During tensile deformation, the plastic deformation occurs preferentially in the ferrite region and local strain generation at the interface of austenite and ferrite phases leads to final failure of the sample [126–128]. The presence of Nb(C,N) precipitates could further promote void formation and link up [106] as shown schematically in Figure 8 [107]. With a further decrease in temperature to 700°C and below, the volume fraction of ferrite phase increases and ductility is improved. From the above discussions on inter-granular fracture, grain boundary sliding is the major embrittlement factor in austenite phase region of Nb containing steels and austenite steels. However, the presence of film like ferrite film along austenite grain boundaries is the major embrittlement factor in the mixed ferrite-austenite phase region of Nb containing steels and plain carbon steels [127].
Similar to Nb(C/N) particles, V combines with N to form VN particles [122,123,129] in the austenite phase which can have a detrimental effect on the hot-ductility property of steel [84,127]. The embrittling effect of V is lower than that for Nb [130] because of its higher solubility in the austenite phase of steel [131].
Addition of a small amount of Ti (0.018 wt-% [106]) is beneficial to reduce transverse cracking in Nb containing steel [105,132]. In the presence of Ti, most of the N would combine with Ti to form coarse TiN precipitates (∼50 nm) [106,123] in the inter-dendritic regions, which subsequently become the austenite grain boundaries. Content of Ti should be well controlled to prevent the formation of micrometre size coarse TiN particles which are prone to initiate cleavage fracture [133]. The TiN precipitates (∼50 nm) can then act as nucleation sites for NbC precipitates at temperatures (∼<1200°C) which limits the formation of fine (20–50 nm) Nb(C/N) precipitates on grain boundaries [83,106,107]. Abushosha et al. [134] observed that the addition of Ti (∼0.02 wt-%) in C–Mn–Al–Nb steel improved hot-ductility at higher temperatures (700°C–1100°C) at a slow cooling rate of ∼0.42°C s−1 (in the study by Abushosha et al. [134], cooling rate is defined for the stage of cooling from the melting temperature of ∼1540°C for 300 s to the tensile deformation temperatures to examine the hot-ductility trough (700°C–1100°C)). This slow cooling rate allows NbC precipitation on coarse TiN (∼50 nm) precipitates [134]. Abushosha et al. [135] determined an empirical relation (Equation (2)) for RA (%) of Ti containing C–Mn–Al steel for the temperature range 700°C–1100°C. For improved ductility, slow cooling rate, coarse particle size and low N levels (for lower volume fraction of TiN precipitates) are recommended.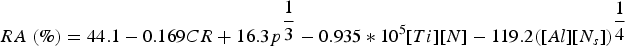
At higher temperature (>1200°C), P is well known to segregate into the inter-dendritic region and causes hot-tearing, discussed earlier. However, for the lower temperature region (<1200°C), presence of P (max. 0.02 wt-% [136]) is beneficial to reduce grain boundary embrittlement in Nb containing steel. P atoms segregate to the austenite grain boundaries [55] and therefore retard the precipitation kinetics of Nb(C/N) in austenite [84,100,110,123,137].
Solid state segregation [138] of residual elements such as Cu and Sn [139–141] to the grain boundaries increases the tendency of transverse cracking in continuous cast steel in the low temperature region (<1200°C) [142]. Before discussing the effect of Cu and Sn on the lower temperature (<1200°C) behaviour, different types of solid state segregation is introduced in brief. Segregation can be divided into equilibrium [143–145] and non-equilibrium [81,146–149] segregation. Equilibrium segregation is thermodynamic in nature where the magnitude of segregation decreases with an increase in temperature. While, non-equilibrium segregation is a kinetic process and relies on the formation of vacancy-impurity complexes during cooling [81,146]. During the cooling process from high temperature, the equilibrium concentration of vacancies and vacancy-impurity complexes are reduced and result in the formation of a vacancy concentration gradient within grains. Owing to the vacancy concentration gradient, there is a net flow of vacancies and vacancy-impurity complexes towards vacancy sinks such as grain boundaries and surfaces. Generally, diffusion of complexes is faster than diffusion of impurities in the matrix [150]. Flow of vacancy-impurity complexes lead to deposition of impurity atoms at the grain boundary (i.e. segregation of residual elements to the grain boundary) [146,151,152].
