Abstract
To lower the iron losses of the hot metal desulphurisation (HMD) process, slag modifiers can be added to the slag. Slag modifiers decrease the apparent viscosity of the HMD slag. Most common slag modifiers in industry contain fluoride as a fluidiser. However, fluoride leads to a higher magnesium consumption and has health, safety and environment issues. Fluoride-free alternatives like nepheline syenite (NS) and fly ash (or pulverised fuel ash, PFA) can decrease the slag’s apparent viscosity. Experiments with HMD slags containing CaF2, NS and PFA and without slag modifier were performed for slags with a high and an average basicity. The melting points of the slags and their viscosities 1250–1600°C were measured. The experimental results are compared with FactSage calculations. PFA and NS are viable alternatives in the industrial HMD process, as reasonable amounts are sufficient to reach the same lower apparent viscosities and melting points as with CaF2.
Introduction
Iron losses during the slag removal are the largest costs of the hot metal desulphurisation (HMD) process. Typically, during the slag skimming, more than half of the removed material is iron rather than slag. This means that, depending on the process scale, 500–3000 kg of iron is removed per heat. On an annual basis, millions of euros worth of iron is skimmed off in the HMD process and only part of that can be recovered during slag processing [1–6].
Iron losses during the HMD process as a result of slag skimming can be divided into entrainment losses and colloidal losses (also called emulsion losses). Entrainment losses refer to iron that is entrained with the slag when it is skimmed off. Colloidal losses refer to iron that is entrapped in the slag in a colloidal form (an emulsion when the slag and iron are both liquid, a suspension when the iron is solid and a gel when the slag is solid), which is removed together with the slag. Figure 1 schematically shows the difference between colloid loss and entrainment loss. In industry, these two types of iron losses are typically of the same magnitude [2,4–6]. Schematic representation of colloidal iron loss (left) and entrained iron loss (right).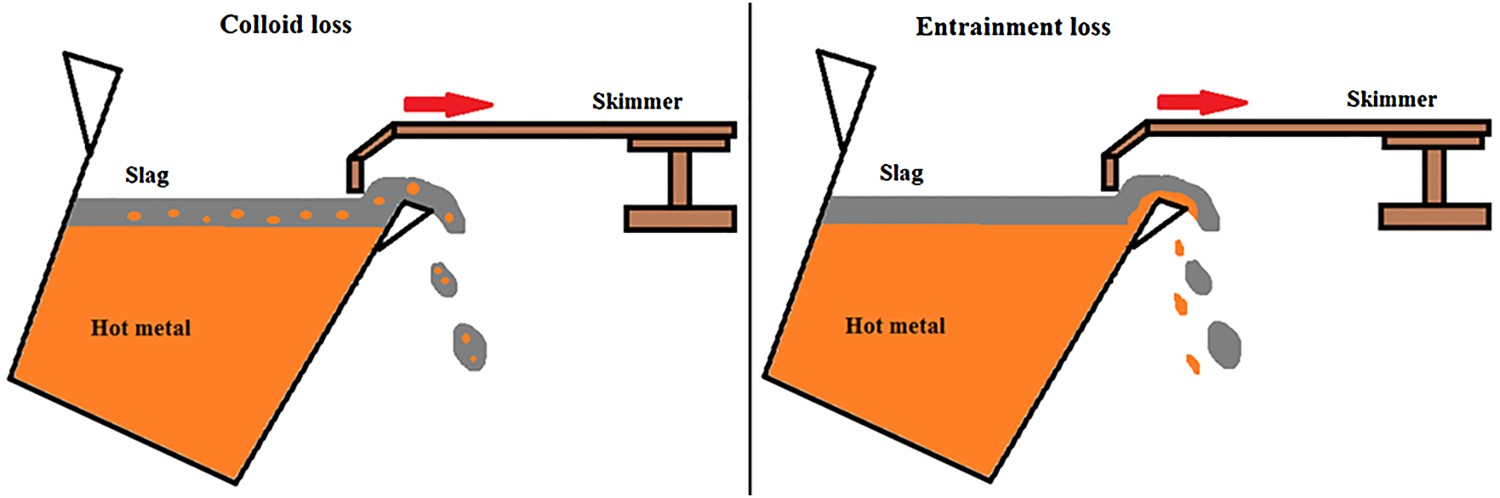
Colloid losses can be reduced by having a less viscous slag, so the entrapped iron will sink back more easily into the metal bath. The liquidity and viscosity of the slag can be made more beneficial by increasing the slag temperature or by changing the slag composition. As it is not economically viable to heat up the slag, many steel plants all over the world use slag modifiers to change the slag composition in order to decrease its apparent viscosity, which depends on the solid fraction and the viscosity of the liquid fraction. Often, fluoride-containing slag modifiers like fluorspar (CaF2), sodium cryolite (Na3AlF6) or potassium cryolite (KAlF4) are used [7]. A problem with fluoride is that it is environmentally unfriendly and in the magnesium-lime co-injection HMD process, fluoride can decrease the magnesium efficiency, because the fluoride reacts with the magnesium to form MgF2 [1]. A fluoride-free slag modifier that would decrease the apparent viscosity of the slag just as effectively would, therefore, be beneficial for the industrial HMD process. In literature nepheline syenite (NS) [4–6] and fly ash (or pulverised fuel ash, PFA) [8] are reported as successful fluoride-free slag modifiers for the HMD process.
In the present paper, it is investigated if NS and PFA are viable alternatives for fluoride-containing slag modifiers in the HMD process.
Theory
HMD slag
Typical slag compositions for BF carryover slag and HMD slag after injection (without slag modifier). Composition in wt-%, excluding iron.
