Abstract
In hot metal desulphurisation (HMD) the slag will hold the removed sulphur. However, the iron that is lost when the slag is skimmed off, accounts for the highest costs of the HMD process. These iron losses are lower when the slag has a lower viscosity, which can be achieved by changing the slag composition. A lower slag basicity decreases the viscosity of the slag, but also lowers its sulphur removal capacity, therefore optimisation is necessary. In this study, the optimal HMD slag composition is investigated, considering both the sulphur removal capacity and the iron losses. In part I the theory is discussed and in part II the optimal slag is validated with plant data, laboratory experiments and a thermodynamic analysis.
Introduction
Since the early days of iron- and steelmaking, sulphur is considered as an unwanted impurity that needs to be removed [1]. Although there are various processes in the modern steelmaking chain where sulphur can be removed, a dedicated hot metal desulphurisation (HMD) process between the blast furnace (BF) and converter (or basic oxygen furnace, BOF) remains necessary. The HMD process benefits from the low oxygen activity of the hot metal at that stage of the process chain (before the BOF). Essentially during the HMD process the dissolved sulphur reacts with reagents (typically magnesium and/or lime) to form sulphides that end up in the slag phase. When the slag is removed after reagent injection, the hot metal is desulphurised [2,3].
As the sulphur is only removed by skimming off the sulphur-containing slag, it is essential for the HMD process that the slag contains all formed sulphides. The mass of sulphur that can be removed with a certain slag, is defined as the ‘sulphur removal capacity’ of the slag. This sulphur removal capacity is different from the thermodynamically defined ‘sulphide capacity’ (CS ), which was introduced by Fincham and Richardson [4]. In the present work the sulphur removal capacity of the slag is used as the criterion for optimising slag regarding sulphur removal.
The largest costs during the HMD process are the iron losses, iron that is skimmed off together with the slag. Depending on the heat size, typically 500–4000 kg iron (0.5–2.5 wt-% of the total iron) is skimmed off per heat [5,6]. By changing the apparent viscosity of the slag (ηslag ), the iron losses can be lowered [6–11]. This means that iron losses partly depend on the slag composition.
The aim of this study is to find the optimal slag for the HMD process, which is defined as a slag with an optimal balance between maximising sulphur removal capacity and minimising iron losses. Because the slag composition changes during the process, as reagents are added, the sulphur removal capacity should be sufficient throughout the process. The slag composition that minimises the iron losses should be reached at the end of the process, so the focus here is on the final slag composition. In order to be acceptable for industry, this optimal slag should not lead to health, safety and environmental issues and should not lead to a large increase in costs. In the present paper, part I of this study, the theory behind the sulphur removal capacity, as well as a theoretical study of HMD iron losses, are presented. This part ends with conclusions about the optimal HMD slag, based on theory. In part II of this study [12] the theory is examined and validated with a Monte Carlo simulation using FactSage [13], plant data analysis and laboratory viscosity and melting point experiments with the optimal slag.
Sulphur removal capacity
Desulphurisation process
In Europe and North America, the magnesium-lime co-injection process is the state-of-the-art HMD process. In this process magnesium and lime are injected into the hot metal. Most of the desulphurisation (> 95%) takes place by the reaction between magnesium and sulphur in the bath (reaction 1). The formed MgS ascends to the slag layer and reacts with lime to form CaS (reaction 2). Only a small portion of the dissolved sulphur directly reacts with lime via reaction 3 [2,14,15].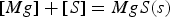
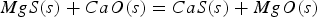
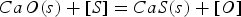
In these reactions [x] means that element x is dissolved in hot metal and (s) indicates solid. Most of the formed sulphides and oxides eventually dissolve in the slag, although a substantial part of the CaS remains in a solid fraction [14]. All solid phases have a lower density than the hot metal and end up in the slag. Since both magnesium and sulphur are dissolved in the metal, reaction 1 is a homogeneous reaction, which is fast. Lime remains a solid throughout the process, making reaction 3 a heterogeneous reaction, which is slower. Besides, reaction 3 is further slowed down by surface passivation of the lime particles. The desulphurisation rate in the co-injection process is therefore controlled by reaction 1 [2,3,14,15]. This leads, via reaction 2, to a heterogeneous slag, which is not necessarily in equilibrium with the hot metal with respect to sulphur distribution. Other sulphur removing processes in steelmaking, including the blast furnace (BF), the Kanbara reactor (KR) HMD process and several secondary metallurgy (post BOF) processes, which include the ladle furnace and the vacuum degasser, are dominated by reaction 3. The slag and metal bath are generally in equilibrium regarding the sulphur distribution. Metallic magnesium is not introduced for desulphurisation of the metal in any of the above-mentioned processes [2,3,15–18].