In theory, there is a critical cooling rate at which non-equilibrium segregation attains its maximum value for a particular solute element during the cooling process [149,153]. Song et al. [81] estimated the critical cooling rate to be between 5°C s−1 to 20°C s−1 for non-equilibrium segregation of Sn to the austenite grain boundaries where the cooling rate is defined for the stage of cooling from the solution treatment temperatures used in the experiments of ∼1320°C for 180 s to the tensile deformation temperatures to examine the low temperature ductility trough (700°C–900°C). For a cooling rate of ∼10°C s−1, Sn segregation to the grain boundaries is a maximum (Figure 10(a)) and results in low hot-ductility for the temperature range of 750°C–800°C (Figure 10(b)). Therefore, it is preferred to avoid the critical cooling rate condition over the temperature range of straightening operations for the continuous-casting process (especially for thin-slab casting where the surface of thin-slab experiences fast cooling rate conditions [118]) [81]. Moreover, using a high level of steel scrap with high residual Sn content (>0.15 wt-% [118]) would not be advised for thin-slab casting. Sn boundary concentration (at.-%) examined under Auger electron spectroscopy (AES) (a) and reduction of area (RA %) for different tensile deformation temperatures (b) of 0.061 wt-% Sn bearing steel specimens treated for different cooling rates (5°C s−1 to 20°C s−1) (taken from Song et al. [81]).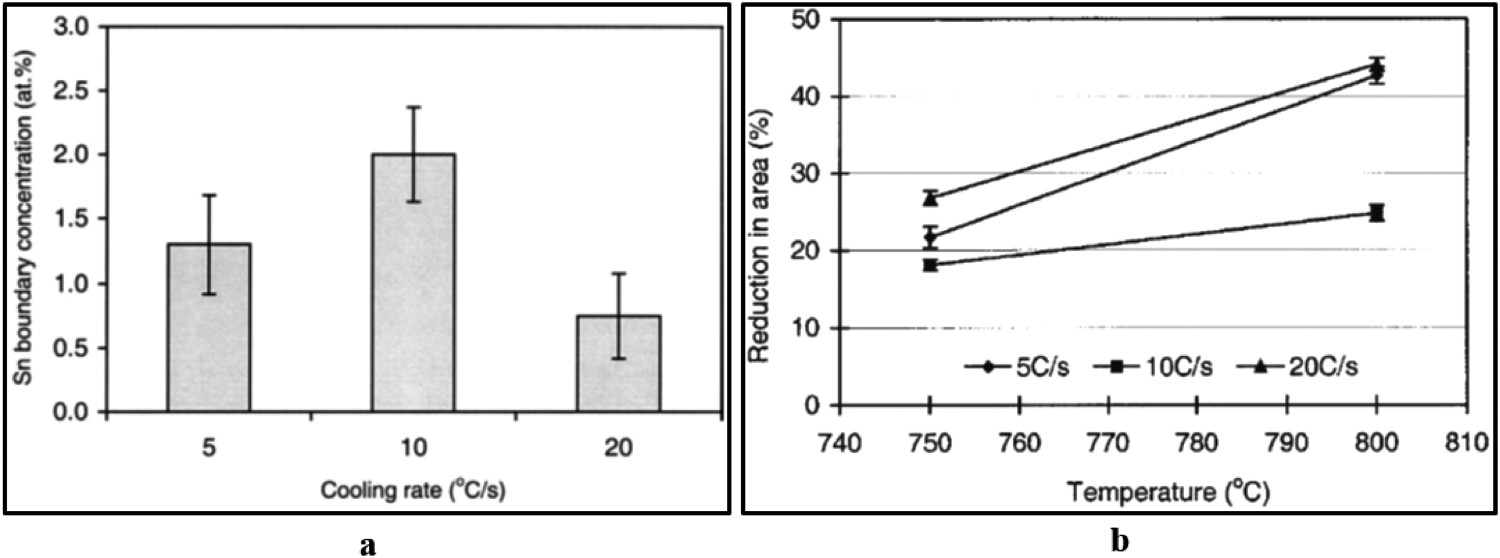
Nagasaki and Kihara [139] and Matsuoka et al. [141] studied the effect of Cu (<1 wt-% [139] and <0.4 wt-% [141]) and Sn (<0.2 wt-% [139] and <0.05 wt-% [141]) on hot-ductility of low-carbon steels. Segregation of Sn weakened the austenite grain boundaries and ferrite-austenite interfaces and the hot-ductility was deteriorated. Nagasaki and Kihara [139] did not observe Cu segregation to the grain boundaries. This is due to higher solubility and lower diffusivity of Cu (solubility ∼9 at.-% at 1094°C [154] and diffusivity ∼2.5×10−14 m2 s−1 at 1315°C [155]) than Sn (solubility ∼0.71 at.-% at 1100°C [156] and diffusivity ∼1.6×10−13 m2 s−1 at 1307°C [157]) in austenite. In the study by Matsuoka et al. [141], cracks are developed along the austenite grain boundaries and proeutectoid ferrite formed along the austenite grain boundaries during high temperature (∼900°C) deformation (strain rate ∼0.001 s−1) of Cu–Sn bearing steel. While in Nagasaki and Kihara’s study [139], no cracks are observed along the proeutectoid ferrite formed on the austenite grain boundaries in low-carbon Cu-Sn bearing steel. This implies that the initiation and growth of cracks are not due to local strain concentration along proeutectoid ferrite formed on the austenite grain boundaries. The mechanism of inter-granular fracture is based on diffusion of vacancies as explained in the work of Sricharoenchai et al. [158]. The embrittlement at a strain rate (∼0.01 s−1) for the temperature range (757°C–807°C) is due to dislocation pile up at the grain boundary and inhibition of boundary migration by Sn [139,158]. As long as the grain boundary is immobile, microvoids are nucleated by diffusion of vacancies to the grain boundary to annihilate the dislocations. Microvoid growth and coalescence leads to formation of cracks along the grain boundary [139,158].