Figure 2 shows that the HMD final slag has a high solid fraction at typical HMD temperatures (1300–1450°C) and that the slag’s solid fraction increases during the HMD process. Note that the exact composition of Table 1 is used for the FactSage calculations that will be presented in this paper, so no slag modifier or FeO, which would decrease the solid fraction, is considered.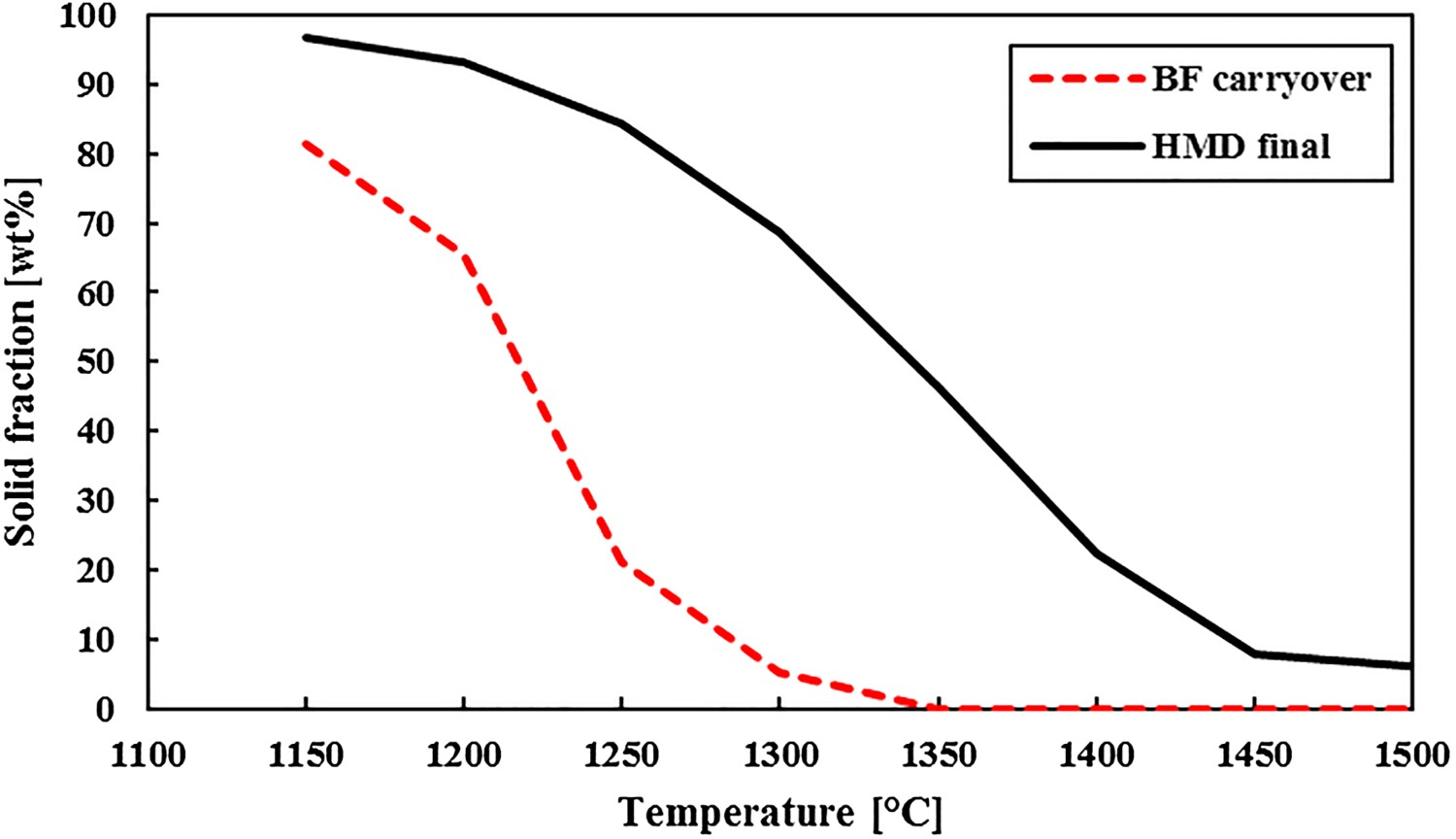
Iron loss
HMD slags with a higher apparent viscosity (η
app, in Pa·s) have higher colloidal iron losses, as entrapped iron droplets cannot easily drip back into the bath. Slags with a very low η
app will have higher entrainment iron losses, because it will be harder to skim off a very liquid slag. Therefore, the most ideal slag would have a low η
app during injection but would be solid during skimming (after all the iron dripped out of the slag). However, such a large physical change of the slag in such a short time will be difficult to accomplish in practice, even with the use of slag modifiers. Since typically the overall iron losses increase at an increasing η
app, a slag with a low η
app is desired. The η
app depends on the volume fraction of the solids (f) and the viscosity of the liquid fraction (η0
). For slags with f < 5 vol.-%, η
app can be determined with the Einstein-Roscoe equation [2,12,13]: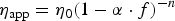
Basicity
The viscosity of the slag is influenced by its basicity. In industry, basicity of the slag is typically determined via B2 (Equation 2) or B4 (Equation 3) [15]: Solid fraction (in wt-%) of typical HMD slag for different B2 values at 1350°C. Determined with FactSage [11].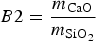
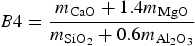
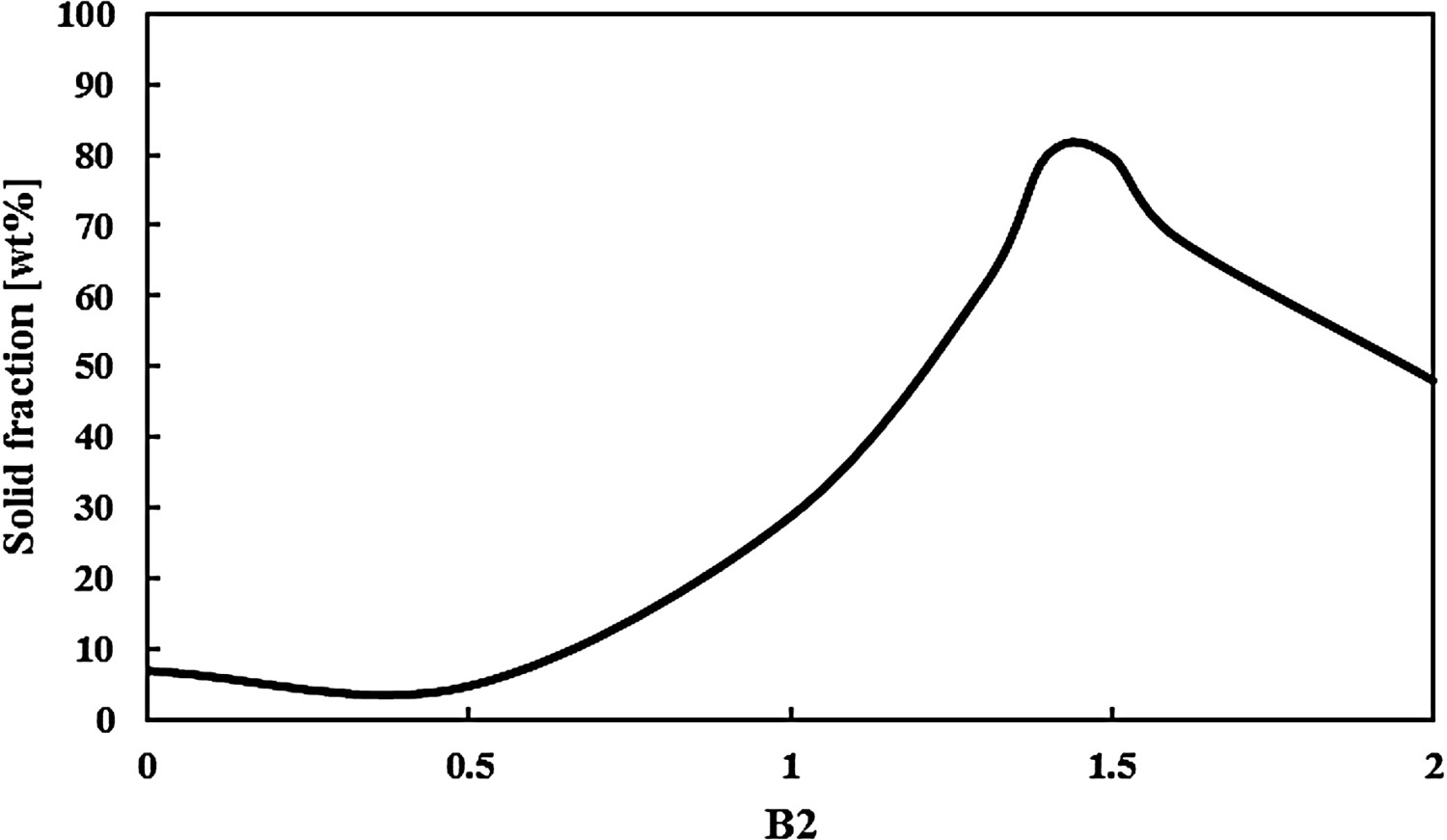
As can be seen in Table 1, a typical HMD slag is basic (for this example B4 = 1.6 and B2 = 1.3). The FactSage calculations for the typical HMD slag from Table 1 at 1350°C, only changing B2, show that the solid fraction is lowered by lowering B2. Furthermore, since f > 10 vol.-%, lowering the solid fraction is the only way to lower η app.
Slag modifiers
Adding acid network formers, like SiO2 or Al2O3, decreases the apparent viscosity of HMD slag, even though they increase the viscosity of the liquid fraction of the slag. Adding alkali metal oxides, like Na2O or K2O, which are strong basic network modifiers, will decrease the apparent viscosity of HMD slag as well. Because alkali metal oxides are network modifiers, they decrease η0 , just like CaO does. Unlike CaO, alkali metal oxides decrease the solid fraction of the slag. Alkali metal oxides are therefore excellent HMD slag modifiers. In addition, they increase the sulphur capacity of the slag [17].