Typical slag compositions for BF carryover slag and HMD slag after reagent injection [6].
Composition and temperature range for HMD slags after reagent injection.
In the slag composition the amount of FeOx (FeO and Fe2O3) has been excluded, as it is difficult to measure the amount of FeOx in the slag. With XRF (X-ray fluorescence) analysis, which is a typical method for slag analysis, all components are oxidised, so no distinction between FeOx dissolved in the slag, and metallic Fe, captured in the slag, can be made. FeOx does have a significant effect on the viscosity and melting point of the slag [17,19]. Figure 1 illustrates the effect of adding FeO to a slag with a balanced composition of 40 wt-% Cao, 30 wt-% SiO2, 15 wt-% Al2O3 and 10 wt-% MgO on the slag’s liquidus temperature (Tliq
) at thermodynamic equilibrium (determined with FactSage 7.3 [20]). The FeOx concentration in BF carryover slag is typically estimated around 1 wt-%, but can be up to 3 wt-% [15,21,22]. Effect of FeO concentration on Tliq
in a slag with a balanced composition of 40 wt-% CaO, 30 wt-% SiO2, 15 wt-% Al2O3 and 10 wt-% MgO. Determined with FactSage 7.3.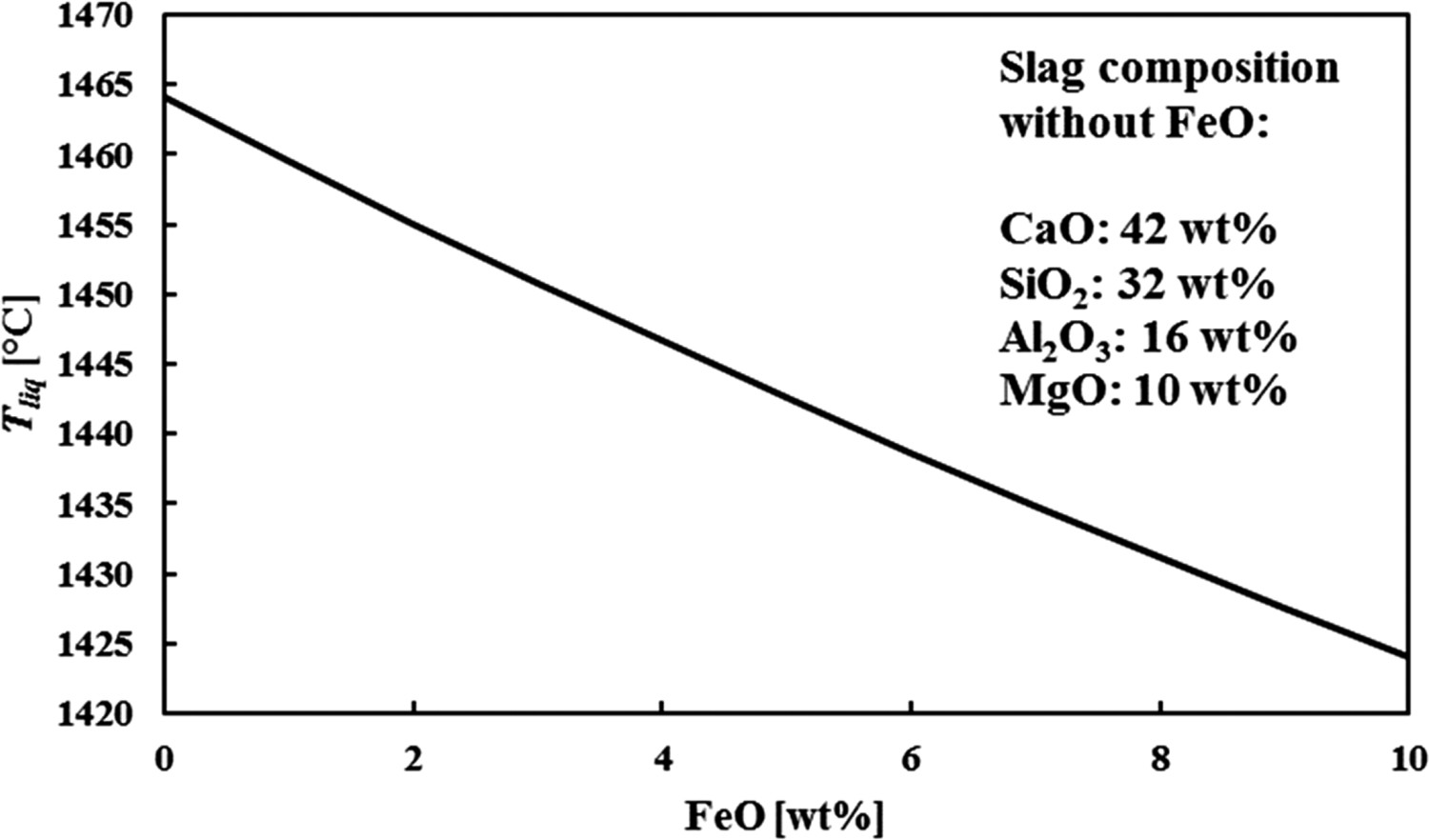
Sulphide capacity
When the oxygen partial pressure (pO2
) < 1 Pa (10−5 atm), the only way for a sulphur atom to enter the slag is to replace an oxygen atom in an oxide (usually CaO). Under these conditions sulphur is only present in the slag as a sulphide. When pO2
> 100 Pa (10−3 atm), sulphur will be present as sulphate in the slag [4]. It is generally accepted that in HMD pO2
is much lower than 10−5 atm (in some literature a pO2
of 10−15 atm. is mentioned [23]), so all sulphur in the slag will be present in the form of sulphides. In this study there is a clear difference between the practical ‘sulphur removal capacity’ and the thermodynamically defined sulphide capacity (CS
), which was introduced by Fincham and Richardson [4]. Here the sulphur removal capacity is defined as the amount of sulphur that can be removed with a certain slag, not necessarily in equilibrium with the hot metal. CS
is defined as ‘the potential capacity of a melt to hold sulphur as a sulphide’ [4,24], which is given in Equation (4). It should be noted that the main difference between the sulphur removal capacity and the sulphide capacity is that the sulphide capacity only takes dissolved sulphides in the liquid slag at equilibrium into account, while the sulphur removal capacity also takes solid sulphides, as well as dissolved sulphides that are not in equilibrium into account. Therefore, the sulphur removal capacity of a slag is a better quantity to judge sulphur removal in operational practice since it is typically higher than its sulphide capacity.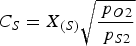
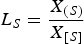
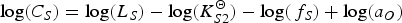
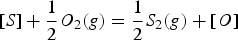
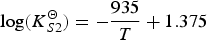
The combination of Equations (6) and (8) gives the impression that for a certain slag composition CS only depends on temperature. Panda et al. [26] showed with FactSage calculations with a private database (CON2) for typical ladle furnace slags, that this is true only at high pO2 values (for ladle furnace slags typically pO2 > 0.1 Pa), or low pS2 values (typically pS2 < 1 Pa). At lower pO2 or higher pS2 values, pO2 and pS2 will influence CS . Jung and Moosavi-Khoonsari [34] stated that the concept of CS , where the amount of sulphides that a slag can contain only depends on its composition and temperature, is only valid if the slag contains a low fraction of sulphides. If the slag contains more sulphides, pO2 and pS2 play a role as well. This means that for processes where relatively low amounts of sulphur need to dissolve in the slag, like desulphurisation in secondary metallurgy, CS is a unique temperature- and composition-dependent property of the slag. In HMD, the slag contains more sulphides (HMD slag can contain up to 15 wt-% CaS), which means that CS under HMD conditions is a function of pO2 and pS2 as well.
In literature, many authors made a model to predict CS
based on the slag composition, often including the optical basicity (Λ), which was defined by Duffy and Ingram [35] as: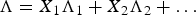
Overview of different models to determine CS , based on optical basicity (Λ).T is in K.
*Hao and Wang used an alternative Λ: Λcorr , which differs for HMD slag 10-13% from Λ.
The two most influential factors for CS