Cu is also known to reduce the kinetics of nucleation and growth of ferrite phase [159,160]. The reasons [159–161] for the decrease in nucleation and growth kinetics are (1) segregation of Cu to the austenite grain boundaries decreases the boundary energy, (2) formation of solute atom clusters of Cu or fine coherent precipitation of Cu at the austenite grain boundaries [161] and (3) solute drag effect by Cu atoms. A delay in the phase transformation of austenite to ferrite would deteriorate the hot-ductility property during continuous casting process [106].
Mintz et al. [137] studied the hot-ductility (deformation temperature from 750°C to 1000°C) of C–Mn–Al steels containing Cu (<0.5 wt-%). At higher temperatures (>750°C) and in the presence of an oxidising environment, Cu in higher concentrations (∼0.5 wt-%) combines with S and O to form copper-oxy-sulphides/copper-sulphide particles along austenite grain boundaries. In presence of O, MnS could oxidise to MnO releasing S to segregate to the grain boundaries (Equation (3)). Because of the oxidation of MnS, the local concentration of S and O increases at the boundary and favour formation of copper-oxy-sulphide/copper-sulphide particles. During hot-deformation, copper-oxy-sulphide/copper-sulphide particles favour inter-granular failure by promoting void formation and the linkage of voids for crack growth [137,162]. With the increase in Ni content, solubility of Cu in austenite phase increases [118] and this avoids the formation of copper-oxy-sulphide/copper-sulphide particles and improves hot-ductility [137].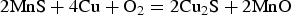
Copper is also well known to cause surface hot-shortness during processing of steel. Cu is more noble than iron and is not oxidised when steel is heated. Therefore, during oxidation, iron is removed preferentially from the surface layers and residual elements build up in the sub-scale layer. Cu as a residual element alone among the others forms a liquid phase when its solubility is exceeded in austenite phase. Under severe oxidation environments (temperatures of 1100°C–1200°C), Cu build up can reach the saturation limit for the steel grade [12]. When the solubility limit of copper is exceeded in the steel microstructure, liquid copper forms at the interface of the iron-oxide scale and steel. Solubility of Cu in austenite is ∼10 wt-% at a temperature of ∼1193°C and ∼0.1 wt-% at a temperature of ∼521°C [3]. This Cu rich liquid phase penetrates along austenite grain boundaries and leads to surface cracks during processing of steel [19]. Cu surface hot-shortness is a bigger issue during hot-rolling than conventional continuous casting process. In hot-rolling process, slabs are pre-heated in a furnace for a long time (1–2 hr) before the first pass of hot-rolling. A prolonged oxidation period (1–2 hr) causes significant liquid Cu enrichment below the iron-oxide scale at the steel surface which further penetrates along the austenite grain boundaries during hot-rolling. Cracking due to Cu hot-shortness is widely observed and reported for hot-working, more than in the conventional continuous casting literature [163,164]. One of the reasons could be that more severe oxidation occurs during reheating immediately prior to hot-working leading to significant liquid Cu enrichment whereas during the conventional continuous casting process there is more limited time at high temperature reducing the amount of oxidation and hence Cu enrichment at the steel surface. Anelli et al. [165] have shown the liquid phase of Cu–Ni–Sn penetrating along austenite grain boundaries in EAF continuous cast blooms (bulk Cu concentration in the range of 0.10 wt-% to 0.17 wt-%). Nb containing plate steels with additions of 0.25 wt-% of Cu and 0.25 wt-% of Ni have shown higher tendency of transverse cracking as compared to steels without Cu and Ni additions [118].