Typical compositions (in wt-%) of NS and PFA.
Fluoride-containing slag modifiers have been widely used for decades in industry. Fluoride decreases the slag’s liquidus temperature and its viscosity. However, fluoride does not act as a network modifier in the sense that it breaks down the SiO2 polymeric network. Instead fluoride tends to be calcium driven in an HMD slag and forms CaF+ ion pairs, which break up divalent calcium ions that bind silicate anions (thus forming bridges between two silicate structures). This effectively lowers the slag’s viscosity [18,19]. This means that fluoride is more effective as a slag modifier for slags with a higher basicity, as these have more calcium-silicate bridges.
The disadvantage of fluoride-containing slag modifiers is that they decrease the desulphurisation efficiency of magnesium. The fluoride reacts with the injected magnesium to form MgF2, thus preventing it from reacting with the dissolved sulphur. This phenomenon has been described in more detail in an earlier publication [1]. In addition to that, fluoride can have a negative influence on human health and the environment. Therefore, the use of fluoride-containing slag modifiers is restricted in several countries.
Experiments
Slag preparation
Compositions (in wt-%) of the synthetic HMD slags with different modifiers used for the experiments, determined by XRF.
The composition of the slags without slag modifiers (master slags) are based on slag composition measurements at Tata Steel in Port Talbot (U.K.). To prepare the master slags, reagents of CaCO3, SiO2, Al2O3, MgO, MnO, K2CO3, Na2CO3 and CaS were weighed and mixed in a Tema mill for 20 s. The mixed reagents were then put into a graphite crucible and heated to 1600°C in an induction furnace to prefuse. After 10 min at 1600°C, the furnace was cooled to room temperature with the crucible and slag inside. Once cooled, the prefused slag was pulverised in a Tema mill for 60 s. The prefused powdered slag was then decarburised in an alumina crucible inside a muffle furnace at 700°C for 18 h to remove any residual carbon that had been absorbed from the graphite crucible during prefusing.