are temperature and, via Λ, CaO content. Figures 2 and 3 show the influence of temperature and CaO, respectively, on the CS
determined by the models from Table 3. A simplified typical HMD slag composition was used (40 wt-% CaO, 35 wt-% SiO2, 9 wt-% MgO, 16 wt-% Al2O3; when changing the CaO concentration the other components were changed in the same ratio). Comparison of CS
determined by the models from Table 3, for different temperatures. Comparison of CS
determined by the models from Table 3, for different CaO concentrations at 1400°C.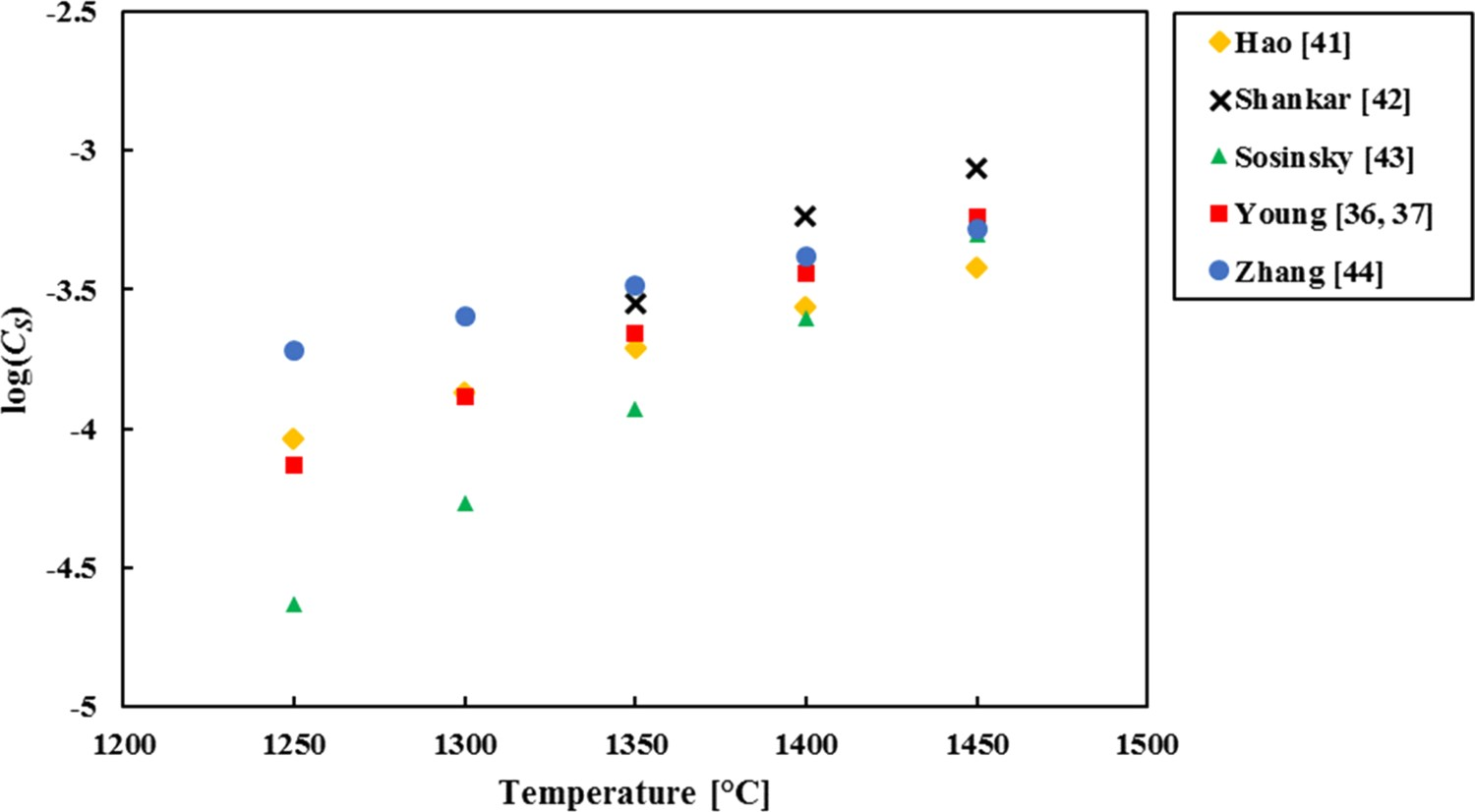
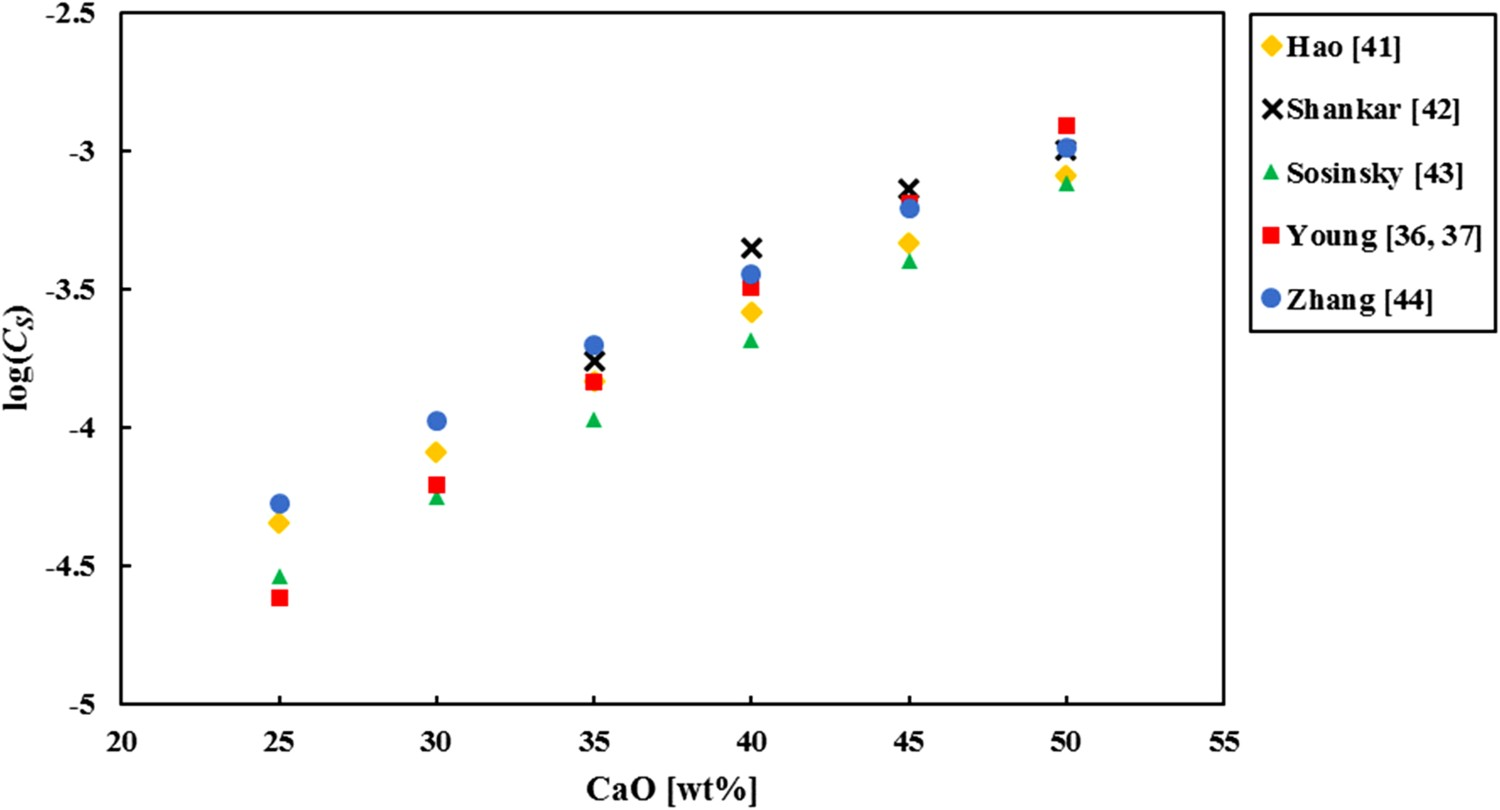
Although the different models give different outcomes for a typical simplified HMD slag, the log(CS) value ranges from −3 to −5, also when the temperature or the CaO content is changed within relevant ranges. Condo et al. [45] measured CS for synthetic typical BF slags, which are comparable to HMD slags in composition, temperature and pO2 , and they also found log(CS) values around −4.
CS in an industrial HMD
To understand the significance of CS
for the HMD process, the apparent CS
is determined for industrial HMD heats. At the industrial HMD process, all sulphur that is removed from the hot metal ends up in the slag. Therefore, when the initial and final sulphur content of the hot metal at the HMD is known and an estimate for the slag weight is made (assuming 1500 kg carryover slag from the BF for a typical heat size of 300 t), LS
can be calculated for every heat. When assuming typical values for fS
= 2.5 and aO
= 5·10−7, CS
can be calculated for every heat with Equation (6). Furthermore, the final composition of the slag can be estimated for every heat by assuming an average BF carryover slag composition and adding the injected reagents to that slag, assuming that all removed sulphur in the slag is CaS. Also, a homogeneous slag is assumed. Figure 4 gives the log(CS) values of 47 129 HMD heats from Tata Steel, IJmuiden, set against the log(CS) values predicted by Young's model (Equation 13) based on the slag composition and temperature. The Figure 4 shows that CS
from Young's model is roughly a factor 10 off from CS
values based on actual industrial desulphurisation results. The actual amount of sulphides in the slag is higher than the CS
prediction with Young's model, meaning that the actual HMD slag contains almost 10 times more sulphur than the slag could contain based on the equilibrium prediction. Also, there is a large scatter for individual heats in the difference between CS
from Young's model and CS
from plant data. It should be noted that the precision of the method to determine CS
for a single industrial heat is not very high, but that cannot explain the scatter entirely. Furthermore, it should be noted that aO
was estimated at 1·10-4, but that literature mentions typical aO
values between 5·10-5 and 4·10-4. However, only at aO
= 2·10-5, Young's model accurately predicts desulphurisation at the industrial HMD (the prediction is still not precise though, as the scatter remains). There is no reference in literature of such low aO
values at HMD, so this cannot be used to tune the results. Also by changing the fs
to 13, Young's model can be made in agreement with the plant data results. Kitamura mentions fs
= 11 for HMD conditions [15], which would bring Young's model more in agreement with the plant data. Finally, also when replacing Young's model for any of the other models listed in Table 4, the results are comparable, meaning that the actual sulphur concentration in the slag is significantly higher than CS
from the model predicts. Therefore, CS
models based on slag composition (translated to Λ) and temperature only, do not give an accurate or precise prediction of desulphurisation at an industrial HMD. Density plot of log(CS) values for 47,129 heats at the Tata Steel IJmuiden HMD stations, where the predicted values from Young’s model are on the X-axis and the actual values based on removed sulphur are on the Y-axis.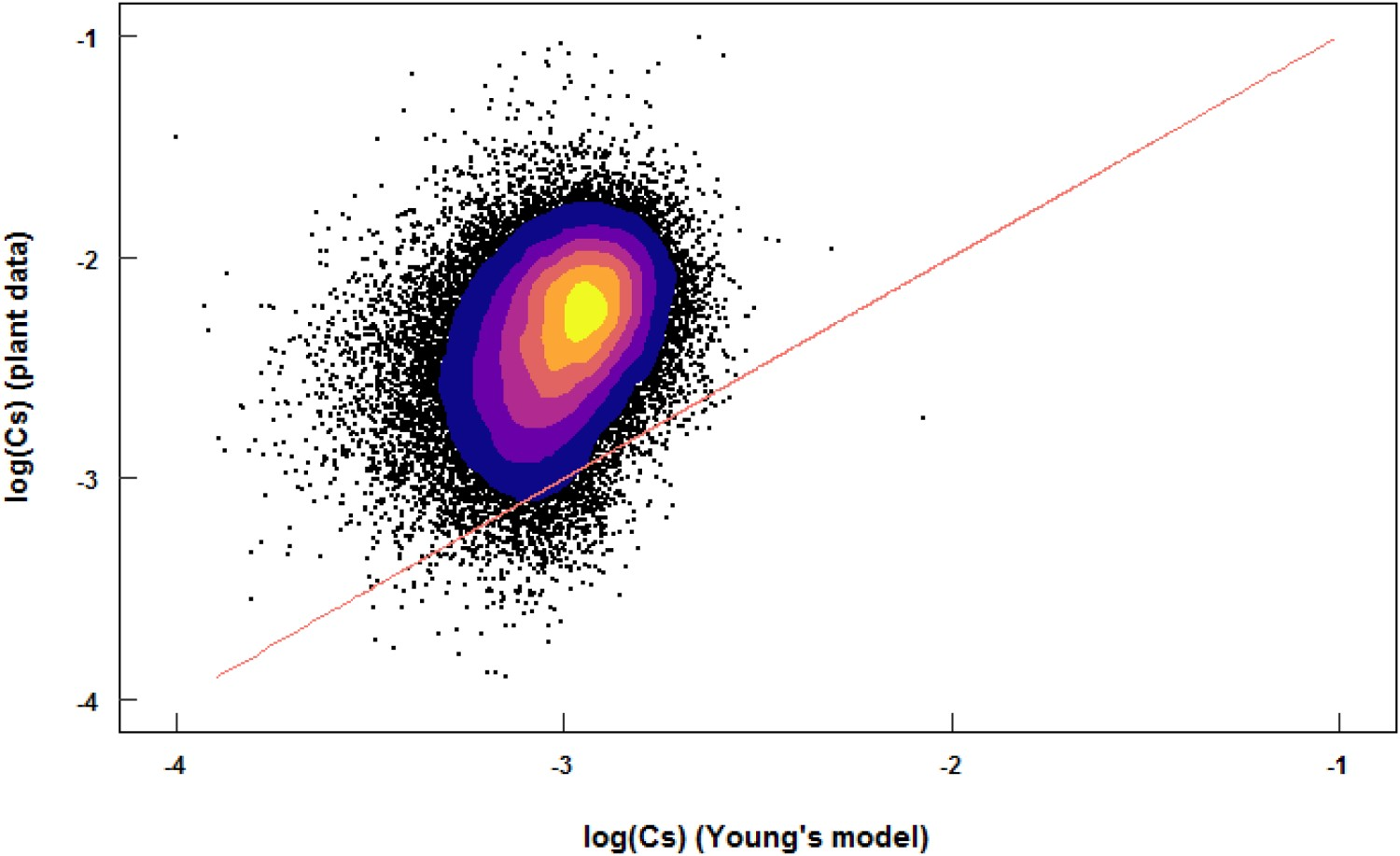
The accuracy of the method to determine CS for a single industrial heat is not very high, because of the rough assumptions made. When applying this method to more than 47 thousand industrial heats, the order of magnitude of the CS values is the same, but the variation cannot be described by Young’s model. It is clear that, despite of the scatter, Young’s model predicts the CS values based on plant data quite well. Young’s model showed the best correlation with the plant data compared to the other models from Table 3. It seems that desulphurisation of hot metal can be predicted by the slag composition (translated to Λ) and temperature only. It has to be noted that when an aO of 3 ppm is used, instead of the 0.5 ppm which is used in the present work, Young’s model (and the other models from Table 3) underestimate CS by a factor 10. The actual CS greatly depends on aO . Therefore, models based on slag composition (or Λ) rely on the assumption that slag and hot metal are in equilibrium.