Discussion
The list of residual elements present in steel scrap is huge [3]. Within this article current metallurgical understanding about the behaviour of various residual elements (such as P, S, Si, Mn, Cu, Sn, Pb, Nb, Ti, V), individually and synergistically at high (>1200°C) and low (<1200°C) temperatures during the casting process, with a focus on local enrichment and cracking caused by the residual elements, is reviewed. There are certain residual elements (such as Zn, Bi, As, Ni, Sb, Cr, etc.) [3] whose effect on casting process of steel have not been reported in detail and therefore is not extensively discussed in this review article. However, significant work is done and reported on the effect of these residual elements (such as Zn, Bi, As, Ni, Sb, Cr, etc.) on further downstream process of steel production (such as hot-working, cold-working and end user properties) [3,41,166]. In the literature [87,89–92], there have been extensive discussion on the hot-tearing problems caused by the inter-dendritic segregation of P, S, Si and Mn during solidification. The extent of inter-dendritic segregation of Cu, Sn and Pb during solidification has not been reported in detail and therefore is not extensively discussed in this review article.
Previous researches [74–76,91–94] have studied the significance of low cooling rates during the solidification stage of casting to minimise the inter-dendritic segregation of solute elements, which also decreases the susceptibility to hot-tearing problems. Cooling rate during solidification of conventional continuous casting of ∼250 mm thick slab casting (∼1°C s−1) is lower than that of ∼50 mm thin-slab casting (∼3.3°C s−1) over the temperature range of 900°C–1350°C [118]. The lower cooling rate conditions might give more time for inter-dendritically segregated solute elements to back-diffuse and prevent hot-tearing problems in conventional continuous thick slab casts.
Steel produced via the TSC process is not recommended with more than 0.15 wt-% Cu and 0.015 wt-% Sn (from scrap use), to avoid surface crack formation. While, in the case of steel strip produced via TRC process a maximum of 0.55 wt-% Cu and 0.16 wt-% Sn has been recommended, therefore allowing low quality scrap to be used [167,168]. Strip casting technologies are relatively new and not widely used for mass steel production for industrial applications as compared to CCC and TSC [167] therefore there is less information available on issues related to residual presence.
Brief summary of problems, countermeasures and advantages and existence of different residual elements studied in literature during casting process.
Conclusions
The present article reviews the effect of a few residual elements (such as P, S, Si, Mn, Cu, Sn, Pb, Nb, Ti, V) on cast steel at different thermal (from liquidus to ∼600°C) and mechanical (bending or straightening) stages during continuous casting and ingot casting process. The effect of different residual elements during casting is a broad topic and involves the understanding of both individual and combined behaviour of residual elements for different thermal and mechanical conditions that arise during the casting process. For the higher temperature region (>1200°C) during and after solidification, inter-dendritic segregation of P, S, Si and Mn have a significant effect and can cause a hot-tearing problem. While for the lower temperature region (<1200°C), inter-granular fracture due to embrittling precipitates (fine (Fe,Mn)S or Nb(C/N) or copper-oxy-sulphide/copper-sulphide particles), surface hot-shortness due to Cu and solid-state segregation of Sn can cause hot-cracking problems. By optimising the cooling rate, deformation temperature (for strand straightening operation during continuous casting process), strain rate and chemical composition, the above mentioned hot-ductility problems can be minimised.
Footnotes
Acknowledgements
Authors would like to thank the academic and industrial partners of the SUSTAIN Manufacturing Hub for their valuable discussions during this study. Authors would like to thank the financial assistance from EPSRC grant EP/S018107/1.
Disclosure statement
No potential conflict of interest was reported by the author(s).