For the synthetic slag with CaF2 a concentration typical for industry was added. For the synthetic slags with high basicity (1.3 and 1.4), an equivalent of 20 kg per heat of NS or PFA, respectively, was added. For the lower basicity slags (2.3 and 2.4), an equivalent of 25 kg per heat of NS or PFA, respectively, was added. For the modified slags, the actual slag modifiers were added to the master slags and prefused again. For slags 2.2–2.4, slag 2.1 was used as the master slag. Slag 2.1a was only used for reference.
Viscosity measurements
For the slag viscosity measurements, a Bähr VIS-403 HF rotational viscometer was used. In this setup, the torque applied to a spindle that is rotating at a constant speed, while being submerged in a known volume of the melted slag, is measured. The viscosity is calculated as the ratio of shear stress (τ, in Pa) to shear rate (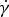
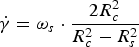
The resulting shear stress is calculated by: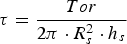
The Bähr VIS-403 HF viscometer was calibrated at room temperature using three certified silicon oils with viscosities between 0.1 and 1.0 Pa.s. Regression analysis was used to determine the calibration curve. The calibration was specific to the rotation speed selected for the tests. The viscosity measurement has a typical error of 10%.
Crucibles made from different materials were tested for the viscosity measurements. A zirconia crucible was not suited, because when the slag inside the crucible was melted, the crucible started to leak at the point where the bottom is glued to the wall. A molybdenum crucible was better suited, although the crucibles initially broke when cooling them down after the experiment due to slag expansion for slag 1.1. This was caused by the phase transformation of dicalcium silicate (C2S) from β to γ during cooling. By doping the other slags with 0.1 wt-% B2O3 (0.14 wt-% for slag 1.4), the crucibles survived cooling down. Lowering the CaO content for slags 2.1–2.4 also helped preventing the β → γ phase transformation of C2S during cooling.
For every experiment, 24 g of prefused powdered slag was put into the crucible and inserted into the rotational viscometer. The oxygen level in the chamber was lowered by an argon purge at 200 mL min–1, to protect the crucible and spindle from oxidation. The temperature inside the furnace was increased to 1600°C after which the rotating spindle was submerged into the molten sample. A constant rotation speed of 400 rev min–1 was maintained throughout the experiment. The sample was then cooled at 10°C min–1 until the sample reached a maximum torque of 25 mNm.
Melting point measurements