Most of the desulphurisation in the magnesium-lime co-injection HMD process takes place in the hot metal itself via reaction (1), as this reaction is much faster than reaction (3). The composition of the slag has no influence on the reaction with Mg. Only reactions (2) and (3) take place at the hot metal-slag interface (at least for a large part). If reaction (1) is significantly faster than reaction (2), the slag and hot metal will not be at equilibrium. Since almost no MgS can be found in industrial HMD slag [14], all MgS that is formed via reaction (1) will react to CaS via reaction (2) during the HMD process. Furthermore, resulphurisation (sulphur from the slag dissolving again in the hot metal) is observed in industry, but this is in the order of magnitude of 1–10 ppm. If the metal and slag were far from equilibrium, larger resulphurisation would be observed in industry, especially for heats that are delayed between HMD and BOF. Magnelöv et al. [7] stated that CS calculated based on Λ is not applicable for the HMD process, because the HMD slag is not homogeneous and fully liquid. This inhomogeneity of the slag is another explanation for the large scatter in Figure 4.
Influence of slag components and temperature on CS, ranging from ▾ (negative) to ▴▴ (very positive)
It should be noted that the effect of MgO is marginal. Under industrial conditions it has a slight positive effect as it often replaces CS -negative components like SiO2 or Al2O3. Note that in this table the slag component MgO is discussed and not the metallic Mg, which is injected during the HMD process to desulphurise the hot metal.
Basicity
The CS depends on the basicity of the slag, which depends on the slag composition. Understanding basicity as having a high concentration of free oxygen, which can be replaced by sulphur more easily, helps understanding that slags with a higher basicity will pick up more sulphur and thus help desulphurisation. This explains the large influence of aO on CS as well. There is no universal quantitative definition of basicity available. Therefore, different empirical definitions are used today, including optical basicity Λ (defined by Young et al. [36,37]) and the CaO/SiO2 (known as B2) ratio (which can be extended with MgO, Al2O3 and P2O5), which is commonly used in steel plants [50,51].
Although basicity is hard to quantify from a scientific point of view, an empirical definition of the basicity, like B2, is sufficient for industrial practice. In a slag with a basicity (B2) below 0.93, which is equal to a molar ratio CaO:SiO2 of 1:1, the CaS formation will be retarded by the lack of free oxygen (O2- ions), which are donated by basic oxides. For completeness MgO (as O2- donator) and Al2O3 (which can act as O2- acceptor) should also be taken into consideration [3,34,50]. Above this minimum slag basicity, there should be enough CaO, stoichiometrically, to react with the MgS, according to reaction (2) (MgS reacting to CaS). Only kinetics (like undissolved lime not being in contact with the hot metal, for example the core of a lime particle [2]) will hamper this reaction. Therefore, in industry some extra lime will be needed on top of the lime required to bring the B2 above 0.93 and the lime required for reaction (2). How much extra lime is required is difficult to quantify on a theoretical basis. Li et al. [52] suggest a minimum B2 of 1.1, based on industrial experience.
Iron loss
Types of iron loss
The definition of iron losses during the HMD process is the amount of Fe that is (unwantedly) removed during the HMD process (mostly during the skimming). Iron losses can mount up to 0.5–2.5% of the total hot metal weight. The total iron losses depend on the ladle size and geometry, larger ladles typically lead to lower iron losses, but also on the slag conditions and the skimming skills of the operator [5–10,22,52,53]. It is hard though to have an accurate number for iron losses (via slag), since the iron distribution in the slag is not homogeneous, so a sample will not give an accurate value [22,53]. Also determining the iron loss by measuring the weight difference before and after skimming is inaccurate by a few hundred kilograms (approximately 5 wt-% of the slag), as the amount of BF carryover slag that was present is unknown and the weight measurements themselves are inaccurate, which makes an accurate mass balance under industrial conditions not possible. There are different types of iron losses: Colloid loss: iron droplets entrapped in the slag in colloidal form (like an emulsion) and removed together with the slag (see Figure 5). Entrainment loss: iron entrained with the slag during skimming (see Figure 5). Dust loss: iron that leaves the system as dust. Skull formation: iron that solidifies at the ladle rim or skimmer paddle and forms skull. Chemical loss: iron that reacts and ends up in the slag. Schematic representation of colloid loss (left) and entrainment loss (right) during skimming in the HMD process [6].
