Abstract
To effectively recycle the phosphorus in P-bearing steelmaking slag and use it as slag phosphate fertilizer, it is necessary to study the effect of slag temperature on occurrence forms and enrichment behaviour of phosphorus in P-bearing steelmaking slag. In this work, the thermodynamic and kinetic behaviours of the formation of phosphorus-rich phases are systematically investigated. The results show that decreasing of the slag temperature (1473—1823 K) has a little effect on the thermodynamic trend of the formation of the 2CaO·SiO2 phase (denoted as C2S) and on the early precipitation of the n2CaO·SiO2–3CaO·P2O5 solid solution (denoted as n C2S–C3P). Additionally, the precipitation of C2S first increases and then decreases, and reaches a maximum value between 1623 and 1653 K. Moreover, the diffusion of phosphorus is a rate-limiting link in the process. The kinetic equation that describes the variation of P2O5 content with time in the phosphorus-rich phase is 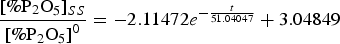
Get full access to this article
View all access options for this article.