For the melting point measurements, a Misura HM2–1600 heating microscope was used. Samples were prepared using a steel die to manually compress prefused powdered slag into cylinders of 3 mm in height and 2 mm in diameter. The samples were then placed onto an alumina plate and inserted into the horizontal tube furnace. The samples were heated to 1100°C at 50°C min–1, after which they were heated to the melting point at 6°C min–1. The device acquires and stores images of the sample at 2°C intervals during the heating cycle. During the heating cycle, all the dimensional parameters were measured automatically in order to identify phase transitions of the material. The sintering temperature of the sample is defined as the temperature where the sample height <95% of the original height. The softening temperature is defined as the temperature where the corners of the sample soften. This is a subjective measure, but it is automatically done by the device’s software, so it is reproducible. The melting temperature of the sample is defined as the temperature where the base of the sample is 3 times larger than the sample height, which is according to the DIN 51730 standard. The melting point measurement has a typical error of ±4°C.
Results
Viscosity measurements
As a reference, a slag sample from the Tata Steel HMD station in Port Talbot (U.K.), where KAlF4 is used as slag modifier (the fluoride content is proportional to 0.6% CaF2 in the post HMD slag), was analysed with the viscometer. Figure 4 gives the apparent viscosity as a function of temperature. It should be noted that, unlike the synthetic slags, the measured slag also contains FeO, which lowers the slag viscosity. Viscosity measurement of HMD slag from Tata Steel Port Talbot (U.K.) at different slag temperatures (October 2018).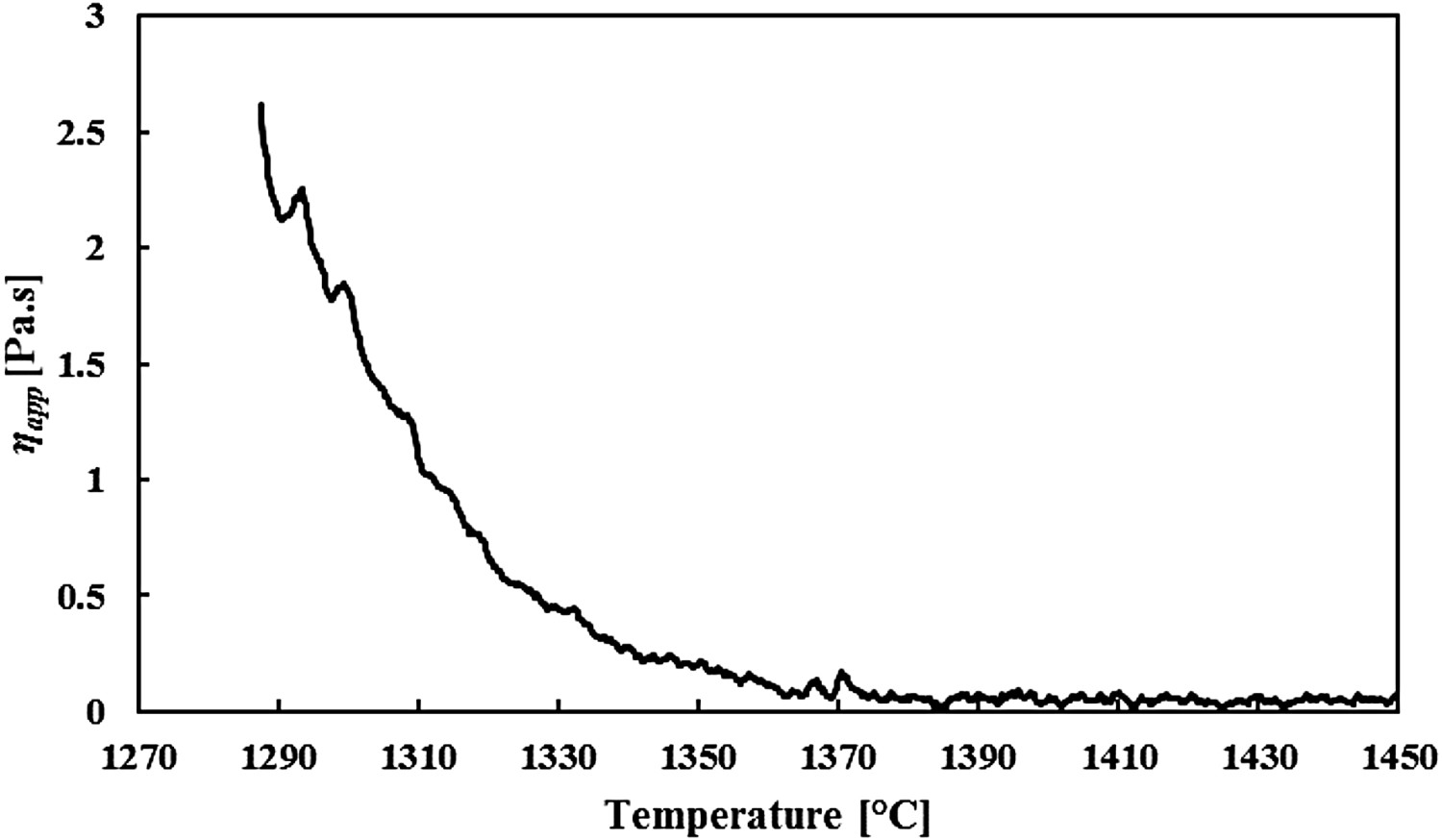
Figure 5 gives the apparent viscosity of slags 1.1–1.4 as a function of temperature. Compared to the real HMD slag from Figure 4, the viscosities of the synthetic slags are much higher at similar temperatures. This is partly because no FeO is added to the slag, but also because of a higher basicity of the synthetic slags (1.1–1.4) compared to the reference slag sample. The measurements clearly show that at lower temperatures PFA and CaF2 significantly lower the slag’s viscosity, whereas NS has no significant effect on the viscosity. At higher temperatures, all slags are liquid, so their viscosities are all around 0.1 Pa·s. Viscosity measurements for slags 1.1 (master), 1.2 (with CaF2), 1.3 (with NS) and 1.4 (with PFA) at different temperatures.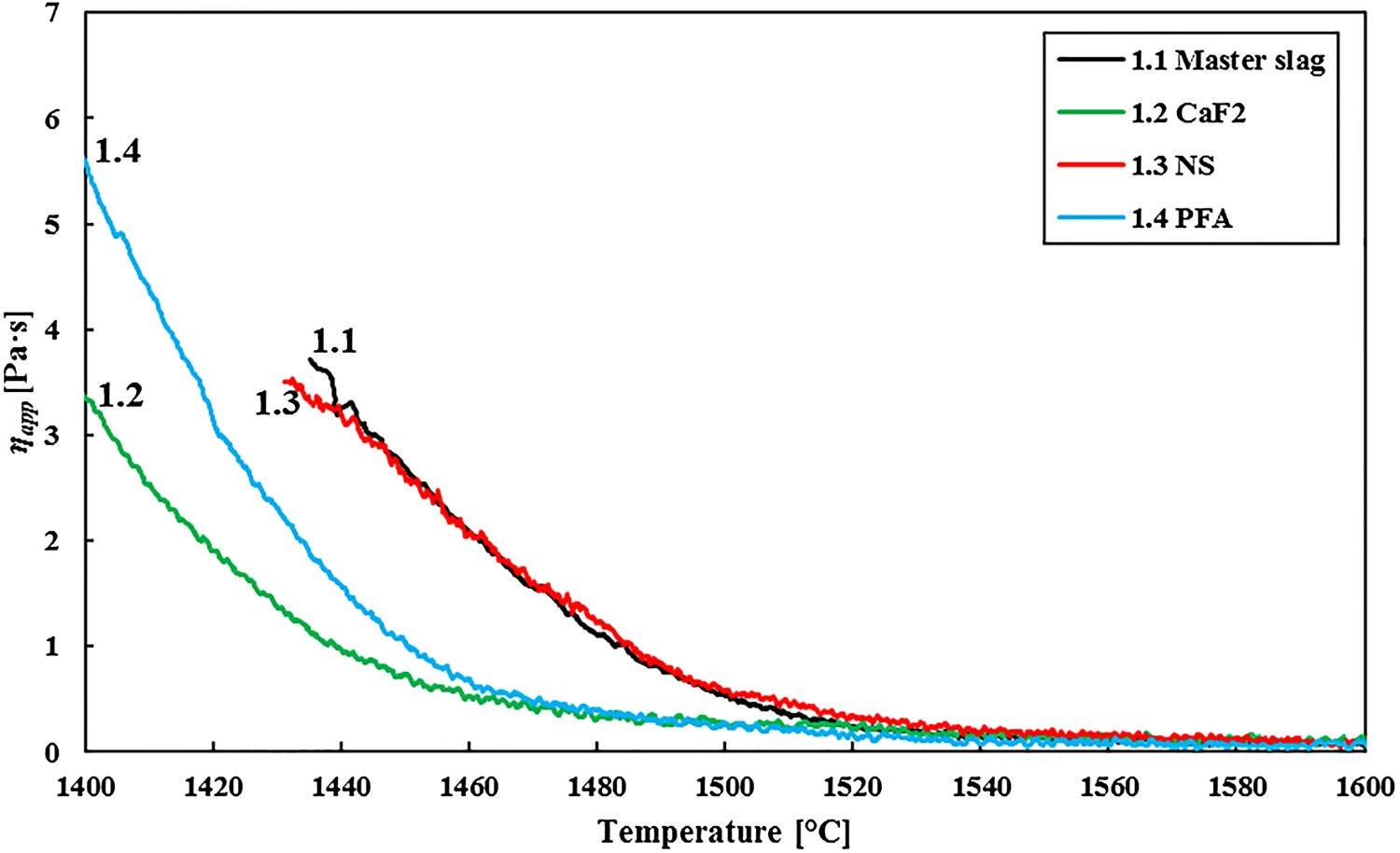
Figure 6 gives the measured viscosities of slags 2.1–2.4, in which more slag modifier is added than in slags 1.1–1.4, at different temperatures. Although the viscosities are lower than for slags 1.1–1.4, they are still higher than for the reference industrial HMD slag sample. The basicity is now comparable, but still no FeO was added to the synthetic slags, which explains the difference with the HMD slag. This measurement clearly shows an effect of all slag modifiers on the slag’s viscosity at lower temperatures (below 1400 °C), where PFA has the largest influence and CaF2 the smallest. Viscosity measurements for slags 2.1 (master slag), 2.2 (CaF2), 2.3 (NS) and 2.4 (PFA) at different temperatures.