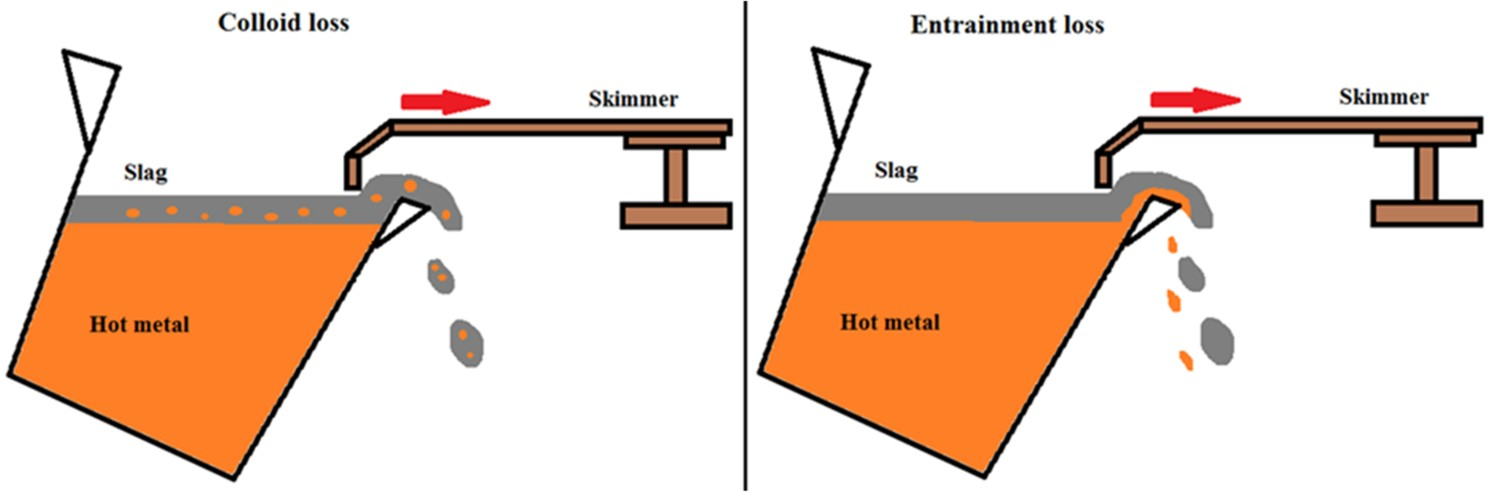
Of these types colloid loss and entrainment loss are the most important. Together they cover > 95% of the total iron loss. SEM analysis of industrial HMD slag, done by Yang et al. [53, 54], shows both small (< 0.5 mm) round iron droplets, typical for colloid loss, and large (> 0.5 mm) irregular shaped iron, typical for entrainment loss. The total amount of iron of both droplet types is in the same order of magnitude. Although this method makes it difficult to exactly quantify the size of colloid loss and entrainment loss, it does prove that both types of iron loss are of comparable size.
Viscosity of the slag: a higher viscosity leads to higher iron losses. Solid fraction: more solids in the slag lead to higher iron losses. A higher solid fraction also increases the slag’s viscosity. Particle size and shape of the solids in the slag: bigger and variable sized particles lead to higher iron losses. Interfacial tension and wettability: a lower interfacial tension between slag and iron leads to higher iron losses. Iron droplet size: smaller iron droplets lead to higher iron losses
Slag properties will not have major influence on
Iron droplets
Changing the slag viscosity has a larger influence on the colloid loss than on the entrainment loss. Therefore, when trying to influence the iron losses via the slag properties, which is the scope of this research, the focus should be on the colloid loss. The iron droplets, present in the slag in colloidal form, do not have a uniform size and shape. Their size and shape depends on the way the droplets are formed. Two mechanisms of how iron droplets are formed are described in literature (see Figure 6). In mechanism I, droplets are formed by iron being entrained by N2 and Mg gas into the slag, where they will get a regular round or oval shape to minimise the surface area. In mechanism II droplets are formed by iron being splashed through the open eye on top of the slag, where it solidifies in an irregular shape [9,53,54]. Han and Holappa [57] showed with hot metal experiments that droplets formed via mechanism I are not spherical, but irregularly shaped (in the experiments most droplets had a diameter, ddrop
< 10 μm). The droplets do become spherical when solidifying. Besides, they define two separate mechanisms within mechanism I: film entrainment and bubble entrainment of the iron. Schematic representation of iron droplet formation mechanisms at HMD. Mechanism I shows droplets entrained by gas bubbles; mechanism II shows droplets launched from the slag eye on top of the slag.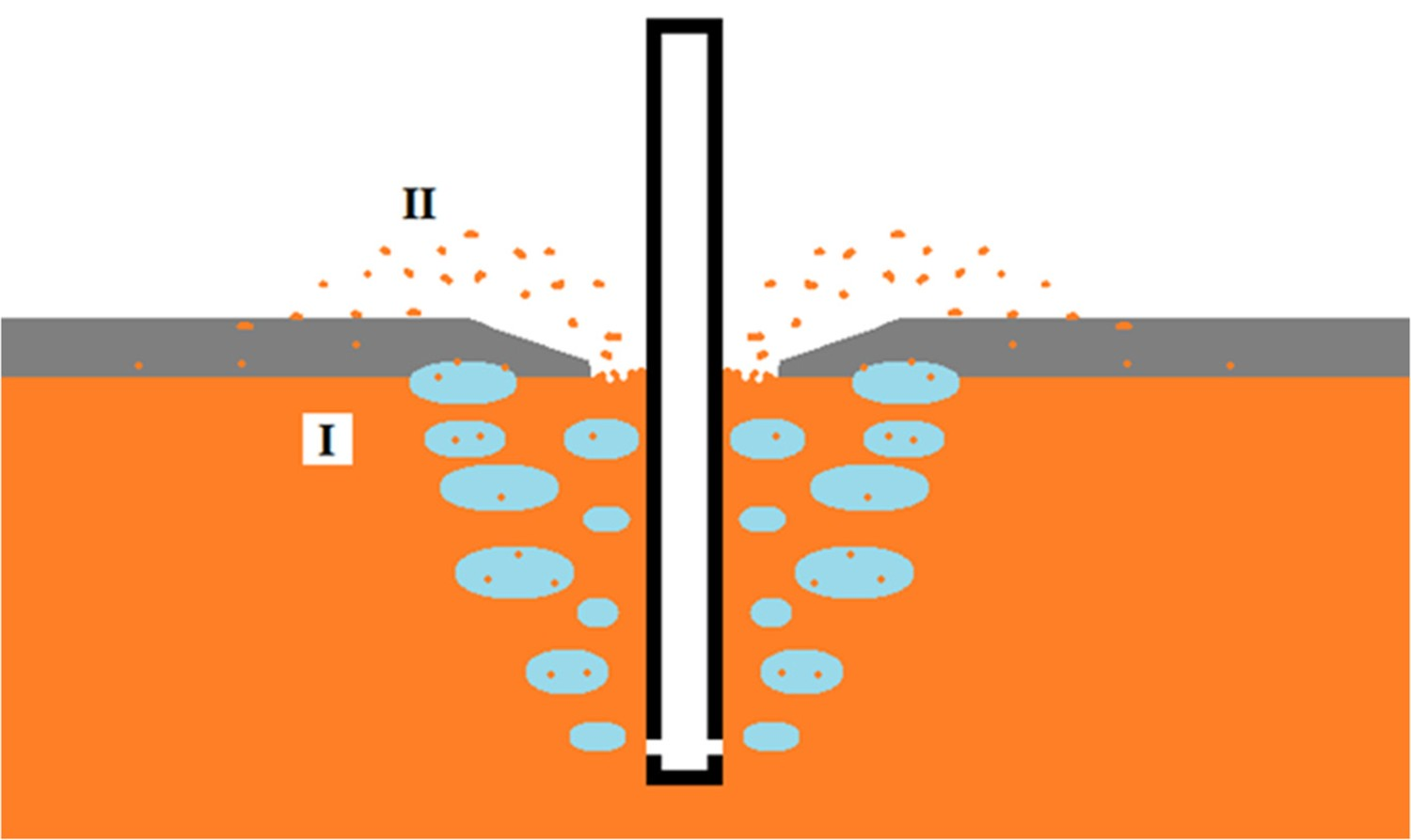
Yang et al. [53] found that when the injection process lasts longer, more iron ends up in the slag via mechanism I (there is an almost linear relationship), while the amount of iron in the slag via mechanism II hardly depends on the injection time at all. This is in contradiction with what Visser [14] suggested, that iron in the slag builds up over time via mechanism II. However, Visser did not consider mechanism I as a significant source of iron and did not investigate both mechanisms. Yang and Visser agree that the total amount of iron in the slag does increase when the injection process lasts longer.