Melting point measurements
The reference HMD slag sample from the Tata Steel HMD station in Port Talbot was analysed with the heating microscope as well, to determine its melting point. Figure 7 shows that the slag starts to soften at 1326°C and melts at 1334°C. Melting point measurement of HMD slag from Tata Steel Port Talbot.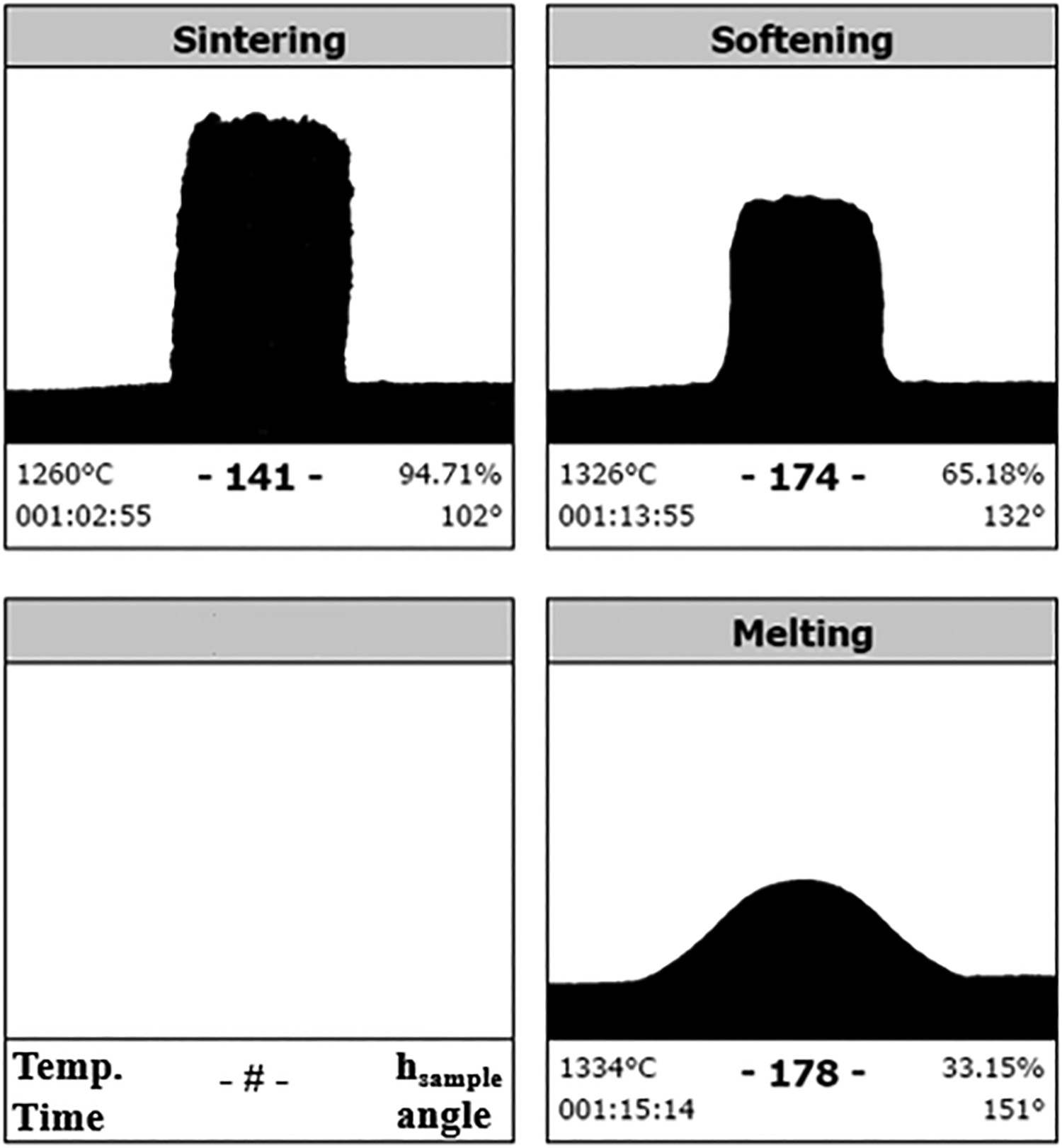
T liq for all synthetic slags, determined with FactSage and their measured T melt, determined with the heating microscope. ΔT gives the difference with the relevant master slag.
Discussion
Experimental results
The large difference between the industrial HMD slag sample and the synthetic slags, both for the apparent viscosity and for the melting temperature, shows that the results with the synthetic slags cannot be used to quantitatively predict the effect of the tested slag modifiers in an industrial HMD slag. This is because the slag sample contains FeO x and has a different basicity from the synthetic slags. Finally, a HMD slag under industrial conditions is never homogeneous, both in composition and temperature. This means that the laboratory results should be discussed semi-quantitatively.
The viscosity measurements show that for a high-basicity slag and high temperatures (see Figure 8, left), NS does not decrease the slag viscosity. At a high basicity, η0
will already be low, while the high η
app is mainly caused by the high solid fraction. Adding alkali oxides, which lower η0
, will not lead to a significantly lower η
app under these conditions. PFA and CaF2 do significantly lower the slag viscosity. At a high basicity, fluoride has a larger effect on η
app, as it breaks down the divalent calcium bridges (see § 2.4). Adding SiO2 and Al2O3 via PFA will lower the basicity, so this is also an effective way to lower the slag viscosity under the given conditions. However, NS also adds SiO2 and Al2O3 to the slag, albeit less than PFA does, so it is an unexpected result that NS does not seem to have any effect at all on η
app at high basicity. Possibly something went wrong with the composition of slag 1.3 (NS), as the NS did not seem to influence the melting point of the high basicity slag either (see Figure 9, left), whereas it did influence T
melt for the medium basicity slag. Measured viscosities for slags 1.1–1.4 at 1450 and 1500°C (left) and for slags 2.1–2.4 at 1350 and 1400°C (right). Difference in melting point (ΔT
melt; left) and break temperature (ΔT
break; right) between the master slag and slag with a slag modifier.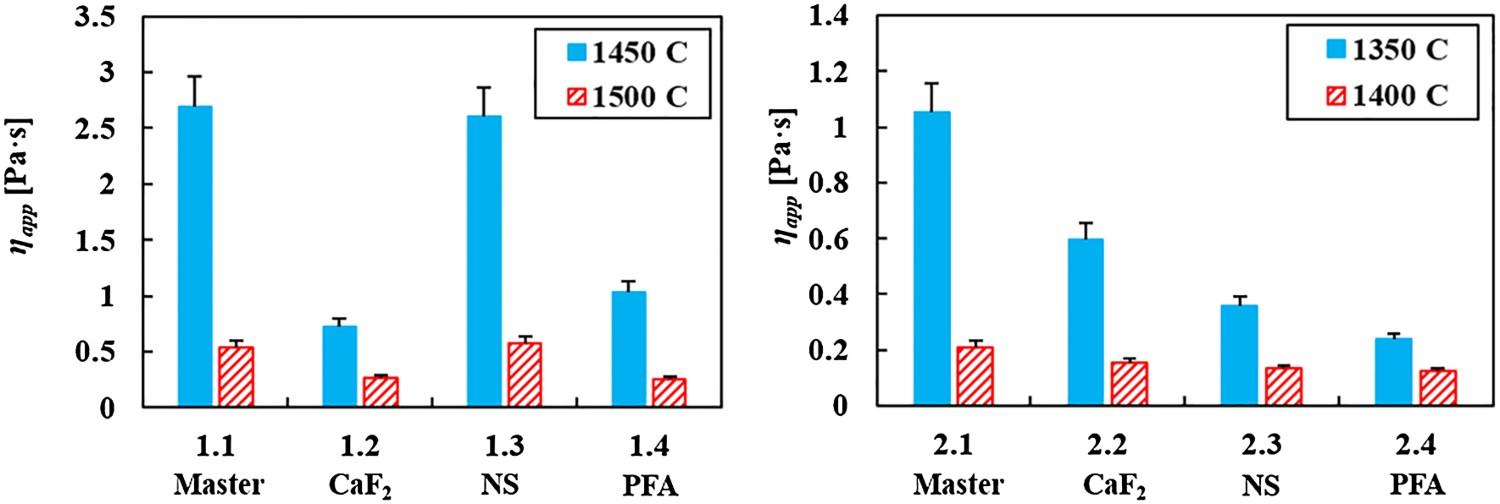
The viscosity measurement results for lower basicity slags and lower temperatures (see Figure 8, right) give another picture. Owing to the lower basicity, most of the slag, with or without slag modifier is already liquid at 1400°C. At 1350°C there are clear differences. All slag modifiers lower the viscosity, but PFA does that most successfully. The difference between CaF2 and PFA can be explained by the fact that at a lower basicity, the divalent calcium bridge breaking by fluoride has less effect on the viscosity, as there are fewer calcium bridges to break. The lower viscosity for slags with PFA compared to slags with NS is more remarkable, as the network-modifying effect of the surplus of alkali metal oxides in NS seems less successful in lowering the viscosity than the surplus of FeOx in PFA. The small difference in basicity between slag 2.3 (NS) and 2.4 (PFA), cannot explain the difference in measured viscosity. Besides as slag 2.3 has a lower B2 than slag 2.4, it would be expected that slag 2.3 has the lowest viscosity, which is not the case.