Viscosity of the slag
It is generally accepted that a lower apparent slag viscosity (ηslag
) leads to lower colloid losses, which usually also leads to lower overall iron losses. Figure 7 shows the estimated iron loss per heat (300 t) for 47,109 heats at the HMD stations at Tata Steel, IJmuiden, for the estimated ηslag
. Iron loss at different ηslag at Tata Steel in IJmuiden. Circles show the individual heats. The boxes stretch from the 25th till the 75th percentile of the distribution. The lines (whiskers) extend to 1.5 times the interquartile range. In red a polynomial trendline.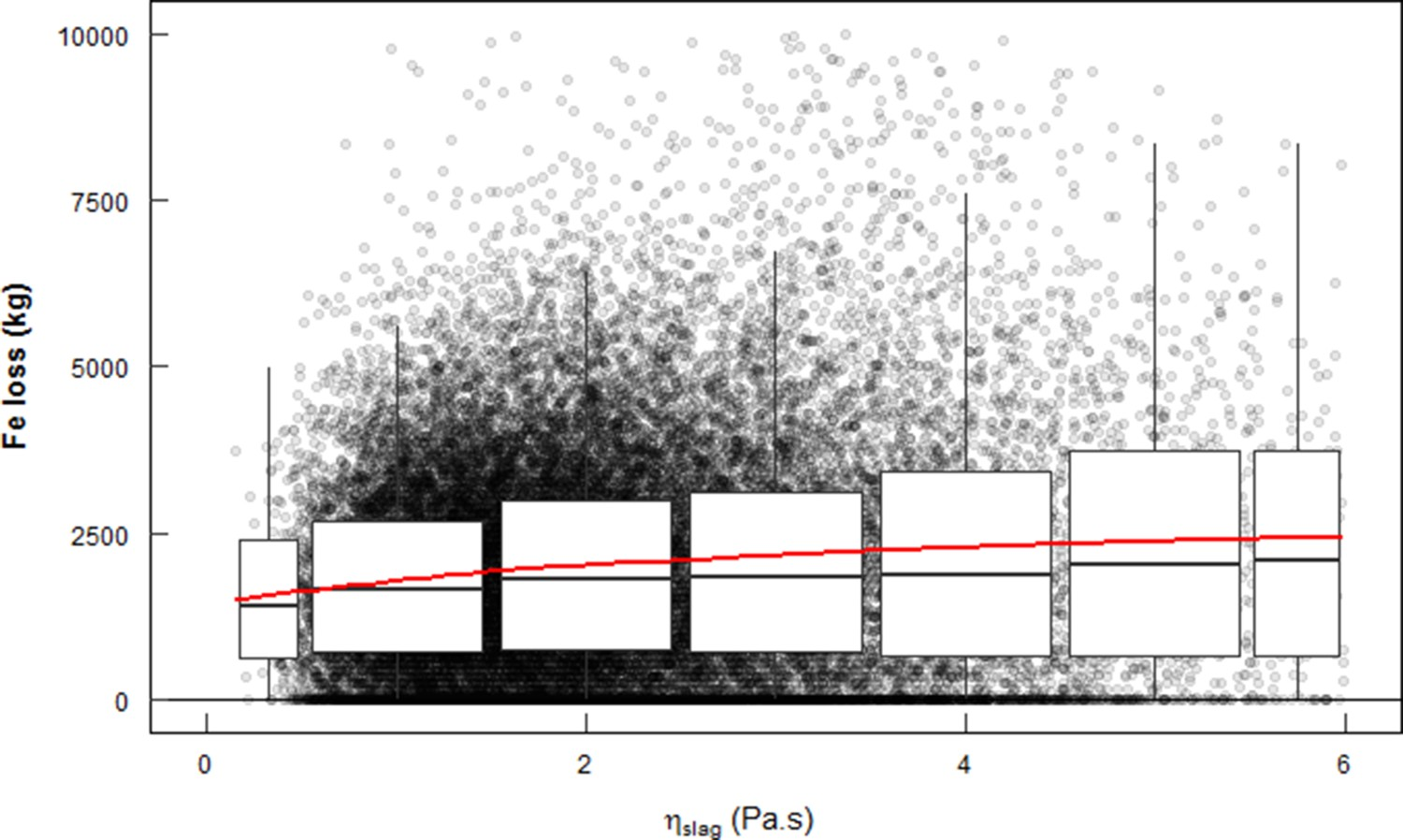
The ηslag
(in Pa·s) is estimated based on the Einstein-Roscoe equation [58] (Equation (15)), which can be used to determine ηslag
for slags.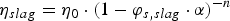
To determine η0 and φs,slag , simplified equations, based on FactSage calculations, were used. These equations depend on temperature and on the fractions of the major slag components (CaO, SiO2, Al2O3, MgO and CaS), where the temperature has the largest influence on both η0 and φs,slag . The slag composition is estimated by taking an average BF carryover slag composition and adding the injected reagents and removed sulphur, assuming that all sulphur becomes CaS and all Mg becomes MgO. The iron loss is estimated by doing a mass balance over every heat, measuring the ladle weight before and after the HMD process and estimating the BF carryover slag (typically 1500 kg) and the amount of slag that remains in the ladle after skimming (typically 500 kg). The method to estimate ηslag and iron loss is inaccurate. Estimating viscosities of industrial slags always leads to large errors, typically > ± 30 % [59]. However, the large amount of data (47 109 heats) makes the trend reliable. It is clear that a higher ηslag leads to higher iron losses.
With the help of Stoke’s law (Equation (16)) the influence of ηslag
on the time an iron droplet needs to settle back from the slag into the metal bath, can be estimated [14,55]. The influence of ηslag
on tsettle
for different ddrop
(ranging from 0.01 to 10 mm) with hslag
= 10 cm.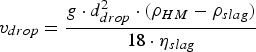
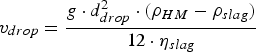
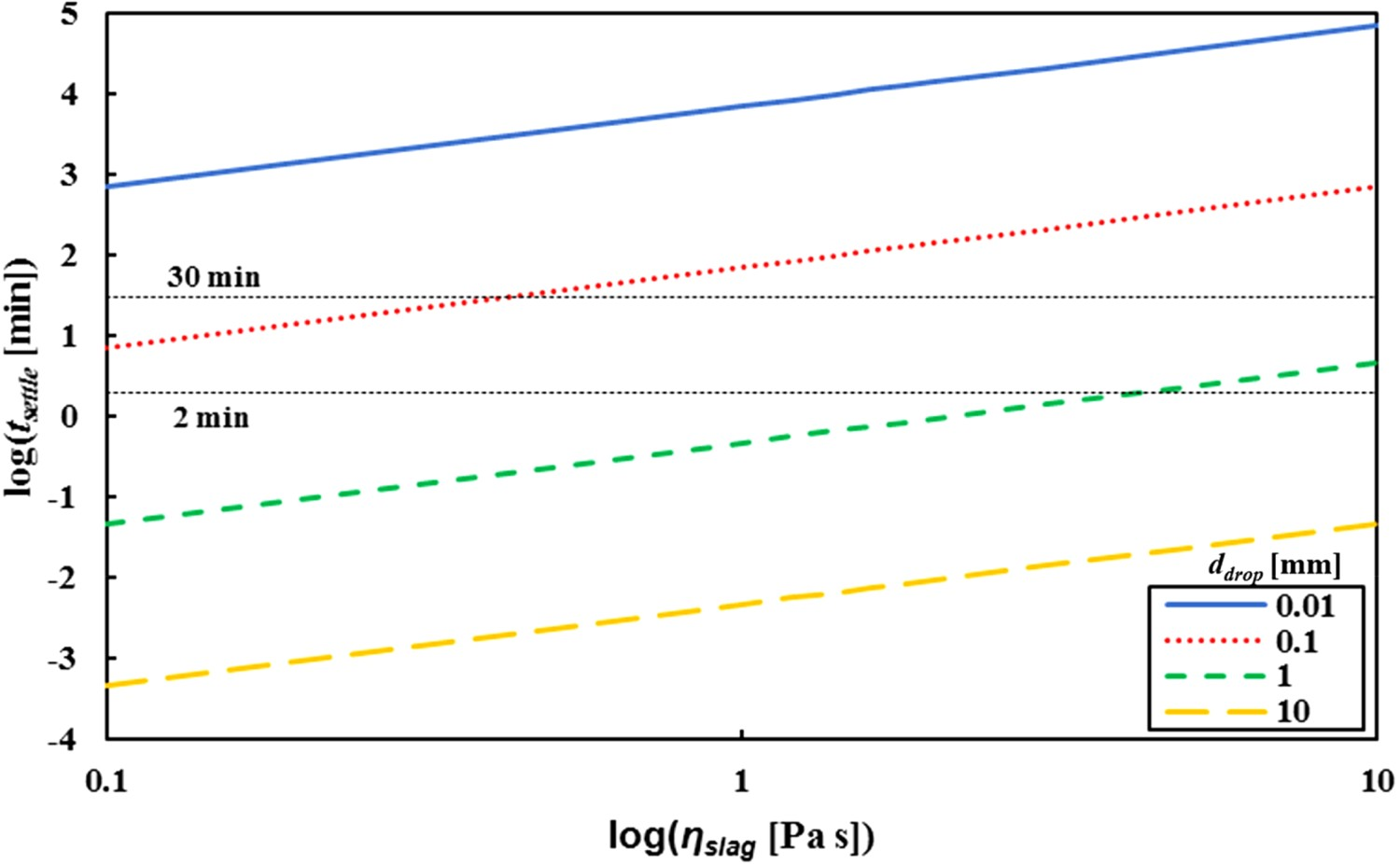
Under industrial conditions, the minimum time between stop reagent injection and start skimming is 2 min (lance lifting, sampling and ladle tilting). Droplets that settle in less than 2 min will therefore never be skimmed off. Under normal conditions, the maximum time between the start of reagent injection and the end of skimming is 30 min. Droplets that take more than 30 min to settle will always be removed together with the slag, if they start on top of the slag. For droplets with a settling time between 2 and 30 min, it depends on the moment they ended up in the slag and on the moment when the skimming starts, whether they are skimmed off or not. Note that the mentioned settling times are valid for a droplet that starts on top of the slag; for droplets that end up in the slag via mechanism I and start at a lower point in the slag, different settling times apply. In this simplified model, the extra friction for droplets that are not spherical, as well as the surface tension a droplet has to overcome when it lands on top of the slag, has been neglected.
Nevertheless Figure 8 shows that, regardless of the circumstances, droplets with ddrop > 2 mm will always settle before skimming starts. Droplets with ddrop < 0.5 mm will never settle in time. This means that by optimising ηslag and the allowed tsettle , within industrial boundaries, only the droplets between 0.5 and 2 mm can be retrieved.
Influence of different slag components on Tliq and η 0, under typical HMD conditions.
Although many components are able to lower Tliq and η0 of the slag, some of them have disadvantages that make them unwanted or restricted for an optimal slag. Halogen-based components (CaF2, CaCl2) are harmful for human health and environment. Besides, fluoride-based components lower the desulphurisation efficiency of magnesium [6]. Adding too much alkali metal oxides (Na2O and K2O), will make the slag less suitable for recycling at the BF, as alkali metals tend to recirculate inside the BF due to their low boiling point, which leads to an unwanted build-up of these elements [16,17]. Furthermore, TiO2 leads to Ti(C,N) formation. Ti(C,N) particles form a layer between the slag and hot metal and make the slag sticky, resulting in higher iron losses [14,64]. Finally, the fact that SiO2 lowers Tliq , but increases η0 , while CaO does the opposite, explains some typical misunderstandings in steelmaking regarding the influence of basicity on ηslag . Einstein-Roscoe’s equation (Equation (15)) shows that for lower temperatures, where part of the slag is solid, lowering the solid fraction by lowering Tliq , lowers ηslag . At higher temperatures, where the slag is fully liquid, only lowering η0 will lower ηslag . In secondary metallurgy, slag temperatures are high (> 1500 °C) and the slags are usually liquid. Under these conditions a higher basicity decreases ηslag . As HMD slag has lower temperatures, typically part of the slag is solid, so a lower basicity (more SiO2) decreases ηslag .
Solid fraction of the slag
It is generally accepted that a lower solid fraction of the slag leads to lower iron losses. Although a fully liquid slag will lead to increased entrainment losses, the decrease in colloid losses will more than make up for that. Industrial data showed that higher temperatures, resulting in a higher liquid fraction of the slag, lead to lower overall iron losses [10]. It should be noted that a substantial amount of the CaS will not dissolve in the HMD slag and, as it has a melting point of 2525 °C, will remain as a solid in the slag.
As the HMD slag is not a homogeneous single phase, the slag will not have a single melting point. Therefore, typically part of the slag is solid, while another part is liquid. The larger the liquid part of the slag is, the lower the iron losses are [4,11,52]. In order to better understand the influence of the slag composition on the liquid fraction of the slag, the slag can be viewed at as if it is homogeneous. With the thermodynamic software FactSage, using a private database [13,20], a ternary diagram is made to show Tliq
for CaO–SiO2–Al2O3 slag with 10 wt-% MgO (typical for HMD slags), which is shown in Figure 9. Liquidus projection of CaO-SiO2-Al2O3 slag with 10 wt-% MgO, determined with FactSage. The lines indicate Tliq
(°C). Typical composition ranges for BF carryover slag (‘BF’ solid line) and final HMD slag (‘HMD’ dashed line) are encircled in the diagram. The dotted line indicates where B2 = 1.1.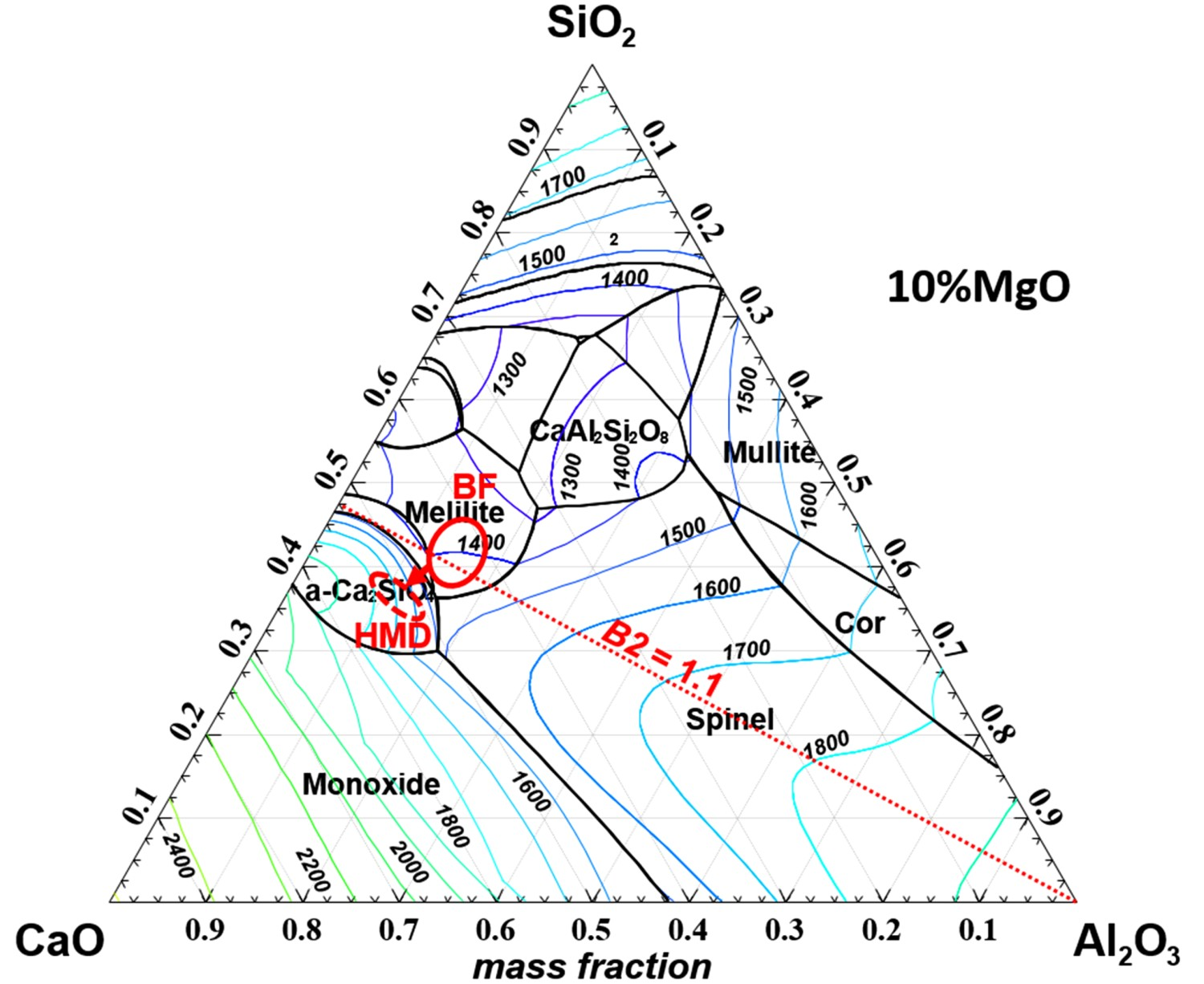
It should be noted that the other slag components all lower Tliq , as can be seen in Table 5. Therefore at the BF and HMD the actual Tliq will be lower than expected based on Figure 9. When keeping the HMD slag composition range from Table 2 in mind, it is clear that lowering the slag’s basicity, so adding more SiO2 and Al2O3, would lower Tliq of the slag. It is remarkable that the composition of BF carryover slag is closer to the ‘sweet spot’ with the lowest Tliq than the HMD slag composition after injection. This is due to the fact that at the BF a liquid slag is favourable and therefore a control target [16,17]. During the HMD process effectively MgO (via reactions 1 and 2) and CaO, which both increase Tliq , are added to the slag.