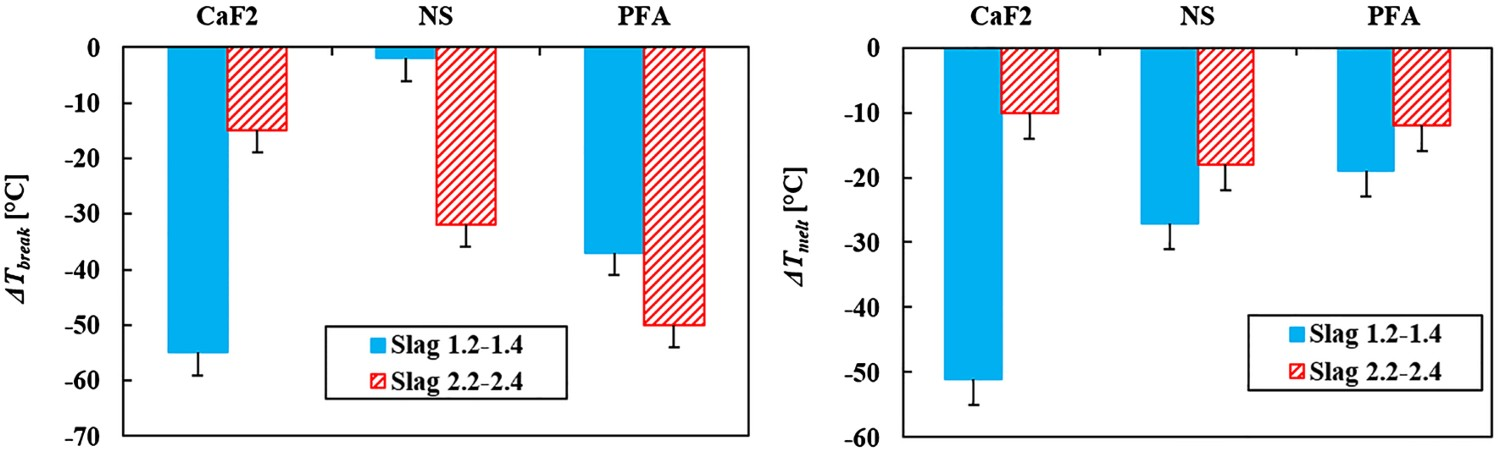
When looking at the effect of the slag modifiers on the slag’s melting point (Figure 9), a similar image as with the viscosity is observed. At high basicity slags CaF2 decreases the slag melting point the most, whereas at a lower slag basicity NS and PFA are more effective in lowering T melt, albeit that the differences are smaller. This suggests that T melt is influenced in a similar way as the viscosity by the different slag modifiers. Note that the measured T melt of slag 1.3 (NS) is doubtful, as its viscosity measurements gave unexpected results as well, as was discussed earlier. As PFA has the highest influence on the slag basicity compared to NS, it consequently has the largest effect on T melt. This is in line with the theory.
The results for slags with different basicity show similar trends and are in accordance with the theory, apart from the measurements of slag 1.3 (NS). Also, the results from the viscosity measurements and the melting point measurements are in agreement with each other. It is therefore safe to conclude that the observed trends for the different slag modifiers are reliable.
Industrial use of slag modifiers
The slag compositions that were measured in this research are representative of adding realistic amounts of slag modifier in an industrial HMD. For a heat size of around 300 t, the synthetic slags represent roughly the addition of 15 kg CaF2, 20 kg of NS and 25 kg of PFA, respectively. The experimental results show that adding a relatively small amount of slag modifier, compared to a total slag weight of around 2500 kg, has a large influence on the slag’s viscosity. This implies that in an industrial HMD process, iron losses can be lowered by a relatively small addition to the slag. When looking purely at the slag modifying aspect, all three slag modifiers can be used effectively in an industrial HMD process.
For an industrial use of slag modifiers, more considerations need to be taken into account. As mentioned in § 2.4, the use of fluoride-containing slag modifiers is restricted due to health, safety and environmental reasons. Besides fluoride addition leads to a higher magnesium consumption. NS and PFA are fluoride-free. Of these two, PFA has the lower cost to achieve the same low viscosity. However, as PFA is a byproduct from coal fired power plants or waste incinerators, its composition and size can in practice be less consistent. Furthermore, the ash could contain unwanted components, like heavy metals, which could make the resulting slag difficult to recycle or even hazardous for health, safety and environmental reasons. NS has, as it is a mined mineral, fewer problems with unwanted or unexpected impurities. Also, as it is mined on all continents, its availability is quite wide. However, the alkali metal oxides in NS, which have the desired effect on the slag’s viscosity, also make the slag less suitable for recycling. One of the few options to recycle HMD slag is to recharge it to the BF. Thus the iron in the slag is recovered and the composition of HMD slag is quite close to that of blast furnace (BF) slag. The high sulphur content of the HMD slag is not problematic, as the BF is an excellent desulphuriser [9]. Alkali metals are problematic for the BF, as they can accumulate inside, due to their low melting point. This can lead to skull formation on the BF walls [20]. The addition of alkali metals, also in the form of oxides, is therefore restricted.
Conclusions
In this research slag modifiers were investigated and compared, the objective being to lower the apparent viscosity of slag in the hot metal desulphurisation process in order to decrease iron loss to the slag. Because of the restrictions of fluoride-containing slag modifiers, two fluoride-free slag modifiers, nepheline syenite and pulverised fuel ash, were compared with CaF2 and with a slag without any slag modifier. The main conclusions of this research on synthetic HMD slags are as follows: Both PFA and NS are viable alternatives for fluoride-containing slag modifiers, lowering the apparent viscosity like CaF2, to reduce iron losses at the HMD. At higher basicity and temperature, fluoride-based slag modifiers are more effective. Under these conditions, alkali metal oxides will not have a significant effect on the slag’s apparent viscosity. Lowering the slag’s basicity by adding SiO2 and Al2O3 does lower the apparent viscosity of the HMD slag. At higher temperatures (typically above 1350°C), the apparent viscosity of the HMD slag is already low enough without slag modifiers. Slag modifiers will not significantly contribute to lower iron losses above these temperatures. Relatively small changes in the HMD slag composition can lead to large effects on the slag’s apparent viscosity, and thus iron loss to the slag. This makes the use of slag modifiers for an industrial HMD viable.