In literature MgO, CaO/SiO2 (B2) and Al2O3 (together with FeOx) are identified as the components with the largest influence on Tliq of HMD slag [52,60,62]. Li et al. [60] suggest that for a mostly liquid HMD slag MgO should be <10 wt-% and Al2O3 should be 12–16 wt-%. The composition range of a typical HMD slag (Table 2) shows that in practice MgO should be as low as possible, while Al2O3 should be increased.
Apart from the slag’s solid fraction, it has been suggested that the size and shape of the solid particles themselves influence the iron losses as well. Larger and more irregularly shaped slag particles will hamper the settling of the iron droplets in the slag. Magnelöv et al. [7,8,11] showed that addition of the slag modifier nepheline syenite makes the HMD slag look more ‘fine-grained’ during the HMD process and that this slag was easier to skim. Also cold samples from that slag, after skimming, showed a finer-grained slag compared to the reference slag, with a comparable composition. However, they could not prove that this finer-grained slag actually led to lower iron losses.
Interfacial tension
Interfacial tension is another factor which can influence the iron losses. When the interfacial tension between the slag and the hot metal droplet decreases, it will lead to more friction when metal droplets descend through the slag layer. Therefore, it is expected that a lower interfacial tension will lead to higher iron losses. Interfacial tensions between slag and hot metal are difficult to measure, as slag and hot metal tend to react, thus changing the initial compositions. In general the effect of dissolved elements on the interfacial tension is known [65].
The composition of the hot metal has a larger effect on the interfacial tension than the composition of the slag. Sulphur and oxygen, being surface active elements, have the largest influence on the interfacial tension. More oxygen or sulphur in the hot metal lead to lower interfacial tensions [65]. Therefore, to lower iron losses, the oxygen and sulphur concentration in hot metal should be as low as possible. Given the purpose of the HMD process, the sulphur and oxygen are always kept as low as possible, regardless their effect on the interfacial tension.
Of the elements that lower the interfacial tension of the hot metal, titanium has the largest influence. However, even though a higher titanium content of the hot metal leads to increased iron losses, the effect of the interfacial tension seems to be negligible. The increased iron losses are mostly attributed to the Ti(C,N) formation.
From all typical slag components, FeO and MnO have the largest influence on the interfacial tension. Under HMD conditions, FeO will reduce to Fe and [O], leading to an increased oxygen concentration in the hot metal, which leads to the lower interfacial tension. MnO will react with Fe to form [Mn] and FeO, which on its turn leads to Fe and [O]. Also the effect of other oxides in the slag on the interfacial tension depends on their ability to supply oxygen to the hot metal [65].
Because only little research was done about the effect of interfacial tension on iron losses, as it is difficult to measure [66], there are no reliable figures available on the influence of interfacial tension on iron losses and it is hard to isolate their effect. In general, interfacial tension is not considered as a major factor for iron losses, as iron losses can be explained without it. Furthermore, elements that have the highest influence on the interfacial tension, sulphur and oxygen, are already kept as low as possible in the HMD process. More exotic elements that increase the interfacial tension, like tungsten, are too expensive to use in industry. Therefore, to find the optimal HMD slag, interfacial tension is not taken into account in this study.
Optimal slag
The optimal HMD slag should be able to contain sufficient sulphur, while leading to the lowest possible iron losses. Under industrial conditions, the sulphur removal capacity of the slag cannot always accurately be predicted by its sulphide capacity (CS ), as the HMD slag is inhomogeneous and as sulphur is also present in the form of solid CaS. However, a comparison between plant data and CS models shows that CS can be used to indicate if a certain slag composition is optimal or not. CaO is the most important component in the slag, regarding sulphur removal capacity. There should be enough CaO to react with the sulphur and, based on industrial experience, B2 (CaO/SiO2) > 1.1.
To minimise the iron losses with an optimal HMD slag, the focus should be on minimising the colloid losses by lowering ηslag . As ηslag has an adverse effect on colloid loss and entrainment loss, the focus in industry should be on the colloid loss. At the same time, entrainment loss can be limited by taking other measures like improving skimming skills of operators, improving skimming control or by skimming automation. To lower ηslag , both η0 and φs,slag should be lowered. This can best be done by minimising the MgO content of the slag, preferably < 10 wt-%, and increasing the slag’s Al2O3 content, preferably 12–16 wt-%. Furthermore, other slag components that lower the η0 and φs,slag , like Na2O, K2O and MnO are desirable, keeping in mind that their use can be limited because of other process requirements. The amount of TiO2 in the slag should be minimised and is ideally 0. For the optimal HMD slag, a B2 of 1.1 is required, to allow the sulphur removal.
For industry this means that the addition of reagents should be optimised, not only from a desulphurisation point of view, but also to create an optimal slag. Furthermore, a slag modifier could help to further optimise ηslag , and thus minimise iron losses.
Conclusions
Based on the fundamentals of hot metal desulphurisation (HMD) slag and industrial data, the following concluding remarks can be made. The sulphide capacity (CS
), as defined by Fincham and Richardson [4], is not applicable for direct industrial use, as the industrial slag is inhomogeneous and not necessarily at equilibrium, but CS
can be used to determine the optimal slag composition. For a sufficient sulphur removal capacity of the slag, the slag should contain at least enough CaO to allow all MgS to react with CaO to form CaS. Besides, a minimal CaO:SiO2 weight ratio (B2) in the slag of 1.1 is required. A lower apparent viscosity of the slag leads to lower overall iron losses. Optimising the HMD slag conditions has a higher impact on colloid losses than on entrainment losses. Therefore, in industry, the focus should be on lowering the colloid losses. Under industrial circumstances, MgO concentration in the HMD slag should be as low as possible and preferably < 10 wt-%. Al2O3 should preferably be 12–16 wt-%.
These remarks on the optimal HMD slag, considering sulphur removal capacity and iron losses, will be validated in part II of this study [12].
List of symbols and abbreviations
Symbols
Oxygen activity-
Basicity CaO/SiO2-
Sulphide capacity –
Droplet diameterm
Henrian sulphur activity coefficient –
Gravity constant (9.81)m·s−2
Slag height m
Reaction equilibrium constant for reaction 7–
Sulphur distribution ratio –
Constant (typically 2.5)–
Partial pressure of x Pa
Temperature K or °C
Liquidus temperature K or °C
Settling times
Settling speed droplet m·s−1
Weight percentage of component xwt-%
Maximum solid fraction –
Viscosity of the liquid fraction of the slagPa·s
Apparent viscosity of the slag Pa·s
Optical basicity–
Density of x kg·m−3
Solid volume fraction of the slag-
Abbreviations
Blast furnace
Basic oxygen furnace, or oxygen steelmaking converter
Electromagnetic force
Hot metal desulphurisation
Kanbara reactor
X-ray fluorescence
